Drugs, Health Technologies, Health Systems
Reimbursement Recommendation
Trastuzumab deruxtecan (Enhertu)
Indication: For treatment of adult patients with unresectable or metastatic HR-positive, HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer who have received at least one endocrine therapy in the metastatic setting and are not considered suitable for endocrine therapy as the next line of treatment.
Sponsor: AstraZeneca Canada Inc.
Final recommendation: Reimburse with conditions
Summary
What Is the Reimbursement Recommendation for Enhertu?
Canada’s Drug Agency (CDA-AMC) recommends that Enhertu be reimbursed by public drug plans for the treatment of adult patients with unresectable or metastatic hormone receptor (HR)-positive, HER2-low (immunohistochemistry [IHC] 1+ or IHC 2+ or in situ hybridization [ISH]-negative) or HER2-ultralow (IHC 0 with membrane staining) breast cancer who have received at least 1 endocrine therapy in the metastatic setting and who are not considered suitable for further endocrine therapy as the next line of therapy if certain conditions are met.
Why Did CDA-AMC Recommend Reimbursement?
The pan-Canadian Oncology Drug Review Expert Review Committee (pERC) determined that Enhertu demonstrates acceptable clinical value compared with chemotherapy in adult patients with unresectable or metastatic HR-positive, HER2-low or HER2-ultralow breast cancer who have received at least 1 endocrine therapy in the metastatic setting and who are not considered suitable for endocrine therapy as the next line of therapy. This determination was sufficient for pERC to recommend that Enhertu be reimbursed. Given that Enhertu is expected to be an alternative to chemotherapy, acceptable clinical value refers to added value versus chemotherapy.
Evidence from a clinical trial demonstrated that, compared with chemotherapy, Enhertu likely results in added clinical benefit in progression-free survival (PFS) in adults with HR-positive metastatic breast cancer (MBC) with low or ultralow HER2 expression who have received 1 or more lines of endocrine-based therapy and no previous chemotherapy for MBC. Overall survival (OS) was consistent with the results observed in the PFS analysis, suggesting a trend toward superiority for Enhertu. Compared to chemotherapy, Enhertu met some of the identified patient needs as it likely delays disease progression and may prolong survival.
Which Patients Are Eligible for Coverage?
Enhertu should only be covered for patients with HR-positive disease who have received at least 1 endocrine therapy in the metastatic setting and are not considered suitable for endocrine therapy as the next line of therapy. Patients should have a good performance status and must not have symptomatic spinal compression, clinically active central nervous system metastases, current interstitial lung disease (ILD) or pneumonitis, or received prior chemotherapy for advanced or MBC.
What Are the Conditions for Reimbursement?
Enhertu should only be reimbursed if initiated and prescribed by health care professionals with expertise in treating advanced breast cancer and if the cost of Enhertu is reduced. Enhertu should be discontinued upon the occurrence of progressive disease or unacceptable toxicity.
Review Background
Disease background: MBC is a life-threatening disease in which breast cancer spreads beyond the breast and nearby lymph nodes to distant organs such as the liver, brain, lungs, or bones. In 2024, there were an estimated 30,800 new cases of breast cancer diagnosed in Canada, where 30,500 new cases were in females and 290 were in males. Around 80% of breast cancers are currently classified as HER2-negative, of those approximately 60% show low HER2 expression (IHC 1+ or IHC 2+ or ISH-negative) and about 20% to 25% exhibit ultralow levels of HER2 (IHC 0 with membrane staining).
Indication and reimbursement request: Trastuzumab deruxtecan (Enhertu) has been approved by Health Canada for the treatment of adult patients with unresectable or metastatic HR-positive, HER2-low (IHC 1+ or IHC 2+ or ISH-negative) or HER2-ultralow (IHC 0 with membrane staining) breast cancer who have received at least 1 endocrine therapy in the metastatic setting and are not considered suitable for endocrine therapy as the next line of treatment. This application was submitted by the sponsor before receiving a Notice of Compliance from Health Canada. The CDA-AMC review reflects the sponsor’s requested reimbursement population, which is aligned with the final approved indication.
