Drugs, Health Technologies, Health Systems
Reimbursement Review
Asciminib (Scemblix)
Sponsor: Novartis Pharmaceuticals Canada Inc.
Therapeutic area: Philadelphia chromosome–positive chronic myeloid leukemia
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Clinical Review
Abbreviations
AE
adverse event
AP
accelerated phase
ATP
adenosine triphosphate
BP
blast phase
CDA-AMC
Canada’s Drug Agency
CI
confidence interval
CML
chronic myeloid leukemia
CP
chronic phase
DMR
deep molecular response
ECOG
Eastern Cooperative Oncology Group
ELN
European LeukemiaNet
FAS
full analysis set
GRADE
Grading of Recommendations Assessment, Development and Evaluation
HRQoL
health-related quality of life
IA4
interim analysis 4
IS
international scale
ITC
indirect treatment comparison
LLSC
Leukemia & Lymphoma Society of Canada
MDASI-CML
MD Anderson Symptom Inventory for chronic myeloid leukemia
MMR
major molecular response
MR
molecular response
MR2
2.0 log reduction in BCR-ABL1 transcripts
MR4
4.0 log reduction in BCR-ABL1 transcripts
MR4.5
4.5 log reduction in BCR-ABL1 transcripts
OS
overall survival
Ph+
Philadelphia chromosome positive
qPCR
quantitative polymerase chain reaction
SAE
serious adverse event
SD
standard deviation
TFR
treatment-free remission
TKI
tyrosine kinase inhibitor
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Table 1: Background Information of Application Submitted for Review
Item | Description |
|---|---|
Drug product | Asciminib (Scemblix), 20 mg and 40 mg, film-coated tablets, oral |
Sponsor | Novartis Pharmaceuticals Canada Inc. |
Indication | Adult patients with Philadelphia chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase who are newly diagnosed or who have previously received 1 or more tyrosine kinase inhibitors. |
Reimbursement request | For the treatment of adult patients with Ph+ CML in chronic phase previously treated with 1 tyrosine kinase inhibitor. |
Health Canada approval status | NOC |
Health Canada review pathway | Project Orbis Type B |
NOC date | July 25, 2025 |
Recommended dose | 80 mg q.d. or 40 mg b.i.d. |
b.i.d. = twice a day; CML = chronic myeloid leukemia; NOC = Notice of Compliance; Ph+ = Philadelphia chromosome positive; q.d. = once a day.
Introduction
Chronic myeloid leukemia (CML) is a myeloproliferative neoplasm characterized by the aberrant and uncontrolled proliferation of mature and maturing granulocytes.1 Blood and bone marrow cells in patients with CML usually contain a characteristic chromosomal abnormality, known as the Philadelphia chromosome, resulting from a reciprocal translocation between chromosomes 9 and 22. In this process, a segment of the ABL1 gene on chromosome 9 detaches and joins the BCR gene on chromosome 22, forming the BCR-ABL1 fusion gene. This results in a shortened chromosome 22, which drives the uncontrolled cell growth associated with CML.1 The incidence rate of CML across all ages and sexes in Canada, excluding Quebec, ranged from 400 to 665 cases annually from 2009 to 2019. This corresponds to an incidence rate of 2.3 per 100,000 people in 2019.2 It is estimated that approximately 90% to 95% of patients with CML are in chronic phase (CP) at diagnosis which usually manifests as leukocytosis and immature myeloid cells in the blood.1,3 The CP is the earliest and most stable stage of the disease, characterized by a high number of mature white blood cells, with limited blast cell proliferation in the bone marrow and peripheral blood.1 Approximately 50% of patients with CML in CP are asymptomatic at presentation and are often diagnosed upon receiving laboratory abnormalities during a routine examination.1 Patients with symptoms most commonly present with anemia, fatigue, splenomegaly, and weight loss. Infrequently, patients are initially diagnosed in advanced CML stages: accelerated phase (AP) or blast phase (BP).1
Tyrosine kinase inhibitors (TKIs) are the initial treatment of choice for the majority of patients with CML.4 Imatinib was the first drug in this class to be approved for patients with CML and is commonly used as the first line of treatment. Some patients with initial responses to imatinib ultimately lose their response; 10% to 15% of patients with CML in CP develop resistance to imatinib within 18 months to 2 years of treatment.4 Dasatinib, nilotinib, and bosutinib are second-generation TKIs approved for treatment of CP or AP CML in patients with resistance or intolerance to prior therapy with a TKI.4 Ponatinib is a third-generation TKI used in patients with a BCR-ABL1 mutation, although it is associated with serious toxicity including cardiovascular, cerebrovascular, and peripheral vascular events.5,6
Asciminib is an oral inhibitor of BCR-ABL1 tyrosine kinase.7 It inhibits the ABL1 kinase activity of the BCR-ABL1 fusion oncoprotein, by specifically targeting the ABL myristoyl pocket (STAMP). Asciminib is administered as an oral tablet at a dosage of 80 mg daily7 and has received a recommendation of reimburse with conditions by Canada’s Drug Agency (CDA-AMC) in 2022 for the treatment of adult patients with Philadelphia chromosome–positive (Ph+) CML in CP previously treated with 2 or more TKIs.8 Asciminib is also currently undergoing review by CDA-AMC for the treatment of adult patients with previously untreated Ph+ CML in CP.9
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects of 20 mg and 40 mg asciminib, oral tablets, for the treatment of adult patients with CML in CP previously treated with 1 TKI.
Perspectives of Patients, Clinicians, and Drug Programs
The information in this section is a summary of input provided by the patient and clinician groups who responded to the call for input from CDA-AMC and from clinical experts consulted by CDA-AMC for the purpose of this review.
Patient Input
Four organizations provided input for this review including the CML Society of Canada, Heal Canada, and a joint submission from the Canadian CML Network and the Leukemia & Lymphoma Society of Canada (LLSC). The CML Society of Canada collected insights through surveys and phone interviews with more than 20 patients from Canada, France, the US, and the UK who had experience with asciminib after at least 1 prior therapy. Heal Canada conducted a survey of 15 people living in Canada with CML and interviewed 16 patients not living in Canada who had experience with asciminib, most of whom accessed the treatment upon TKI failure. The joint submission gathered responses from an online survey of 70 patients living in Canada with Ph+ CML in CP, all of whom had at least 1 prior therapy and 20 patients provided details on their experience with asciminib treatment. Additional input from the LLSC included in-person interviews with 2 patients living with CML and a round table discussion with 3 patients receiving treatment with asciminib. Across submissions, details on the proportion of patients with Ph+ CML in CP were not consistently identified.
All submissions highlighted the significant physical and emotional burdens of CML, including stress, fatigue, pain, and sleep disturbances, which affect daily activities, work, and social life. Heal Canada noted that most patients were asymptomatic before diagnosis and that lifelong medication is often undesirable, as many hope to discontinue treatment while maintaining remission. The joint submission emphasized the emotional toll of CML and the challenge of finding an effective treatment with manageable side effects. Many patients undergo a trial and error process with their health care providers to identify the most suitable therapy. Patients reported experience with various TKIs (e.g., bosutinib, dasatinib, imatinib), as well as other treatments such as allopurinol, bone marrow or stem cell transplant, chemotherapy, hydroxyurea, and radiation. Commonly reported TKI side effects across submissions included bone, joint, or muscle pain; diarrhea; fatigue; headache; and nausea and vomiting.
Across submissions, patients and caregivers prioritize treatments that improve symptom control, reduce side effects, and enhance quality of life, even if some side effects must be managed. The joint submission emphasized the importance of having multiple treatment options for CML. Key considerations when evaluating new therapies included quality of life during treatment, the number and severity of side effects, and treatment convenience.
Side effects experienced by patients treated with asciminib varied across submissions and included cold-like symptoms; bone, joint, or muscle pain; brain fog; fatigue; headache; low blood cell count; and skin rash. Across submissions, patients who had experience with asciminib reported various benefits including improved blood counts, reduced fatigue, and enhanced quality of life, with milder side effects compared to other TKIs. Patients found asciminib easy to use, with fewer disruptions to their daily lives, with those resistant or intolerant to other TKIs experiencing notable benefits. The CML Society of Canada highlighted the ability of asciminib to target a broader range of mutations, increasing the likelihood of achieving deep molecular response (DMR) (commonly defined as a 4 log reduction in BCR-ABL1 transcripts [MR4] or 4.5 log reduction in BCR-ABL1 transcripts [MR4.5]10) and improving patients’ quality of life. The joint submission reported that more than half of respondents on asciminib experienced moderate to significant improvements in daily routines and mental health, with nearly half also noting benefits in personal and work life compared to other treatments.
Clinician Input
Input From Clinical Experts Consulted for This Review
The clinical experts shared that therapy goals for patients with Ph+ CML in CP include achieving the best molecular response (MR) possible with the fewest side effects. For some patients, their goal is treatment-free remission (TFR) and for others it is improved health-related quality of life (HRQoL). The clinical experts emphasized that these goals are particularly important for patients receiving second-line treatment, as the need for second-line treatment indicates prior TKI therapy was unsuccessful due to resistance or intolerance, highlighting the need for alternative therapeutic options. The clinical experts noted that, although in most cases the disease responds to treatment when managed appropriately and with patient adherence, there are instances where the disease does not respond to treatment or becomes refractory over time. The clinical experts noted that nonadherence may occur due to a variety of reasons including side effects that are not being addressed, patient age, and personal beliefs. The clinical experts stated that some patients feel worse on treatment than before diagnosis, making it essential to minimize side effects and optimize quality of life, especially given the long-term, often lifelong nature of therapy. The experts stated that this is particularly relevant for patients starting a second-generation TKI, which are known to have more side effects than imatinib. The experts stated that individual responses and side effects experienced by patients can vary, and as such, having a broad range of therapeutic options is essential.
Although the clinical experts noted that the treatment under review is for first-line or second-line therapy, they would reserve asciminib for cases where treatment with a second-generation TKI was unsuccessful, whether due to resistance, including mutation-driven resistance, or less commonly, intolerance. The clinical experts added that resistance or intolerance to a treatment can only be confirmed after trying at least 1 other therapy for comparison. One clinical expert noted that it would be appropriate to recommend that patients try other treatments before initiating asciminib because the long-term safety profile of asciminib is not yet available, which may be a concern for many patients, and because of its currently higher cost relative to other available treatments. The clinical experts shared that subsequent TKIs, including asciminib, may be slightly more effective in patients with high Sokal scores. They also noted that patients with multiple comorbid conditions would be most in need of intervention, as some options may be safer than others based on individual health conditions.
The experts stated that most provinces and territories use the 2020 European LeukemiaNet (ELN)5 recommendations in clinical practice to assess response to treatment. The clinical experts noted that in both clinical trials and clinical practice, objective MRs to treatment corresponding to a reduction in BCR-ABL transcript levels are clinically relevant outcomes. One clinical expert noted that newer TKIs have not been able to improve TFR or survival beyond improvements made with first-generation TKIs; emphasizing that patients may still achieve long-term survival even if all treatment milestones are not met. As such, attempting TFR may become less important in the case of treatment resistance. One expert noted that many other factors can influence how a patient improves including coping mechanisms, mental outlook, and available support systems.
The clinical experts noted that asciminib may be discontinued due to disease progression, treatment resistance or intolerance, a suboptimal response to treatment, or a deep and sustained MR allowing for TFR. Other reasons to discontinue treatment include undergoing a stem cell transplant or the development of a separate terminal illness where treating CML is no longer appropriate. The clinical experts highlighted the importance of having CML experts or hematologists in consultation with CML experts diagnosing and monitoring the disease due to its rarity and the expense of the drugs involved.
Clinician Group Input
Two clinician groups (43 clinicians in total) provided input: the Canadian CML Physicians Interest Group and the Ontario Health (Cancer Care Ontario) Hematology Drug Advisory Committee. Input was gathered via virtual discussions with information gathered from literature reviews and collective clinical experiences.
The Canadian CML Physicians Interest Group noted that the introduction of TKIs transformed CML from a fatal disease into a manageable chronic condition. Both submissions noted that in Canada, standard first-line treatment for CML in CP includes imatinib, dasatinib, nilotinib, or bosutinib, though access varies by province and territory. Additional TKIs like asciminib and ponatinib are both approved for later lines of therapy, including for patients with the T315I mutation, which presents resistance to first-line options. Across submissions, the main treatment goals for patients with CML in CP include prolonging survival, preventing disease progression to AP or BP CML, achieving response milestones such as a major MR (MMR), improving or maintaining quality of life, improving blood counts, minimizing treatment-related toxicities, reducing splenomegaly and other disease symptoms, and offering eligible patients the opportunity to attempt TFR. For individual patients, treatment goals evolve over time and across lines of therapy but are based on patient desires and disease-specific characteristics. Both submissions noted that despite the transformative impact of TKI therapy in CML, treatment failure remains a persistent issue across all lines of therapy, leading to continued CML-related mortality. As such, the clinician groups noted that there is a need for treatments that are better tolerated and have superior efficacy.
The input noted that patients best suited for treatment with asciminib in the second-line setting are those that fit the ASC2ESCALTE study’s inclusion criteria regardless of whether their disease progression was caused by resistance, intolerance, or lack of response.
The Canadian CML Physicians Interest Group stated that most clinicians in Canada follow the 2020 ELN5 recommendations for treating CML as guidelines for treatment decisions. Response assessments begin with a baseline quantitative polymerase chain reaction (qPCR) and mutation testing if resistance is suspected. During the first 12 months of treatment, qPCR is performed every 3 months, with treatment changes recommended for intolerance or unmet molecular milestones. After 12 months, BCR-ABL1 levels are monitored every 3 months to 6 months, with increased monitoring and mutation testing if response is lost. The input noted that BCR-ABL1 transcript levels serve as a surrogate marker for treatment efficacy and long-term survival, with lower levels linked to reduced progression risk and improved outcomes. Patients who sustain MR4 or deeper for at least 2 years may be eligible for TFR.
Both inputs agreed that asciminib should be discontinued in cases of response failure (BCR-ABL1 on the international scale [IS] > 10% at 3 or 6 months or > 1% at 12 months or later) or if toxicity persists despite dose changes. Experienced hematologists should oversee initial treatment and early monitoring, while pharmacy and nursing teams can support oral medication management, adverse event (AE) monitoring, and treatment adherence.
Drug Program Input
The drug programs identified the following jurisdictional implementation issues: relevant comparators, considerations for initiation of therapy, considerations for prescribing of therapy, generalizability, funding algorithm, care provision issues, and system and economic issues. Refer to Table 4 for more details.
Clinical Evidence
Systematic Review
Description of Studies
One single-arm, ongoing, phase II, open-label, dose-escalation study (the ASC2ESCALATE study) of oral asciminib in first-line and second-line treatment of adult patients with CML in CP was included in this review. This report will focus on the second-line cohort only. The primary objective of the ASC2ESCALATE study was to evaluate the efficacy, safety, and tolerability of asciminib (starting dose of 80 mg once daily) for the treatment of adult patients with CML in CP previously treated with 1 TKI. Dose escalations were guided by the patients’ BCR-ABL1 results at 6 months and 12 months. Patients with BCR-ABL1 on the IS less than or equal to 1% at 6 months remained on their current dose, while those above this threshold increased their dose to 200 mg once daily. At 12 months, patients who did not achieve MMR, defined as a 3.0 log reduction in BCR-ABL1 transcripts (BCR-ABL1 on the IS ≤ 0.1%) increased their dose from 80 mg once daily to 200 mg once daily, or from 200 mg once daily to 200 mg twice daily. If MMR was still not achieved and it was deemed in the patient’s best interest, the investigator could discontinue the study treatment and switch to an alternative therapy. The ASC2ESCALATE study includes a 28-day screening period, a 36-month treatment period, and a 30-day safety follow-up phase. As of the November 15, 2024, clinical cut-off date for interim analysis 4 (IA4), a preliminary assessment was conducted on 101 evaluable patients in the second-line cohort. Of these, 63 patients (62.4%) had reached the 6-month follow-up, with additional data available for ██ ███████ patients who reached the 9-month time point. Efficacy end points of interest for this review included the proportion of patients who achieved an MMR, the proportion of patients who achieved MR, defined as 2.0 (MR2), 4.0 (MR4), and 4.5 (MR4.5) log reductions in BCR-ABL1 transcripts (≤ 1%, ≤ 0.01%, and ≤ 0.0032%, respectively), duration of and time to MMR, HRQoL as measured by the MD Anderson Symptom Inventory Questionnaire for chronic myeloid leukemia (MDASI-CML), overall survival (OS), as well as safety outcomes including AEs, serious AEs (SAEs), and notable harms such as pancreatitis and ischemic heart conditions.
The mean age of patients enrolled was 51.2 years (range, 18 years to 89 years), and most patients had an Eastern Cooperative Oncology Group (ECOG) Performance Status score of 0 or 1 (98.0%). All patients had received a prior TKI, the most common of which were dasatinib (n = 45 [44.6%]) and imatinib (n = 43 [42.6%]). The reasons for discontinuing the prior TKI were due to lack of efficacy (n = 57 [56.4%]) and lack of tolerability (n = 44 [43.6%]).
Efficacy Results
Major Molecular Response
The proportion of patients with an MMR was measured at 1, 3, 6, and 9 months in 101 patients with adequate follow-up. At 1 month, MMR was observed in 1.1% of patients (1 of 94; 95% confidence interval [CI], 0.0% to 5.8%). At 3 months, the MMR rate was 39.5% (34 of 86; 95% CI, 29.2% to 50.7%). By 6 months, 44.4% of patients (28 of 63; 95% CI, 31.9% to 57.5%) had an MMR. At 9 months, █████ ██████████ ███ █████ ██ █████) had an MMR.
Results for subgroup analyses by reason for prior TKI discontinuation (lack of efficacy or intolerance) were generally consistent with the primary analysis at the 9-month time point ██████ ████ ███ █████ ██ ██████ ███ █████ ████ ███ █████ ██ ███████ ██████████████.
MR2 Response
The proportion of patients with MR2 was █████ ███████ ███ ███ █████ ██ ██████ at 1 month, ████████████ ███ ███ █████ ██ ██████ at 3 months █████ ███████ ███ ███ █████ ██ ██████ at 6 months, and █████ ███████ ███ ███ █████ ██ ██████ at 9 months.
MR4 Response
The proportion of patients with MR4 was 0% (0 of 94; 95% CI, 0.0% to 3.9%) at 1 month, 11.6% (10 of 86; 95% CI, 5.7% to 20.4%) at 3 months, 25.4% (16 of 63; 95% CI, 15.3% to 37.9%) at 6 months, and █████ ███████ ███ ████████ ██ ██████ at 9 months.
