Peer-Reviewed Article
Canadian Trends in Estimated Pharmaceutical Drug Purchases and Projections: 2023
This article has been peer reviewed.
Authors: Mina Tadrous, Pooyeh Graili, Kaleen N. Hayes, Heather L. Neville, Joanne Houlihan, Fiona Clement, Jason R. Guertin, Michael R. Law, Tara Gomes
Abstract
Background: Comprehensive data identifying trends in the pharmaceutical market are limited. Last year, our group presented the first forecast. We report the first annual update of estimated pharmaceutical drug purchases in Canada. The goal is to identify factors that may influence future spending to support public and private decision-makers in predicting the growth of national drug purchases.
Methods: We conducted a time series analysis of annual estimated pharmaceutical drug purchases across Canada between 2001 and 2021 using IQVIA’s Canadian Drugstore and Hospital Purchases Audit. We calculated total estimated pharmaceutical drug purchases and relative percentage change annually, stratified by sector (retail and hospital), and forecast annual spending to 2023. We also assessed recent drug approvals, policies, or legislation that may influence drug spending.
Results: Total drug purchases for 2021 were approximately $35.4 billion, 8.3% higher than in 2020 (7.3% growth in the retail sector; 12.4% growth in the hospital sector). Overall, total spending for the top 25 drugs accounted for 31.2% and 52.3% of total spending in the retail and hospital sectors, respectively. Spending has grown over the past 2 decades, with an annual average growth of 5.4% and 7.4% for the retail and hospital sectors, respectively. The forecast for the retail sector is continued moderate levels of growth in drug spending (7% to 8% annually), with higher rates of growth (12% to 13% annually) in the hospital setting.
Interpretation: We project continued growth in overall drug purchases across the entire Canadian market. New approvals of specialty and oncology drugs and generic formulations of the top 25 drugs are expected to influence drug purchases in 2022–2023. If no action is taken to curb sustained growth in pharmaceutical spending in Canada, costs may necessitate a shift in spending from other public budgets or to private industry and directly to patients. The opportunity cost of continued increased spending on pharmaceuticals must be considered.
Introduction
Total public health care spending in Canada was forecasted by CIHI to reach more than $308 billion in 2021, representing 12.7% of the country’s gross domestic product.1 Public spending on prescription drugs was expected to account for 6.9% ($16.5 billion) of national public health care purchases — a $1.1 billion increase compared with the 2020 projections.1 Public drug spending accounted for 44% of total drug spending in Canada in 2020.1
In 2021, our group published the first of what we aim to be an annual report highlighting spending on drugs in Canada.2 We found that between 2001 and 2020 drug spending increased on average by 5.3% annually in the retail sector and by 7.1% annually in the hospital sector, and we projected that spending was likely to continue increasing until at least 2023.2 Although drug spending has been historically driven by medications for chronic conditions, our results highlighted that biologics and biosimilars had become the key contributors to drug spending.2 Furthermore, based on the available information about novel treatments approved by Health Canada and the US FDA combined with the historical growth in spending,2-7 we projected that drug spending for the years 2021 to 2023 would increase by 3% to 4% annually in the retail sector and by 7% to 8% annually in the hospital sector.2
Budget planning for drug spending is an annual process and requires forecasting of potential growth. We aim to conduct this analysis annually to help inform decision-makers and formulary managers in developing their annual budgets and strategies. Approvals for novel pharmaceutical technologies with the potential to change budget trajectories are expected in the coming years; therefore, an annual update of both the spending forecast and a Horizon Scan are needed. External and independent assessments of drug spending across the country can help support public and private decision-makers better anticipate how various factors could influence drug spending as novel technologies driving spending are expected to continue. In this first update of our planned annual report, we present trends and forecasts of retail and in-hospital estimated pharmaceutical drug purchases across Canada. We also provide a Horizon Scan looking at new innovative drugs that could potentially change the spending trajectory.
Methods
Drug Purchases and Projection
We conducted a retrospective time series analysis of annual estimated pharmaceutical drug purchases (i.e., drug purchases) across Canada between January 1, 2001, and December 31, 2021, using IQVIA’s Canadian Drugstore and Hospital (CDH) Purchases Audit. These data estimate purchasing costs and unit volumes of all pharmaceutical products purchased by the Canadian retail and hospital sectors.8 This audit is derived from a sample of outlets within these sectors (more than one-third of retail and 86% of hospital sectors) in each province and territory, which are projected to represent total pharmaceutical purchasing at the national level using proprietary methods. We included all pharmaceutical purchasing of all dosage forms and formulations over the study period. Purchasing may be made through a wholesaler or directly from the manufacturer, and therefore may include mark-ups, but does not capture discounts and rebates. Although CDH projections will not capture any subsequent rebates or volume discounts credited to the outlets afterward, it does reflect all up-front discounts for each specific invoice. Due to the aggregated nature of the data provided, research ethics board approval was not required.
We describe total estimated pharmaceutical drug purchases across Canada annually (using calendar years) over the study period, stratified by sector (retail versus hospital). We calculated the annual growth in purchases as the relative percentage change from the previous year. We used exponential smoothing models, applying Holt’s method, to forecast annual estimated pharmaceutical drug purchases in 2022 and 2023. Costs were reported as nominal annual costs, and nominal costs were used for projection models. In a sensitivity analysis, we inflated annual costs before 2021 to 2021 values using the all-items consumer price indexes.9 Finally, we identified the 25 medications with the highest total purchases in the calendar year 2021 in the retail and hospital sectors separately.
Horizon Scan
An Environmental Scan of global and national drug spending and pipeline reports was conducted to identify therapeutic classes with potentially major impacts on pharmaceutical spending.10-13 All medications approved by Health Canada in 2021 and the first quarter of 2022, as well as generic drugs and biosimilars currently under review, were assessed for anticipated budget impacts.14 New drug approvals by the US FDA in 2021 and the first quarter of 2022 were also reviewed to identify drugs that may soon enter the Canadian market.15
Drugs with potential high impact on future spending (via upward pressure or downward pressure) were flagged based on the following criteria: disease prevalence, current medication use and availability in the therapeutic area, anticipated cost, and public plan reimbursement recommendations by CADTH. Based on the list of drugs with the highest potential for impact on the Canadian market, the authors made a consensus recommendation on whether the projected spending might be more likely on the high, middle, or low end of the projected confidence interval (CI) range estimated from the forecast analysis for the next 2 years.
Results
Trends in Canadian drug purchases from 2001 to 2021 in the retail and hospital sectors are presented in Figure 1 and Figure 2, respectively. Graphs that include values accounting for inflation can be found in Appendix 1 (Figure 4 and Figure 5).
Overall Spending
In 2021, total drug purchases in Canada grew 8.3%, reaching $35.4 billion (versus $32.7 billion in 2020). This was higher than our previous forecasted rate of 4.2%. The retail sector accounted for 84.5% of drug purchases in 2021 ($29.9 billion), and the hospital sector accounted for 15.5% ($5.5 billion). Retail and hospital purchases in 2021 increased from 2020 by 7.3% and 12.4%, respectively. The rates in both sectors were also higher than our forecasted rates of 6.9% and 3.8%, respectively. Figure 3 illustrates the trends in the annual change in purchases across both sectors.
Retail and Hospital Spending
In the retail sector, total drug purchases increased by 184%, from $10.5 billion in 2001 to $29.9 billion in 2021. In the hospital sector, total drug purchases grew 309% from $1.3 billion in 2001 to $5.5 billion in 2021. Among retail purchases, the average annual growth was higher in earlier years (6.7% between 2001 and 2011) compared with 2012 onward (4.1% between 2012 and 2021); 2011 was the only year with a decrease in annual purchases (−2.9%). Over the entire period (2001 to 2021), the average annual growth was 5.4% and 7.4% for the retail and hospital sectors, respectively.
In the hospital sector, the average annual growth was similar to the retail environment between 2001 and 2011 (6.5%) but grew quicker between 2012 and 2021 (8.4%); there was no year with a reported decrease in annual spending. The drug purchases for hospital-administered drugs grew 12.7%, 18.4%, 6.9%, and 12.4% in 2018, 2019, 2020, and 2021, respectively. In comparison, drug purchases in the retail sector experienced a smaller growth of 3.4%, 5.2%, 3.8%, and 7.3% in 2018, 2019, 2020, and 2021, respectively.
Figure 1: Total Estimated Pharmaceutical Drug Purchases in Canada for the Retail Sector From 2001 to 2021
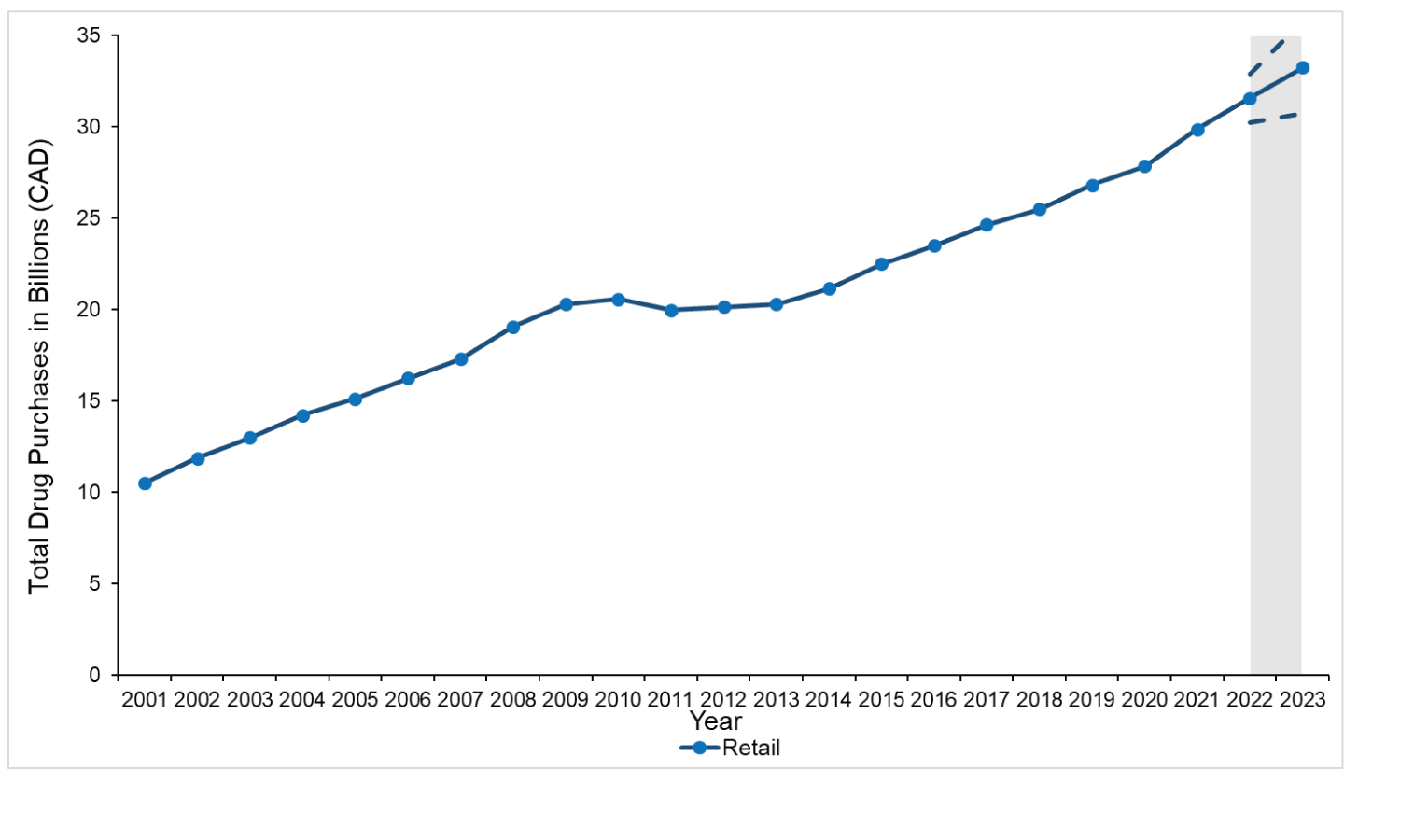
Note: The grey zone illustrates forecasted purchases in the retail sector (solid line) with 95% confidence interval estimates (dashed lines) for 2022 and 2023.