Drug under review: Enhertu is a HER2-targeted antibody drug conjugate. It is available as an IV infusion and the dosage recommended in the product monograph is 5.4 mg/kg once every 3 weeks until disease progression or unacceptable toxicity.
Treatment costs: At the submitted price of $2,440.00 per 100 mg vial, the per 28-day cost of trastuzumab deruxtecan is expected to be $13,013 per patient, based on the Health Canada–recommended dosage and an average weight of 69 kg.
Highlights of Input From Interested Parties
The patient groups, Breast Cancer Canada – McPeak Sirois Group for Clinical Research, Canadian Breast Cancer Network, and Rethink Breast Cancer noted the following regarding impacts of the disease, unmet needs, and important outcomes:
Patient group inputs highlighted physical impacts of MBC with significant or debilitating impact on quality of life included pain, nausea, insomnia, fatigue, depression, lack of concentration, memory loss, and diarrhea. Additionally, patient groups highlighted negative social impacts of the condition, such as restricted employment opportunities; increased caregiving demands; reduced physical activity; and reduced social interaction, community involvement, and time with family and loved ones.
Currently available chemotherapy options provide limited efficacy with considerable toxicity.
Treatment goals are to delay disease progression, reduce symptom burden, improve quality of life, and delay chemotherapy.
The clinician groups Ontario Health (Cancer Care Ontario) Breast Cancer Drug Advisory Committee and REAL Alliance, and the clinical experts consulted by CDA-AMC noted the following regarding unmet needs arising from the disease and place in therapy for the drug under review:
The clinical experts consulted by CDA-AMC identified an unmet need for effective, well-tolerated therapies for patients because available chemotherapy provides limited benefit with considerable toxicity.
Enhertu is anticipated to be used as monotherapy in the MBC setting, after progression on at least 1 endocrine-based therapy with or without CDK4/6 inhibitors and no previous chemotherapy for MBC.
The participating public drug programs raised potential implementation issues related to considerations for initiation, prescribing of therapy, generalizability of trial populations to broader populations, care provision issues, system and economic issues, and potential need for a provisional funding algorithm.
Recommendation
With a vote of 17 in favour to 1 against, pERC recommends that trastuzumab deruxtecan be reimbursed for the treatment of adult patients with unresectable or metastatic HR-positive, HER2-low (IHC 1+ or IHC 2+ or ISH-negative) or HER2-ultralow (IHC 0 with membrane staining) breast cancer who have received at least 1 endocrine therapy in the metastatic setting and who are not considered suitable candidates for further endocrine therapy as the next line of therapy, only if the conditions listed in Table 1 are met.
Table 1: Reimbursement Conditions and Reasons
Reimbursement condition | Reason | Implementation guidance |
|---|---|---|
Initiation | ||
1. Treatment with trastuzumab deruxtecan should be reimbursed in adult patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+ or ISH-negative) or HER2-ultralow (IHC 0 with membrane staining) breast cancer who meet all the following criteria: 1.1. have HR-positive disease (ER and/or PgR ≥ 1%) 1.2. have received at least 1 endocrine therapy in the metastatic setting 1.3. are not considered suitable candidates for endocrine therapy as the next line of therapy. | Evidence from the DESTINY-Breast06 trial showed that treatment with trastuzumab deruxtecan compared with physician’s choice of chemotherapy resulted in clinical benefit in patients with these characteristics. | pERC agreed with the clinical experts that it would be reasonable to use the discretion of the treating clinician to identify patients who are not considered suitable candidates for endocrine therapy. In the DESTINY-Breast06 trial, patients in the target patient population were eligible to receive trastuzumab deruxtecan if they had progressed within 6 months after initiation of first-line endocrine therapy plus a CDK4/6 inhibitor for the treatment of metastatic disease; or if they progressed on 2 or more lines of endocrine therapy (disease recurrence in the first 24 