MR4.5 Response
The proportion of patients with MR4.5 was 0% (0 of 94; 95% CI, 0.0% to 3.9%) at 1 month, 2.3% (2 of 86; 95% CI, 0.3% to 8.2%) at 3 months, 9.5% (6 of 63; 95% CI, 3.6% to 19.6%) at 6 months, and ████ █████████ ███ ████ ██ ██████ at 9 months.
Duration of MMR
Results for this outcome were not available as of IA4.
Time to MMR
Results for this outcome were not available as of IA4.
Overall Survival
Results for this outcome were not available as of IA4.
MD Anderson Symptom Inventory for Chronic Myeloid Leukemia
Results for this outcome were not available as of IA4.
Harms Results
Adverse Events
At the time of IA4, most patients (95.0%) experienced at least 1 AE. Headaches and nausea were the most frequently reported AEs experienced by 22.8% and 20.8% of patients, respectively. Cough, diarrhea, fatigue, and hypertension were each reported as AEs by 16 patients (15.8%). Approximately one-third (31.7%) of patients experienced AEs of grade 3 or higher, the most common of which was hypertension (8.9%).
Serious AEs
Three patients (3.0%) experienced SAEs of grade 3 or higher due to cardiac disorders, gastrointestinal disorders, general disorders, and administration site conditions.
Withdrawals Due to AEs
Three patients (3.0%) discontinued treatment due to AEs, with 1 patient reporting nausea and vomiting of grade 3 or higher.
Mortality
There were no deaths reported as of IA4.
Notable Harms
AEs of special interest in the ASC2ESCALATE study included gastrointestinal toxicity, experienced by 51.5% of patients, followed by hypersensitivity (21.8%), acute pancreatitis including isolated pancreatic enzyme elevations (16.8%), and myelosuppression (11.9%).
Critical Appraisal
There were notable issues with the study design of the ASC2ESCALATE study, specifically related to the single-arm and open-label nature, as well as the dose-escalation design. Considering the nature of Ph+ CML in the CP, which does not spontaneously reverse, the single-arm, noncomparative design was not deemed inappropriate; however, the lack of comparative evidence versus other second-line treatments (e.g., bosutinib, dasatinib, nilotinib, and imatinib) remains a concern and precludes the ability to assess the comparative effectiveness and safety relative to other available TKIs in Canadian clinical practice. The ASC2ESCALATE study was also open label, potentially increasing the risk of detection bias and performance bias. The reported AEs in the study were consistent with the known and documented profile for asciminib; thus, it is unlikely that the results for harms were biased by the open-label design. Lastly, the ASC2ESCALATE study used a dose-escalation design primarily to evaluate the safety and tolerability of increasing doses of asciminib in patients with inadequate response to the standard dose. This type of design is not usually intended to establish the overall effectiveness or comparative harms of the treatment but rather to identify whether higher doses can be administered safely, and was intended to achieve deeper responses with dose escalation. However, the escalated dose of 200 mg is not within the dosage range approved by Health Canada for asciminib. Furthermore, only 7 patients received an escalated dose of asciminib as of IA4. According to the FDA, MMR is a surrogate end point used for traditional approval for therapies in CML.11 According to ELN (2020) and National Comprehensive Cancer Network (2024) guidelines, sustained MMR (and DMR) are strong indicators of long-term outcomes including long-term cytogenetic remission and a reduced rate of disease progression.5,12 However, the sustainability of MMR observed in patients treated with asciminib in the ASC2ESCALATE study could not be assessed from the interim analysis. Formal statistical tests for efficacy outcomes were not conducted for the ASC2ESCALATE study, and as such, no P values were calculated. The threshold for a positive study outcome was observing a 95% CI for MMR rate at 12 months with a lower limit larger than 30% to reject the null hypothesis. As of the most recent interim analysis (IA4), descriptive results were provided for molecular outcomes (MMR, MR4.5, MR4, and MR2) through to 9 months and harms outcomes (AEs and SAEs) at 6 months. At 6-month and 9-month time points, the proportion of patients with MMR was suggestive of benefit according to the hypothesis. However, a limited number of patients were included in the primary efficacy population (N = 101), and only 63 and ██ patients had MR data available at 6 months and 9 months, respectively, which limits the generalizability of the findings. The evidence provided for the ASC2ESCALATE study at IA4 was considered immature and there were no data available for OS, time to event (time to or duration of MMR), and HRQoL (MDASI-CML). Improvements in quality of life were considered important to patients, clinician groups, and clinical experts consulted for this review, and as such, the lack of such results as of the most recent interim analysis is a notable limitation.
The ASC2ESCALTE trial was conducted exclusively in the US; however, the clinical experts consulted by CDA-AMC noted that the study population was generally consistent with the population expected to receive treatment in Canada. The clinical experts noted that some eligibility criteria such as ECOG Performance Status score may have been restrictive, selecting for ideal patients with less severe CML (98% had an ECOG Performance Status score ≤ 1), which may not reflect the general patient population but is typical of clinical trials. The clinical experts also stated that patients with an ECOG Performance Status score of 2 or higher, as well as those with CML in AP or BC — who were excluded from the ASC2ESCALATE trial — could receive asciminib if it was deemed the best treatment option. The clinical experts also highlighted that there was an overrepresentation of white patients (82.2%) in the ASC2ESCALATE study, which is not representative of what would be seen in the population of patients living in Canada. The absence of data beyond 9 months limits the ability to assess the durability of treatment responses and long-term safety. Although asciminib is available in the third-line setting, this absence of data is particularly important for this therapeutic class, because AEs may emerge only after prolonged use, with cumulative incidence increasing over time. One clinical expert highlighted the example of nilotinib, where cardiovascular risks became apparent only after a decade on the market.
GRADE Summary of Findings and Certainty of the Evidence
For the pivotal study identified in the sponsor’s systematic review, Grading of Recommendations Assessment, Development and Evaluation (GRADE) was used to assess the certainty of the evidence for outcomes considered most relevant to inform expert committee deliberations, and a final certainty rating was determined as outlined by the GRADE Working Group.13,14 Although GRADE guidance is not available for noncomparative studies, the CDA-AMC review team assessed the pivotal single-arm study for study limitations (which refers to internal validity or risk of bias), inconsistency across studies, indirectness, imprecision of effects, and publication bias to present these important considerations. Because the lack of a comparator arm does not allow for a conclusion to be drawn on the effect of the intervention versus any comparator, the certainty of evidence for single-arm studies starts at very low certainty with no opportunity for rating up.
Table 2 presents the GRADE summary of findings for asciminib for the treatment of patients with CML in CP in the second-line setting. The selection of outcomes for GRADE assessment was based on the sponsor’s summary of clinical evidence, consultation with clinical experts, and input received from patient and clinician groups and public drug plans. The following list of outcomes was finalized in consultation with expert committee members:
MR (MMR and MR2)
survival (OS)
quality of life (MDASI-CML)
notable harms (pancreatitis and ischemic heart disease).
Table 2: Summary of Findings for Asciminib for Patients With CML in Chronic Phase (Second Line)
Outcome and follow-up (months) | Patients (study), N | Effect | Certaintya | What happens |
|---|---|---|---|---|
MR | ||||
Proportion of patients with MMR, defined as a 3.0 log reduction in BCR-ABL1 transcripts (BCR-ABL1 on the IS ≤ 0.1%) at 12 months Follow-up: NA | NA (1 single-arm trial) | NA | NAb | There is no evidence about the effect of asciminib on the proportion of patients with MMR at 12 months compared to any active comparator. |
Proportion of patients with MMR, defined as a 3.0 log reduction in BCR-ABL1 transcripts (BCR-ABL1 on the IS ≤ 0.1%) at visit, n (%) Follow-up: 9 months | ██ ██ ██████████ ██████ | █████ ████ ███ ████ ██ █████ | Lowc,d,e | The evidence is very uncertain about the effect of asciminib on the proportion of patients with MMR when compared with any comparator. |
Proportion of patients with MR2, defined as a 2.0 log reduction in BCR-ABL1 transcripts (BCR-ABL1 on the IS ≤ 1.0%) at visit, n (%) Follow-up: 9 months | ██ ██ ██████████ ██████ | █████ ████ ███ ████ ██ █████ | Lowc,d,e | The evidence is very uncertain about the effect of asciminib on the proportion of patients with MR2 when compared with any comparator. |
OS | ||||
OS Follow-up: NA | NA (1 single-arm trial) | NA | NAb | There is no evidence about the effect of asciminib on OS. |
Health-related quality of life | ||||
MDASI-CML Follow-up: NA | NA (1 single-arm trial) | NA | NAb | There is no evidence about the effect of asciminib on quality of life. |
Harms | ||||
Notable harm: pancreatitis (clinical) Follow-up: 6 months | 101 (1 single-arm trial) | 0 per 1,000 | Very lowc,f | The evidence is very uncertain about the effect of asciminib on pancreatitis when compared with any comparator. |
Notable harm: ischemic heart disease Follow-up: 6 months | 101 (1 single-arm trial) | 10 per 1,000 | Very lowc,f | The evidence is very uncertain about the effect of asciminib on ischemic heart disease when compared with any comparator. |
CDA-AMC = Canada's Drug Agency; CI = confidence interval; CML = chronic myeloid leukemia; GRADE = Grading of Recommendations Assessment, Development and Evaluation; IS = international scale; MDASI-CML = MD Anderson Symptom Inventory for chronic myeloid leukemia; MMR = major molecular response; MR = molecular response; MR2 = 2.0 log reductions in BCR-ABL1 transcripts; NA = not applicable; OS = overall survival.
Note: All serious concerns with study limitations (which refer to internal validity or risk of bias), indirectness, imprecision of effects, and publication bias are documented in the table footnotes.
aIn the absence of a comparator group, conclusions about efficacy relative to any comparator cannot be drawn and the certainty of evidence is started at very low and cannot be rated up.
bThe ASC2ESCALATE trial is still ongoing. Data for the primary end point (the proportion of patients with MMR) and secondary end points (OS, MDASI-CML) at 12 months were not reported as of the most recent interim analysis.
cRated down 1 level for serious study limitations as results are based on the most recent interim analysis. Although not necessarily due to bias, interim analyses can overestimate treatment effects.
dRated down 1 level for serious imprecision; evidence from 1 single-arm trial with small sample size (N = 42 patients at 9 months).
eDespite the study limitations resulting in the certainty of evidence starting as “very low,” the outcomes of MMR and MR2 are demonstrative of an antitumour effect, which is supported by the FDA, and the proportion of patients with MR was considered clinically meaningful by the clinical experts consulted for this review. As such, given the effect size, which was believed to be large and clinically important, the CDA-AMC review team considered the certainty of this evidence to be higher.
fRated down 1 level for serious risk of bias due to potential bias arising from the open-label nature of the study and the subjective nature of the outcome.
Long-Term Extension Studies
No long-term extension studies were submitted by the sponsor for this review.
Indirect Comparisons
No indirect treatment comparisons (ITCs) were submitted by the sponsor for this review.
Studies Addressing Gaps in the Evidence From the Systematic Review
Description of Studies
One retrospective chart review study (Atallah et al.15) conducted in the US in adult patients living with Ph+ CML in CP (N = 255) was submitted by the sponsor. Eligible patients did not have a T315I mutation and were treated with asciminib after prior treatment with 1 TKI. Time to achieving or maintaining MMR, MR2, and MR4.5 was evaluated using Kaplan-Meier analyses. Subgroup analyses were conducted based on the reason for first TKI discontinuation (intolerance versus resistance) and by TKI generation (first generation versus second generation).
The mean age was 60.5 years (standard deviation [SD] = 9.5 years) and 43.5% of patients were female and 56.5% were male. A total of 20.8% of patients identified as Black or African American, 16.5% as Hispanic or Latino, 56.1% as white, and 6.7% as other. At CP CML diagnosis, 22.0% had low-risk, 57.6% intermediate-risk, and 18.4% high-risk Sokal scores (2.0% unknown). A total of 23.1%, 59.6%, and 17.3% had an ECOG Performance Status score of 0, 1, or at least 2, respectively. For initial treatment, 49.8% received imatinib, while 34.5%, 10.6%, and 5.1% received dasatinib, nilotinib, and bosutinib, respectively. First-line treatment had a mean duration of ████ ██████ (SD = ████ months), during which, █████ of patients had an MMR or better. Among subgroups, 43.5% discontinued their first-line TKI due to intolerance and 23.5% due to treatment resistance.
Efficacy Results
Patients Remaining on Treatment
Based on Kaplan-Meier analysis, the estimated probability of patients remaining on asciminib treatment at 48 weeks was 95.0% (95% CI, 91.3% to 97.1%). The probability of remaining on treatment with asciminib at 48 weeks among those who discontinued their first TKI due to intolerance (n = 111) or treatment failure (n = 60) was 97.2% (95% CI, 91.5% to 99.1%) and 92.2% (95% CI, 80.1% to 97.1%), respectively. By initial TKI type, the probability of remaining on treatment with asciminib at 48 weeks was 93.4% (95% CI, 87.2% to 96.6%) in patients previously on a first-generation TKI (n = 127) and 96.4% (95% CI, 91.0% to 98.7%) in those previously on a second-generation TKI (n = 128).
Molecular Outcomes
Major Molecular Response
At week 48, MMR was achieved or maintained by 68.3% of patients (95% CI, 61.8% to 74.5%). The median time to MMR was 30.7 weeks. At week 48, the proportion of patients who achieved or maintained MMR was 68.4% (95% CI, 58.9% to 77.5%) in patients with intolerance to prior TKI therapy, and 55.8% (95% CI, 42.9% to 69.7%) in patients with prior treatment failure. A total of 73.6% (95% CI, 65.4% to 81.3%) of patients initially treated with a second-generation TKI, and 61.5% (95% CI, 51.7% to 71.5%) with a first-generation TKI, had achieved or maintained an MMR.
MR2 Response
At week 48, 84.0% of patients (███ ███ █████ ██ ██████ achieved or maintained MR2, and the median time to MR2 was ████ █████. At week 48, the proportion of patients who achieved or maintained MR2 was 90.7% ████ ███ █████ ██ ██████ among those who discontinued their first TKI due to treatment failure and 79.5%; (███ ███ █████ ██ ██████ in those who discontinued due to intolerance. A total of 86.7% (███ ███ █████ ██ ██████ of patients initially treated with a second-generation TKI, and 80.4% ████ ███ █████ ██ ██████ of patients who received a first-generation TKI, had achieved or maintained MR2.
MR4 Response
At week 48, MR4 was achieved or maintained in 40.6% of patients ████ ███ █████ ██ ███████ At week 48, the proportion of patients who achieved or maintained MR4 was 44.9% ████ ███ █████ ██ ██████ in patients with prior TKI intolerance and 15.9% ████ ███ ████ ██ ██████ with prior TKI failure. MR4 was achieved or maintained by 43.2% ████ ███ █████ ██ ██████ of patients who received a second-generation TKI as first-line therapy and 37.3% ████ ███ █████ ██ ██████ of patients who received a first-generation TKI as first-line therapy.
Harms Results
While on treatment with asciminib, the most commonly reported AEs were fatigue (8.6%), headache (7.1%), rash (4.3%), and abdominal pain (2.4%). Gastrointestinal AEs included nausea (11.8%), vomiting (6.7%), and diarrhea (4.7%). Cytopenia occurred in 2.4% of patients. No cases of stroke, myocardial infarction, or heart failure were reported.
Critical Appraisal
The sponsor submitted data on a retrospective chart review study of the efficacy and safety of asciminib in the real-world setting. However, the absence of study protocols and statistical analysis plans limited the ability to fully assess the study design, outcome measurements, and analytical methods. Chart reviews rely on the accuracy and completeness of clinical documentation. The evaluation of real-world MR may have been based on heterogeneous criteria and inconsistent assessment schedules. As such, dates of diagnosis, progression, or death may have been inconsistently recorded, potentially leading to inaccurate results.
By week 48, ██ ████████ █████ in the overall cohort had either experienced an event or were censored. As censoring could have resulted from various factors including loss to follow-up or disease progression, the potential for biased response estimates cannot be excluded, particularly as the specific reasons and timing for censoring were not reported.
Although the sponsor defined treatment failure based on existing clinical guidelines, physicians may have applied their own criteria when identifying resistance to first-line TKI therapy. Differences in clinical decision-making and patient management across institutions could have confounded results. AEs were recorded during treatment with asciminib, but because physicians may only document AEs that lead to treatment changes (e.g., dose adjustments), underreporting is likely.
The retrospective chart review was conducted exclusively in the US with patients selected by physicians participating in a panel which may limit the generalizability of the findings to Canadian clinical practice. Clinical experts noted that in Canadian clinical practice, in the first-line setting, patients are more likely to receive dasatinib and less likely to receive nilotinib or bosutinib compared to the patients included in the chart review. Despite these differences in treatment patterns, the results suggested that asciminib was associated with consistent benefit in MR across all subgroups, regardless of whether patients initially received a first-generation or second-generation TKI.
Conclusions
One phase II, single-arm, open-label trial (the ASC2ESCALATE study) provided evidence for the efficacy and safety of asciminib in adult patients with Ph+ CML in CP that is intolerant to or has failed to respond to prior treatment with 1 TKI. In the ASC2ESCALATE study, ███ of patients experienced MMR by 9 months, which is likely a clinically important result, although the evidence was uncertain due to the substantial limitations driven by the single-arm study design, short follow-up, small sample size, and interim results. Nonetheless, the observed improvements in MMR at the interim time points in the trial were recognized as clinically meaningful according to the clinical experts consulted for this review based on their experience with TKIs in the second-line setting in this patient population. Asciminib has been available in Canada since 2022, and no new safety concerns were identified in the ASC2ESCALATE study.
Despite the potentially clinically significant results for MR outcomes, and tolerable safety profile, the absence of data for other outcomes that are important to patients and clinicians such as OS, time to and duration of MMR and TFR, and HRQoL limits the ability to draw conclusions on the durability and importance of any clinical benefit with asciminib, and the impact of asciminib on these outcomes in this population remains unknown. Furthermore, there are no direct or indirect comparative data on the effectiveness and safety of asciminib versus other treatment options in Canada in this patient population, which is a significant limitation of the evidence.
The MR findings of the ASC2ESCALATE study were supported by additional evidence from a retrospective chart review that provided a real-world evaluation of molecular outcomes in adult patients with CP CML treated with asciminib after prior TKI therapy, and no new safety concerns were identified; however, this study was associated with significant limitations, and can only be considered supportive of the overall effect of asciminib.
Introduction
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects of asciminib (Scemblix), film-coated tablets, 20 mg and 40 mg, oral, for the treatment of adult patients previously treated with 1 TKI for CML in CP.
Disease Background
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following has been summarized and validated by the review team.