Source: This figure is based on information licensed from IQVIA: Canadian Drugstore and Hospital Purchases Audit for 2001 to 2021 estimates of real-world activity. All rights reserved.
Figure 2: Total Estimated Pharmaceutical Drug Purchases in Canada for the Hospital Sector From 2001 to 2021
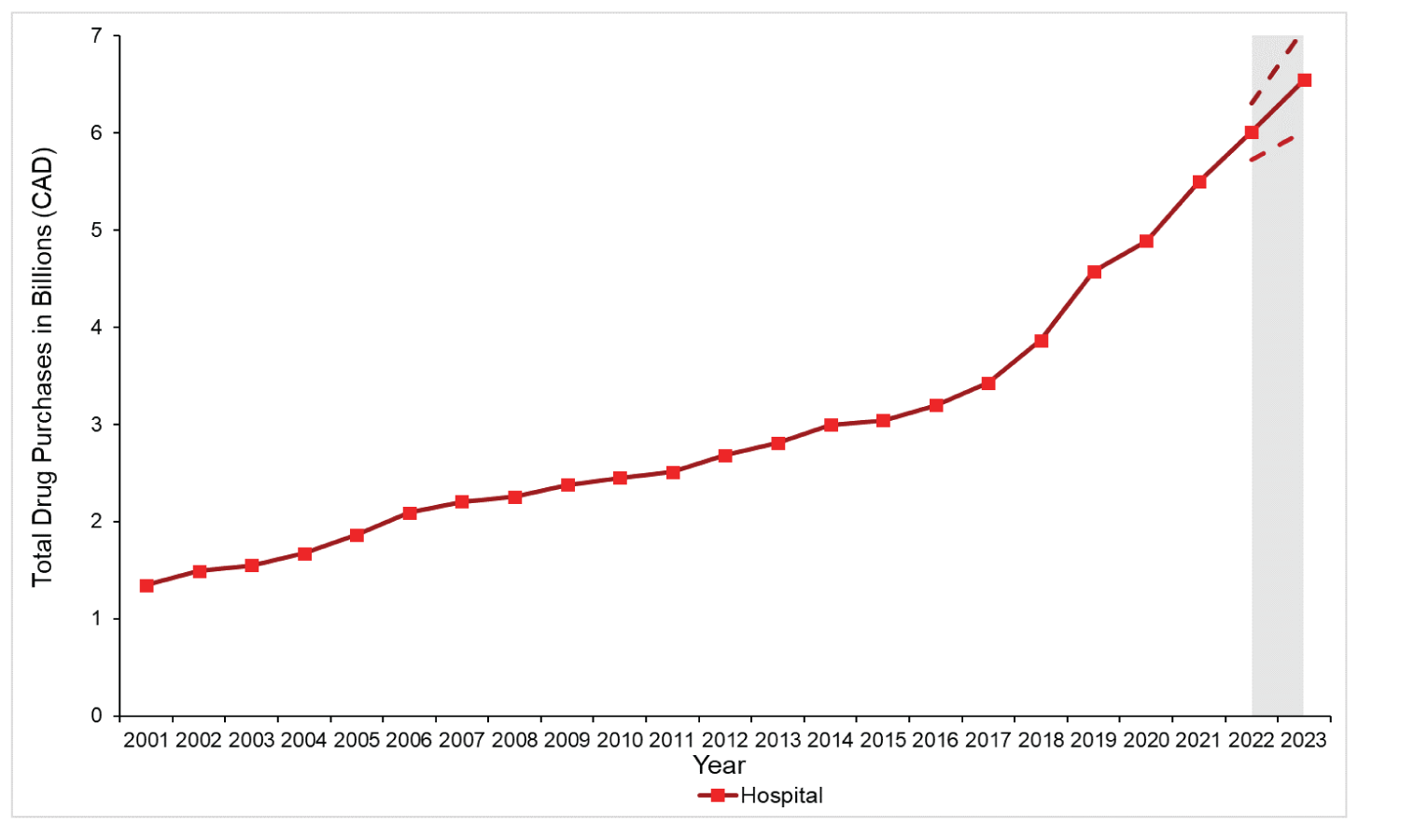
Note: The grey zone illustrates forecasted purchases in the hospital sector (solid line) with 95% confidence interval estimates (dashed lines) for 2022 and 2023.
Source: This figure is based on information licensed from IQVIA: Canadian Drugstore and Hospital Purchases Audit for 2001 to 2021 estimates of real-world activity. All rights reserved.
Drug Expenditure Forecast for 2022 and 2023
We forecast a yearly increase (all sectors combined) in drug purchases of 6.2% in 2022 and 5.9% in 2023 (Figure 3). We project that purchases for hospital-administered drugs will increase annually by 9.4% and 8.8% for 2022 and 2023, respectively; in the retail setting, purchases will increase between 5.6% and 5.3% annually for 2022 and 2023, respectively. When considered in absolute costs, we forecast an increase of $33.2 billion (95% CI, $30.7 billion to $35.8 billion) for retail purchases and $6.5 billion (95% CI, $6.0 billion to $7.0 billion) for hospital purchases by 2023 (Figure 1 and Figure 2).
Figure 3: Annual Growth in Estimated Pharmaceutical Drug Purchases From the Previous Year (2002 to 2021) for the Retail and Hospital Sectors
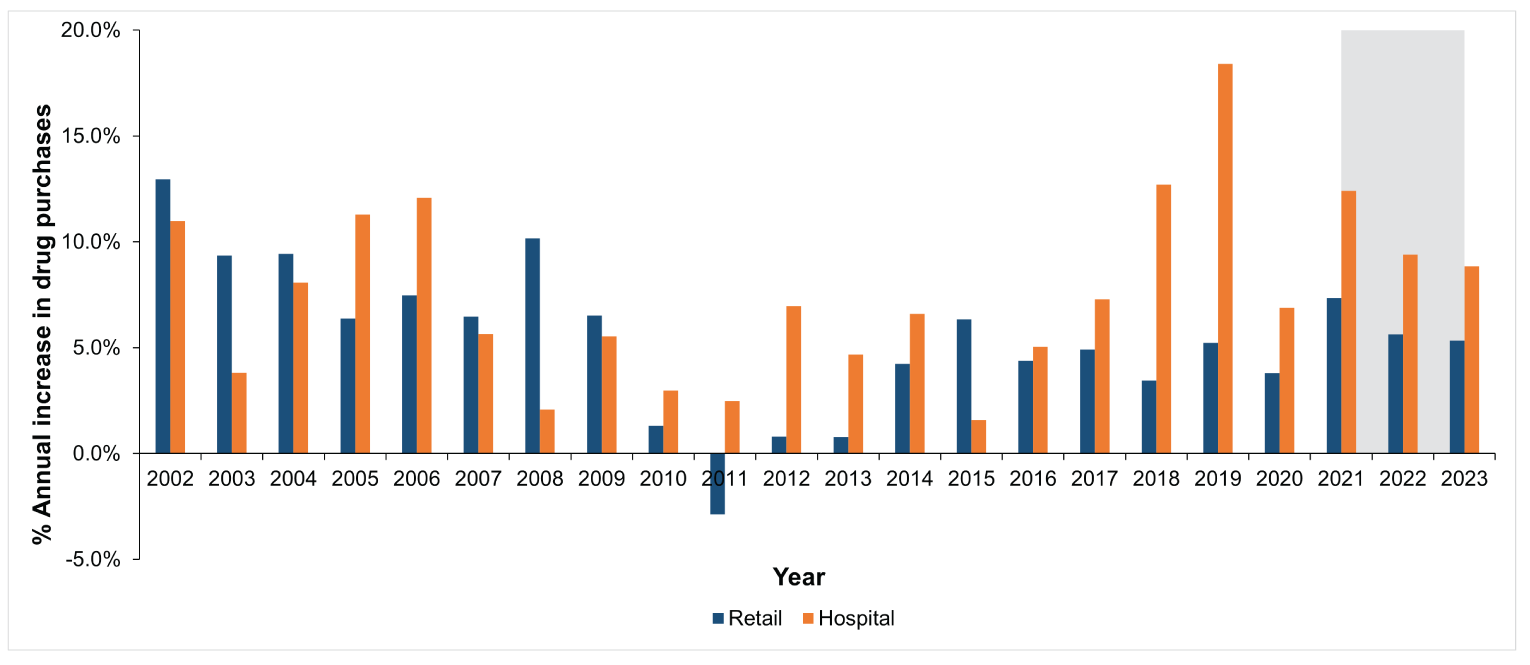
Note: Blue bars represent the retail sector and orange bars represent the hospital sector. The grey zone illustrates forecasted annual growth for 2022 and 2023.
Source: This figure is based on information licensed from IQVIA: Canadian Drugstore and Hospital Purchases Audit for 2001 to 2021 estimates of real-world activity. All rights reserved.
Top 25 Drugs by Overall Purchases in 2021
Table 1 provides the top 25 drugs by purchases in retail and hospital settings across the country for the year 2021. In terms of purchases, infliximab ($1.3 billion), adalimumab ($979 million), and ustekinumab ($636.3 million) were the top 3 drugs in the retail setting, whereas pembrolizumab ($518.8 million), daratumumab ($254.2 million), and nivolumab ($245.7 million) were the top 3 drugs in the hospital setting. Overall, total purchases for the top 25 drugs in both markets in 2021 accounted for $12.2 billion ($9.3 billion and $2.9 billion in the retail and hospital sectors, respectively), which accounted for 31.2% and 52.3% of total spending in each sector, respectively. Only 3 drugs were in the top 25 of both lists: a vascular endothelial growth factor-A (VEGF-A) antagonist (aflibercept: number 4 in retail spending and number 17 in hospital spending) and 2 kinase inhibitors (ibrutinib: number 13 in retail spending and number 7 in hospital spending; palbociclib: number 24 in retail spending and number 10 in hospital spending).