months of adjuvant endocrine therapy counted as 1 line). To ensure reliable differentiation of an IHC 0 without membrane staining, IHC 0 with membrane staining, and IHC 1+, testing centres in Canada need to implement adequate education and training to ensure patients with HER2-low and HER2-ultralow disease are correctly identified. If a recent tissue sample is not available, HER2-low or HER2-ultralow status may be tested using archival metastatic tissue samples. Time-limited opportunities to switch to trastuzumab deruxtecan may be reasonable during the initial funding period. Drug plan input states that sequencing of sacituzumab govitecan and trastuzumab deruxtecan is currently not funded and the intention is to remain consistent with this approach for this file. |
2. Patients should have a good performance status. | Patients with an ECOG PS score of 0 to 1 were included in the DESTINY-Breast06 trial. | Patients with an ECOG PS score of 2 may be treated at the discretion of the treating clinician. |
3. Patients must not have any of the following: 3.1. symptomatic spinal compression 3.2. clinically active CNS metastases 3.3. current ILD or pneumonitis 3.4. received prior chemotherapy for advanced or MBC; prior chemotherapy in the neoadjuvant or adjuvant setting is permitted, if patients completed systemic chemotherapy more than 12 months before developing advanced or metastatic disease. | This CDA-AMC review did not identify any evidence that patients with symptomatic spinal cord compression, active CNS metastases, current ILD or pneumonitis, or prior chemotherapy treatment that would benefit from trastuzumab deruxtecan as they were excluded from the DESTINY-Breast06 trial. Patients in the DESTINY-Breast06 trial were allowed to receive prior chemotherapy in the neoadjuvant or adjuvant setting, if patients completed treatment more than 12 months before developing advanced or metastatic disease. | — |
Discontinuation | ||
4. Trastuzumab deruxtecan must be discontinued upon the occurrence of any of the following: 4.1. progressive disease per RECIST 1.1 4.1.2. assessment for disease progression must be based on clinical and radiographic evaluation every 2 to 3 months, or as per clinician’s discretion. 4.2. unacceptable toxicity. | This CDA-AMC review identified no evidence that continuing treatment with trastuzumab deruxtecan in patients whose disease has progressed is effective. In the DESTINY-Breast06 trial, tumour response was assessed every 6 weeks for the first 12 months, and every 9 weeks thereafter. | — |
Prescribing | ||
5. Trastuzumab deruxtecan must only be initiated and prescribed by health care professionals with expertise in treating advanced breast cancer. | This will ensure that trastuzumab deruxtecan is prescribed for appropriate patients and that adverse effects are managed in an optimal and timely manner. | — |
Pricing | ||
6. A reduction in price. | Using the CDA-AMC base-case analysis, the ICER for trastuzumab deruxtecan was $453,869 per QALY gained when compared with chemotherapy in the requested reimbursement population. A band 3a price reduction would be required to achieve cost-effectiveness at a $50,000 per QALY threshold. A band 3a price reduction would be required to achieve cost-effectiveness at a $100,000 per QALY threshold. Price reductions for any given willingness-to-pay threshold are available in the CDA-AMC Main Report and Supplemental Material documents. | The CDA-AMC analysis is based on public list prices for all treatments. Further price reductions may be required if there are price arrangements (discounts) currently in place for any treatment included in the economic analysis. |
CDA-AMC = Canada’s Drug Agency; CNS = central nervous system; ECOG = Eastern Cooperative Oncology Group Performance Status; ER = estrogen receptor; HR = hormone receptor; ICER = incremental cost-effectiveness ratio; IHC = immunohistochemistry; ILD = interstitial lung disease; ISH = in situ hybridization; MBC = metastatic breast cancer; pERC = pan-Canadian Oncology Drug Review Expert Review Committee; PgR = progesterone receptor; QALY = quality-adjusted life-year; RECIST 1.1 = Response Evaluation Criteria in Solid Tumours Version 1.1.
aFor the statement regarding the size of the price reduction required, band 1 = 1% to 24%, band 2 = 25% to 49%, band 3 = 50% to 74%, and band 4 = 75% or greater.