CML is a myeloproliferative neoplasm characterized by the aberrant and uncontrolled proliferation of mature and maturing granulocytes.1 Blood and bone marrow cells in patients with CML usually contain a characteristic chromosomal abnormality, known as Philadelphia chromosome, resulting from a reciprocal translocation between chromosomes 9 and 22 where a part of the ABL1 gene on chromosome 9 attaches to the BCR gene on chromosome 22, forming the abnormal BCR-ABL1 fusion gene. This results in a shortened chromosome 22 (the Philadelphia chromosome) which drives the uncontrolled cell growth associated with CML.1 This BCR-ABL fusion gene produces an overactive tyrosine kinase enzyme. This abnormal activity continuously stimulates cell cycle pathways leading to unchecked proliferation of bone marrow cells.3
CML accounts for approximately 15% to 20% of all adult cases of leukemia.16 CML can occur in all age groups; however, most cases are diagnosed in patients aged 60 years or older, with a median age at presentation of 66 years in the US.1,17 Primary risk factors for CML include high-dose radiation exposure, increasing age, and male sex.18,19 It is estimated that approximately 90% to 95% of patients with CML are in CP at diagnosis which usually manifests as leukocytosis and immature myeloid cells in the blood.1,3 The CP is the earliest and most stable stage of the disease, characterized by a high number of mature white blood cells, with limited blast cell proliferation in the bone marrow and peripheral blood.1 Approximately 50% of patients with CML in CP are asymptomatic at presentation and are often diagnosed upon receiving laboratory abnormalities during a routine examination.1 Patients with symptoms most commonly present with anemia, fatigue, splenomegaly, and weight loss. Infrequently, patients are initially diagnosed in advanced CML stages: AP or BP.1 These stages show more immature blood cells in blood and bone marrow. They also involve worsening symptoms and additional cytogenetic abnormalities. CML diagnoses are confirmed by identification of the Philadelphia chromosome, BCR-ABL1 fusion gene, or BCR-ABL1 fusion mRNA using conventional cytogenetics, fluorescence in situ hybridization analysis, or reverse transcription polymerase chain reaction.20
The incidence rate of CML across all ages and sexes in Canada, excluding Quebec, ranged from 400 to 665 cases annually from 2009 to 2019.2 This corresponds to an incidence rate of 2.3 per 100,000 people in 2019.2 The 20-year prevalence rate in 2018 in Canada (excluding Quebec) was higher in male individuals (17.1 per 100,000 people) than in female individuals (13.3 per 100,000 people).21 The 20-year prevalence of CML across all ages and sexes in Canada, excluding Quebec, ranged from 3,785 cases in 2015 to 4,325 cases in 2018. This corresponds to a 20-year prevalence rate of 15.2 per 100,000 people in 2018.21
Standards of Therapy
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following has been summarized and validated by the review team.
TKIs targeting the BCR-ABL kinase represent the standard of care for patients with newly diagnosed CML in CP.4 Following the development of the first TKIs more than 20 years ago, 10-year survival rates have improved from approximately 20% to approximately 80% to 90%.22 Imatinib was the first drug in this class to be approved and is commonly used as the first line of treatment in this patient population.1 However, mutations to the adenosine triphosphate (ATP) binding site of BCR-ABL are associated with drug resistance and a high risk of progression, with approximately 10% to 15% of patients with CML in CP developing resistance to imatinib within 18 months to 2 years of treatment.1,4 Dasatinib, nilotinib, and bosutinib are second-generation TKIs that produce faster and deeper responses than imatinib and have a much smaller spectrum of resistance mutations.4 These agents produce similar rates of MMR (BCR-ABL1 on the IS ≤ 0.1%) and have similar progression-free survival and OS when used as second-line therapies.5,6 Ponatinib is a third-generation TKI designed to fit into the ATP binding domain of mutant forms of BCR-ABL and is known to bind firmly even in the presence of mutations such as T315I that are associated with resistance to first-generation and second-generation TKIs. As such, ponatinib is used in patients with CML in all phases who have a BCR-ABL1 T315I mutation and in patients where no other TKI is indicated.4 Ponatinib is associated with serious toxicity including cardiovascular, cerebrovascular, and peripheral vascular events as well as the elevation of pancreatic enzymes, pancreatitis, dermatitis, and fatigue.5,6
Evidence suggests that approximately one-third of patients have switched to an alternative TKI in randomized studies of CML in CP; mainly due to toxicity or suboptimal response.23 Patients with Ph+ CML who experience resistance or intolerance to imatinib in the first-line setting may receive second-generation TKIs such as bosutinib, dasatinib, or nilotinib in the second-line setting. Patients who receive a second-generation TKI in the first-line setting may receive an alternative second-generation TKI in the second-line setting. Rarely, patients may be stepped back onto imatinib in the second-line setting if the change is being made for intolerance.5,6
The overarching goals of care for all patients with Ph+ CML in CP are to achieve and maintain clinical remission, prevent progression to advanced disease stages such as AP or BP, and minimize treatment-related toxicity.24 Lifelong treatment with a TKI is often necessary, and for patients with CML in CP who have experienced failure with a prior TKI, achieving and sustaining an MMR is crucial to reducing the risk of disease progression and ensuring continuous suppression of the leukemic clone.
Drug Under Review
Asciminib is an oral inhibitor of BCR-ABL1 that selectively binds to a myristoyl pocket, inducing conformational changes typically produced by myristate binding to the N terminus of ABL1.7 This results in blocked kinase activity, preventing tumour cell proliferation. Asciminib demonstrates activity against both wild-type and mutated BCR-ABL1, including the T315I mutation.7 Asciminib is administered as an oral tablet at a dosage of 80 mg daily.7
The proposed Health Canada indication for asciminib is for the treatment of adult patients with newly diagnosed or previously treated Ph+ CML in CP and asciminib is currently undergoing review by Health Canada under Project Orbis, with a Notice of Compliance date of July 25, 2025.7
The sponsor’s reimbursement request is for the treatment of adult patients with Ph+ CML in CP previously treated with 1 TKI.7 CDA-AMC is simultaneously conducting a review of asciminib for use in treating adult patients with newly diagnosed Ph+ CML in CP (first-line setting).9 In 2022, asciminib also received a recommendation for reimbursement by CDA-AMC for the treatment of adult patients with Ph+ CML in CP that has previously been treated with 2 or more TKIs.8
Key characteristics of asciminib are summarized in Table 3 with other treatments available for Ph+ CML in CP in the second-line setting.
Table 3: Key Characteristics of Asciminib, Bosutinib, Nilotinib, Dasatinib, and Ponatinib
Item | Asciminib | Bosutinib | Nilotinib | Dasatinib | Ponatinib |
|---|---|---|---|---|---|
Mechanism of action | Asciminib is a potent inhibitor of ABL/BCR-ABL1 tyrosine kinase and inhibits the ABL1 kinase activity of the BCR-ABL1 fusion protein, by specifically targeting the ABL myristoyl pocket. | Bosutinib inhibits the activity of the oncogenic BCR-ABL kinase that promotes CML, through binding of the ATP binding site of the BCR-ABL oncoprotein. | Nilotinib inhibits the activity of the oncogenic BCR-ABL kinase that promotes CML, through binding of the ATP binding site of the BCR-ABL oncoprotein. | Dasatinib inhibits the activity of the oncogenic BCR-ABL kinase that promotes CML, binding both the inactive and active conformations of the enzyme. | Ponatinib is a potent pan–BCR-ABL inhibitor with structural elements, including a carbon–carbon triple bond that enables high-affinity binding to native BCR-ABL and mutant forms of the ABL kinase. |
Indicationa | Proposed: For the treatment of adult patients with newly diagnosed or previously treated CP Ph+ CML. | For the treatment of adult patients with newly diagnosed CP Ph+ CML. For the treatment of adult patients with AP, BP, or CP Ph+ CML with resistance or intolerance to prior TKI therapy. | For the treatment of adult patients with newly diagnosed CP Ph+ CML. For the treatment of adult patients with CP and AP Ph+ CML with resistance or intolerance to prior TKI therapy including imatinib. | For the treatment of adult patients with newly diagnosed CP Ph+ CML. For the treatment of adult patients with CP, AP, or BP Ph+ CML with resistance or intolerance to prior therapy including imatinib. | For the treatment of adult patients with CP, AP, or BP CML or Ph+ ALL for whom other TKI therapy is not appropriate, including CML or Ph+ ALL that is T315I mutation positive or where there is prior TKI resistance or intolerance. |
Route of administration | Oral | Oral | Oral | Oral | Oral |
Recommended dose | 80 mg q.d. or 40 mg b.i.d. at 12-hour intervals | 500 mg b.i.d. with food | 400 mg b.i.d. | 100 mg q.d. | Starting dosage: 45 mg q.d. |
Serious adverse effects or safety issues | QT interval prolongation; hypertension; embryo–fetal toxicity; myelosuppression; pancreatic toxicity; hepatitis B reactivation | Gastrointestinal toxicity, hepatic toxicity, cardiac toxicity, pancreatic toxicity, fluid retention, hemorrhage, QT interval prolongation, myelosuppression, tumour lysis syndrome, hepatitis B reactivation, second primary malignancies | Cardiac deaths, QT interval prolongation, ischemic heart disease, ischemic cerebrovascular events, peripheral arterial occlusive disease, hepatoxicity, pancreatitis, myelosuppression | Myelosuppression, hemorrhage, fluid retention, congestive heart failure, and pulmonary arterial hypertension | Arterial occlusions, venous thromboembolism, heart failure, hemorrhage events, hepatotoxicity, myelosuppression, and pancreatitis |
ALL = acute lymphoblastic leukemia; AP = accelerated phase; ATP = adenosine triphosphate; b.i.d. = twice a day; BP = blast phase; CML = chronic myeloid leukemia; CP = chronic phase; Ph+ = Philadelphia chromosome positive; q.d. = once a day; TKI = tyrosine kinase inhibitor.
aHealth Canada–approved indication.
Sources: Asciminib draft product monograph,7 bosutinib product monograph,25 nilotinib product monograph,26 dasatinib product monograph,27 and ponatinib product monograph.28
Perspectives of Patients, Clinicians, and Drug Programs
The full patient and clinician group submissions received are available in the consolidated patient and clinician group input document for this review on the project website.
Patient Group Input
This section was prepared by the review team based on the input provided by patient groups.
Four organizations provided input for this review including the CML Society of Canada, Heal Canada, and a joint submission from the Canadian CML Network and the LLSC. The CML Society of Canada collected insights through surveys and phone interviews with more than 20 patients from Canada, France, the UK, and the US who had experience with asciminib after at least 1 prior therapy. Heal Canada conducted a survey of 15 people living in Canada patients with CML and interviewed 16 people not living in Canada patients who had experience with asciminib, most of whom accessed the treatment upon TKI failure. The joint submission gathered responses from an online survey of 70 people living in Canada with Ph+ CML in CP, all of whom had at least 1 prior therapy and 20 patients provided details on their experience with asciminib treatment. Additional input from the LLSC included in-person interviews with 2 patients living with CML and a round table discussion with 3 patients receiving treatment with asciminib. Across submissions, details on the proportion of patients with Ph+ CML in CP were not consistently identified.
All submissions highlighted the significant physical and emotional burden of CML, including stress, fatigue, pain, and sleep disturbances, which affect daily activities, work, and social life. Heal Canada noted that most patients were asymptomatic before diagnosis and that lifelong medication is often undesirable, as many hope to discontinue treatment while maintaining remission. The joint submission emphasized the emotional toll of CML and the challenge of finding an effective treatment with manageable side effects. Many patients undergo a trial and error process with their health care providers to identify the most suitable therapy. Patients reported experience with various TKIs (e.g., bosutinib, dasatinib, imatinib), as well as other treatments such as allopurinol, bone marrow or stem cell transplant, chemotherapy, hydroxyurea, and radiation. Commonly reported TKI side effects across submissions included bone, joint, or muscle pain; diarrhea; fatigue; headache; and nausea and vomiting.
Across submissions, patients and caregivers prioritize treatments that improve symptom control, reduce side effects, and enhance quality of life, even if some side effects must be managed. The joint submission emphasized the importance of having multiple treatment options for CML. Key considerations when evaluating new therapies included quality of life during treatment, the number and severity of side effects, and treatment convenience.
Side effects experienced by patients treated with asciminib varied across submissions and included cold-like symptoms; bone, joint, or muscle pain; brain fog; fatigue; headache; low blood cell count; and skin rash. Across submissions, patients who had experience with asciminib also reported various benefits including improved blood counts, reduced fatigue, and enhanced quality of life, with milder side effects compared to other TKIs. Patients found asciminib easy to use, with fewer disruptions to their daily lives, with those resistant or intolerant to other TKIs experiencing notable benefits. The CML Society of Canada highlighted the ability of asciminib to target a broader range of mutations, increasing the likelihood of achieving a DMR (MR4.5) and improving patients’ quality of life. The joint submission reported that more than half of respondents on asciminib experienced moderate to significant improvements in daily routines and mental health, with nearly half also noting benefits in personal and work life compared to other treatments.
Clinician Input
Input From Clinical Experts Consulted for This Review
All CDA-AMC review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 2 clinical specialists with expertise in the diagnosis and management of Ph+ CML in CP.
Unmet Needs
The clinical experts shared that therapy goals for patients with Ph+ CML in CP include achieving the best MR possible with the fewest side effects. For some patients, their goal is TFR and for others it is improved HRQoL. The clinical experts emphasized that these goals are particularly important for patients receiving second-line treatment, as the need for second-line treatment indicates prior TKI therapy was unsuccessful due to resistance or intolerance, highlighting the need for alternative therapeutic options. The clinical experts noted that, although in most cases the disease responds to treatment when managed appropriately and with patient adherence, there are instances where the disease does not respond to treatment or becomes refractory over time. The clinical experts noted that nonadherence may occur due to a variety of reasons including side effects that are not being addressed, patient age, and personal beliefs. The clinical experts stated that some patients feel worse on treatment than before diagnosis, making it essential to minimize side effects and optimize quality of life, especially given the long-term, often lifelong nature of therapy. The experts stated that this is particularly relevant for patients starting a second-generation TKI, which are known to have more side effects than imatinib. The experts stated that individual responses and side effects experienced by patients can vary, and as such, having a broad range of therapeutic options is essential.
Place in Therapy
Although the clinical experts noted that the treatment under review is for first-line or second-line therapy, they would reserve asciminib for cases where treatment with a second-generation TKI was unsuccessful, whether due to resistance, including mutation-driven resistance, or less commonly, intolerance. The clinical experts added that resistance or intolerance to a treatment can only be confirmed after trying at least 1 other therapy for comparison. One clinical expert noted that it would be appropriate to recommend that patients try other treatments before initiating asciminib because the long-term safety profile of asciminib is not yet known, which may be a concern for many patients, and given its currently higher cost relative to other available treatments.
Patient Population
The clinical experts shared that subsequent TKIs, including asciminib, may be slightly more effective in patients with high Sokal scores. They also noted that patients with multiple comorbid conditions would be most in need of intervention, as some options may be safer than others based on individual health conditions.
Assessing the Response Treatment
The experts stated that most provinces and territories use the ELN 2020 guidelines5 to assess response to treatment in clinical practice. The clinical experts noted that in both clinical trials and clinical practice, objective MRs to treatment corresponding to a reduction in BCR-ABL transcript levels are clinically relevant outcomes. One clinical expert noted that newer TKIs have not been able to improve TFR or survival beyond improvements made with first-generation TKIs; emphasizing that patients may still achieve long-term survival even if all treatment milestones are not met. As such, attempting TFR may become less important in the case of treatment resistance. One expert noted that many other factors can influence how a patient improves including coping mechanisms, mental outlook, and available support systems.
Discontinuing Treatment
The clinical experts noted that asciminib may be discontinued due to disease progression, treatment resistance or intolerance, a suboptimal response to treatment, or a deep and sustained MR allowing for TFR. Other reasons to discontinue treatment include undergoing a stem cell transplant or the development of a separate terminal illness where treating CML is no longer appropriate.
Prescribing Considerations
The clinical experts highlighted the importance of having CML experts or hematologists in consultation with CML experts diagnosing and monitoring the disease due to its rarity and the expense of the drugs involved.
Clinician Group Input
This section was prepared by the review team based on the input provided by clinician groups.
Two clinician groups consisting of 43 clinicians provided input for this review, including the Canadian CML Physicians Interest Group and the Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee. Input was gathered via virtual discussions with information gathered from literature reviews and collective clinical experiences.
The Canadian CML Physicians Interest Group noted that the introduction of TKIs transformed CML from a fatal disease into a manageable chronic condition. Both submissions noted that in Canada, standard first-line treatment for CML in CP includes imatinib, dasatinib, nilotinib, or bosutinib, though access varies by province and territory. Additional TKIs like asciminib and ponatinib are both approved for later lines of therapy, including for patients with the T315I mutation, which presents resistance to first-line options. Across submissions, the main treatment goals for patients with CML in CP include prolonging survival, preventing disease progression to AP or BP CML, achieving MR milestones (e.g., MMR or DMR), improving or maintaining quality of life, improving blood counts, minimizing treatment-related toxicities, reducing splenomegaly and other disease symptoms, and offering eligible patients the opportunity to attempt TFR. For individual patients, treatment goals evolve over time and across lines of therapy but are based on patient desires and disease-specific characteristics. Both submissions noted that despite the transformative impact of TKI therapy in CML, treatment failure remains a persistent issue across all lines of therapy, leading to continued CML-related mortality. As such, the clinician groups noted that there is a need for treatments that are better tolerated and have superior efficacy.
The input noted that patients best suited for treatment with asciminib in the second-line setting are those that fit the ASC2ESCALTE study’s inclusion criteria regardless of whether their disease progression was caused by resistance, intolerance, or lack of response.
The Canadian CML Physicians Interest Group stated that most clinicians in Canada follow the ELN (2020)5 recommendations for treating CML as guidelines for treatment decisions. Response assessments begin with a baseline qPCR and mutation testing if resistance is suspected. During the first 12 months of treatment, qPCR is performed every 3 months, with treatment changes recommended for intolerance or unmet molecular milestones. After 12 months, BCR-ABL1 levels are monitored every 3 months to 6 months, with increased monitoring and mutation testing if response is lost. The input noted that BCR-ABL1 transcript levels serve as a surrogate marker for treatment efficacy and long-term survival, with lower levels linked to reduced progression risk and improved outcomes. Patients who sustain MR4 or deeper for at least 2 years may be eligible for TFR.
Both inputs agreed that asciminib should be discontinued in cases of response failure (BCR-ABL1 > 10% at 3 or 6 months or > 1% at 12 months or later), persistent toxicity despite dose adjustments, or intolerance. Experienced hematologists should oversee initial treatment and early monitoring, while pharmacy and nursing teams can support oral medication management, AE monitoring, and treatment adherence.