Table 1: Top 25 Drugs by Purchases in Retail and Hospital Settings for the Calendar Year 2021 and Trends Compared With 2020
Rank | Retail | Hospital | ||||
|---|---|---|---|---|---|---|
Drug | Total estimated pharmaceutical drug purchases in 2021 | Trend from 2020 | Drug | Total estimated pharmaceutical drug purchases in 2021 | Trend from 2020 | |
1 | Infliximab | $1,273,113,789 | Up | Pembrolizumab | $518,807,002 | Up |
2 | Adalimumab | $978,987,507 | Up | Daratumumab | $254,230,664 | Up |
3 | Ustekinumab | $636,290,643 | Up | Nivolumab | $245,654,407 | Down |
4 | Aflibercept | $608,972,710 | Up | Haemagglutinin (nonspecific) | $164,311,416 | Up |
5 | Semaglutide | $576,044,914 | Up | Rituximab | $160,022,931 | Down |
6 | Apixaban | $410,725,793 | Up | Pertuzumab: Trastuzumab | $138,664,496 | Up |
7 | Methylphenidate | $340,706,844 | Up | Ibrutinib | $120,818,606 | Up |
8 | Ranibizumab | $325,530,299 | Up | Trastuzumab | $110,287,917 | Down |
9 | Empagliflozin | $320,537,190 | Up | Durvalumab | $106,020,619 | Up |
10 | Metformin: sitagliptin | $317,062,227 | Down | Palbociclib | $92,539,410 | Up |
11 | Rivaroxaban | $290,607,683 | Up | Bevacizumab | $90,680,807 | Down |
12 | Lisdexamfetamine | $288,792,626 | Up | Vaccine, pneumococcal conjugate | $88,725,538 | Up |
13 | Ibrutinib | $275,252,945 | Up | Osimertinib | $82,691,346 | Up |
14 | Vedolizumab | $274,476,001 | Up | Ipilimumab | $73,638,520 | Up |
15 | Etanercept | $268,976,817 | Down | Darbepoetin alfa | $71,675,858 | Up |
16 | Budesonide: formoterol | $251,975,825 | Down | Nusinersen | $62,287,791 | Down |
17 | Paliperidone palmitate | $234,005,797 | Up | Aflibercept | $61,455,247 | Up |
18 | Rosuvastatin | $233,956,220 | Up | Erythropoietin alpha | $60,796,021 | Down |
19 | Golimumab | $231,374,597 | Up | Trastuzumab emtansine | $59,868,119 | — |
20 | Insulin glargine | $224,952,661 | Down | Alteplase | $59,159,609 | Up |
21 | Sofosbuvir: velpatasvir | $219,433,495 | Down | Vaccine, HPV types 6, 11, 16, 18, 3 | $56,676,885 | Down |
22 | Sitagliptin | $200,224,171 | Down | Vaccine, rotavirus | $54,616,577 | Up |
23 | Denosumab | $186,499,982 | — | Bictegravir, emtricitabine, and tenofovir alafenamide | $51,108,504 | — |
24 | Palbociclib | $180,998,587 | — | Antipoliomyelitic vaccine, diphtheria toxoid, Haemophilus influenzae B, pertussis, and tetanus purified toxoid | $49,942,835 | — |
25 | Ocrelizumab | $178,010,209 | — | Enoxaparin | $40,336,899 | — |
Total of top 25 | $9,327,509,532 | $2,875,018,024 | ||||
Total 2021 spending | $29,877,770,437 | $5,498,567,931 | ||||
Top 25% of total | 31.20% | 52.30% | ||||
Note: The drugs with a dash are new entries to the top 25 drugs in 2021.
Source: This table is based on information licensed from IQVIA: Canadian Drugstore and Hospital Purchases Audit for 2001 to 2021 estimates of real-world activity. All rights reserved.
Horizon Scan
Fifteen global reports were reviewed to determine trends for drug spending in Canada (Appendix 1, Table 3). In Table 2, we present the drugs that are anticipated to have the most impact on drug purchases in 2022 and 2023. Here, we summarize major themes for new and upcoming therapies in Canada. We first highlight treatments we believe will have the largest impact on increased spending (“upward pressure”) then discuss potential mitigating factors (“downward pressure”). A complete list of drug approvals for Health Canada and the US FDA in 2021 and the first quarter of 2022 are presented in Table 4, and Table 5, respectively. The FDA approved 50 novel drugs in 2021 compared with 53 in 2020.7 Health Canada approved 78 drugs in 2021 compared with 84 new drugs in 2020, of which 40 were novel drugs.16
Table 2: Selected Drugs and Biologicals That Received Health Canada and/or FDA Approval in 2021 and Early 2022 and Are Expected to Affect Drug Spending
Category | Drug | Brand name or reference product | Indication | Health Canada approval date | FDA approved |
|---|---|---|---|---|---|
Diabetes | Insulin aspart biosimilar | Kirsty and Trurapi (for NovoRapid) | Diabetes | October 2021 | No |
Insulin lispro biosimilar | Lyumjev, Liprelog, and Admelog (for Humalog) | Diabetes | January 2021 | Lyumjev: yes Liprelog: no Admelog: yes | |
Canagliflozin, dapagliflozin, empagliflozin, combinations generics | NA | SGLT2 inhibitors for diabetes with additional indications (established cardiovascular disease, nephropathy, heart failure) | Under review | No (tentative approval) | |
Sitagliptin, linagliptin, combinations generics | NA | DPP-4 inhibitors for diabetes | Under review | No (tentative approval) | |
Liraglutide generic | NA | GLP-1 receptor agonist for diabetes and established cardiovascular disease | Under review | No | |
Weight management | Semaglutide | Wegovy | GLP-1 receptor agonist for chronic weight management in adults | November 2021 | Yes |
Liraglutide | Saxenda | GLP-1 receptor agonist for chronic weight management in adolescents aged 12 and older | February 2021 | Yes | |
Migraine | Eptinezumab | Vyepti | CGRP antagonist for prevention of migraine | January 2021 | Yes |
Lasmiditan | Reyvow | 5-HT1F receptor agonist for the treatment of acute migraine | Not under review | Yes | |
Cystic fibrosis | Elexacaftor, ivacaftor, tezacaftor | Trikafta | Cystic fibrosis with F508del mutation in adolescents and adults 12 years and older | June 2021 | Yes |
HIV | Abacavir, dolutegravir, and lamivudine generic | NA | HIV | Under review | No |
Dolutegravir generic | NA | HIV | Under review | No (tentative approval) | |
Emtricitabine, rilpivirine, and tenofovir alafenamide generic | NA | HIV | Under review | No | |
Macular degeneration | Ranibizumab biosimilar | Byooviz (for Lucentis) | Age-related macular degeneration, diabetic macular edema, diabetic retinopathy | March 2022 | Yes |
ALS | Sodium phenylbutyrate and ursodoxicoltaurine | Albrioza | ALS | Under review | No |
Spinal muscular atrophy | Risdiplam | Evrysdi | SMA, infants aged 2 months and older up to adults aged 25 years | April 2021 | Yes |
Anticoagulant | Apixaban generic | NA | Prevention and treatment of VTE, prevention of stroke in atrial fibrillation | Under review | No |
Immune diseases | Tofacitinib generic | NA | JAK inhibitor for rheumatoid arthritis, psoriatic arthritis, ulcerative colitis | Under review | No (tentative approval) |
Adalimumab, multiple biosimilars | NA | Multiple indications (e.g., rheumatoid arthritis, Crohn disease, ulcerative colitis) | 2021 | Yes | |
Multiple sclerosis | Teriflunomide generic | NA | Relapsing remitting MS | May 2022 | Yes |
Ofatumumab | Kesimpta | Relapsing remitting MS | January 2021 | Yes | |
Ponesimod | Ponvory | Relapsing remitting MS | April 2021 | Yes | |
Diroximel fumarate | Vumerity | Relapsing remitting MS | Not under review | Yes | |
Schizophrenia | Paliperidone long-acting injection generic | NA | Schizophrenia and related psychotic disorders | Under review | Yes |
Oncology | Brexucabtagene autoleucel | Tecartus | CAR T-cell therapy for relapsed or refractory mantle cell lymphoma | June 2021 | Yes |
Ciltacabtagene autoleucel | Carvykti | CAR T-cell therapy for relapsed or refractory multiple myeloma | Under review | Yes | |
Encorafenib | Braftovi | Additional indication of metastatic colorectal cancer | March 2021 | Yes | |
Trastuzumab deruxtecan | Enhertu | Unresectable or metastatic HER2-positive breast cancer after treatment with trastuzumab emtansine | April 2021 | Yes | |
Daratumumab | Darzalex | Additional indications in multiple myeloma and amyloidosis | April 2021 | Yes | |
Sacituzumab govitecan | Trodelvy | Advanced or metastatic triple negative breast cancer | September 2021 | Yes | |
Isatuximab | Sarclisa | Relapsed and refractory multiple myeloma | September 2021 | Yes | |
Venetoclax | Venclexta | Additional indications for first-line CML and AML | January 2021 | Yes | |
Selinexor | Xpovio | First oral selective inhibitor of nuclear export for multiple myeloma | Under review | Yes | |
Ibrutinib generic | Imbruvica | Multiple indications | Under review | Yes | |
Carfilzomib generic | Kyprolis | Relapsed multiple myeloma | Under review | Yes | |
Pomalidomide generic | Pomalyst | Relapsed multiple myeloma | Under review | Yes |
ALS = amyotrophic lateral sclerosis; AML = acute myeloid leukemia; CAR = chimeric antigen receptor; CGRP = calcitonin gene-related peptide; CML = chronic myelogenous leukemia; DPP-4 = dipeptidyl peptidase-4; GLP-1 = glucagon-like peptide-1; MS = multiple sclerosis; NA = not applicable; SGLT2 = sodium-glucose cotransporter-2; SMA = spinal muscular atrophy; VTE = venous thromboembolism.
Potential for Upward Pressure
In oncology, chimeric antigen receptor (CAR) T-cell therapies are anticipated to make the largest impact on spending. A new CAR T-cell therapy was recently approved for the treatment of mantle cell lymphoma, and another is under review by Health Canada for multiple myeloma, a relatively common cancer. Daratumumab (Darzalex) is the number 2 ranked drug in the hospital sector, and purchases have increased over the past 2 years. It will likely impact spending even more in coming years because it has now been approved for additional indications of multiple myeloma and amyloidosis therapy.
Certain therapeutic areas are likely to increase spending due to the prevalence of these conditions in the Canadian population. Drugs for migraine treatment continue to increase drug spending as the new calcitonin gene-related peptide inhibitors take over a higher market share for the prevention of chronic migraine. Lasmiditan (Reyvow), a first-in-class medication indicated for acute treatment of migraine that was approved by the FDA in 2019, is not yet under review by Health Canada. Semaglutide (Wegovy), the number 5 ranked drug in the retail sector, has been approved for chronic weight management in adults and is currently under review by CADTH for this indication. Liraglutide (Saxenda) has an expanded indication for weight management in adolescents 12 years and older; however, CADTH did not recommend reimbursement by public plans. New medications for multiple sclerosis continue to be approved, such as ofatumumab (a once-monthly injection) and ponesimod, both of which are expected to increase the cost of treatment overall. Of note, ocrelizumab, which is a treatment for multiple sclerosis that is infused every 6 months, is a new addition to the top 25 in retail spending this year. Finally, aducanumab (Aduhelm) received FDA approval for the treatment of Alzheimer disease in 2021. Approximately 76,000 Canadians are diagnosed with dementia each year,17 and aducanumab costs approximately US$28,000 annually. The medication is presently under review by Health Canada, but it remains unclear whether it will be approved and funded in Canada.
For rare diseases, the increasing number of therapies with high price tags are expected to drive costs, particularly in diseases that previously had no or few treatment options available. Notable examples include sodium phenylbutyrate and ursodoxicoltaurine for amyotrophic lateral sclerosis and risdiplam for spinal muscular atrophy. In addition, elexacaftor, ivacaftor, and tezacaftor (Trikafta) was approved in 2021 for cystic fibrosis and received a positive recommendation for reimbursement by CADTH for adults and adolescents aged 12 years and older. Trikafta received an expanded indication for pediatric patients aged 6 years and older in 2022 and another positive recommendation to reimburse by CADTH, indicating continued growth for this product.