Rationale for the Recommendation
Clinical Value
Based on the totality of the clinical evidence, pERC concluded that trastuzumab deruxtecan demonstrates acceptable clinical value compared with chemotherapy in adult patients with unresectable or metastatic HR-positive, HER2-low or HER2-ultralow breast cancer who have received at least 1 endocrine therapy in the metastatic setting and who are not considered suitable candidates for endocrine therapy as the next line of therapy. Given that trastuzumab deruxtecan is expected to be an alternative to chemotherapy, acceptable clinical value refers to added value versus chemotherapy.
Evidence from the DESTINY-Breast06 trial demonstrated that treatment with trastuzumab deruxtecan likely results in added clinical benefit in PFS in adults with HR-positive MBC with low or ultralow HER2 expression who have received 1 or more lines of endocrine-based therapy and no previous chemotherapy for MBC. Treatment with trastuzumab deruxtecan was associated with statistically significant and clinically meaningful improvements in PFS compared with physician’s choice of chemotherapy in both the HER2-low subpopulation (primary analysis; median PFS = 13.2 months versus 8.1 months; hazard ratio = 0.62, 95% confidence interval [CI], 0.52 to 0.75) and the intention-to treat [ITT] population (key secondary analysis; median PFS = 13.2 months versus 8.1 months; hazard ratio = 0.64, 95% CI, 0.54 to 0.76). Exploratory subgroup results for PFS in the HER2-ultralow population suggested a median PFS of 13.2 months with trastuzumab deruxtecan and 8.3 months with chemotherapy with a hazard ratio of 0.78 (95% CI, 0.5 to 1.21). OS was consistent with the results observed in the PFS analysis, suggesting a trend toward superiority for trastuzumab deruxtecan (ITT population; median OS = ████ months versus ████ months; hazard ratio = ████, 95% CI, ████ to ████). There was uncertainty in the OS results due to imprecise estimates and limited generalizability to clinical practice in Canada, as postprogression use of trastuzumab deruxtecan in patients receiving physician’s choice of chemotherapy was less frequent in the trial than in routine care. pERC considered that a greater proportion of patients in the trastuzumab deruxtecan group reported adjudicated ILD or pneumonitis, as well as gastrointestinal toxicities. pERC discussed the need for proactive monitoring and early intervention to support the management of ILD or pneumonitis and other toxicities in clinical practice. Overall, pERC agreed with the clinical experts and input from patient groups that the adverse effects associated with trastuzumab deruxtecan were significant but manageable in the appropriate clinical setting.
Patients and clinicians identified the need for effective treatment options that delay disease progression, reduce symptom burden, and improve quality of life. pERC concluded that, compared to chemotherapy, trastuzumab deruxtecan met some of the identified needs as it likely delays disease progression and may prolong survival. Although treatment with trastuzumab deruxtecan in the DESTINY-Breast06 trial may result in little to no difference in patients’ health-related quality of life (HRQoL) compared with chemotherapy, patient input suggested a treatment that delays disease progression and may extend survival with manageable side effects has the potential to improve patients’ HRQoL.
Further information on the committee’s discussion around clinical value is provided in the Summary of Deliberation section.
Developing the Recommendation
The determination of acceptable clinical value was sufficient for pERC to recommend reimbursement of trastuzumab deruxtecan. As part of the deliberation on whether to recommend reimbursement, the committee also considered unmet clinical need, unmet nonclinical need, and health inequity. Information on this discussion is provided in the Unmet Clinical Need and Distinct Social and Ethical Considerations domains in the Summary of Deliberation section.
Because pERC recommended that trastuzumab deruxtecan be reimbursed, the committee also deliberated on whether reimbursement conditions should be added to address important economic considerations, health system impacts, or social and ethical considerations, or to ensure clinical value is realized. The resulting reimbursement conditions, with accompanying reasons and implementation guidance, are stated in Table 1.