Drug Program Input
The drug programs provide input on each drug being reviewed through the reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by for this review are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Relevant comparators | |
How does second-line asciminib compare to currently funded treatments for adult patients with CML in CP? | The clinical experts noted that asciminib appears similar in efficacy with less short-term side effects but unknown long-term safety. |
Considerations for initiation of therapy | |
Patients in the ASC2ESCALATE study were diagnosed with CML in CP (per WHO criteria) with no history of AP or BC. Should patients with CML in AP or BP be eligible for treatment with asciminib? | The clinical experts noted that asciminib should be considered a viable treatment option for these patients when it is deemed the most appropriate choice for an individual patient, particularly given the precedent of using other agents in this setting. However, the experts also note that it is unclear whether asciminib would be more effective than any other treatment currently used for this patient population due to the lack of evidence in this specific population. |
Patients included in the ASC2ESCALATE study had an ECOG PS score of 0 to 2. Can patients with an ECOG PS score > 2 receive treatment with asciminib? | Yes, patients with an ECOG PS score of ≥ 2 would be eligible to receive treatment with asciminib according to the clinical experts. |
The following patients were excluded from the ASC2ESCALATE study:
Would there be any consideration for patients with these exclusions to receive asciminib? | The clinical experts noted that if a patient previously discontinued asciminib due to intolerance (rather than resistance), rechallenging with asciminib may be appropriate if it is later determined to be the most tolerable option after trying other therapies. Additionally, while largely anecdotal, its use in patients with multiple prior intolerances (particularly in the third-line or later settings) has been observed in real-world practice and should remain an option, though its relevance may decrease over time. In cases of relapse or progression post transplant, where treatment options are limited and data for any drug are weak, asciminib should not be excluded, particularly if asciminib was the treatment used that resulted in the patient becoming transplant eligible. Also, patients with a T315I mutation should not be excluded from receiving asciminib. |
Can asciminib be given again to patients who relapsed while off therapy? | The clinical experts stated that it is reasonable to expect that asciminib can be given again to patients who relapsed while off therapy based on data from other TKIs in comparable treatment settings. |
Considerations for prescribing of therapy | |
The recommended total daily dose is 80 mg which can be taken orally either as 80 mg once daily at approximately the same time each day, or as 40 mg twice daily at approximately 12-hour intervals. In the ASC2ESCALATE study, patients may be dose-escalated to 200 mg once daily if BCR-ABL1 is > 1% at 6 months and may be dose-escalated to 200 mg twice daily if BCR-ABL1 is > 0.1% at 12 months. | This is a comment from the drug programs to inform pERC deliberations. |
Generalizability | |
In the event of a positive funding recommendation, should patients receiving an alternative TKI be eligible to switch to asciminib when funding becomes available? | The clinical experts stated that patients receiving an alternative TKI should only be eligible to switch to asciminib if there is a valid reason for switching. |
Funding algorithm | |
Request an initiation of a rapid provisional funding algorithm. | This is a comment from the drug programs to inform pERC deliberations. |
Drug may change place in therapy of comparator drugs. | This is a comment from the drug programs to inform pERC deliberations. |
Other care provision issues | |
Asciminib is associated with potential drug–drug, drug–food, and drug–herb interactions. | This is a comment from the drug programs to inform pERC deliberations. |
System and economic issues | |
There is confidential pricing for bosutinib; and nilotinib, dasatinib, and imatinib are available as generics. | This is a comment from the drug programs to inform pERC deliberations. |
AP = accelerated phase; ATP = adenosine triphosphate; BC = blast crisis; BP = blast phase; CML = chronic myeloid leukemia; CP = chronic phase; ECOG PS = Eastern Cooperative Oncology Group; HSCT = hematopoietic stem cell transplant; pERC = pan-Canadian Oncology Drug Review Expert Review Committee; PS = Performance Status; TKI = tyrosine kinase inhibitor.
Clinical Evidence
The objective of this Clinical Review report is to review and critically appraise the clinical evidence submitted by the sponsor on the beneficial and harmful effects of asciminib (Scemblix), 20 mg and 40 mg, oral tablets in the treatment of adult patients with Ph+ CML in CP previously treated with 1 TKI. The focus will be placed on comparing asciminib to relevant comparators and identifying gaps in the current evidence.
A summary of the clinical evidence included by the sponsor in the review of asciminib is presented in the first section with the critical appraisal of the evidence included at the end of the section. This main section, the systematic review, includes the pivotal study that was selected according to the sponsor’s systematic review protocol. The assessment of the certainty of the evidence in this section using the GRADE approach follows the critical appraisal of the evidence. The second section includes a retrospective chart review that was considered by the sponsor to address important gaps in the systematic review evidence. No indirect evidence or long-term extension studies were included by the sponsor.
Included Studies
Clinical evidence from the following is included in the review and appraised in this document:
1 pivotal study identified in the systematic review (the ASC2ESCALATE study)
1 additional study addressing gaps in evidence.
Systematic Review
Contents within this section have been informed by materials submitted by the sponsor. The following has been summarized and validated by the review team.
Description of Studies
Characteristics of the ASC2ESCALATE study are summarized in Table 5. One ongoing, phase II, open-label, multicentre, single-arm, dose-escalation study was included in the systematic review, the ASC2ESCALATE study.29 The study had 2 cohorts and investigated the efficacy of asciminib in both the first-line and second-line setting. This report will focus on the second-line cohort only. The primary objective of the ASC2ESCALATE study was to evaluate the efficacy, safety, and tolerability of asciminib (starting dose of 80 mg once daily) for the treatment of adult patients with CML in CP previously treated with 1 TKI.
In the second-line cohort, 101 patients were recruited from 85 sites across the US. The primary analysis was to occur after 12 months of study treatment, although results were not available at the time of this submission. Therefore, the current review is based on the results of an interim analysis (IA4) conducted at 6 months (with 9-month visit results added for MRs post database lock), with a data cut-off date of November 15, 2024.
The study consisted of a 28-day screening period, a 36-month treatment period, and a 30-day safety follow-up phase after the last dose of study treatment (Figure 1).
Table 5: Details of Studies Included in the Systematic Review
Detail | ASC2ESCALATE study |
|---|---|
Designs and populations | |
Study design | Phase II, open-label, multicentre, single-arm, dose-escalation study |
Locations | 85 sites in the US |
Patient enrolment dates | Start date: November 11, 2022 End date: October 17, 2027 |
Enrolled (N) | N = 101 (second-line cohort) |
Inclusion criteria |
|
Exclusion criteria |
|
Drugs | |
Intervention | All eligible patients were initially treated with asciminib 80 mg q.d. At 6 months:
At 12 months:
In patients with toxicity of ≥ grade 3 or persistent grade 2 toxicity refractory to optimal management, dose escalation was not considered, and the same dose of asciminib was continued. |
Comparator(s) | NA |
Study duration | |
Screening phase | 28 days |
Treatment phase | 36 months |
Follow-up phase | 30 days following the last dose of study treatment |
Outcomes | |
Primary end point | Proportion of patients achieving MMR at 12 months |
Secondary and exploratory end points | Secondary:
Exploratory:
|
Publication status | |
Publications | |
1L = first line; 2L = second line; ALT = alanine transaminase; AP = accelerated phase; AST = aspartate transaminase; ATP = adenosine triphosphate; BC = blast crisis; b.i.d. = twice a day; CML = chronic myeloid leukemia; CP = chronic phase; DMR = deep molecular response; ECOG = Eastern Cooperative Oncology Group; ELN = European LeukemiaNet; HSCT = hematopoietic stem cell transplant; MDASI-CML = MD Anderson Symptom Inventory for chronic myeloid leukemia; MMR = major molecular response; MR = molecular response; MR2 = 2.0 log reductions in BCR-ABL1 transcripts; MR4 = 4.0 log reductions in BCR-ABL1 transcripts; MR4.5 = 4.5 log reductions in BCR-ABL1 transcripts; NA = not applicable; OS = overall survival; q.d. = once a day; TKI = tyrosine kinase inhibitor; ULN = upper limit of normal.
Note: The ASC2ESCALATE study included both 1L and 2L cohorts; however, only the 2L cohort criteria are presented to align with the sponsor’s reimbursement request.
aScheduled time points include 3, 6, 12, 18, 24, 30, and 36 months.
bOutcome was unavailable as of the data cut-off date for the interim analysis (interim analysis 4).
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the 2L cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Figure 1: Study Design of the ASC2ESCALATE Study
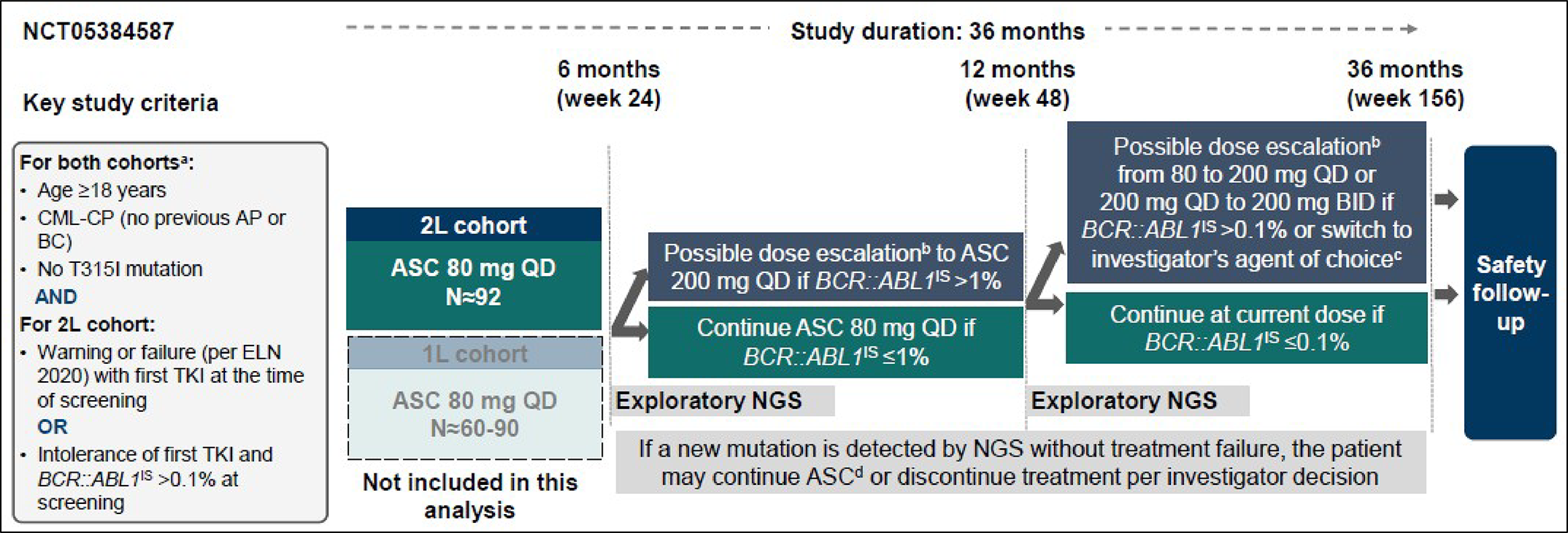
1L = first line; 2L = second line; AP = accelerated phase; ASC = asciminib; BC = blast crisis; BID = twice a day; CML = chronic myeloid leukemia; CP = chronic phase; ELN = European LeukemiaNet; IS = international scale; NGS = next-generation sequencing; QD = once a day; TKI = tyrosine kinase inhibitor.
Note: The patient will be taken off the study and switched to an investigator’s drug of choice if it is in the best interest of the patient.
aFor newly diagnosed patients with CML in CP (1L cohort), treatment with 1 prior TKI (imatinib, dasatinib, nilotinib, or bosutinib) for ≤ 4 weeks was allowed.
bFor any grade 3 or 4 toxicity, or persistent grade 2 toxicity unresponsive to optimal management, the dose escalation did not apply, and patients were continued on the current asciminib dosage.
cPatients switching to investigator’s drug of choice were taken off of the study.
dAt the same dose unless meeting dose escalation criteria.
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the 2L cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31
Populations
Inclusion and Exclusion Criteria
Eligibility criteria for the ASC2ESCALATE trial are summarized in Table 5. Briefly, the ASC2ESCALATE study included patients with CML in CP receiving second-line therapy, with no history of AP or BP and no known presence of a T315I mutation. Eligible patients had either warning signs or treatment resistance to first-line TKI therapy at screening.
Warning was defined as:
BCR-ABL1 on the IS greater than 1% but less than or equal to 10% after 6 months of first-line treatment, or
BCR-ABL1 on the IS greater than 0.1% but less than or equal to 1% after 12 months of first-line treatment.
Treatment resistance was defined as:
BCR-ABL1 on the IS greater than 10% after 6 to 12 months of first-line treatment,
BCR-ABL1 on the IS greater than 1% after 12 months of first-line treatment, or
loss of MMR after 12 months of first-line treatment.
Patients intolerant to first-line TKI therapy were also eligible.
The study excluded patients previously treated with 2 or more ATP binding site TKIs or those who had used asciminib before.
Interventions
All eligible patients began treatment with asciminib 80 mg once a day. At 6 months, patients proceeded with 1 of the following:
continued the same dose, if BCR-ABL1 on the IS was less than or equal to 1%
increased to 200 mg once a day, if BCR-ABL1 on the IS was greater than 1%.
At 12 months, patients proceeded with 1 of the following:
continued the current dose, if MMR was achieved
increased to 200 mg once a day, if MMR was not achieved at 80 mg once a day
increased to 200 mg twice a day, if MMR was not achieved at 200 mg once a day
switched to investigator's drug of choice, if MMR was not achieved and it was in the patient's best interest.
Dose increases did not apply to those with grade 3 or grade 4 toxicity or persistent grade 2 toxicity. These patients remained on their current asciminib dose. For patients who were unable to tolerate the protocol-specified dosing schedule, dose interruptions and/or reductions were recommended or required to enable continuation of study treatment.
Study treatment was required to be discontinued if it was stopped before the planned duration, either by the patient or investigator, particularly if continuation may have harmed the patient's well-being. Discontinuation may have occurred due to AEs, patient ineligibility, noncompliance, protocol deviations, or use of prohibited treatments. Specific criteria requiring discontinuation included treatment failure (e.g., confirmed loss of MMR or BCR-ABL1 on the IS > 1% after 12 months), disease progression (e.g., progression to AP, blast crisis, or CML-related death), and pregnancy. In some cases, the detection of new mutations may also lead to discontinuation based on the investigator’s judgment.
Concomitant and Prohibited Medications
Concomitant medications and therapies deemed necessary for the supportive care and safety of the patient were allowed, provided their use was documented in the patient records and on the appropriate case report form, including the medication’s duration (start and end dates or if continuing at final exam). This included blood and platelet transfusions for patients with anemia and with thrombocytopenia. Specific data on these concomitant therapies were not available as of the most recent IA4 analysis.
Prohibited medications included other anticancer agents and strong CYP3A inducers or inhibitors. Concomitant administration of drugs with a known risk of torsades de pointes (a type of polymorphic ventricular tachycardia) should be prohibited with asciminib doses of 200 mg once or twice daily.
Outcomes
A list of efficacy end points assessed in this clinical review report is provided in Table 6, followed by descriptions of the outcome measures. Summarized end points are based on outcomes included in the sponsor’s summary of clinical evidence as well as any outcomes identified as important to this review according to the clinical experts consulted for this review and input from patient and clinician groups and public drug plans. Using the same considerations, the CDA-AMC review team selected end points that were considered to be most relevant to inform expert committee deliberations and finalized this list of end points in consultation with members of the expert committee. The end points considered most important to the expert committee deliberations (MMR, MR2, OS, notable harms of pancreatitis and ischemic heart disease) were assessed using GRADE. Other efficacy end points (MR4, MR4.5, time to and duration of MMR, HRQoL as assessed by the MDASI-CML) are summarized in the report as supportive information.
Table 6: Outcomes Summarized From the Study Included in the Systematic Review
Outcome measure | Time point | ASC2ESCALATE study |
|---|---|---|
Proportion of patients achieving MMRa | At 12 months (ongoing) | Primary |
Proportion of patients achieving MMR at visit | At 3 and 6 months (as of IA4)b | Secondary |
Proportion of patients achieving MR2 at visit | At 3 and 6 months (as of IA4)b | Secondary |
Proportion of patients achieving MR4 at visit | At 3 and 6 months (as of IA4)b | Secondary |
Proportion of patients achieving MR4.5 at visit | At 3 and 6 months (as of IA4)b | Secondary |
Duration of MMRa | 36 months (ongoing) | Secondary |
Time to MMRa | 36 months (ongoing) | Secondary |
OSa | 36 months (ongoing) | Secondary |
MDASI-CMLa | Through 36 months (ongoing) | Exploratory |
IA4 = interim analysis 4; MDASI-CML = MD Anderson Symptom Inventory for chronic myeloid leukemia; MMR = major molecular response; MR = molecular response; MR2 = 2.0 log reductions in BCR-ABL1 transcripts; MR4 = 4.0 log reductions in BCR-ABL1 transcripts; MR4.5 = 4.5 log reductions in BCR-ABL1 transcripts; OS = overall survival.
Note: Statistical testing did not adjust for multiple comparisons for any end point listed in this table.
aResults for this outcome was not available as of the IA4 data cut-off date of November 15, 2024. As such, a time point of 6 months and 9 months was used.
bThe results for this outcome were also reported at 9 months, despite it not being a predefined time point.
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (IA4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Molecular Outcomes
Major Molecular Response
The primary outcome of the ASC2ESCALATE study was the proportion of patients with MMR receiving second-line therapy at 12 months using the full analysis set (FAS). The MMR rate was defined as at least a 3.0 log reduction in BCR-ABL1 transcripts (BCR-ABL1 on the IS ≤ 0.1%). There were no data reported for the primary outcome as of the most recent interim analysis (IA4). Data as of IA4 included results for up to 9 months for the MMR rate.
Molecular Response
MR2.0, MR4.0, and MR4.5 rates are defined by 2.0, 4.0, and 4.5 log reductions in BCR-ABL1 transcripts (corresponding to BCR-ABL1 on the IS ≤ 1%, ≤ 0.01%, and ≤ 0.0032%, respectively). These responses are confirmed through duplicate real-time quantitative polymerase chain reaction (RQ-PCR) analysis. Data were available for up to 9 months for MR2.0, MR4.0, and MR4.5 rates as of IA4.
The rate of a molecular end point at a visit is the proportion of patients meeting the end point at that visit. The rate of a molecular end point by a visit includes the proportion of patients who met the end point at or before the visit. Only molecular end points at visits were available as of IA4.
Time to MMR and Duration of MMR
Time to MMR was defined as the time from treatment start to the first documentation of that specific end point. Duration of MMR was defined as the time from when it was first documented to the earliest of loss of the end point, disease progression to AP or BP, or CML-related death.
There were no data reported for time-to-event outcomes as of IA4.