Potential for Downward Pressure
A limited number of approved or upcoming generic medications and biosimilars have been identified as having the potential to exert downward pressure on drug spending. In oncology, 2 new generic medications have been approved for multiple myeloma (carfilzomib and pomalidomide) and are expected to have an impact. Generic ibrutinib may impact both retail (ranked number 13) and hospital (ranked number 7) spending, but savings will be partially blunted by the expected replacement with a new kinase inhibitor, zanubrutinib. Among medications for diabetes, insulin aspart and lispro have new biosimilar market entries to join Trurapi (insulin aspart biosimilar for NovoRapid) and Admelog (insulin lispro biosimilar for Humalog), which could lead to reduced spending on insulin.18 Furthermore, several generic drugs in the sodium-glucose cotransporter-2 (SGLT2) inhibitor, dipeptidyl peptidase-4 (DPP-4) inhibitor, and glucagon-like peptide-1 (GLP-1) receptor agonist classes are under review and expected to significantly decrease costs for diabetes treatment overall. Although several generic antiretroviral products for HIV are also under review, it is anticipated the abacavir, dolutegravir, and lamivudine combination will exert the highest impact because it is 1 of the recommended initial therapies in HIV treatment guidelines.19 Based on market share, generic apixaban (number 6 in retail spending) for venous thromboembolism and generic tofacitinib for immune diseases such as rheumatoid arthritis and ulcerative colitis will likely reduce costs. Finally, the first market entry for a ranibizumab biosimilar (number 8) and the availability of several adalimumab biosimilars (number 2) are expected to decrease spending substantially in the retail sector.20
Interpretation
Spending on pharmaceuticals has grown considerably over the past 2 decades in Canada, leading to a total market size of more than $35 billion in 2021. In 2021, the annual growth compared to the previous year in spending reached 7.3% for retail drug purchases and 12.4% for hospital drug purchases, far surpassing growth rates seen the year prior and our previous forecast in 2021. This is particularly apparent in the retail sector where the annual growth in spending between 2020 and 2021 was higher than has been observed for the past decade. This expansion is likely driven by new product approvals, higher market entry prices for new drugs, and growing utilization. Thus, we anticipate continued increases in the retail sector combined with accelerated growth in the hospital sector, leading to projected total annual drug purchases nearing $40 billion by 2023. Importantly, the anticipated growth in the number of new therapies and associated costs will continue to put pressure on constrained government budgets across the country, leading to a need for strategies that balance access to novel therapeutics against limited resources.
It is expected that the pressure from growth in the available therapies identified in the pipeline are likely to continue to affect the degree of change in spending in the coming years. We did note fewer approvals in comparison to 2021, which was a record-breaking year. Overall, we believe the growth in spending will be on the lower end of the predicted range in the outpatient retail setting due to the introduction of new generics or biosimilar formulations for several top 25 therapies, including adalimumab, ranibizumab, 2 direct oral anticoagulants, DPP-4 inhibitors, SGLT2 inhibitors, GLP-1 agonists, and insulin biosimilars. However, much of the effect of biosimilars will depend on their uptake in Canada. Presently, most Canadian provincial public drug plans require biologic-naive patients to initiate a biosimilar formulation, and half of these drug plans have introduced mandatory non-medical biosimilar switching policies over the past several years.21 If a large province, such as Ontario, implemented similar policies, we anticipate this would have a large effect on total national spending projections. Finally, despite this downward pressure, diabetes therapies have particular potential to be “blockbuster” retail pharmacy drugs due to the high prevalence of diabetes and the expanded scope of many diabetes drugs to include weight loss. Therefore, it will be important to monitor the ongoing use of these medications to ensure appropriate use and manage spending.22
Importantly, the validity of our projections remains uncertain since they were mostly based on data collected before the COVID-19 pandemic. This also explains why our current projections for 2023 are higher than our previous forecast. The pandemic has substantially impacted health care across the country, including drug spending. For example, at the time of publishing our first report, only 1 treatment, remdesivir, was approved by Health Canada and the FDA for a COVID-19 indication but spending on this drug had yet to occur. Since then, many treatments, including a series of COVID-19 vaccines, were not only developed but are approved and being used across the country. Spending on these new treatments may dramatically increase drug spending across Canada, maybe even beyond the upper bound of our projections for 2022 and 2023. However, other factors directly and indirectly related to the COVID-19 pandemic may have the opposite impact. The second, third, fourth, and fifth waves of COVID-19 have caused various levels of delayed treatments within hospitals across the country and have led to reduced screening of chronic conditions and cancers, potentially reducing the use of health care resources, including pharmaceuticals, at least in the short-term. We did observe that 16 of the top 25 drugs experienced expansion in their total purchases compared with last years’ report in 2020.
Our analysis has limitations that warrant discussion. First, we do not have information on the rebates that manufacturers provide to public and private drug plans or hospital purchasers because these contracts are kept confidential. These discounts can be sizable — for example, pan-Canadian Pharmaceutical Alliance (pCPA) negotiations yielded $1.24billion in savings in 2017–2018.23 However, our results still represent the total purchases in the current drug system, and we do not anticipate major differences in the proportion of rebates to total purchases in recent years. The purchase prices do account for up-front discounts. Rebates are also re-invested to pay for future drug spending and thus are paid forward in spending. Additionally, rising prices have important consequences on patient co-pays depending on payment method and plan structure.24 Second, our data had no information on payers (i.e., public insurance, private insurance, or out-of-pocket) and thus only characterizes global spending in Canada and the future impact for all payers.
Conclusion
Overall pharmaceutical drug purchases in Canada for 2021 were higher than our predicted increases in our previous report. This was for both the retail and hospital sectors and was caused by the combined impact of higher utilization and higher prices for new therapies. Because the nature of drugs is driving the change, there is a continued need for rigorous assessment and price negotiation by public payers to ensure these investments represent good value for money. We project continued growth in overall drug purchases across the entire Canadian market. Specifically, the forecast for the retail sector is continued moderate levels of growth in drug spending (7% to 8% annually), with higher rates of growth (12% to 13% annually) in the hospital setting.
References
1.Canadian Institute for Health Information. National Health Expenditure Trends | CIHI. [accessed 2022 Jul 25]. https://www.cihi.ca/en/national-health-expenditure-trends
2.Tadrous M, Shakeri A, Hayes KN, Neville HL, Houlihan J, Clement F, Guertin JR, Law MR, Gomes T. Canadian Trends and Projections in Prescription Drug Purchases: 2001–2023. Canadian Journal of Health Technologies. 6377 [accessed 2022 Jul 25];1(11). doi:10.51731/CJHT.2021.209
3.Health Canada. Approved in 2020: Drugs for human use - Canada.ca. 2022 Jul 26 [accessed 2022 Jul 25]. https://www.canada.ca/en/health-canada/services/publications/drugs-health-products/drug-medical-device-highlights-2020/approved-2020-drugs-human-use.html
4.Morgan SG, Bathula HS, Moon S. Pricing of pharmaceuticals is becoming a major challenge for health systems. BMJ (Clinical research ed.). 2020 [accessed 2022 Jul 24];368. https://pubmed.ncbi.nlm.nih.gov/31932289/. doi:10.1136/BMJ.L4627 PubMed
5.Health Canada. Mid-year update: Health products approved in 2021 - Canada.ca. 2022 Jul 26 [accessed 2022 Jul 25]. https://www.canada.ca/en/health-canada/services/drugs-health-products/highlights-reports/mid-year-update-2021.html
6.Morgan S, Cunningham C. Population Aging and the Determinants of Healthcare Purchases: The Case of Hospital, Medical and Pharmaceutical Care in British Columbia, 1996 to 2006. Healthcare Policy. 2011;7(1):68–79. https://www.longwoods.com/product/22525 PubMed
7.US Food and Drug Administration. Advancing Health Through Innovation: New Drug Therapy Approvals 2021. 2021. www.fda.gov
8.Brogan IMS. Canadian drug store and hospital purchases audit database. Montreal: IMS Brogan. 2007.
9.Statistics Canada. Consumer Price Index, annual average, not seasonally adjusted.
10.Tichy EM, Schumock GT, Hoffman JM, Suda KJ, Rim MH, Tadrous M, Wiest MD, Matusiak LM, Clark JS, Cuellar S, et al. National trends in prescription drug purchases and projections for 2020. American journal of health-system pharmacy : AJHP : official journal of the American Society of Health-System Pharmacists. 2020 [accessed 2022 Jul 24];77(15):1213–1230. https://pubmed.ncbi.nlm.nih.gov/32412055/. doi:10.1093/AJHP/ZXAA116
11.Telus Health. Health T. The Drug Pipeline: What private plans can expect in 2021. 2021.
12.Patented Medicine Prices Review Board. Meds Pipeline Monitor 2019 - Canada.ca. Ottawa (ON): PMPRB. 2020 [accessed 2022 Jul 24]. https://www.canada.ca/en/patented-medicine-prices-review/services/reports-studies/meds-pipeline-monitor-2019.html
13.ExpressScripts Canada. Prescription Drug Trend Report. 2022. https://www.express-scripts.ca/sites/default/files/2022-04/ExpressScriptsCanada2022DrugTrendReport.pdf
14.Government of Canada. Generic submissions under review - Canada.ca. [accessed 2022 Jul 25]. https://www.canada.ca/en/health-canada/services/drug-health-product-review-approval/generic-submissions-under-review.html
15.US Food and Drug Administration. Novel Drug Approvals for 2021 | FDA. [accessed 2022 Jul 25]. https://www.fda.gov/drugs/new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2021
16.Health Canada. DRUG AND MEDICAL DEVICE HIGHLIGHTS. 2020. https://www.canada.ca/content/dam/hc-sc/documents/services/publications/drugs-health-products/drug-medical-device-highlights-2020/drug-medical-device-highlights-2020.pdf
17.Public Health Agency of Canada. Dementia in Canada, including Alzheimer’s disease : highlights from the Canadian Chronic Disease Surveillance System. 2017 [accessed 2022 Jul 25]:5.
18.Schneider T, Gomes T, Hayes KN, Suda KJ, Tadrous M. Comparisons of Insulin Spending and Price Between Canada and the United States. Mayo Clinic proceedings. 2022 [accessed 2022 Aug 5];97(3):573–578. https://pubmed.ncbi.nlm.nih.gov/35135692/. doi:10.1016/J.MAYOCP.2021.11.028 PubMed
19.Panel on Antiretroviral Guidelines for Adults N-. Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents with HIV Developed by the DHHS Panel on Antiretroviral Guidelines for Adults and Adolescents-A Working Group of the Office of AIDS Research Advisory Council (OARAC) How to Cite the Adult and Adolescent Guidelines: Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents with HIV. Department of Health and Human. [accessed 2022 Aug 5].
20.SMART & BIGGAR. Biosimilars approved in Canada. [accessed 2022 Jul 26]. https://www.smartbiggar.ca/insights/biosimilars
21.Siu EC, Tomalin A, West K, Anderson S, Wyatt G. An ever-evolving landscape: an update on the rapidly changing regulation and reimbursement of biosimilars in Canada. GaBI Journal. 2019 [accessed 2022 Jul 24];8(3):107–118. doi:10.5639/GABIJ.2019.0803.014
22.Ibekwe A, Perras C, Mierzwinski-Urban M. Monoclonal Antibodies to Prevent Migraine Headaches - PubMed. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2016. https://pubmed.ncbi.nlm.nih.gov/30855775/
23.Ontario Government. Ontario Public Drug Programs Ministry of Health and Long-Term Care. 2017.