Summary of Deliberation
pERC considered all domains of value of the deliberative framework before developing its recommendation: clinical value, unmet clinical need, distinct social and ethical considerations, economic considerations, and impacts on health systems. For further information about the domains of value, refer to Expert Committee Deliberation at Canada’s Drug Agency.
The committee considered the following key discussion points, organized by the 5 domains of value.
Clinical Value
Appropriate comparators: pERC noted that single-agent chemotherapy was an appropriate comparator for trastuzumab deruxtecan.
Efficacy versus chemotherapy: One phase III, open-label, randomized controlled trial (DESTINY-Breast06) (N = 866) demonstrated that treatment with trastuzumab deruxtecan (5.4 mg/kg every 3 weeks) was associated with statistically significant improvements in PFS compared with physician’s choice of chemotherapy in both the HER2-low subpopulation and the ITT population. At a median follow-up time of 18.6 months, the 18-month between-group difference in PFS rates in the ITT population was █████; 95% CI, ████ to █████. OS outcomes were supportive of the results observed in the PFS analysis, suggesting a trend toward superiority for trastuzumab deruxtecan. However, the interpretation of OS data were limited by a ██████████ █████ information fraction in the ITT population (█████ of expected events observed), imprecision in between-group estimates (████ ███ ████ ████████ █████ ███████ █████ ██ ██ ███████ ███ ████), and limited generalizability to clinical practice in Canada due to less frequent postprogression use of trastuzumab deruxtecan in the trial compared to routine care. Due to a relatively short OS follow-up time, long-term survival benefit remains uncertain. Treatment with trastuzumab deruxtecan may result in little to no difference in patients’ HRQoL compared with physician’s choice of chemotherapy, although there was uncertainty due to the descriptive nature of the analysis and substantial attrition rates.
Efficacy in HER2-ultralow subgroup: The exploratory subgroup analyses for PFS and OS in the HER2-ultralow subgroup of the DESTINY-Breast06 trial were not powered to detect between-group differences and were limited by small sample sizes resulting in imprecise estimates (i.e., CIs encompassing small effects near the null). Despite uncertainty in the subgroup results, there was consistency in the direction of effects for PFS, OS, and objective response rate, favouring trastuzumab deruxtecan over chemotherapy. Additionally, point estimates were directionally aligned with PFS and OS results in the ITT and HER2-low populations. Due to the low number of patients at risk beyond 24 months, long-term survival was uncertain. pERC heard from clinical experts consulted by CDA-AMC that patients with HER2-low and HER2-ultralow MBC are managed similarly in clinical practice, and that outcomes with trastuzumab deruxtecan are expected to be comparable between these groups.
Clinical importance of treatment effects: pERC discussed the clinical meaningfulness of PFS in the MBC setting. pERC agreed with the clinical experts that the improvements in PFS of the magnitude observed in the DESTINY-Breast06 trial are of clinical importance in a heavily pretreated patient population, given limited efficacy of available chemotherapy options and attrition of patients with each successive line of therapy. Additionally, pERC acknowledged that patients with HER2-ultralow disease currently have no HER2-targeted treatment options.
Harms results: Comparative safety from the DESTINY-Breast06 trial indicated that the occurrence of treatment-emergent adverse events (AEs) of any grade and serious AEs was similar across treatment groups. The most common treatment-emergent AEs that occurred more commonly in the trastuzumab deruxtecan group were nausea, alopecia, anemia, vomiting, diarrhea, and constipation. The product monograph includes a serious warning about ILD and pneumonitis. According to the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) assessment, there is high certainty that trastuzumab deruxtecan results in an increase in ILD or pneumonitis cases compared with physician’s choice of chemotherapy. pERC considered that for some patients, lung damage may not be reversible and, although infrequent, it can potentially be fatal. pERC discussed the need for proactive monitoring and early intervention to support the management of ILD or pneumonitis in clinical practice. Trastuzumab deruxtecan is currently available in later lines for patients with HER2-low MBC, and no new safety signals were observed in the DESTINY-Breast06 trial. Overall, pERC agreed with the clinical experts and input from patient groups that the adverse effects associated with trastuzumab deruxtecan were significant but manageable in the appropriate clinical setting.