Survival Outcomes
Overall Survival
OS was defined as the time from enrolment to death from any cause during the study. If a patient is alive, OS is censored at the latest known date of survival before the data cut-off date. All deaths are considered, even if they occur after interruptions or discontinuation of study treatment.
There were no data reported for survival outcomes as of the most recent interim analysis (IA4).
HRQoL Outcomes
MD Anderson Symptom Inventory for Chronic Myeloid Leukemia
The MDASI-CML is a 26-item self-administered questionnaire for adult patients with CML. Twenty of the items measure the severity of disease-related symptoms and are scored from 0 (not present) to 10 (as bad as you can imagine) and 6 items that measure symptom interference with daily life scored from 0 (did not interfere) to 10 (interfered completely), with higher scores indicating more severe symptoms and significant impact on daily life. The validity, reliability, and minimal important difference of the MDASI-CML are summarized in Table 7.
There were no data reported for HRQoL outcomes as of IA4.
Harms Outcomes
Safety and tolerability were assessed at each visit based on the incidence of AEs, serious AEs, deaths, AEs leading to discontinuation, AEs leading to dose reduction or interruption, and AEs of special interest. AEs were coded using the standard Medical Dictionary for Regulatory Activities and graded by the investigator according to the National Cancer Institute Common Toxicity Criteria for Adverse Events, Version 5.0. All SAEs were collected from the initiation of the study intervention until at least 30 days after discontinuing study treatment.
Table 7: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
MDASI-CML32 | A 26-item self-administered questionnaire suitable for adult patients with CML.32 | In a study32 using 3 cohorts including 35 adult patients with Philadelphia chromosome–positive CML to identify symptoms; 18 expert clinicians, patients, and caregivers to refine items; and 152 patients followed for 1 year to validate the questionnaire: Validity: Content validity supported by patient input and cognitive debriefing. Concurrent validity shown by moderate negative Spearman correlations (−0.269 to −0.436; P < 0.01). Known-group validity demonstrated by moderate to large effect size differences between TKIs. Construct validity supported by 3-factor model (residual SD = 0.045 < the reciprocal of the square root of the sample size of 0.081).32 Reliability: Internal consistency was good (alpha = 0.80 to 0.95). Test-retest reliability was high (intraclass correlation coefficient ≥ 0.89 over 2 weeks).32 Responsiveness: No evidence identified from the literature for this disease area. | No MID identified from the literature for this disease area. |
CML = chronic myeloid leukemia; MDASI-CML = MD Anderson Symptom Inventory for chronic myeloid leukemia; MID = minimal important difference; SD = standard deviation; TKI = tyrosine kinase inhibitor.
Statistical Analysis
Statistical analyses of the efficacy end points are summarized in Table 8.
Sample Size and Power Calculations
The null hypothesis of the primary end point assumed that no more than 30% of patients would achieve MMR at 12 months. Using a 1-sided alpha of 2.5% and 80% power, the required sample size was calculated. A total of 92 patients was estimated, accounting for an anticipated 10% dropout rate.
Statistical Testing
No statistical testing was performed for efficacy end points. For each end point, the point estimate and 95% CI (Clopper–Pearson method) were presented. Missing values were considered nonresponders.
For patients with dose escalation, the MMR rate and 95% CI at month 12 will be presented using the Clopper–Pearson method.
Table 8: Statistical Analysis of Efficacy End Points in ASC2ESCALATE Study
End point | Statistical model | Adjustment factors | Handling of missing data | Sensitivity analyses |
|---|---|---|---|---|
Proportion of patients with MMR at 12 monthsa |
| NA |
|
|
Proportion of patients with MMR, MR2, MR4, and MR4.5 at and by all scheduled data collection time pointsb |
| NA |
| NA |
Duration of and time to MMRa |
| NA | NA | NA |
OSa |
| NA | NA | NA |
MDASI-CMLa |
| NA | NA | NA |
CI = confidence interval; IA4 = interim analysis 4; MDASI-CML = MD Anderson Symptom Inventory for chronic myeloid leukemia; MMR = major molecular response; MR = molecular response; MR2 = 2.0 log reductions in BCR-ABL1 transcripts; MR4 = 4.0 log reductions in BCR-ABL1 transcripts; MR4.5 = 4.5 log reductions in BCR-ABL1 transcripts; NA = not available; OS = overall survival; PCR = polymerase chain reaction.
aData were not reported as of the IA4 data cut-off date of November 15, 2024.
bData were reported at 1-month, 3-month, 6-month, and 9-month time points as of the IA4 data cut-off date of November 15, 2024.The rate of a molecular end point at a visit is the proportion of patients meeting the end point at that visit. The rate of a molecular end point by a visit includes the proportion of patients who met the end point at or before the visit. Only molecular end points at visits were available as of IA4.
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (IA4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Subgroup Analyses
No subgroup analyses were defined in the study’s statistical analysis plan. However, IA4 data included MMR results by reason for discontinuation of first-line treatment (resistance or intolerance).
Sensitivity Analysis
A sensitivity analysis was planned to recalculate the primary end point without applying the imputation rule for patients with missing polymerase chain reaction evaluations at month 12.
Analysis Populations
The analysis populations of the ASC2ESCALATE study are provided in Table 9. The efficacy outcomes were analyzed based on the FAS population. The safety outcomes were analyzed using the safety analysis set, defined as patients who received at least 1 dose of study treatment.
Table 9: Analysis Populations of ASC2ESCALATE Study
Study | Population | Definition | Application |
|---|---|---|---|
ASC2ESCALATE | Full analysis set |
|
|
Safety set |
|
|
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Protocol Amendments and Deviations
The original study protocol for the ASC2ESCALATE study was amended twice up to the IA4 data cut-off date. The first amendment (March 2023) included the addition of a first-line cohort of patients with newly diagnosed CML in CP, inclusion of patients identified based on warning criteria to prior TKI, and clarification of dose modification guidelines for hepatoxicity. The second amendment (August 2024) included extending the study treatment duration from 24 months to 36 months for longer-term MR data, revising the assessment schedule to remove unnecessary visits, and adding the availability of asciminib 100 mg tablets.
Results for protocol deviations were not available as of IA4.
Results
Patient Disposition
Patient disposition for the ASC2ESCALATE study is summarized in Table 10. At the time of IA4 (data cut-off date of November 15, 2024), 101 patients were assigned treatment, all of whom were treated. As of IA4, 9 patients (8.9%) discontinued treatment, of which 6 (5.9%) discontinued less than 6 months after initiating treatment. The primary reasons for discontinuation included AEs in 4 patients (4.0%), and patient decision in 3 patients (3.0%).
As of IA4, █ ██████ patients entered the safety follow-up period, with ███████ completing it, and || patients ██████ discontinuing the safety follow-up, citing loss to follow-up by ██████ patient and ███████ due to patient decision.
Table 10: Summary of Patient Disposition From the ASC2ESCALATE Study
Patient disposition | ASC2ESCALATE study |
|---|---|
Asciminib N = 101 | |
Assigned treatment, N | 101 |
Treated, n (%) | 101 (100) |
Patient status, n (%) | |
Treatment ongoing | 92 (91.1) |
Completed treatment | 0 |
Discontinued from treatment, n (%) | 9 (8.9) |
< Month 6 | ██████ |
≥ Month 6 and < month 12 | ███████ |
≥ Month 12 | || |
Reason for discontinuation, n (%) | |
Adverse eventa | 4 (4.0) |
Lost to follow-up | 1 (1.0) |
Patient decision | 3 (3.0) |
Physician decision | 1 (1.0) |
Safety, n (%) | |
Entered safety follow-up | ██████ |
Completed safety follow-up | ██████ |
Discontinued safety follow-up | ██████ |
Reason for safety discontinuation, n (%) | |
Lost to follow-up | ██████ |
Patient decision | ██████ |
FAS, N | 101 |
SAS, N | 101 |
FAS = full analysis set; SAS = safety analysis set.
Note: The data cut-off date for this 6-month efficacy analysis was November 15, 2024.
aOne of these patients had adverse events occur > 30 days after their last asciminib dose.
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Baseline Characteristics
The baseline characteristics outlined in Table 11 are limited to those that are most relevant to this review or were felt to affect the outcomes or interpretation of the study results.
The mean age of patients in the ASC2ESCALATE study was ████ years (range, 18 years to 89 years) and most patients (████%) were younger than 65 years. There were more male patients (56.4%) than female patients (43.6%) enrolled. A total of 1% of patients identified as American Indian or Alaska Native [wording from original source], 4% as Asian, 8.9% as Black or African American, 82.2% as white, and 4% were unknown. Most patients had an ECOG Performance Status score of 0 or 1 (████%), and ███% had a score of 2. The mean time since CML diagnosis was █████ weeks (SD = ██████). Baseline mutations included E450Q/M244V, E459G, and V299L (1% each). All patients had received prior TKI therapy, consisting primarily of dasatinib (44.6%) and imatinib (42.6%). The primary reasons for discontinuation of prior TKI therapy were lack of efficacy (56.4%) and lack of tolerability (43.6%), with most patients (66.3%) having had more than 12 months of prior TKI therapy.
Table 11: Summary of Baseline Characteristics From the ASC2ESCALATE Study
Characteristic | ASC2ESCALATE study |
|---|---|
Asciminib N = 101 | |
Age (years), n (%) | |
Mean (SD) | ████ ███████ |
Median (range) | 50.0 (18 to 89) |
Age group, n (%) | |
< 65 years | ██ ██████ |
≥ 65 years | ██ ██████ |
Sex, n (%) | |
Female | 44 (43.6) |
Male | 57 (56.4) |
Race, n (%) | |
American Indian or Alaska Native | 1 (1.0) |
Asian | 4 (4.0) |
Black or African American | 9 (8.9) |
White | 83 (82.2) |
Unknown | 4 (4.0) |
Ethnicity, n (%) | |
Hispanic or Latino | 12 (11.9) |
Not Hispanic or Latino | 88 (87.1) |
Unknown | 1 (1.0) |
BMI (kg/m2) | |
Mean (SD) | ████ ██████ |
ECOG PS score, n (%) | |
0 | ██ ██████ |
1 | ██ ██████ |
2 | ██████ |
Time since diagnosis of CML (weeks) | |
Mean (SD) | █████ ████████ |
Median (range) | ████ ███ ████ |
Baseline mutations, n (%)a | |
E450Q/M244V | 1 (1.0) |
E459G | 1 (1.0) |
V299L | 1 (1.0) |
No amplification | ██████ |
Not evaluable | ██████ |
Evidence of extramedullary involvement, n (%) | |
No | ██ ██████ |
Yes | ██████ |
Location of extramedullary involvement, n (%) | |
Spleen | ██████ |
Prior TKI therapy, n (%) | |
Bosutinib | 5 (5.0) |
Dasatinib | 45 (44.6) |
Imatinib | 43 (42.6) |
Nilotinib | 10 (9.9) |
Duration of prior TKI (months), n (%) | |
< 6 months | 18 (17.8) |
≥ 6 months to < 12 months | 16 (15.8) |
≥ 12 months | 67 (66.3) |
Reason for discontinuing prior TKI, n (%) | |
Lack of efficacy | ██ ██████ |
Lack of tolerability | ██ ██████ |
BMI = body mass index; CML = chronic myeloid leukemia; ECOG = Eastern Cooperative Oncology Group; PS = Performance Status; SD = standard deviation; TKI = tyrosine kinase inhibitor.
Notes: The data cut-off date for this 6-month efficacy analysis was November 15, 2024.
Racial categories used in the table are as reported in the source and may not align with Canada's Drug Agency inclusive language guidelines.
aMutational analysis was performed for all patients by Sanger sequencing at screening. Patients with nonevaluable or no amplification mutational analysis were confirmed not to have T315I mutation.
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Exposure to Study Treatments
As shown in Table 12, at IA4, the mean duration of exposure was ████ weeks (SD = █████), with a median duration of 26.1 weeks (range, 6 weeks to 100 weeks). Regarding duration of exposure, 42.6% of patients were exposed for less than 24 weeks, while 57.4% were exposed for 24 weeks or more. The average daily dose was ████ mg (SD = █████), with a median dose of 80.0 mg (range, 41 mg to 140 mg).
████████████ (████%) and ██ ███████ patients had at least 1 dose interruption or dose reduction, respectively. The mean duration of dose interruption was ████ days (SD = █████). Seven patients (6.9%) had their dose increased from 80 mg once daily to 200 mg once daily. Of these, 3 patients had dose escalation based on their response at week 24, and 4 patients based on their response at week 48.
Table 12: Summary of Patient Exposure From the ASC2ESCALATE Study (Safety Set)
Exposure | ASC2ESCALATE study |
|---|---|
Asciminib N = 101 | |
Duration of exposure | |
Duration of exposure (weeks) | |
Mean (SD) | ████ ███████ |
Median (range) | 26.1 (6 to 100) |
Duration of exposure categories, n (%) | |
< 24 weeks | 43 (42.6) |
≥ 24 weeks | 58 (57.4) |
≥ 48 weeks | 17 (16.8) |
≥ 96 weeks | 1 (1.0) |
Dose intensity | |
Average daily dose (mg) | |
Mean (SD) | ████ ███████ |
Median (range) | ████ ███ ██ ████ |
Dose intensity (mg/day) | |
Mean (SD) | ████ ███████ |
Median (range) | 80 (30 to 140) |
Dose adjustments | |
Dose reduction | |
Patients with ≥ 1 dose reduction, n (%) | ██ ██████ |
Adverse event | ██ ██████ |
Dosing error | ██████ |
Dose interruption | |
Patients with ≥ 1 dose interruption, n (%) | ██ ██████ |
Adverse event | ██ ██████ |
Dosing error | ██████ |
Physician decision | ██████ |
Patient decision | ██████ |
Duration of dose interruption (days), mean (SD) | ████ ███████ |
Dose escalation, n (%) | |
Number of patients with dose escalation | 7 (6.9) |
80 mg q.d. to 200 mg q.d. at week 24 | 3 (3.0) |
80 mg q.d. to 200 mg q.d. at week 48 | 4 (4.0) |
q.d. = once a day; SD = standard deviation.
Note: The data cut-off date for this 6-month efficacy analysis was November 15, 2024.
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Subsequent Treatment
Subsequent treatments have not yet been detailed for the study population in the ASC2ESCALATE study.
Efficacy
Efficacy outcomes assessed at IA4 (data cut-off date of November 15, 2024) in the second-line cohort of the ASC2ESCATE study for the FAS are displayed in Table 13.
Major Molecular Response
The proportion of patients with MMR was measured at 1, 3, 6, and 9 months in 101 patients with adequate follow-up. At 1 month, 1.1% of patients (1 of 94; 95% CI, 0.0% to 5.8%) had an MMR. At 3 months, the MMR rate was 39.5% (34 of 86; 95% CI, 29.2% to 50.7%). By 6 months, 44.4% of patients (28 of 63; 95% CI, 31.9% to 57.5%) had an MMR and at 9 months, █████ ███████████ ███ █████ ██ ██████ ███ █ ███.
MMR by Reason for Discontinuing Prior TKI
Subgroup analysis for the proportion of patients with MMR was conducted based on the reason patients discontinued their previous TKI (resistance or intolerance). Among patients who discontinued their prior TKI due to lack of efficacy, the proportion of patients with MMR at 1 month was ████ ██████ ███ ███ ████ ██ ███████ at 3 months was █████ ███████ ███ ███ █████ ██ ██████, at 6 months was █████ ███████ ███ ███ █████ ██ ███████ and at 9 months was █████ ███████ ███ ███ █████ ██ ███████.
For patients who stopped their prior TKI due to intolerance, MMR was not observed in any of the ██ patients ██████ ███ ███ ████ ██ █████ at 1 month. At months 3, 6, and 9, the proportion of patients with MMR was █████ ███████ ███ ███ █████ ██ ████████████ ███████ ███ ███ █████ ██ ███████ ███ █████ ██████ ███ ███ █████ ██ ███████ respectively.
MR2 Response
The proportion of patients with MR2 was █████ (██████ ███ ███ █████ ██ ██████ at 1 month, █████ (██████ ███ ███ █████ ██ ██████ at 3 months, █████ (██████ ███ ███ █████ ██ ██████ at 6 months, and █████ ███████ ███ ███ █████ ██ ██████ at 9 months.
MR4 Response
The proportion of patients with MR4 was 0% (0 of 94; 95% CI, 0.0% to 3.9%) at 1 month, 11.6% (10 of 86; 95% CI, 5.7% to 20.4%) at 3 months, 25.4% (16 of 63; 95% CI, 15.3% to 37.9%) at 6 months, and █████ ██████; ███ ████████ ██ ██████ at 9 months.
MR4.5 Response
The proportion of patients with MR4.5 was 0% (0 of 94; 95% CI, 0.0% to 3.9%) at 1 month, 2.3% (2 of 86; 95% CI, 0.3% to 8.2%) at 3 months, 9.5% (6 of 63; 95% CI, 3.6% to 19.6%) at 6 months, and ████ █████████ ███ ████ ██ ██████ at 9 months.
Duration of MMR
Results for the duration of MMR were not available as of IA4.
Time to MMR
Results for time to MMR were not available as of IA4.
Overall Survival
Results for OS were not available as of IA4.
MD Anderson Symptom Inventory for Chronic Myeloid Leukemia
Results for MDASI-CML were not available as of IA4.
Table 13: Summary of Key Efficacy Results From ASC2ESCALATE Study (FAS)
Variable | Asciminib N = 101 | |
|---|---|---|
n of N (%)a | 95% CI | |
MMR | ||
At 1 month | 1 of 94 (1.1) | 0.0 to 5.8 |
At 3 months | 34 of 86 (39.5) | 29.2 to 50.7 |
At 6 months | 28 of 63 (44.4) | 31.9 to 57.5 |
At 9 months | █████ ██████ | █████ ██ █████ |
MRR by reason for discontinuing prior TKI | ||
Lack of efficacy | ||
At 1 month | ████ █████ | ████ ██ █████ |
At 3 months | █████ ██████ | █████ ██ █████ |
At 6 months | █████ ██████ | █████ ██ █████ |
At 9 months | █████ ██████ | █████ ██ █████ |
Lack of tolerability | ||
At 1 month | ████ █████ | ████ ██ ████ |
At 3 months | █████ ██████ | █████ ██ █████ |
At 6 months | █████ ██████ | █████ ██ █████ |
At 9 months | ████ ██████ | █████ ██ █████ |
MR2 | ||
At 1 month | 44 of 94 (46.8) | █████ ██ █████ |
At 3 months | 73 of 86 (84.9) | █████ ██ █████ |
At 6 months | 52 of 63 (82.5) | █████ ██ █████ |
At 9 months | █████ ██████ | █████ ██ █████ |
MR4 | ||
At 1 month | 0 of 94 (0.0) | 0.0 to 3.9 |
At 3 months | 10 of 86 (11.6) | 5.7 to 20.4 |
At 6 months | 16 of 63 (25.4) | 15.3 to 37.9 |
At 9 months | █████ ██████ | █████ ██ █████ |
MR4.5 | ||
At 1 month | 0 of 94 (0.0) | 0.0 to 3.9 |
At 3 months | 2 of 86 (2.3) | 0.3 to 8.2 |
At 6 months | 6 of 63 (9.5) | 3.6 to 19.6 |
At 9 months | ████ █████ | ████ ██ █████ |
CI = confidence interval; FAS = full analysis set; MMR = major molecular response; MR = molecular response; MR2 = 2.0 log reductions in BCR-ABL1 transcripts; MR4 = 4.0 log reductions in BCR-ABL1 transcripts; MR4.5 = 4.5 log reductions in BCR-ABL1 transcripts; RQ-PCR = real-time quantitative polymerase chain reaction; TKI = tyrosine kinase inhibitor.