24.Memedovich KA, Manns B, Beall R, Hollis A, Clement F. The impact of pharmaceutical rebates on patients’ drug purchases. CMAJ. 2019 [accessed 2022 Jul 25];191(11):E308–E312. https://www.cmaj.ca/content/191/11/E308. doi:10.1503/CMAJ.181041/-/DC1 PubMed
Appendix 1: Additional Figures and Tables
Note that this appendix has not been copy-edited.
Figure 4: Total Estimated Pharmaceutical Drug Purchases in Canada for the Retail Sector From 2001 to 2021
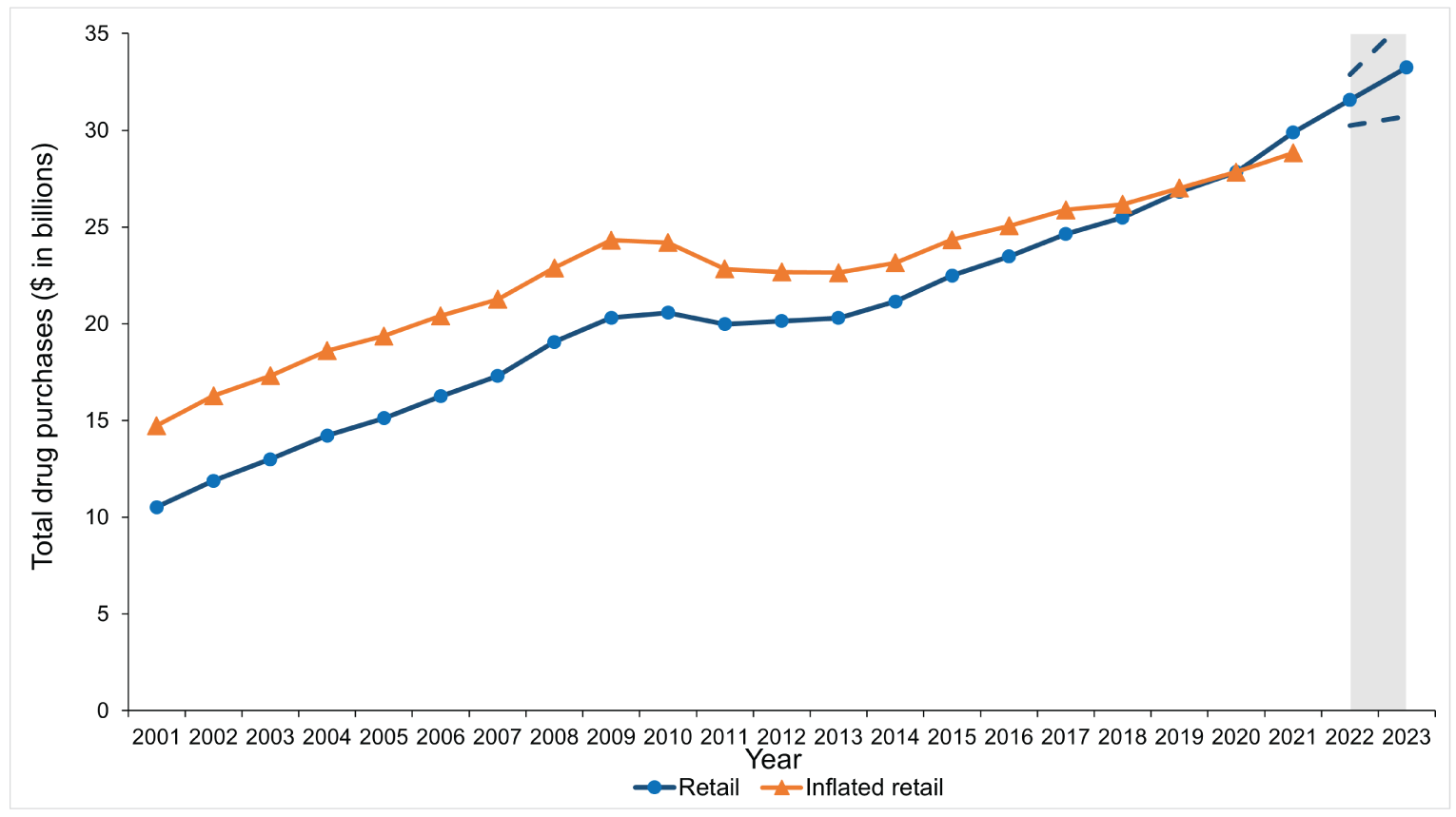
Note: The grey zone illustrates forecasted purchases in the retail sector (solid line) with 95% confidence interval estimates (dashed lines) for 2022 and 2023. Blue line with circles = non–inflated-adjusted purchases; orange line with triangles = inflated-adjusted purchases.
Source: This figure is based on information licensed from IQVIA: Canadian Drugstore and Hospital Purchases Audit for the period of 2001 to 2021 estimates of real-world activity. All rights reserved.
Figure 5: Total Estimated Pharmaceutical Drug Purchases in Canada for the Hospital Sector From 2001 to 2021
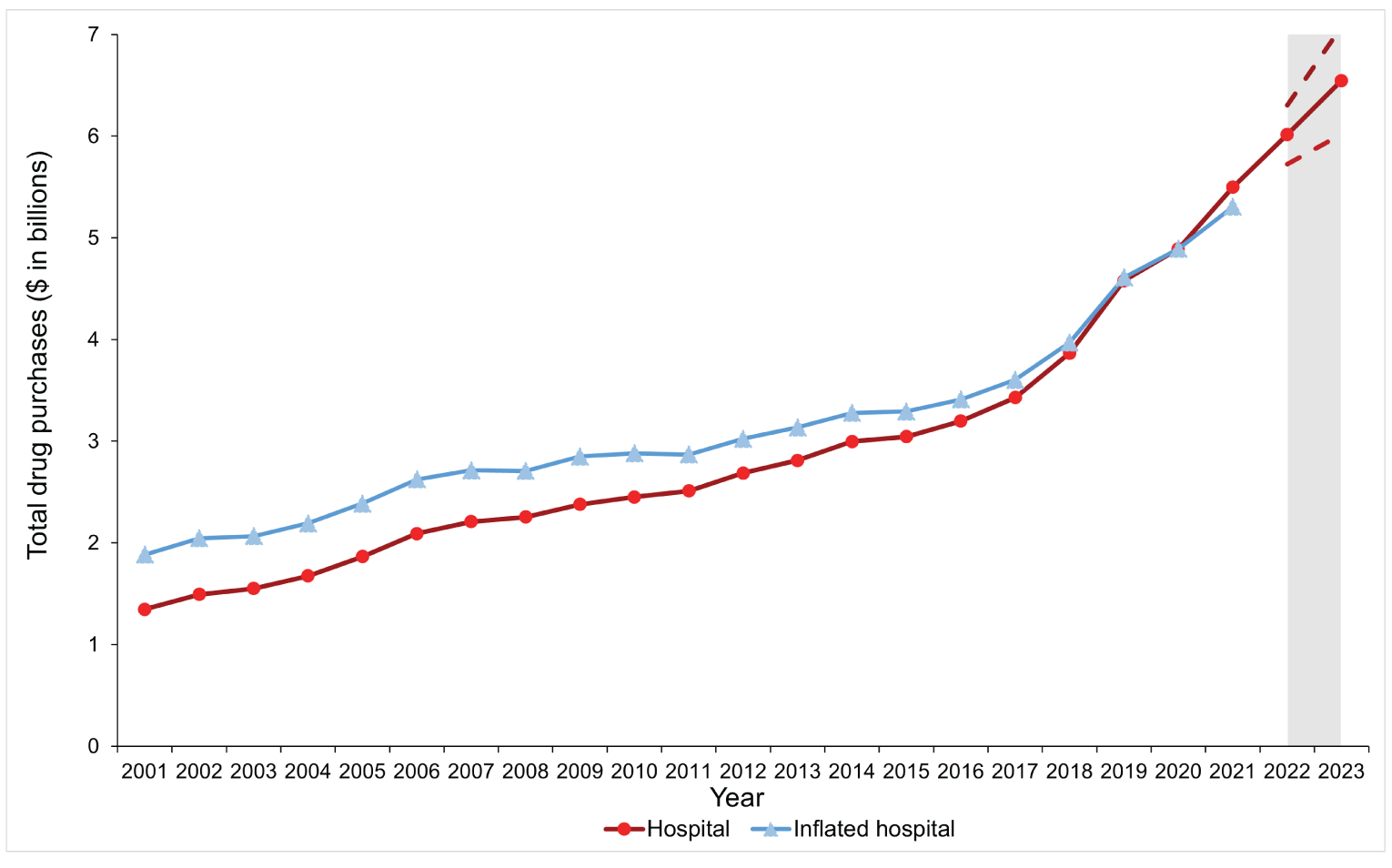
Note: The grey zone illustrates forecasted purchases in the hospital sector (solid line) with 95% confidence interval estimates (dashed lines) for the years 2022 and 2023. Red line with circles = non–inflated-adjusted purchases; light blue line with triangles = inflated-adjusted purchases.
Source: This figure is based on information licensed from IQVIA: Canadian Drugstore and Hospital Purchases Audit for the period of 2001 to 2021 estimates of real-world activity. All rights reserved.