Certainty of the evidence: The committee noted that PFS results from the DESTINY-Breast06 trial were consistent between the ITT and HER2-low populations, demonstrating improvements supported by “high” to “moderate” certainty of evidence based on the GRADE assessment by CDA-AMC. Trastuzumab deruxtecan likely improves OS with “moderate” levels of certainty due to imprecision, according to the GRADE assessment. As noted, pERC heard from the clinical experts that postprogression use of trastuzumab deruxtecan in patients treated with physician’s choice of chemotherapy, is more common in clinical practice in Canada than in the trial, which may limit the generalizability of the OS results. The assessment of HRQoL (European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 global health status/quality of life scale) was rated as low certainty due to a very serious risk of study limitations (missing data and bias due to the open-label study design). Results for serious AEs and ILD or pneumonitis cases were associated with “moderate” and “high” certainty of evidence, respectively.
Clinical value: Based on the preceding considerations, pERC determined there was added clinical value versus single agent chemotherapy.
Unmet Clinical Need
Input on unmet clinical need: Within the HER2-negative population, the recently defined HER2-low and HER2-ultralow subtypes represent distinct biological subsets with limited treatment options and a continued need for effective therapies. The clinical experts consulted by CDA-AMC noted that patients with HER2-low and HER2-ultralow subtypes, whose disease has progressed following endocrine therapy, are typically managed with cytotoxic chemotherapy. Patients and clinicians highlighted the need for effective treatment options that delay disease progression, improve survival, and defer the use for chemotherapy. Patients with HER2-ultralow disease, currently have no HER2-targeted treatment options.
Severity of the disease: pERC noted that unresectable or MBC is an aggressive disease associated with a significant impact on HRQoL and a minority of patients surviving for 5 years or longer. Patient groups highlighted the negative physical, social, mental, emotional, and financial impacts of MBC on patients’ and caregivers’ lives.
Availability of treatment options: Patients who have received at least 1 endocrine therapy in the metastatic setting and who are not considered suitable candidates for endocrine therapy as the next line of therapy, are typically managed with cytotoxic chemotherapy with limited efficacy. pERC acknowledged the unmet therapeutic need for novel treatments.
Distinct Social and Ethical Considerations
Ethical implications: pERC acknowledges input from patient and clinician groups as well as clinical experts consulted by CDA-AMC, highlighting that while HER2 testing is currently performed as the standard of care for patients with MBC, some implementation barriers may exist while laboratories in Canada update reporting standards to classify the spectrum of HER2-negative status, including HER2-low and HER2-ultralow. The need for IV administration every 3 weeks may create barriers, especially in rural and remote areas. Patients and families may face indirect costs such as travel, time off work, and caregiving.
Economic Considerations
Health impacts of trastuzumab deruxtecan versus relevant comparators: Trastuzumab deruxtecan is predicted to be associated with a gain of 0.35 life-years and 0.31 quality-adjusted life-years (QALYs) compared to chemotherapy.
Cost of trastuzumab deruxtecan versus relevant comparators: Trastuzumab deruxtecan is predicted to be associated with higher costs to the health care systems than chemotherapy (incremental costs = $138,930), primarily driven by increased costs associated with drug acquisition.
Key findings of the economic evaluation: Based on the submitted evidence using the sponsor’s cost-utility analysis, the CDA-AMC base-case analysis estimated that the incremental cost-effectiveness ratio for trastuzumab deruxtecan was $453,869 per QALY gained when compared with chemotherapy in the requested reimbursement population (Figure 1).