Note: The data cut-off date for this 6-month efficacy analysis was November 15, 2024. Data are based on the number of patients with adequate follow-up (patients who have assessments within corresponding analysis time interval and patients who discontinue earlier).
an = number of patients who achieved outcome at scheduled visits; N = number of patients with adequate follow-up (including participants who had RQ-PCR assessments within corresponding analysis time intervals or participants who discontinued earlier).
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Harms
Harms data reported in this section are from the data cut-off date of November 15, 2024. A summary of harms reported in the safety population in the ASC2ESCALATE study is provided in Table 14.
Adverse Events
At the time of IA4, most patients (95.0%) experienced at least 1 AE. Headaches and nausea were the most frequently reported AEs experienced by 22.8% and 20.8% of patients, respectively. Cough, diarrhea, fatigue, and hypertension were each reported as AEs by 16 patients (15.8%). Nearly one-third (31.7%) of patients experienced any grade 3 or higher AEs, the most frequent of which was hypertension (8.9%).
Serious AEs
█████ ████████ ██████ ███████████ ████ ███ ██ ███████ ██████████ ████████████████ ██████████ ███████ ██████████ ███ ██████████████ ████ ███████████.
Withdrawals Due to AEs
Three patients (3.0%) discontinued treatment due to AEs, with 1 patient reporting nausea and vomiting of grade 3 or higher.
Mortality
There were no deaths reported as of IA4.
Notable Harms
AEs of special interest in the ASC2ESCALATE study included ████████████████ ████████ ███████████ ██ █████ ██ █████████ ████████ ██ ████████████████ ████████ █████ ████████████ █████████ ████████ ██████████ ██████ ██████████ ████████ ███ ████████████████ ████████.
Table 14: Summary of Harms Results From ASC2ESCALATE Study (Safety Set)
AEs | ASC2ESCALATE study |
|---|---|
Asciminib n = 101 | |
Most common AEs, occurring in ≥ 10% of patients, n (%) | |
Patients with ≥ 1 AE | 96 (95.0) |
Headache | 23 (22.8) |
Nausea | 21 (20.8) |
Cough | 16 (15.8) |
Diarrhea | 16 (15.8) |
Fatigue | 16 (15.8) |
Hypertension | 16 (15.8) |
Lipase increased | 14 (13.9) |
Arthralgia | 13 (12.9) |
Myalgia | 13 (12.9) |
Vomiting | 13 (12.9) |
Abdominal pain | 11 (10.9) |
SAEs, n (%) | |
Patients with ≥ 1 SAE | ██████ |
Cardiac disorders | ██████ |
Gastrointestinal disorders | ██████ |
General disorders and administration site conditions | ██████ |
Deaths, n (%) | |
Patients who died | ██████ |
AEs of special interest, n (%) | |
Gastrointestinal toxicity | ██ ██████ |
Hypersensitivity | ██ ██████ |
Acute pancreatitis (including isolated pancreatic enzyme elevations) | ██ ██████ |
Acute pancreatitis (clinical) | ██████ |
Myelosuppressiona | ██ ██████ |
Hepatotoxicity (including laboratory terms) | ██████ |
Hemorrhage | ██████ |
Edema and fluid retention | ██████ |
Phototoxicity | ██████ |
Ischemic heart conditions | ██████ |
AE = adverse event; SAE = serious adverse event.
Note: The data cut-off date for this 6-month efficacy analysis was November 15, 2024. Data are based on the number of patients with adequate follow-up (patients who have assessments within corresponding analysis time interval and patients who discontinue earlier).
aMyelosuppression includes erythropenia, leukopenia, thrombocytopenia, cytopenias affecting ≥ 1 lineage.
Sources: The ASC2ESCALATE study’s 6-month efficacy key outputs for the second-line cohort: final (interim analysis 4) and sponsor’s summary of clinical evidence.9,31 Details included in the table are from the sponsor’s summary of clinical evidence.
Critical Appraisal
Internal Validity
The ASC2ESCALATE study is an ongoing, phase II, multicentre, open-label, single-arm, dose-escalation study of asciminib in the second-line setting. Although inclusion and exclusion criteria of the study were stated, selection procedures, such as patients who were no longer eligible for the trial after screening, were not described.
There were notable issues with the study design of the ASC2ESCALATE study, specifically related to the single-arm and open-label nature, as well as the dose-escalation design. Considering the nature of Ph+ CML in the CP, which does not spontaneously reverse, the single-arm, noncomparative design was not deemed inappropriate; however, the lack of comparative evidence versus other second-line treatments (e.g., bosutinib, dasatinib, nilotinib, and imatinib) remains a concern and precludes the ability to assess the comparative effectiveness and safety relative to other available TKIs in Canadian clinical practice. The ASC2ESCALATE study was also open label, whereby the investigator and the study participants were aware of their treatment status, potentially increasing the risk of detection bias and performance bias. As such, the assessment of subjective end points such as AEs could be biased as the knowledge of the intervention can impact patient expectations and perceptions about the harms of treatment. However, the reported AEs were consistent with the known and documented profile for asciminib. Therefore, it is unlikely that the results for harms were biased by the open-label design. MMR and other MR outcomes are objective and are therefore less likely to be subject to such bias. Lastly, the ASC2ESCALATE study used a dose-escalation design primarily to evaluate the safety and tolerability of increasing doses of asciminib in patients with inadequate response to the standard dose. This type of design is not usually intended to establish the overall effectiveness or comparative harms of the treatment but rather to identify whether higher doses can be administered safely, and was intended to achieve deeper responses with dose escalation. However, the escalated dose of 200 mg is not within the dosage range approved by Health Canada for asciminib. Furthermore, only 7 patients received an escalated dose of asciminib as of IA4.
According to the FDA, MMR is a surrogate end point used for traditional approval for therapies in CML.11 According to ELN (2020) and National Comprehensive Cancer Network (2024) guidelines, with consistent medication adherence, sustained MMR (and DMR) are strong indicators of long-term outcomes including long-term cytogenetic remission and a reduced rate of disease progression.5,12 However, the sustainability of MMR observed in patients treated with asciminib in the ASC2ESCALATE study could not be assessed from the interim analysis.
Formal statistical tests for efficacy outcomes were not conducted for the ASC2ESCALATE study, and as such, no P values were calculated. The threshold for a positive study outcome was observing a 95% CI for MMR rate at 12 months with a lower limit larger than 30% to reject the null hypothesis. As of the most recent interim analysis (IA4), descriptive results were provided for molecular outcomes (MMR, MR4.5, MR4, and MR2) through to 9 months and harms outcomes (AEs and SAEs) at 6 months. At 6-month and 9-month time points, the proportion of patients with MMR was suggestive of benefit according to the hypothesis. However, a limited number of patients were included in the primary efficacy population (N = 101), and only 63 and ██ patients had MR data available at 6 months and 9 months, respectively, which limits the generalizability of the findings. The rate of discontinuation from treatment through to 9 months was relatively low (9%), and nearly all patients remained ongoing in the trial. In the assessment of MR rates, patients with missing data were categorized as not having achieved a response which is a conservative assumption in a single-arm design. Therefore, the estimated rates for MR by different thresholds are likely valid and representative of the study population.
The evidence provided for the ASC2ESCALATE study at IA4 was considered immature and there were no data available for OS, time to event (time to or duration of MMR), and HRQoL (MDASI-CML). Thus, the clinical benefit of the treatment, whether in terms of survival, disease control, or patient quality of life could not be established in this patient population based on the evidence submitted. Improvements in quality of life were considered important to patients, clinician groups, and clinical experts consulted for this review, and as such, the lack of such results as of the most recent interim analysis is a notable limitation.
External Validity
The ASC2ESCALTE trial was conducted exclusively in the US; however, the clinical experts consulted by CDA-AMC noted that the study population was generally consistent with the population expected to receive treatment in Canada. The clinical experts noted that some eligibility criteria such as ECOG Performance Status score may have been restrictive, selecting for ideal patients with less severe CML (98% had an ECOG Performance Status score ≤ 1), which may not reflect the general patient population but is typical of clinical trials. The clinical experts also stated that patients with an ECOG Performance Status score of 2 or higher, as well as those with CML in AP or BC — who were excluded from the ASC2ESCALATE trial — could receive asciminib if it was deemed the best treatment option. The clinical experts also highlighted that there was an overrepresentation of white patients (82.2%) in the ASC2ESCALATE study, which is not representative of what would be seen in the population of patients living in Canada.
The absence of data beyond 9 months limits the ability to assess the durability of treatment responses and long-term safety. Although asciminib is available in the third-line setting, this absence of data is particularly important for this therapeutic class, because AEs may emerge only after prolonged use, with cumulative incidence increasing over time. One clinical expert highlighted the example of nilotinib, where cardiovascular risks became apparent only after a decade on the market.
GRADE Summary of Findings and Certainty of the Evidence
Methods for Assessing the Certainty of the Evidence
For the pivotal study identified in the sponsor’s systematic review, GRADE was used to assess the certainty of the evidence for outcomes considered most relevant to inform expert committee deliberations, and a final certainty rating was determined as outlined by the GRADE Working Group.13,14
High certainty: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: We are moderately confident in the effect estimate — the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. We use the word “likely” for evidence of moderate certainty (e.g., “X intervention likely results in Y outcome”).
Low certainty: Our confidence in the effect estimate is limited — the true effect may be substantially different from the estimate of the effect. We use the word “may” for evidence of low certainty (e.g., “X intervention may result in Y outcome”).
Very low certainty: We have very little confidence in the effect estimate — the true effect is likely to be substantially different from the estimate of effect. We describe evidence of very low certainty as “very uncertain.”
Although GRADE guidance is not available for noncomparative studies, the CDA-AMC review team assessed the pivotal single-arm study for study limitations (which refers to internal validity or risk of bias), inconsistency across studies, indirectness, imprecision of effects, and publication bias to present these important considerations. Because the lack of a comparator arm does not allow for a conclusion to be drawn on the effect of the intervention versus any comparator, the certainty of evidence for single-arm studies started at very low certainty.
When possible, certainty was rated in the context of the presence of an important (nontrivial) treatment effect; if this was not possible, certainty was rated in the context of the presence of any treatment effect (i.e., the clinical importance is unclear). In all cases, the target of the certainty of evidence assessment was based on the point estimate and where it was located relative to the threshold for a clinically important effect (when a threshold was available) or to the null. For this review, the target of the certainty of evidence assessment was based on the presence or absence of any (non-null) effect for each outcome measure.
Results of GRADE Assessments
The GRADE assessments included an evaluation of the main outcomes considered important by clinicians, patient groups, and committee members. Table 2 presents the GRADE summary of findings for asciminib for the treatment of patients with CML in CP in the second-line setting.
Long-Term Extension Studies
No long-term extension studies were submitted for the review of asciminib in the second-line setting.
Indirect Evidence
No ITCs were submitted for the review of asciminib in the second-line setting.
Studies Addressing Gaps in the Systematic Review Evidence
Description of Studies
One retrospective, noninterventional, physician panel-based medical chart review by Atallah et al.15 was submitted by the sponsor to provide additional real-world insights of clinical outcomes and tolerability of asciminib. The chart review was conducted in the US in adult patients living with CML in CP (N = 255). Eligible patients did not have a T315I mutation and were treated with asciminib after prior treatment with 1 TKI.
Outcomes
Outcomes evaluated in the study included time to treatment discontinuation, time to and proportion of patients achieving MRs (MR2, MMR, MR4.5), and harms. Time to treatment discontinuation was defined as the time from asciminib initiation to treatment discontinuation, as defined by the treating physician (event), or the end of the observation period (censoring). Time to MR2, MMR, and MR4.5 was defined as the time from asciminib initiation to first response assessment documenting the response level of interest (event), the earliest of treatment discontinuation, or the end of the observation period (censoring).
Statistical Analysis
The study was descriptive, and no formal hypothesis testing was conducted. The proportion of patients remaining on treatment and the proportion of patients achieving or maintaining MRs were analyzed using Kaplan-Meier methods, with 95% CIs at 48 weeks after asciminib initiation. MR outcomes were evaluated in patients who underwent MR testing at least every 3 months. The time to treatment discontinuation was defined as the duration from asciminib initiation to discontinuation, as determined by the treating physician, or censored at the end of the observation period (defined as the earliest of hematopoietic stem cell transplant, progression to AP or BP, loss to follow-up, date of electronic case report form completion, or death from any cause).
Subgroup Analysis
Subgroup analyses were conducted based on the reason for first TKI discontinuation (treatment failure or intolerance, as assessed by the physician) and type of first TKI received (first generation [imatinib] or second generation [dasatinib, nilotinib, or bosutinib]). Suggested definitions for treatment failure were aligned with National Comprehensive Cancer Network guidelines (BCR-ABL1 > 10% at 6 months or > 1% at 12 months).
Results
Patient Disposition
Among the 255 included patients, the mean age was 60.5 years (SD = 9.5 years), with 43.5% identifying as female and 56.5% as male. A total of 20.8% identified as Black or African American, 16.5% as Hispanic or Latino, 56.1% as white, and 6.7% as other. At diagnosis, 22% had low-risk, 57.6% intermediate-risk, and 18.4% high-risk Sokal scores. A total of ████%, ████%, and 17.3% had an ECOG Performance Status score of 0, 1, or at least 2, respectively. Imatinib was the initial TKI treatment for nearly half (49.8%) of patients, with the remainder receiving second-generation TKIs: dasatinib (34.5%), nilotinib (10.6%), or bosutinib (5.1%). The mean duration of initial TKI treatment was ████ months (SD = ████ months). Fewer than ████ ███████ had MMR or better during their first-line treatment, and ████% maintained this response at the end of their first-line treatment. A total of 43.5% of patients discontinued their first TKI due to intolerance, while 23.5% discontinued due to treatment failure. After starting asciminib, the median follow-up was 73.6 weeks, during which no patients progressed to AP or BP and 1 underwent hematopoietic stem cell transplant.
Efficacy
Patients Remaining on Treatment
Based on Kaplan-Meier analysis, the estimated probability of patients remaining on asciminib treatment at 48 weeks was 95.0% (95% CI, 91.3% to 97.1%).
The probability of remaining on treatment with asciminib at 48 weeks among those who discontinued their first TKI due to intolerance (n = 111) or treatment failure (n = 60) was 97.2% (95% CI, █████ ██ ████%) and 92.2% (95% CI, █████ ██ ████%), respectively. By initial TKI type, the probability of remaining on treatment with asciminib at 48 weeks was 93.4% (95% CI, █████ ██ ████%) in patients previously on a first-generation TKI (n = 127) and 96.4% (95% CI, █████ ██ ████%) in those previously on a second-generation TKI (n = 128).
Molecular Outcomes
Major Molecular Response
At week 48, MMR was achieved or maintained by 68.3% of patients (95% CI, 61.8% to 74.5%). The median time to MMR was 30.7 weeks.
At week 48, the proportion of patients who achieved or maintained MMR was 68.4% (95% CI, 58.9% to 77.5%) in patients with intolerance to prior TKI therapy, and 55.8% (95% CI, 42.9% to 69.7%) in patients with prior treatment failure. A total of 73.6% (95% CI, 65.4% to 81.3%) of patients initially treated with a second-generation TKI, and 61.5% (95% CI, 51.7% to 71.5%) with a first-generation TKI, had achieved or maintained an MMR.
MR2
At week 48, 84.0% of patients (95% CI, █████ ██ ████%) achieved or maintained MR2, and the median time to MR2 was ████ weeks.
At week 48, the proportion of patients who achieved or maintained MR2 was 90.7% (95% CI, █████ ██ ████%) among those who discontinued their first TKI due to treatment failure, and 79.5%; (95% CI, █████ ██ ████%) in those who discontinued due to intolerance. A total of 86.7% (95% CI, █████ ██ ████%) of patients initially treated with a second-generation TKI, and 80.4% (95% CI, █████ ██ ████%) of patients who received a first-generation TKI, had achieved or maintained MR2.
MR4
At week 48, MR4 was achieved or maintained in 40.6% of patients (95% CI, █████ ██ ████%).
At week 48, the proportion of patients who achieved or maintained MR4 was 44.9% (95% CI, █████ ██ ████%) in patients with prior TKI intolerance and 15.9% (95% CI, ████ ██ ████%) with prior TKI failure. MR4 was achieved or maintained by 43.2% (95% CI, █████ ██ ████%) of patients who received a second-generation TKI as first-line therapy, and 37.3% (95% CI, █████ ██ ████%) of patients who received a first-generation TKI as first-line therapy.
Harms
While on treatment with asciminib, the most commonly reported AEs were fatigue (8.6%), headache (7.1%), rash (4.3%), abdominal pain (2.4%), and cytopenia (2.4%). Gastrointestinal AEs included nausea (11.8%), vomiting (6.7%), and diarrhea (4.7%). No cases of stroke, myocardial infarction, or heart failure were reported.
Critical Appraisal
The sponsor submitted data on a retrospective chart review study of the efficacy and safety of asciminib in the real-world setting. However, the absence of study protocols and statistical analysis plans limited the ability to fully assess the study design, outcome measurements, and analytical methods.
Chart reviews rely on the accuracy and completeness of clinical documentation. The evaluation of real-world MR may have been based on heterogeneous criteria and inconsistent assessment schedules. As such, dates of diagnosis, progression, or death may have been inconsistently recorded, potentially leading to inaccurate results.
In the overall cohort, ██ █████ patients had either experienced the event of interest or were censored by the 48-week time point. As censoring could occur for a range of reasons such as loss to follow-up or progression to AP or BP, the potential for biased response estimates cannot be excluded, particularly as the specific reasons and timing for censoring were not reported.
Although the sponsor defined treatment failure based on existing clinical guidelines, physicians may have applied their own criteria when identifying resistance to first-line TKI therapy. Differences in clinical decision-making and patient management across institutions could have confounded results. AEs were recorded during treatment with asciminib, but because physicians may only document AEs that lead to treatment changes (e.g., dose adjustments), underreporting is likely.