Table 3: Fifteen Global Reports Reviewed to Determine Trends for Drug Spending in Canada
Report | Link |
|---|---|
Advancing Health Through Innovation: New Drug Therapy Approvals 2021 | Advancing Health Through Innovation: New Drug Therapy Approvals 2021 (fda.gov) |
2022 Drug Plan Trends Report: High-cost drugs, DEI shaping drug plans | 2022 Drug Plan Trends Report: High-cost drugs, DEI shaping drug plans | Benefits Canada.com |
Biosimilars approved in Canada February 2022 | |
CADTH Policy Intel 2021 US FDA New Drug Therapy Approvals: A Summary | Not available |
2022 Biosimilars Report: The US Journey and Path Ahead | |
2022 Prescription Drug Trend Report | ExpressScriptsCanada2022DrugTrendReport.pdf (express-scripts.ca) |
Drug and Medical Device Highlights, 2020 | |
Global Trends in R&D Overview Through 2021 | |
2021 Category watch: The Impact of New Drugs. A closer look at diabetes, migraine and multiple sclerosis | |
2021 TELUS Health Drug Data Trends and National Benchmarks | |
The Drug Pipeline: What private plans can expect in 2021 |
Table 4: Health Canada Drug Approvals Between January 1, 2021, and March 31, 2022
Generic name | Therapeutic area | Approval date | Submission outcome |
|---|---|---|---|
Acetylsalicylic acid, atorvastatin calcium, ramipril | Cardiac therapy | March 2021 | Cancelled by sponsor |
Ad26.COV2.S (recombinant) | Vaccines | November 2021 | Issued NOC |
Adalimumab | Immunosuppressants | January 2022 | Issued NOC |
Adalimumab | Immunosuppressants | December 2021 | Issued NOC |
Adalimumab | Immunosuppressants | January 2021 | Issued NOC |
Alfa 1-proteinase inhibitor (human) | Antihemorrhagics | November 2021 | Issued NOC |
Amifampridine | Other nervous system drugs | June 2021 | NOC invalid |
Amisulpride | Psycholeptics | February 2021 | Cancelled by sponsor |
Amivantamab | Antineoplastic agents | March 2022 | Issued NOC under the NOC/c Guidance |
Anifrolumab | Immunosuppressants | November 2021 | Issued NOC |
Aprotinin | Antihemorrhagics | February 2022 | Cancelled by sponsor |
Avalglucosidase alfa | Other alimentary tract and metabolism | November 2021 | Issued NOC |
Avanafil | Urologicals | October 2021 | Issued Notice of Non-compliance - Withdrawal |
Azacitidine | Antineoplastic agents | January 2021 | Issued NOC |
Belumosudil mesylate | Immunosuppressants | March 2022 | Issued NOC |
Bendamustine hydrochloride | Antineoplastic agents | January 2022 | Issued NOC |
Bevacizumab | Antineoplastic agents | November 2021 | Issued NOC |
Bevacizumab | Antineoplastic agents | November 2021 | Issued NOC |
Bevacizumab | Antineoplastic agents | November 2021 | Issued NOC |
Bevacizumab | Antineoplastic agents | September 2021 | Cancelled by sponsor |
Bevacizumab | Antineoplastic agents | September 2021 | Issued NOC |
Bevacizumab | Antineoplastic agents | January 2021 | Issued NOC |
Bimekizumab | Immunosuppressants | February 2022 | Issued NOC |
Binimetinib | Antineoplastic agents | March 2021 | Issued NOC |
Brexucabtagene autoleucel | Antineoplastic agents | June 2021 | Issued NOC |
Brilliant blue G | Ophthalmologicals | January 2021 | Issued NOC |
Budesonide, formoterol fumarate dihydrate, glycopyrronium bromide | Drugs for obstructive airway diseases | July 2021 | Issued NOC |
Bupivacaine | Anesthetics | August 2021 | Cancelled by sponsor |
Bupivacaine, meloxicam | Anti-inflammatory and antirheumatic products | March 2022 | Issued NOC |
ChAdOx1-S (recombinant) | Vaccines | November 2021 | Issued NOC |
Chlormethine | Antineoplastic agents | June 2021 | Issued NOC |
Colchicine | Antigout preparations | August 2021 | Issued NOC |
Copanlisib dihydrochloride | Antineoplastic agents | December 2021 | Cancelled by sponsor |
Corynebacterium diphtheria CRM-197 protein, pneumococcal polysaccharide serotypes 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F, 22F, 23F, and 33F | Vaccines | November 2021 | Issued NOC |
Cyclosporine | Ophthalmologicals | February 2021 | Issued NOC |
Cytarabine, daunorubicin hydrochloride | Antineoplastic agents | April 2021 | Issued NOC |
Dequalinium chloride | Gynecological anti-infectives and antiseptics | July 2021 | Issued NOC |
Diamorphine hydrochloride | Other nervous system drugs | February 2022 | Issued NOC |
Dostarlimab | Antineoplastic agents | December 2021 | Issued NOC under the NOC/c Guidance |
Drospirenone | Sex hormones and modulators of the genital system | November 2021 | Issued NOC |
Drospirenone, estetrol monohydrate | Sex hormones and modulators of the genital system | March 2021 | Issued NOC |
Elasomeran | Vaccines | September 2021 | Issued NOC |
Elexacaftor, ivacaftor, tezacaftor | Other respiratory system products | June 2021 | Issued NOC |
Emapalumab | Immunosuppressants | February 2021 | Cancelled by sponsor |
Encorafenib | Antineoplastic agents | March 2021 | Issued NOC |
Encorafenib | Antineoplastic agents | March 2021 | Issued NOC |
Enfortumab vedotin | Antineoplastic agents | October 2021 | Issued NOC |
Eptinezumab | Analgesics | January 2021 | Issued NOC |
Favipiravir | Antivirals for systemic use | February 2022 | Cancelled by sponsor |
Ferric pyrophosphate citrate | Antianemic preparations | April 2021 | Issued NOC |
Fibrinogen (human), thrombin (human) | Antihemorrhagics | March 2021 | Issued NOC |
Filgotinib | Immunosuppressants | January 2021 | Cancelled by sponsor |
Filgrastim (r-metHuG-CSF) | Immunostimulants | October 2021 | Issued NOC |
Finerenone | Diuretics | October 2021 | Cancelled by sponsor |
Fostemsavir tromethamine | Antivirals for systemic use | October 2021 | Issued NOC |
Gallium oxodotreotide | Diagnostic radiopharmaceuticals | February 2022 | Issued NOC |
Goserelin acetate | Endocrine therapy | October 2021 | Cancelled by sponsor |
Haemagglutinin, neuraminidase antigen | Vaccines | January 2021 | Issued NOC |
Hyaluronidase (human recombinant), immunoglobulin (human), | Immune sera and immunoglobulins | January 2022 | Issued NOC |
Idecabtagene vicleucel | Antineoplastic agents | May 2021 | Issued NOC under the NOC/c Guidance |
Inclisiran sodium | Lipid modifying agents | July 2021 | Issued NOC |
Infigratinib phosphate | Antineoplastic agents | September 2021 | Issued NOC under the NOC/c Guidance |
Infliximab | Immunosuppressants | December 2021 | Issued NOC |
Insulin aspart | Drugs used in diabetes | October 2021 | Issued NOC |
Insulin lispro | Drugs used in diabetes | September 2021 | Issued NOC |
Lasmiditan | Analgesics | January 2021 | Cancelled by sponsor |
Leuprolide mesylate | Endocrine therapy | November 2021 | Issued NOC |
Lumasiran | Other alimentary tract and metabolism products | March 2022 | Issued NOC |
Lurbinectedin | Antineoplastic agents | September 2021 | Issued NOC under the NOC/c Guidance |
Luspatercept | Antianemic preparations | February 2021 | Issued NOC |
Macitentan, tadalafil | Antihypertensives | October 2021 | Issued NOC |
Mesalazine | Antidiarrheals, intestinal anti-inflammatory/anti-infective agents | January 2022 | Issued NOC |
Mesalazine | Antidiarrheals, intestinal anti-inflammatory/anti-infective agents | May 2021 | Issued NOC |
Methylene blue | Diagnostic agents | May 2021 | Issued Notice of Non-compliance - Withdrawal |
Nirmatrelvir, ritonavir | Antivirals for systemic use | January 2022 | Issued NOC |
Ofatumumab | Antineoplastic agents | January 2021 | Issued NOC |
Ospemifene | Sex hormones and modulators of the genital system | July 2021 | Issued NOC |
Palovarotene | Other drugs for disorders of the musculoskeletal system | January 2022 | Issued NOC |
Pegfilgrastim | Immunostimulants | July 2021 | Cancelled by sponsor |
Pegvaliase | Other alimentary tract and metabolism products | March 2022 | Issued NOC |
Pemigatinib | Antineoplastic agents | September 2021 | Issued NOC under the NOC/c Guidance |
Pertuzumab, trastuzumab | Antineoplastic agents | March 2021 | Issued NOC |
Pitolisant | Other nervous system drugs | May 2021 | Issued NOC |
Ponesimod | Immunosuppressants | April 2021 | Issued NOC |
Pralsetinib | Antineoplastic agents | June 2021 | Issued NOC under the NOC/c Guidance |
Progesterone | Sex hormones and modulators of the genital system | April 2021 | Issued NOC |
Ranibizumab | Ophthalmologicals | March 2022 | Issued NOC |
Recombinant haemagglutinin-strain A (H1N1) virus-like particles, Recombinant haemagglutinin-strain A (H3N2) virus-like particles, Recombinant haemagglutinin-strain B (Victoria lineage) virus-like particles, Recombinant haemagglutinin-strain B (Yamagata lineage) virus-like particles | Vaccines | September 2021 | Cancelled by sponsor |
Recombinant influenza strain A H1N1 HA protein, recombinant influenza strain A H3N2 HA protein, recombinant influenza strain B (Victoria lineage) HA protein, recombinant influenza strain B (Yamagata lineage) HA protein | Vaccines | January 2021 | Issued NOC |
Risdiplam | Other drugs for disorders of the musculoskeletal system | April 2021 | Issued NOC |
Rituximab | Antineoplastic agents | March 2021 | Issued NOC |
Roxadustat | Antianemic preparations | October 2021 | Cancelled by sponsor |
Sacituzumab govitecan | Antineoplastic agents | September 2021 | Issued NOC |
SARS-CoV-2 recombinant spike protein | Vaccines | February 2022 | Issued NOC |
Selpercatinib | Antineoplastic agents | June 2021 | Issued NOC under the NOC/c Guidance |
Semaglutide | Drugs used in diabetes | November 2021 | Issued NOC |
Solriamfetol hydrochloride | Psychoanaleptics | May 2021 | Issued NOC |
Somatrogon | Pituitary, hypothalamic hormones, and analogues | October 2021 | Issued NOC |
Sotorasib | Antineoplastic agents | September 2021 | Issued NOC under the NOC/c Guidance |
Tafamidis | Other nervous system drugs | July 2021 | Issued NOC |
Tafasitamab | Antineoplastic agents | August 2021 | Issued NOC |
Tazarotene | Antipsoriatics | July 2021 | Issued NOC |
Tecovirimat monohydrate | Antivirals for systemic use | November 2021 | Issued NOC |
Tepotinib | Antineoplastic agents | May 2021 | Issued NOC under the NOC/c Guidance |
Tildrakizumab | Immunosuppressants | May 2021 | Issued NOC |
Tozinameran | Vaccines | September 2021 | Issued NOC |
Tralokinumab | Other dermatological preparations | October 2021 | Issued NOC |
Trastuzumab | Antineoplastic agents | January 2022 | Issued NOC |