Figure 1: Estimate of the ICER Used by pERC to Inform the Price Condition
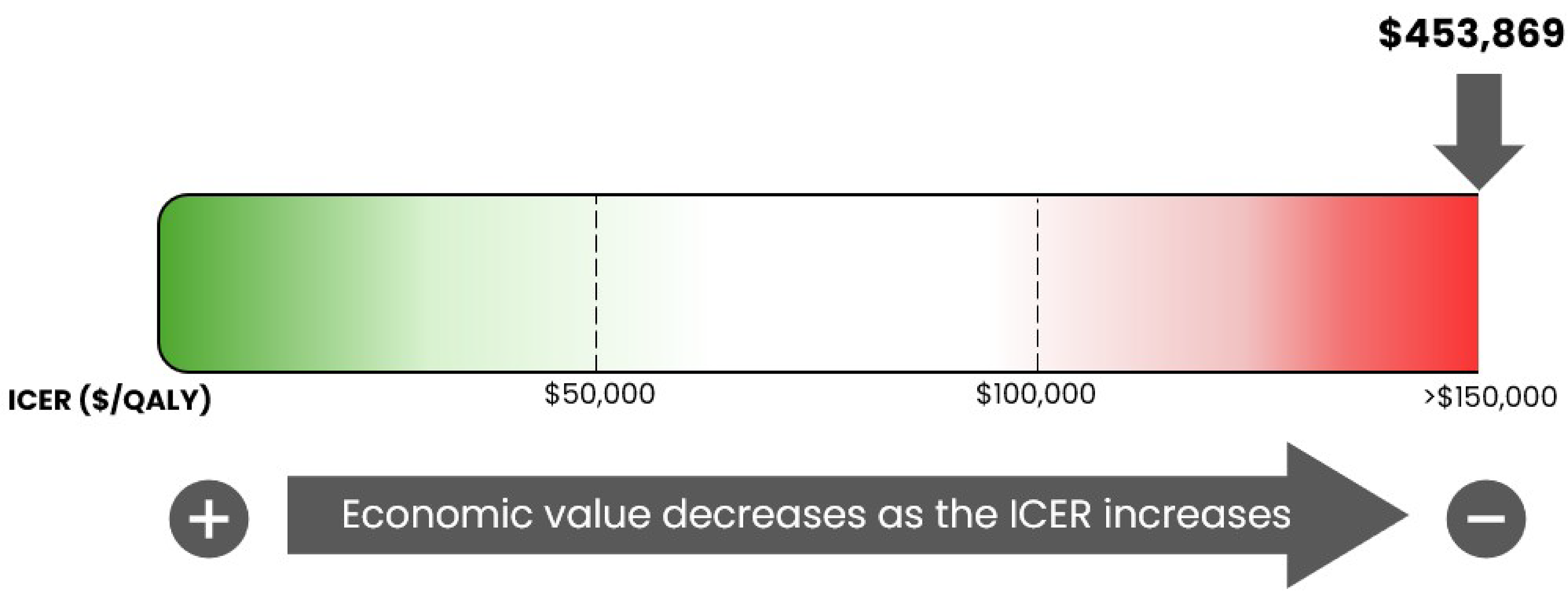
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
Certainty of the evidence: The cost-effectiveness of trastuzumab deruxtecan compared to chemotherapy is highly uncertain due to uncertainty in the long-term efficacy estimates and the costs and benefits of subsequent therapy. pERC noted that the CDA-AMC base case may overestimate incremental life-years and QALYs and underestimate subsequent therapy costs for trastuzumab deruxtecan versus chemotherapy relative to clinical practice in Canada owing to less frequent postprogression use of trastuzumab deruxtecan in the DESTINY-Breast06 trial compared to routine care in Canada. If incremental QALYs gained with trastuzumab deruxtecan are lower than predicted in the CDA-AMC base case, further price reductions than those described in Table 1 may be required.
Other considerations: pERC noted that the CDA-AMC base case reflects the requested reimbursement population and that the cost-effectiveness of trastuzumab deruxtecan for the full Health Canada indication is unknown. pERC further noted that the structure of the sponsor’s economic model did not permit the cost-effectiveness of trastuzumab deruxtecan to be estimated separately for the HER2-low and ultralow subgroups and that whether there is a difference in the cost-effectiveness of trastuzumab deruxtecan between these subgroups is unknown.