The retrospective chart review was conducted exclusively in the US with patients selected by physicians participating in a panel which may limit the generalizability of the findings to Canadian clinical practice. Clinical experts noted that in Canadian clinical practice, in the first-line setting, patients are more likely to receive dasatinib and less likely to receive nilotinib or bosutinib compared to the patients included in the chart review. Despite these differences in treatment patterns, the results suggested that asciminib was associated with consistent benefit in MR across all subgroups, regardless of whether patients initially received a first-generation or second-generation TKI.
Discussion
Summary of Available Evidence
The evidence included in this review consisted of 1 study that was included in the systematic review (the ASC2ESCALATE study), and 1 study addressing gaps. No long-term extensions or ITCs were submitted.
The ASC2ESCALATE study (N = 101) is an ongoing, phase II, single-arm, open-label, dose-escalation study of asciminib in patients with CML in CP who have received 1 prior TKI. The primary objective of the ASC2ESCALATE study was to determine efficacy, safety, and tolerability of asciminib (starting dose of 80 mg once daily). Patients with the T315I mutation or previous treatment with at least 2 ATP-competitive TKIs were excluded from the study. The primary end point of the ASC2ESCALATE trial was MMR at 12 months, with secondary end points consisting of the proportion of patients with MR2, MR4, and MR4.5; duration and time to MMR; and OS. Exploratory end points included HRQoL. Results for this report were based on IA4 (data cut-off date of November 15, 2024), for which data were only available for 9 months of follow-up for MR end points. At the time of IA4, there were no results available for duration of MMR, time to MMR, OS, or HRQoL. The average age of patients in the ASC2ESCALATE study was 51.2 years with ████% of patients having prior treatment with dasatinib or imatinib; and the primary reasons for discontinuation of therapy were lack of efficacy (56.4%) and lack of tolerability (43.6%).
One retrospective chart review study (Atallah et al.15) conducted in the US in adult patients living with CML in CP (N = 255) was submitted by the sponsor to provide additional real-world evidence on the efficacy and safety of asciminib in the second line. The median age was 62 years, ████% of patients had prior treatment with dasatinib or imatinib. Most patients had an ECOG Performance Status score of 0 at baseline (████%). Eligible patients did not have a T315I mutation and were treated with asciminib after treatment with 1 prior TKI.
Interpretation of Results
Efficacy
Patients with Ph+ CML in CP emphasized the heavy physical and emotional impact of the disease, including stress, fatigue, pain, and sleep issues that disrupt daily life, work, and social interactions. Patients noted difficulties with finding an effective treatment with tolerable side effects and noted the desire to be able to stop treatment while staying in remission, particularly because treatment is often lifelong. Key treatment outcomes valued by patients included effective disease control with minimal adverse effects and improved HRQoL. Similarly, clinician input from both the clinician group and clinical experts consulted by CDA-AMC emphasized the need for treatments that maintain or improve the patient’s quality of life, minimize long-term toxicities, and increase the chances of safely discontinuing TKI therapy, leading to TFR.
The clinical experts consulted for this review noted that molecular outcomes are important outcomes of treatment in clinical practice, and the FDA supports the use of molecular outcomes as a surrogate end point for traditional approval for therapies for CML.11 The primary end point of the ASC2ESCALATE trial was the proportion of patients achieving MMR at 12 months; however, results at 12 months were not available at the time of this review. As such, the efficacy of asciminib in the second-line treatment of patients with Ph+ CML in CP was assessed at 9 months. The results suggested improvements in MMR rates among patients who received asciminib, with ██% of patients achieving MMR at 9 months. Additionally, asciminib was associated with improvements in other MR rates at various BCR-ABL1 levels (i.e., MR2, MR4, and MR4.5). According to the experts, MR2 is essentially equivalent to complete cytogenetic remission in which no detectable Ph+ cells are present in the bone marrow sample, indicating a strong response to therapy. However, the findings of the ASC2ESCALATE study must be interpreted in consideration of the single-arm design which typically cannot support causal conclusions about the effect of a drug versus any comparator. In the evaluation of asciminib, the certainty of evidence for the MMR and MR2 results in the ASC2ESCALATE study was considered low, as opposed to “very low,” which is the typical rating for certainty of evidence in single-arm studies. This is because CML in CP is unlikely to spontaneously regress, and reductions in BCR-ABL1 transcript levels are not expected in the absence of effective therapy. Thus, any substantial decline in fusion gene expression can reasonably be attributed to the intervention, suggesting a biologically plausible and highly specific treatment effect. However, as noted, these results are derived from descriptive data from an interim analysis of a single-arm study with a small sample size of only ██ patients contributing data at 9 months, significantly limiting the ability to interpret the results. As such, results should only be viewed as supportive of the overall effect of asciminib. Results of the retrospective chart review in a similar population were consistent with the ASC2ESCALATE study findings for MR rates over 48 weeks; however, the methodological limitations of this study must be considered.
The ELN (2020) guidelines suggest that attaining MMR is associated with nearly 100% CML-specific survival, given that disease progression becomes rare once this level of cytoreduction is reached.5 The clinical experts agreed that an MMR does not confer a significant OS advantage compared to patients who reach MR2. The clinical experts noted that the early molecular results in the ASC2ESCALATE study are consistent with what they would expect to see for a second-generation TKI used in the second-line setting; however, it is uncertain if the MMR and MR2 results derived at 9 months in the ASC2ESCALATE study will be sustained over time.
The clinical experts consulted on this review noted that TFR has become a key outcome in assessing response to TKIs in CML, and that in their experience, approximately half of patients treated with a given TKI are ultimately able to achieve TFR. According to the ELN guidelines, attempting TFR is primarily based on patient selection, duration and depth of MR, and monitoring capacity. The guidelines suggest an optimal stop recommendation is a minimum of 5 years of TKI therapy and a DMR (MR4.5) of at least 2 years (if MR4.5 is observed) or at least 3 years (if MR4 is observed).5 There were no data for TFR in the ASC2ESCALATE study; however, MR4 and MR4.5 were observed in ████% and ███% of patients with adequate follow-up at 9 months, respectively. It is unclear whether the number of patients with an observed MR4.5 or MR 4 in the ASC2ESCALATE study will continue to increase with longer follow-up, or whether DMR will be sustained in these patients. Additionally, the clinical experts consulted for this review noted that some laboratories in Canada are unable to assess deeper MRs (e.g., MR4.5).
As previously noted, the results from the ASC2ESCALATE study were immature at IA4 and there was no evidence provided for key outcomes such as survival (OS), time-to-event measures (time to treatment failure or duration of MMR), and HRQoL (MDASI-CML). As such, the clinical benefit of asciminib for these outcomes remains unestablished in this patient population. Given that MMR, time-to-event measures, and HRQoL outcomes are highly valued by the patients, clinician groups, and clinical experts consulted, the lack of these data represents a notable limitation in assessing the treatment's effectiveness compared to any other treatment.
In the absence of comparative evidence from the ASC2ESCALATE study and given that the sponsor did not submit an ITC comparing asciminib to other TKIs used in the second line, the relative efficacy of asciminib to other treatments in this patient population remains unknown.
Harms
The open-label design of the ASC2ESCALATE study may introduce bias in reporting subjective harms, potentially affecting the reliability of the safety data. Asciminib has been approved for use in Canada since 2022, and no new safety signals were identified in the ASC2ESCALATE study; however, the follow-up was short and the study did not provide any information on long-term harms that have been identified with other TKIs. The experts stated that some AEs (cardiac, vascular, and pulmonary AEs), may appear for the first time after many years of TKI therapy, thus, the cumulative incidence continues to increase after many years of follow-up. Additionally, the clinical experts noted that second-generation TKIs may cause more frequent or severe AEs than imatinib. Given the improved prognosis and near-normal life expectancy in patients with CML, as well as patients often requiring lifelong treatment, long-term safety monitoring is essential but was not evaluated in the ASC2ESCALATE study. The harms results of the retrospective chart review also provided evidence supportive of the ASC2ESCALATE study results. As there was no comparator in the ASC2ESCALATE study and the sponsor did not provide ITCs, the relative safety of asciminib compared to other treatments in this patient population remains unknown.
Conclusion
One phase II, single-arm, open-label trial (the ASC2ESCALATE study) provided evidence for the efficacy and safety of asciminib in adult patients with Ph+ CML in CP that is intolerant to or has failed to respond to prior treatment with 1 TKI. In the ASC2ESCALATE study, ██% of patients experienced MMR by 9 months, which is likely a clinically important result, although the evidence was uncertain due to the substantial limitations driven by the single-arm study design, short follow-up, small sample size, and interim results. Nonetheless, the observed improvements in MMR at the interim time points in the trial were recognized as clinically meaningful according to the clinical experts consulted for this review based on their experience with TKIs in the second-line setting in this patient population. Asciminib has been available in Canada since 2022, and no new safety concerns were identified in the ASC2ESCALATE study.
Despite the potentially clinically significant results for MR outcomes, and tolerable safety profile, the absence of data for other outcomes that are important to patients and clinicians such as OS, time to and duration of MMR, TFR, and HRQoL limits the ability to draw conclusions on the durability and importance of any clinical benefit with asciminib, and the impact of asciminib on these outcomes in this population remains unknown. Furthermore, there are no direct or indirect comparative data on the effectiveness and safety of asciminib versus other treatment options in Canada in this patient population, which is a significant limitation of the evidence.
The MR findings of the ASC2ESCALATE study were supported by additional evidence from a retrospective chart review that provided a real-world evaluation of molecular outcomes in adult patients with CP CML treated with asciminib after prior TKI therapy, and no new safety concerns were identified; however, this study was associated with significant limitations, and can only be considered supportive of the overall effect of asciminib.
References
1.Van Etten RA, Atallah E. Larson RA, Rosmarin AG, eds. Chronic myeloid leukemia: Pathogenesis, clinical manifestations, and diagnosis. UpToDate; 2025. Accessed March 15, 2025. https://www.uptodate.com/contents/chronic-myeloid-leukemia-pathogenesis-clinical-manifestations-and-diagnosis
2.Table 13-10-0747-01. Number of new cases and age-standardized rates of primary cancer, by cancer type and sex. Statistics Canada; 2025. Accessed 2025 Mar 24. https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1310074701&pickMembers%5B0%5D=2.1&pickMembers%5B1%5D=3.53&cubeTimeFrame.startYear=2009&cubeTimeFrame.endYear=2022&referencePeriods=20090101%2C20220101
3.Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2022 update on diagnosis, therapy, and monitoring. Am J Hematol. 2022;97(9):1236-1256. doi:10.1002/ajh.26642 PubMed
4.Schiffer C, Atallah E. Larson RA, Rosmarin AG, eds. Overview of the treatment of chronic myeloid leukemia. UpToDate; 2022. Accessed 2025 Mar 15. http://www.uptodate.com
5.Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34(4):966-984. doi:10.1038/s41375-020-0776-2 PubMed
6.Hochhaus A, Breccia M, Saglio G, et al. Expert opinion-management of chronic myeloid leukemia after resistance to second-generation tyrosine kinase inhibitors. Leukemia. 2020;34(6):1495-1502. doi:10.1038/s41375-020-0842-9 PubMed
7.Scemblix (asciminib): 20 mg and 40 mg, oral tablets [product monograph]. 2025;
8.CADTH Drug Reimbursement Expert Review Committee final recommendation: asciminib (Scemblix - Novartis Pharmaceuticals Canada Inc.). CADTH; 2022. Accessed 2025 Mar 11. https://www.cadth.ca/sites/default/files/DRR/2022/PC0275%20Scemblix%20-%20CADTH%20Final%20Rec.pdf
9.CDA-AMC Sponsor Summary of Clinical Evidence Template [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Scemblix (asciminib): 20 mg and 40 mg, oral tablets. Novartis Pharmaceuticals Canada; 2025.
10.Hehlmann R. The New ELN Recommendations for Treating CML. Journal of Clinical Medicine. 2020;9(11):3671. doi:10.3390/jcm9113671 PubMed
11.Food and Drug Administration. Clinical Trial Endpoints for the Approval of Cancer Drugs and Biologics: Guidance for Industry. 2018. Accessed May 12, 2025. https://www.fda.gov/media/71195/download
12.Shah NP, Bhatia R, Altman JK, et al. Chronic myeloid leukemia, version 2.2024, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2024;22(1):43-69. PubMed
13.Balshem H, Helfand M, Schünemann HJ, et al. GRADE guidelines: 3. Rating the quality of evidence. Journal of clinical epidemiology. 2011;64(4):401-6. doi:10.1016/j.jclinepi.2010.07.015 PubMed
14.Santesso N, Glenton C, Dahm P, et al. GRADE guidelines 26: informative statements to communicate the findings of systematic reviews of interventions. Journal of clinical epidemiology. 2020;119:126-135. doi:10.1016/j.jclinepi.2019.10.014 PubMed
15.Atallah EL, Sadek I, Wei D, et al. Treatment with Asciminib after a Prior Tyrosine Kinase Inhibitor in Patients with Chronic-Phase Chronic Myeloid Leukemia. [internal sponsor manuscript draft]: In Drug Reimbursement Review sponsor submission: Scemblix (asciminib): 20 mg and 40 mg, oral tablets. 2025.
16.Flis S, Chojnacki T. Chronic myelogenous leukemia, a still unsolved problem: pitfalls and new therapeutic possibilities. Drug Des Devel Ther. 2019;Volume 13:825-843. doi:10.2147/DDDT.S191303 PubMed
17.Hoffmann VS, Baccarani M, Hasford J, et al. The EUTOS population-based registry: incidence and clinical characteristics of 2904 CML patients in 20 European Countries. Leukemia. 2015;29(6):1336-1343. doi:10.1038/leu.2015.73 PubMed
18.Canadian Cancer Society. Risk for chronic leukemia. 2024;
19.Hehlmann R. Chronic Myeloid Leukemia in 2020. HemaSphere. 2020;4(5):e468. doi:10.1097/HS9.0000000000000468 PubMed
20.Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391-2405. doi:10.1182/blood-2016-03-643544 PubMed
21.Table 13-10-0751-01. Number of prevalent cases and prevalence proportions of primary cancer by prevalence duration, cancer type, attained age group and sex. Statistics Canada; 2022. Accessed April 16, 2025. https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1310075101&pickMembers%5B0%5D=2.1&pickMembers%5B1%5D=3.2&pickMembers%5B2%5D=4.53&pickMembers%5B3%5D=5.4&cubeTimeFrame.startYear=2015&cubeTimeFrame.endYear=2018&referencePeriods=20150101%2C20180101
22.Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2018 update on diagnosis, therapy and monitoring. Am J Hematol. 2018;93(3):442-459. doi:10.1002/ajh.25011 PubMed
23.Schiffer C, Atallah E. Larson RA, Rosmarin AG, eds. Chronic myeloid leukemia in chronic phase: Initial treatment. UpToDate; 2025. Accessed 2025 Mar 18. http://www.uptodate.com
24.Negrin R, Schiffer C, Atallah E. Larson RA, Rosmarin AG, eds. Treatment of chronic myeloid leukemia in chronic phase after failure of initial therapy. UpToDate; 2025. Accessed 2025 Mar 18. http://www.uptodate.com
25.PrBosulif® (bosutinib): 100 mg, 400 mg, 500 mg, oral tablets [product monograph]. Pfizer Canada; 2021.
26.PrTasinga® (nilotinib): 150 mg, 200 mg, oral capsules [product monograph]. Novartis Pharmaceuticals Canada; 2015.
27.PrSprycel® (dasatinib): 20 mg, 50 mg, 70 mg, 80 mg, 100 mg, 140 mg, oral tablets [product monograph]. Bristol-Myers Squibb Canada; 2021.
28.PrIclusig® (ponatinib): 15 mg, 45 mg, oral tablets [product monograph]. ARIAD Pharmaceuticals; 2019.
29.Atallah EL, Mauro MJ, Sasaki K, et al. Dose-escalation of second-line and first-line asciminib in chronic myeloid leukemia in chronic phase: the ASC2ESCALATE Phase II trial. Future Oncology. 2024:1-11. doi:10.1080/14796694.2024.2402680 PubMed
30.Inc NPC. NCT05384587: Asciminib Monotherapy, with Dose Escalation, for 2nd and 1st Line Chronic Myelogenous Leukemia (ASC2ESCALATE). ClinicalTrials.gov. Accessed March 19, 2025. https://clinicaltrials.gov/study/NCT05384587
31.6-Month Efficacy Key Outputs for 2L Cohort: A Phase II Multicenter, Open-label, Single-arm Dose Escalation Study of Asciminib Monotherapy in 2nd and 1st Line Chronic Phase – Chronic Myelogenous Leukemia (ASC2ESCALATE) (IA4, DCO: November 15, 2024). Novartis Pharmaceuticals Canada; 2025.
32.Williams LA, Gonzalez AGG, Ault P, et al. Measuring the symptom burden associated with the treatment of chronic myeloid leukemia. Blood, The Journal of the American Society of Hematology. 2013;122(5):641-647. PubMed
Pharmacoeconomic Review
Abbreviations
AE
adverse event
BIA
budget impact analysis
CDA-AMC
Canada’s Drug Agency
CMA
cost-minimization analysis
CML
chronic myeloid leukemia
CP
chronic phase
Ph+
Philadelphia chromosome positive
TKI
tyrosine kinase inhibitor
Economic Review
The objective of the economic review is to review and critically appraise the pharmacoeconomic evidence submitted by the sponsor on the cost and budget impact of asciminib compared to tyrosine kinase inhibitors (TKIs) for the treatment of Philadelphia chromosome–positive (Ph+) chronic myeloid leukemia (CML) in chronic phase (CP) previously treated with 1 or more TKIs.1 Additional information about the sponsor’s submission is summarized in Appendix 2.
Item | Description |
|---|---|
Drug product | Asciminib (Scemblix), 20 mg and 40 mg oral tablets |
Indication | Adult patients with Philadelphia chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase who are newly diagnosed or who have previously received 1 or more tyrosine kinase inhibitors. |
Submitted price | Asciminib: $63.00 per 20 mg tablet Asciminib: $85.00 per 40 mg tablet |
Health Canada approval status | NOC |
Health Canada review pathway | Project Orbis Type B |
NOC date | July 25, 2025 |
Reimbursement request | For the treatment of adult patients with Ph+ CML in chronic phase previously treated with 1 tyrosine kinase inhibitor. |
Sponsor | Novartis Pharmaceuticals Canada Inc. |
Submission history | Previously reviewed: Yes Indication: For the treatment of adult patients with Ph+ CML in chronic phase previously treated with 2 or more tyrosine kinase inhibitors. Recommendation date: August 5, 2022 Recommendation: Reimburse with clinical criteria and/or conditions |
CML = chronic myeloid leukemia; NOC = Notice of Compliance; Ph+ = Philadelphia chromosome positive.