Trastuzumab deruxtecan | Antineoplastic agents | April 2021 | Issued NOC under the NOC/c Guidance |
Treosulfan | Antineoplastic agents | June 2021 | Issued NOC |
Treosulfan | Antineoplastic agents | June 2021 | Issued a Notice of Deficiency - Withdrawal |
Trientine hydrochloride | All other therapeutic products | April 2021 | Issued NOC |
Triheptanoin | Other alimentary tract and metabolism | February 2021 | Issued NOC |
Virus-like particles (VLP) of SARS-CoV-2 spike protein | Vaccines | February 2022 | Issued NOC |
Vitamin D3 | Vitamins | February 2021 | Issued NOC |
Vitamin K1 | Antihemorrhagics | June 2021 | Issued a Notice of Deficiency - Withdrawal |
Zanubrutinib | Antineoplastic agents | July 2021 | Issued NOC |
Zanubrutinib | Antineoplastic agents | March 2021 | Issued NOC |
Table 5: FDA Drug Approvals Between January 1, 2021, and March 31, 2022
Generic name | Brand name | Indication | Sponsor | Approval date |
|---|---|---|---|---|
Abacavir sulphate; dolutegravir sodium; lamivudine | Triumeq PD | HIV | Viiv Hlthcare | March 30, 2022 |
Abrocitinib | Cibinqo | Atopic dermatitis | Pfizer | January 14, 2022 |
Acetaminophen | Acetaminophen | Pain | Rising | January 15, 2021 |
Acetaminophen | Acetaminophen | Pain | B Braun Medical Inc. | February 18, 2021 |
Aducanumab-AVWA | Aduhelmbla | Alzheimer disease | Biogen Inc. | June 7, 2021 |
Amifampridine | Ruzurgi | Congenital myasthenic syndromes, Lambert–Eaton myasthenic syndrome | Jacobus Pharm Co Inc. | February 1, 2022 |
Amivantamab-VMJW | Rybrevant | Non–small cell lung cancer | Janssen Biotech | May 21, 2021 |
Amlodipine besylate | Norliqva | Multiple indications | Cmp Dev Llc | February 24, 2022 |
Amphetamine; amphetamine aspartate/ dextroamphetamine sulphate | Dyanavel XR 10 | Attention deficit disorder | Tris Pharma Inc. | November 4, 2021 |
Amphetamine; amphetamine aspartate/ dextroamphetamine sulphate | Dyanavel XR 15 | Attention deficit disorder | Tris Pharma Inc. | November 4, 2021 |
Amphetamine; amphetamine aspartate/ dextroamphetamine sulphate | Dyanavel XR 20 | Attention deficit disorder | Tris Pharma Inc. | November 4, 2021 |
Amphetamine; amphetamine aspartate/ dextroamphetamine sulphate | Dyanavel XR 5 | Attention deficit disorder | Tris Pharma Inc. | November 4, 2021 |
Anifrolumab-FNIA | Saphnelo | Lupus | Astrazeneca Ab | July 30, 2021 |
Argatroban | Argatroban in sodium chloride | Thrombocytopenia | Accord Hlthcare | June 7, 2021 |
Asciminib hydrochloride | Scemblix | Oncology | Novartis | October 29, 2021 |
Asparaginase erwinia chrysanthemi (recombinant)-RYWN | Rylaze | Oncology | Jazz Pharms | June 30, 2021 |
Atogepant | Qulipta | Migraine prevention | Abbvie Inc. | September 28, 2021 |
Atropine sulphate | Atropine | Multiple indications | Paragon Bioteck | March 15, 2022 |
Avacopan | Tavneos | Anti-neutrophil cytoplasmic autoantibody-associated vasculitis | Chemocentryx | October 7, 2021 |
Avalglucosidase alfa-NGPT | Nexviazyme | Late-onset Pompe disease | Genzyme Corp | August 6, 2021 |
Azelastine hydrochloride | Astepro Allergy | Allergy | Bayer Hlthcare | June 17, 2021 |
Azelastine hydrochloride | Children's Astepro Allergy | Allergy | Bayer Hlthcare | June 17, 2021 |
Baclofen | Lyvispah | Muscle relaxant | Saol Theraps Res Ltd. | November 22, 2021 |
Baclofen | Fleqsuvy | Muscle relaxant | Azurity | February 4, 2022 |
Belumosudil mesylate | Rezurock | Chronic graft vs. host disease | Kadmon Pharms Llc | July 16, 2021 |
Belzutifan | Welireg | Von Hippel–Lindau disease-associated renal cell carcinoma | Merck Sharp Dohme | August 13, 2021 |
Bendamustine hydrochloride | Bendamustine | Oncology | Dr Reddys Labs Ltd. | November 12, 2021 |
Bendamustine hydrochloride | Vivimusta | Oncology | Slayback Pharma Llc | March 16, 2022 |
Benzoyl peroxide; tretinoin | Twyneo | Acne | Sol-Gel Technologies | July 26, 2021 |
Bortezomib | Bortezomib | Multiple myeloma | Intas Pharms USA | August 20, 2021 |
Brincidofovir | Tembexa | Smallpox | Chimerix | June 4, 2021 |
Brincidofovir | Tembexa | Smallpox | Chimerix | June 4, 2021 |
Budesonide | Tarpeyo | Steroid | Calliditas | December 15, 2021 |
Bupivacaine | Posimir | Multiple indications | Durect | February 1, 2021 |
Bupivacaine; meloxicam | Zynrelef | Postsurgical pain | Heron Theraps Inc. | May 12, 2021 |
Cabazitaxel | Cabazitaxel | Oncology | Accord Hlthcare | December 29, 2021 |
Cabotegravir | Apretude | HIV | Viiv Hlthcare | December 20, 2021 |
Cabotegravir sodium | Vocabria | Long-acting HIV | Viiv Hlthcare | January 21, 2021 |
Cabotegravir; rilpivirine | Cabenuva kit | Long-acting HIV | Viiv Hlthcare | January 21, 2021 |
Carbidopa; levodopa | Dhivy | Parkinson disease | Avion Pharms | November 12, 2021 |
Carbon dioxide | Carbon dioxide | Multiple indications | Middlesex Gases Technologies, Inc. | October 18, 2021 |
Carbon dioxide | Carbon | Multiple indications | Continental Carbonic Products, Inc. | February 25, 2022 |
Casimersen | Amondys | Duchenne muscular dystrophy | Sarepta Theraps Inc. | February 25, 2021 |
Celecoxib; tramadol hydrochloride | Seglentis | Pain | Kowa Pharms | October 15, 2021 |
Citalopram hydrobromide | Citalopram | Multiple indications | Almatica | January 31, 2022 |
Clindamycin phosphate | Xaciato | Antibiotic | Dare | December 7, 2021 |
Cyclophosphamide | Cyclophosphamide | Oncology | Eugia Pharma Speclts | August 25, 2021 |
Cyclosporine | Verkazia | Immunosuppressant | Santen | June 23, 2021 |
Dabigatran etexilate mesylate | Pradaxa | Anticoagulant | Boehringer Ingelheim | June 21, 2021 |
Daptomycin | Daptomycin | Antibiotic | Hospira Inc. | June 21, 2021 |
Daptomycin | Dapzura Rt | Antibiotic | Baxter Hlthcare Corp | January 25, 2022 |
Daridorexant hydrochloride | Quviviq | Insomnia | Idorsia Pharmaceuticals Ltd. | January 7, 2022 |
Dasiglucagon hydrochloride | Zegalogue | Severe hypoglycemia | Zealand Pharma | March 22, 2021 |
Dasiglucagon hydrochloride | Zegalogue (autoinjector) | Severe hypoglycemia | Zealand Pharma | March 22, 2021 |
Dexmethylphenidate hydrochloride; serdexmethylphenidate chloride | Azstarys | Attention deficit disorder | Commave Therap | March 2, 2021 |
Dextroamphetamine | Xelstrym | Attention deficit disorder | Noven Pharms Inc. | March 22, 2022 |
Difelikefalin acetate | Korsuva | Itching in chronic kidney disease | Cara Therap | August 23, 2021 |
Dihydroergotamine mesylate | Trudhesa | Migraine | Impel Neuropharma | September 2, 2021 |
Diltiazem hydrochloride | Diltiazem hydrochloride in dextrose 5% | Multiple indications | Exela Pharma | October 28, 2021 |
Dolutegravir lamivudine tenofovir alafenamide | Dolutegravir lamivudine tenofovir alafenamide | HIV | Cipla Usa Inc. | March 30, 2022 |
Dolutegravir sodium | Dolutegravir | HIV | Macleods Pharms Ltd. | March 16, 2021 |
Dolutegravir; lamivudine; tenofovir disoproxil fumarate | Dolutegravir, lamivudine and tenofovir disoproxil fumarate | HIV | Lupin Pharms | June 25, 2021 |
Donepezil hydrochloride | Adlarity | Alzheimer disease | Corium | March 11, 2022 |
Dostarlimab-GXLY | Jemperlibla | Endometrial cancer | Glaxosmithkline | April 22, 2021 |
Dostarlimab-GXLY | Jemperli | Oncology | Glaxosmithkline Llc | August 17, 2021 |
Drospirenone; estetrol | Nextstellis | Oral contraceptive | Mayne Pharma | April 15, 2021 |
Efgartigimod alfa | Vyvgart | Generalized myasthenia gravis | Argenx Bv | December 17, 2021 |
Ephedrine hydrochloride | Rezipres | Multiple indications | Eton | June 14, 2021 |
Estradiol; norethindrone acetate; relugolix | Myfembree | Heavy menstrual bleeding due to fibroids | Myovant Sciences | May 26, 2021 |
Evinacumab-DGNB | Evkeeza | Homozygous familial hypercholesterolemia | Regeneron Pharmaceuticals | February 11, 2021 |
Ezetimibe; rosuvastatin calcium | Roszet | Hyperlipidemia | Althera Pharms | March 23, 2021 |
Faricimab-SVOA | Vabysmobla | Neovascular (Wet) age-related macular degeneration, diabetic macular edema | Genentech Inc. | January 28, 2022 |
Fexinidazole | Fexinidazole | Trypanosoma brucei gambiense sleeping sickness | Sanofi | July 16, 2021 |
Filgrastim-AYOW | Releukobla | Neutropenia | Kashiv Biosciences Llc | February 25, 2022 |
Finasteride; tadalafil | Entadfi | Benign prostatic hypertrophy | Veru | December 9, 2021 |
Finerenone | Kerendia | Chronic kidney disease | Bayer Hlthcare | July 9, 2021 |
Fingolimod lauryl sulphate | Tascenso ODT | Multiple sclerosis | Handa | October 18, 2021 |
Fosdenopterin hydrobromide | Nulibry | Molybdenum cofactor deficiency type A | Origin | February 26, 2021 |
Gallium GA-68 gozetotide | Locametz | Diagnostic agent for prostate cancer | Aaa Usa Novartis | March 23, 2022 |
Ganaxolone | Ztalmy | Cyclin-dependent kinase-like 5 deficiency seizures | Marinus Pharamceuticals, Inc. | March 18, 2022 |
Glecaprevir; pibrentasvir | Mavyret | Hepatitis C | Abbvie Inc. | June 10, 2021 |
Glycopyrrolate | Dartisla ODT | Peptic ulcer | Edenbridge Pharms | December 16, 2021 |
Helium | Helium, USP | Multiple indications | General Air Service and Supply Company | July 26, 2021 |
Hydroxychloroquine sulphate | Hydroxychloroquine | Multiple indications | Novitium Pharma | January 14, 2022 |
Ibrexafungerp citrate | Brexafemme | Vaginal candidiasis | Scynexis | June 1, 2021 |
Inclisiran sodium | Leqvio | Hypercholesterolemia | Novartis | December 22, 2021 |
Infigratinib phosphate | Truseltiq | Oncology | Helsinn Hlthcare | May 28, 2021 |
Ketotifen fumarate | Acuvue | Allergy | Johnson Johnson Visn | February 25, 2022 |
Lanreotide acetate | Lanreotide | Gastroenteropancreatic tumours | Invagen Pharms | December 17, 2021 |
Leuprolide mesylate | Camcevi | Prostate cancer | Foresee Pharms | May 25, 2021 |
Levoketoconazole | Recorlev | Antifungal | Strongbridge | December 30, 2021 |
Levothyroxine sodium | Levothyroxine | Hypothyroidism | Custopharm Inc. | May 17, 2021 |
Lonapegsomatropin-TCGD | Skytrofa | Endogenous growth hormone insufficiency | Ascendis Pharma Encocrinology Div A/S | August 25, 2021 |
Loncastuximab tesirine-LPYL | Zynlontabla | B-cell lymphoma | Adc Therapeutics Sa | April 23, 2021 |
Lorazepam | Loreev | Multiple indications | Almatica | August 27, 2021 |
Lutetium LU-177 vipivotide tetraxetan | Pluvicto | Prostate-specific membrane antigen-positive metastatic castration-resistant prostate cancer | Aaa Usa Novartis | March 23, 2022 |