Impacts on Health Systems
Anticipated budget impact: CDA-AMC estimated that 3,453 patients would be eligible for treatment over a 3-year period based on the requested reimbursement population (year 1 = 1,125; year 2 = 1,151; year 3 = 1,177), of whom █████ are expected to receive trastuzumab deruxtecan (year 1 = ███; year 2 = ███; year 3 = ███). The estimated incremental budget impact of reimbursing trastuzumab deruxtecan for the requested reimbursement population is predicted to be approximately $70 million for the first 3 years, with an expected expenditure of $155 million on trastuzumab deruxtecan.
Number of eligible patients: pERC discussed that there is uncertainty in the number of patients considered unsuitable for endocrine therapy because clinical expert input indicated that the selection of such patients will be guided by clinical factors. If the proportion of patients who are considered eligible for trastuzumab deruxtecan in the second line is higher than predicted by the sponsor, costs associated with trastuzumab deruxtecan may be higher in the second line but lower in later lines.
Testing procedure considerations: pERC noted that HER2 testing is currently performed as the standard of care for patients with MBC and acknowledged that evaluating HER2 status before the initiation of trastuzumab deruxtecan would be required. pERC also noted that some implementation barriers may exist while laboratories in Canada update reporting standards to classify the spectrum of HER2-negative status, including HER2-low and HER2-ultralow. The committee highlighted the need for equitable and uniform access to testing and reporting with the updated classification for all patients, including those in rural or remote areas. pERC also noted the possibility of interobserver variability leading to imprecision of testing results when assessing HER2-low and HER2-ultralow status.
Costs related to HER2 testing. Some existing patients with HER2-negative, IHC 0 status may require the review of their previous archival pathology samples to reclassify into HER2-ultralow or HER2-null, which will require additional health care resources (e.g., pathologist time). The pathology experts consulted by CDA-AMC expect that the impact of reclassification on health care resources will be minimal; however, pERC notes that such costs were not included in either the cost-effectiveness analysis or budget impact analysis. If testing costs are incurred by the public payer, costs to the health care systems will be higher than estimated in the CDA-AMC analyses.
Sources of Information Used by the Committee
To make its recommendation, the committee considered the following information (links to the full documents for the review can be found on the project webpage):
the CDA-AMC review of the clinical and pharmacoeconomic evidence submitted by the sponsor, as well as relevant ethical issues related to trastuzumab deruxtecan (refer to the Main Report and Supplemental Material document)
the sponsor’s comments on the draft report and the CDA-AMC responses
patients' perspectives gathered by 3 patient groups: Breast Cancer Canada – McPeak Sirois Group for Clinical Research, Canadian Breast Cancer Network, and Rethink Breast Cancer (refer to the Patient and Clinician Group Input document)
input from 2 clinician groups, Ontario Health (Cancer Care Ontario) Breast Cancer Drug Advisory Committee and REAL Alliance (refer to the Patient and Clinician Group Input document)
input from public drug programs that participate in the reimbursement review process (refer to the Supplemental Material document)
input from 2 clinical experts with expertise in the management of breast cancer and 1 expert pathologist with expertise in testing procedures consulted by CDA-AMC.
pERC Information
Members of the Committee
Dr. Catherine Moltzan (Chair), Dr. Kelvin Chan (Vice-Chair), Paul Agbulu, Dr. Phillip Blanchette, Dr. Matthew Cheung, Dr. Michael Crump, Annette Cyr, Dr. Jennifer Fishman, Dr. Jason Hart, Terry Hawrysh, Dr. Yoo-Joung Ko, Dr. Aly-Khan Lalani, Amy Peasgood, Dr. Anca Prica, Dr. Michael Raphael, Dr. Adam Raymakers, Dr. Patricia Tang, Dr. Pierre Villeneuve, and Danica Wasney.
Meeting date: October 8, 2025
Regrets: One expert committee member did not attend.
Conflicts of interest: None.
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.