Maralixibat chloride | Livmarli | Cholestatic pruritus in people with Alagille syndrome | Mirum | September 29, 2021 |
Maribavir | Livtencity | Post-transplant cytomegalovirus | Takeda Pharms Usa | November 23, 2021 |
Melphalan flufenamide hydrochloride | Pepaxto | Multiple myeloma | Oncopeptides Ab | February 26, 2021 |
Micafungin sodium | Micafungin | Antifungal | Par Sterile Products | June 16, 2021 |
Micafungin sodium | Micafungin | Antifungal | Teva Pharms Usa Inc. | July 30, 2021 |
Midazolam | Midazolam in 0.9% sodium chloride | Sedative | Inforlife | March 22, 2021 |
Mirabegron | Myrbetriq granules | Overactive bladder | Apgdi | March 25, 2021 |
Mitapivat sulphate | Pyrukynd | Hemolytic anemia | Agios Pharms Inc. | February 17, 2022 |
Mobocertinib succinate | Exkivity | Non–small cell lung cancer | Takeda Pharms Usa | September 15, 2021 |
Mometasone furoate | Nasonex 24hr Allergy | Allergy | Perrigo Pharma Intl | March 17, 2022 |
Mometasone furoate; olopatadine hydrochloride | Ryaltris | Allergy | Glenmark Specialty | January 13, 2022 |
Naloxone hydrochloride | Kloxxado | Opioid overdose | Hikma | April 29, 2021 |
Naloxone hydrochloride | Zimhi | Opioid overdose | Adamis Pharms Corp | October 15, 2021 |
Naloxone hydrochloride | Naloxone | Opioid overdose | Kaleo Inc. | February 28, 2022 |
Nivolumab;relatlimab-RMBW | Opdualag | Oncology | Bristol Myers Squibb | March 18, 2022 |
Norepinephrine bitartrate | Norepinephrine bitartrate in 5% dextrose | Multiple indications | Baxter Hlthcare Corp | January 15, 2021 |
Odevixibat | Bylvay | Itching in progressive familial intrahepatic cholestasis | Albireo | July 20, 2021 |
Olanzapine; samidorphan l-malate | Lybalvi | Bipolar disorder, schizophrenia | Alkermes Inc. | May 28, 2021 |
Oritavancin diphosphate | Kimyrsa | Antibiotic | Melinta Therap | March 12, 2021 |
Paclitaxel | Paclitaxel | Oncology | Hbt Labs Inc. | June 30, 2021 |
Pacritinib citrate | Vonjo | Myelofibrosis | Cti Biopharma Corp | February 28, 2022 |
Pafolacianine sodium | Cytalux | Imaging agent for ovarian cancer | On Target Labs | November 29, 2021 |
Pegcetacoplan | Empaveli | Paroxysmal nocturnal hemoglobinuria | Apellis Pharms | May 14, 2021 |
Pemetrexed | Pemetrexed | Oncology | Hospira Inc. | January 8, 2021 |
Pemetrexed | Pemetrexed | Oncology | Hospira Inc. | February 23, 2021 |
Pemetrexed | Pemetrexed | Oncology | Sandoz Inc. | May 6, 2021 |
Piflufolastat F-18 | Pylarify | Diagnostic imaging in prostate cancer | Progenics Pharms Inc. | May 26, 2021 |
Pilocarpine hydrochloride | Vuity | Presbyopia | Abbvie Inc. | October 28, 2021 |
Ponesimod | Ponvory | Multiple sclerosis | Janssen Pharms | March 18, 2021 |
Posaconazole | Noxafil Powdermix kit | Antifungal | Msd Merck Co | May 31, 2021 |
Ranibizumab | Susvimobla | Age-related macular degeneration | Genentech Inc. | October 22, 2021 |
Ranolazine | Aspruzyo sprinkle | Angina | Spil | February 28, 2022 |
Rivaroxaban | Xarelto | Anticoagulant | Janssen Pharms | December 20, 2021 |
Ropeginterferon alfa-2b-NJFT | Besremi | Polycythemia vera | Pharmaessentia Corp | November 12, 2021 |
Ruxolitinib phosphate | Opzelura | Myelofibrosis | Incyte Corp | September 21, 2021 |
Selexipag | Uptravi | Pulmonary arterial hypertension | Actelion | July 29, 2021 |
Semaglutide | Wegovy | Weight management | Novo | June 4, 2021 |
Sertraline hydrochloride | Sertraline | Multiple indications | Almatica | October 4, 2021 |
Sirolimus | Fyarro | Immunosuppressant | Aadi | November 22, 2021 |
Sirolimus | Hyftor | Immunosuppressant | Nobelpharma | March 22, 2022 |
Sitagliptin | Sitagliptin | Diabetes | Zydus Worldwide Dmcc | September 2, 2021 |
Sodium benzoate; sodium phenylacetate | Sodium phenylacetate and sodium benzoate | Hyperammonemia | Maia Pharms Inc. | June 10, 2021 |
Sofosbuvir; velpatasvir | Epclusa | Hepatitis C | Gilead Sciences Inc. | June 10, 2021 |
Sotorasib | Lumakras | Oncology | Amgen Inc. | May 28, 2021 |
Succinylcholine chloride | Succinylcholine | Skeletal muscle relaxant | Hikma | August 20, 2021 |
Sutimlimab-JOME | Enjaymo | Cold agglutinin disease | Bioverativ Therapeutics Inc. | February 4, 2022 |
Tebentafusp-TEBN | Kimmtrak | T-cell therapy for unresectable or metastatic uveal melanoma | Immunocore Ltd. | January 25, 2022 |
Technetium TC-99m succimer | Nephroscan | Renal diagnostic agent | Theragnostics | February 18, 2022 |
Tepotinib hydrochloride | Tepmetko | Metastatic non–small cell lung cancer | Emd Serono Inc. | February 3, 2021 |
Testosterone undecanoate | Tlando | Multiple indications | Antares Pharma Inc. | March 28, 2022 |
Tezepelumab-EKKO | Tezspire | Asthma | Astrazeneca Ab | December 17, 2021 |
Tisotumab vedotin-TFTV | Tivdakbla | Cervical cancer | Seagen | September 20, 2021 |
Tivozanib hydrochloride | Fotivda | Renal cell carcinoma | Aveo Pharms | March 10, 2021 |
Topiramate | Eprontia | Epilepsy | Azurity | November 5, 2021 |
Torsemide | Soaanz | Diuretic | Sarfe Pharms | June 14, 2021 |
Tralokinumab | Adbrybla | Atopic dermatitis | Leo Pharma As | December 27, 2021 |
Treprostinil | Yutrepia | Pulmonary arterial hypertension | Liquidia Technologies, Inc. | November 4, 2021 |
Triamcinolone acetonide | Xipere | Steroid | Bausch And Lomb Inc. | October 22, 2021 |
Trilaciclib dihydrochloride | Cosela | Chemotherapy-induced myelosuppression | G1 Therap | February 12, 2021 |
Umbralisib tosylate | Ukoniq | Lymphoma | Tg Theraps | February 5, 2021 |
Vancomycin | Vancomycin | Antibiotic | Xellia Pharms Aps | August 26, 2021 |
Varenicline tartrate | Tyrvaya | Smoking cessation | Oyster Point Pharma | October 15, 2021 |
Vericiguat | Verquvo | Heart failure | Merck Sharp Dohme | January 19, 2021 |
Viloxazine hydrochloride | Qelbree | Attention deficit disorder | Supernus Pharms | April 2, 2021 |
Voclosporin | Lupkynis | Lupus | Aurinia | January 22, 2021 |
Vosoritide | Voxzogo | Achondroplasia | Biomarin Pharm | November 19, 2021 |
Voxelotor | Oxbryta | Sickle cell anemia | Global Blood Theraps | December 17, 2021 |
Appendix 2: Author Information
Corresponding Author
Mina Tadrous
Leslie Dan Faculty of Pharmacy, University of Toronto
144 College St., Toronto, Ontario
mina.tadrous@utoronto.ca
Author Information
Mina Tadrous, PharmD, PhD;1-3 Pooyeh Graili, MD, MPH, MBE;2 Kaleen N. Hayes, PharmD;4 Heather L. Neville, BSc Pharm, MSc;5 Joanne Houlihan, BSc Pharm;6 Fiona Clement, PhD;7,8 Jason R. Guertin, PhD;9,10 Michael R. Law, PhD;11 Tara Gomes, PhD;2,3,12
1Women’s College Research Institute, Women’s College Hospital, Toronto, Ontario, Canada
2Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, Ontario, Canada
3ICES, Toronto, Ontario, Canada
4Brown University, Providence, Rhode Island, US
5Queen Elizabeth II Health Sciences Centre, Nova Scotia Health Authority, Halifax, Nova Scotia, Canada
6Nova Scotia Cancer Care Program, Nova Scotia Health Authority, Halifax, Nova Scotia, Canada
7Department of Community Health Sciences, Cumming School of Medicine, University of Calgary, Calgary, Alberta, Canada
8O’Brien Institute for Public Health, University of Calgary, Calgary, Alberta, Canada
9Axe Santé des populations et Pratiques optimales en santé, Centre de recherche du CHU de Québec-Université Laval, Québec, Canada
10Department of Social and Preventive Medicine, Université Laval, Québec, Canada
11Centre for Health Services and Policy Research, School of Population and Public Health, University of British Columbia, Vancouver, British Columbia, Canada
12Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Toronto, Ontario, Canada
Author Disclosures
Dr. Gomes received salary support through a Canada Research Chair in Drug Policy Research and Evaluation and grant funding from the Ontario Ministry of Health. Dr. Law received salary support through a Canada Research Chair in Access to Medicines. Dr. Law has consulted for Health Canada, the Health Employees’ Union, the Conference Board of Canada, and provided expert witness testimony for the Attorney General of Canada and the Federation of Post-secondary Educators. Dr. Guertin holds a research career award from the Fonds de recherche du Québec – Santé (FRQS). Dr. Guertin has consulted for the Institut national d’excellence en santé et services sociaux (INESSS). Dr. Tadrous has consulted for CADTH and Green Shield Canada. Ms. Neville is a member of the CADTH Canadian Drug Expert Committee. Ms. Houlihan is a member of the Provincial Advisory Committee for CADTH. Dr. Tadrous reported consulting fees from CADTH and Green Shield Canada and funding from the Ontario Ministry of Health.
Acknowledgements
All authors were involved in the design, interpretation of results, writing, conceptualization of recommendations, and revision of the manuscript. MT was involved in the implementation of the study and had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. MT is the guarantor of the content of the manuscript, including the data and analysis.
The statements, findings, conclusions, views, and opinions contained and expressed in the report are based in part on data obtained under license from IQVIA, Canada Inc. concerning the following information service(s): IQVIA’s Canadian Drugstore and Hospital Purchases Audit, data period January 1, 2001, and December 31, 2021. All rights reserved. The statements, findings, conclusions, views, and opinions expressed herein are not necessarily those of IQVIA Canada Inc. or any of its affiliated or subsidiary entities.
ISSN: 2563-6596
Disclaimer: This article is subject to copyright and other intellectual property rights and may only be used for non-commercial, personal use, or private research and study. This material is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. This article should not be used as a substitute for professional medical advice or for the application of professional judgment in any decision-making process. Users may use this document at their own risk.
This article – unlike others published in the journal -- was submitted by an author group outside CADTH. It was peer reviewed by 2 external reviewers, revised by the corresponding author, and accepted for publication in the journal. Care has been taken to ensure that the information presented is original and has not been submitted elsewhere.