Key Messages
Asciminib is available as 20 mg and 40 mg tablets.1 At the submitted price of $63.00 per 20 mg tablet and $85.00 per 40 mg tablet, the 28-day cost of asciminib is expected to be $4,760 per patient, based on the Health Canada–recommended dosage.2
Based on the results of Canada’s Drug Agency (CDA-AMC)’s base case, asciminib is expected to be associated with lower drug acquisition costs for the health care system compared to ponatinib (incremental savings = –$66,120) and expected to have higher drug acquisition costs compared to all the other TKIs, ranging from $6,487 (versus bosutinib) to $54,484 (versus imatinib). Due to the absence of evidence, it is uncertain whether there will be differences in nondrug costs related to management of adverse events (AEs) and disease monitoring.
CDA-AMC estimates that the budget impact of reimbursing asciminib for the treatment of adult patients with Ph+ CML in CP previously treated with at least 1 TKI will be approximately $8 million over the first 3 years of reimbursement compared to the amount currently spent on comparators, with an estimated expenditure of $65 million on asciminib over this period. The actual budget impact of reimbursing asciminib will depend on the percentage of patients covered by public drug plans and the eligible patient population.
Summary of the Submitted Economic Evaluation
The sponsor submitted a cost-minimization analysis (CMA) comparing asciminib to TKIs from the perspective of a public drug plan payer in Canada over a 1-year time horizon.2 The modelled population comprised adult patients with Ph+ CML in CP who were previously treated with 1 or more TKIs, which is aligned with the reimbursement request and was based on the participants in the ASC2ESCALATE trial.3 The sponsor’s base-case analysis included costs related to drug acquisition (submitted price for asciminib and public list prices for comparators), disease monitoring (i.e., health care resource utilization), and AEs.2
In the sponsor’s base case, asciminib was associated with incremental costs of $8,824 relative to the “All TKIs” pooled comparator, which reflects the sponsor’s distribution of TKIs currently used in clinical practice in Canada.2 Specifically, asciminib was associated with incremental cost savings of $70,481 relative to ponatinib and increased costs compared to all the other TKIs, with incremental costs ranging from $4,364 (versus bosutinib) to $53,656 (versus imatinib).2 Additional information about the sponsor’s submission is summarized in Appendix 2.
CDA-AMC identified several key issues with the sponsor’s analysis (refer to Table 2; full details are provided in Appendix 3). A revised base case was therefore developed.
Table 2: Key Issues With the Sponsor’s Economic Submission
Issue | What evidence is there to inform this issue? | How was this issue addressed by CDA-AMC? | Did CDA-AMC explore uncertainty in a scenario analysis? |
|---|---|---|---|
The clinical similarity of asciminib to relevant comparators in the second-line setting is unknown. | The sponsor presented interim results from an ongoing phase II, single-arm, dose-escalation study (the ASC2ESCALATE study), evaluating the efficacy, safety, and tolerability of asciminib in the reimbursement request population. The sponsor did not provide evidence showing similar efficacy of asciminib to the comparators in the second-line setting.3 | CDA-AMC could not address the absence of comparative clinical evidence. | No scenario analysis was conducted due to a lack of clinical data. |
The type and frequency of AEs for asciminib and comparators in the second-line setting is unknown. | The types and frequencies of AEs were derived from the ASC4FIRST trial, which evaluated the efficacy and safety of asciminib vs. other TKIs in patients with newly diagnosed CML.4,5 The sponsor assumed that the safety data from first-line treatment would be generalizable to patients in the second-line setting. | CDA-AMC excluded costs associated with AE management from the analysis. | No scenario analysis was conducted due to a lack of clinical data. |
The disease monitoring frequency for asciminib and comparators in the second-line setting is unknown. | The frequencies of disease monitoring were informed by a physician survey that assessed health care resource use among patients with CML receiving third-line treatment. The sponsor assumed that these data would be generalizable to patients in the second-line setting.2,6 | CDA-AMC excluded costs associated with disease monitoring from the analysis. | No scenario analysis was conducted due to a lack of clinical data. |
The time horizon is inappropriate. | The sponsor applied a 1-year time horizon to estimate annual treatment costs for asciminib without clinical evidence or treatment effect data to support this time horizon. | CDA-AMC could not address this limitation in the absence of comparative clinical data to inform an appropriate duration. | No scenario analysis was conducted — best available evidence was used in the CDA-AMC base case. |
The dosages modelled for comparators may not align with clinical practice. | The sponsor used dosing consistent with the ASC4FIRST clinical trial; however, in clinical practice, lower dosages are often used to manage AEs. | CDA-AMC did not address this issue, as the dosages used were consistent with those specified in the Health Canada product monographs. | No scenario analysis was conducted — best available evidence was used in the CDA-AMC base case. |
The sponsor’s analysis relies on publicly accessible list prices and does not reflect existing confidential prices potentially negotiated by public plans. | Confidential pricing agreements exist for ponatinib and bosutinib. Thus, the current unit cost paid by public drug plans may be lower than the submitted prices. | CDA-AMC was unable to incorporate the presence of confidential negotiated prices in its reanalysis. | No scenario analysis was conducted. The presence and magnitude of potential cost savings for drug plans remain uncertain. |
AE = adverse event; CDA-AMC = Canada’s Drug Agency; CML = chronic myeloid leukemia; TKI = tyrosine kinase inhibitor; vs. = versus.
Note: Full details of the issues identified by CDA-AMC are provided in Appendix 3.
CDA-AMC Assessment of Cost
The CDA-AMC base case excluded costs associated with disease monitoring and AE management. The CDA-AMC base case was derived by making changes to model parameter values and assumptions (refer to Table 6), in consultation with clinical experts. Detailed information about the base case is provided in Appendix 3.
The total acquisition cost of asciminib is expected to be $62,093 over a 1-year period, compared to $7,609 to $128,213 for the comparators (Table 3). The use of asciminib is expected to result in incremental savings of $68,120 relative to ponatinib and incremental costs compared to all other comparators, with incremental costs ranging from $6,487 (versus bosutinib) to $54,484 (versus imatinib). This increase in health care spending results from higher drug acquisition costs associated with asciminib.
Table 3: Summary of CDA-AMC Results
Drug | Total costs ($)a,b |
|---|---|
Imatinib | 7,609 |
Nilotinib | 45,427 |
Dasatinib | 48,311 |
All TKIsc | 51,138 |
Bosutinib | 55,606 |
Asciminib | 62,093 |
Ponatinib | 128,213 |
CDA-AMC = Canada’s Drug Agency; TKI = tyrosine kinase inhibitor.
aIncludes drug acquisition costs only.
bTotal costs over a 1-year period.
cThis is a pooled comparator that reflects the sponsor’s distribution of TKIs currently used in clinical practice in Canada: 8% imatinib, 6% nilotinib, 50% dasatinib, 31% bosutinib, 5% ponatinib.
Summary of the Budget Impact
The sponsor submitted a budget impact analysis (BIA) to estimate the 3-year (2026 to 2028) budget impact of reimbursing asciminib for the treatment of adult patients with Ph+ CML in CP previously treated with 1 or more TKIs.7 The sponsor assumed that the payer would be CDA-AMC–participating public drug plans and derived the size of the eligible population using an epidemiologic approach.7 The price of asciminib was aligned with the price included in the sponsor’s economic evaluation, while the prices of comparators were based on the publicly available list prices. Additional information pertaining to the sponsor’s submission is provided in Appendix 4.
CDA-AMC identified a number of issues with the sponsor’s estimated budget impact and made changes to model parameters and assumptions in consultation with clinical experts to derive the CDA-AMC base case (Appendix 4). CDA-AMC estimated that 5,162 patients would be eligible for treatment with asciminib over a 3-year period (year 1 = 1,667; year 2 = 1,720; year 3 = 1,775). The estimated incremental budget impact of reimbursing asciminib is expected to be approximately $8 million over the first 3 years, with an expected expenditure of $65 million on asciminib. The actual budget impact of reimbursing asciminib will depend on the percentage of patients covered by public drug plans and eligible patient population.
Conclusions
At the submitted price, asciminib is expected to be more costly than all comparators apart from ponatinib for the treatment of adult patients with Ph+ CML in CP previously treated with 1 or more TKIs. Confidential pricing agreements exist for ponatinib and bosutinib, and a price reduction for asciminib may be required such that no additional costs are incurred by the health care system.
The budget impact of reimbursing asciminib to the public drug plans in the first 3 years is estimated to be approximately $8 million. The 3-year expenditure on asciminib (i.e., not accounting for current expenditure on comparators) is estimated to be $65 million. The estimated budget impact is highly uncertain due to lack of comparative evidence against the comparators, assumptions related to public coverage, access to care, and the eligible patient population.
References
1.Novartis Pharmaceuticals Canada Inc. Scemblix (asciminib): tablets, 20 mg and 40 mg, asciminib (as asciminib hydrochloride), oral [product monograph]. June 21, 2022. Updated July 25, 2024.
2.Novartis Pharmaceuticals Canada Inc. Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Scemblix (asciminib) tablets, 20 mg and 40 mg, asciminib (as asciminib hydrochloride), oral. March 10, 2025.
3.Atallah EL, Mauro MJ, Sasaki K, et al. Dose-escalation of second-line and first-line asciminib in chronic myeloid leukemia in chronic phase: The ASC2ESCALATE Phase II trial. Future Oncol. 2024:1-11. doi:10.1080/14796694.2024.2402680 PubMed
4.National Institute for Health and Care Excellence. Asciminib for treating chronic myeloid leukaemia after 2 or more tyrosine kinase inhibitors (NICE Technology appraisal guidance TA813) [sponsor supplied reference].
5.Novartis Pharmaceuticals Canada Inc. Clinical Study Report: CABL001J12301. A phase III, multi-center, open-label, randomized study of oral asciminib versus Investigator selected TKI in patients with newly diagnosed Philadelphia Chromosome Positive Chronic Myelogenous Leukemia in Chronic Phase. Primary endpoint analysis at week 48 [internal sponsor's report]. August 7, 2024.
6.Novartis Pharmaceuticals Canada Inc. Novartis Market Research (Data on file) [sponsor supplied reference]. Novartis Pharmaceuticals Canada Inc.; 2025.
7.Novartis Pharmaceuticals Canada Inc. Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Scemblix (asciminib) tablets, 20 mg and 40 mg, asciminib (as asciminib hydrochloride), oral. March 10, 2025.
8.Novartis Pharmaceuticals Canada Inc. Drug Reimbursement Review sponsor submission: Scemblix (asciminib) tablets, 20 mg and 40 mg, asciminib (as asciminib hydrochloride), oral [internal sponsor's package]. March 10, 2025.
9.Ontario Ministry of Health. Exceptional Access Program (EAP). Accessed May 1, 2025. http://www.health.gov.on.ca/en/pro/programs/drugs/odbf/odbf_except_access.aspx
10.Ontario Ministry of Health. Ontario drug benefit formulary/comparative drug index. Accessed May 1, 2025. https://www.formulary.health.gov.on.ca/formulary/
11.Government of Ontario. OHIP Schedule of Benefits and fees [sponsor supplied reference]. http://www.ontario.ca/page/ohip-schedule-benefits-and-fees
12.CADTH. Guidelines for the economic evaluation of health technologies: Canada. 4th ed. 2017. Accessed June 6, 2025. https://www.cadth.ca/dv/guidelines-economic-evaluation-health-technologies-canada-4th-edition
13.Statistics Canada. Population estimates on July 1, by age and gender [sponsor supplied reference]. 2018. https://www150.statcan.gc.ca/t1/tbl1/en/cv.action?pid=1710000501
14.Statistics Canada. Canada's population estimates: Strong population growth in 2023 [sponsor supplied reference]. 2024. https://www150.statcan.gc.ca/n1/daily-quotidien/240327/dq240327c-eng.htm
15.National Cancer Institute. Chronic Myeloid Leukemia (CML) People Alive with Cancer (U.S. Prevalence) on January 1, 2021 [sponsor supplied reference]. https://seer.cancer.gov/statistics-network/explorer/application.html?site=97&data_type=5&graph_type=11&compareBy=sex&chk_sex_1=1&series=9&age_range=1&advopt_precision=1&hdn_view=1&advopt_show_apc=on&advopt_display=2#resultsRegion1
16.Eden RE, Coviello JM. Chronic Myelogenous Leukemia. StatPearls Publishing; 2023. Accessed January 30, 2025. https://www.ncbi.nlm.nih.gov/books/NBK531459/
17.Soverini S, Bassan R, Lion T. Treatment and monitoring of Philadelphia chromosome-positive leukemia patients: recent advances and remaining challenges. J Hematol Oncol. 2019;12(1):39. doi:10.1186/s13045-019-0729-2 PubMed
18.Kota VK, Wei D, Yang D, et al. Treatment Patterns and Modifications of Tyrosine Kinase Inhibitors (TK I) Therapy in Early Lines in Patients with Chronic Myeloid Leukemia in Chronic Phase (CML-CP): Real-World Analysis from a Large Commercial Claims Database in the United States (US). Blood. 2023;142(Supplement 1):5190-5190. doi:10.1182/blood-2023-180789
19.IQVIA. PharmaStat. 2023. Accessed April 11, 2025. https://www.iqvia.com/
20.Sutherland G, Dihn T. Understanding the gap: A pan-Canadian analysis of prescription drug insurance coverage. The Conference Board of Canada; 2017. Accessed June 11, 2025. https://www.conferenceboard.ca/e-library/abstract.aspx?did=9326
21.Isabelle G-A, Wanlin C, Chad L. Understanding the gap 2.0: A pan-Canadian analysis of prescription drug insurance coverage. The Conference Board of Canada; 2022. Accessed May 26, 2025. https://www.conferenceboard.ca/product/understanding-the-gap-2-0-a-pan-canadian-analysis-of-prescription-drug-insurance-coverage-2/
22.Canadian Cancer Society. Leukemia statistics. 2024. Accessed June 3, 2025. https://cancer.ca/en/cancer-information/cancer-types/leukemia/statistics
23.The Chronic Myelogenous Leukemia Society of Canada. What is CML? Overview of CML, its symptoms and diagnosis. Accessed June 3, 2025. https://cmlsociety.org/what-is-cml/
Appendix 1: Cost Comparison Table
Please note that this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical experts and CDA-AMC–participating public drug plans. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 4: Cost Comparison for Ph+ CML-CP in 2L
Treatment | Strength | Form | Price ($) | Recommended dosage | Daily cost ($) | 28-day cost ($) |
|---|---|---|---|---|---|---|
Asciminib | 20 mg 40 mg | Tablet | 63.0000a 85.0000a | 40 mg twice daily or 80 mg once daily | 170.00 | 4,760 |
Tyrosine kinase Inhibitors | ||||||
Bosutinib | 100 mg 500 mg | Tablet | 38.9787b 152.2403b | 400 mg once daily | 155.91 | 4,366 |
Dasatinib | 20 mg 50 mg 70 mg 80 mg 100 mg | Tablet | 32.8823c 66.1782c 72.9337c 117.3255c 132.2671c | 100 mg once daily | 132.27 | 3,703 |
Imatinib | 100 mg 400 mg | Tablet | 5.2079c 20.8314c | 400 once mg daily | 20.83 | 583.28 |
Nilotinib | 150 mg 200 mg | Tablet | 24.8355c 34.5709c | 300 mg twice daily | 99.34 | 2,782 |
Ponatinib | 15 mg 45 mg | Tablet | 157.0815b 351.0267b | 45 mg once daily | 351.03 | 9,829 |
2L = second line; CP = chronic phase; Ph+ CML = Philadelphia chromosome–positive chronic myeloid leukemia.
aSponsor’s submitted price.8
bOntario Exceptional Access Program (accessed May 2025).9
cOntario Drug Benefit Formulary (accessed May 2025).10
Appendix 2: Additional Details of the Sponsor’s Submission
Please note that this appendix has not been copy-edited.
Table 5: Key Components of the Sponsor’s Economic Evaluation
Component | Description |
|---|---|
Treatment information | |
Drug under review | Asciminib (Scemblix), 20 mg and 40 mg oral tablets |
Submitted price of drug under review | Asciminib: $63.00 per 20 mg tablet Asciminib: $85.00 per 40 mg tablet |
Regimen | 40 mg twice daily or 80 mg once daily. Treatment should be continued as long as clinical benefit is observed or until unacceptable toxicity occurs. |
Per-cycle cost of drug under review | $4,760 per patient per 28-day cycle |
Model information | |
Type of economic evaluation | CMA |
Treatment | Asciminib |
Included comparators |
|
Perspective | Publicly funded health care payer |
Time horizon | 1 year |
Modelled population | Adult patients with Ph+ CML in CP who were previously treated with 1 or more TKIs. Derived from the second-line cohort of the ASC2ESCALATE study.3 |
Model health states | NA |
Data sources | |
Comparative efficacy | No comparative clinical evidence is available for asciminib relative to its comparators in the second-line setting. |
Resource use and costs |
|
Summary of the submitted results | |
Base-case results |
|
Scenario analysis results | Nonea |
AE = adverse event; CIHI = Canadian Institute for Health Information; CP = chronic phase; FISH = fluorescence in situ hybridization; HCRU = health care resource use; ODB = Ontario Drug Benefit; OP = outpatient; PCR = polymerase chain reaction; Ph+ CML = Philadelphia chromosome–positive chronic myeloid leukemia; TKI = tyrosine kinase inhibitor.
aSubmitted scenarios included changes in the incidence of neutropenia, incidence of platelet count decrease, incidence of thrombocytopenia, incidence of anemia, incidence of neutrophil count decrease, incidence of hypertension, and incidence of alanine aminotransferase increase for some comparators; none had a meaningful impact on cost differences.
Appendix 3: Additional Details of CDA-AMC Reanalyses
Please note that this appendix has not been copy-edited.
Clinical Data in the Economic Model
Assuming comparable clinical efficacy and safety between asciminib and ponatinib, nilotinib, dasatinib, bosutinib, and imatinib, the sponsor submitted a CMA comparing costs associated with drug acquisition, health care resource utilization, and AE management for adults with Ph+ CML in CP previously treated with 1 or more TKIs.2 However, based on the clinical review by CDA-AMC, there are no comparative data on the effectiveness or safety of asciminib relative to other treatment options available in Canada for this patient population. The available evidence is limited to a single-arm trial (ASC2ESCALATE), which lacked a direct comparator, limiting the ability to draw conclusions about comparative efficacy and safety.3 As a result, the assumption of clinical equivalence is subject to a high degree of uncertainty.
Key Issues of the Submitted Economic Evaluation
CDA-AMC identified the following key issues with the sponsor’s analysis:
The clinical similarity of asciminib to relevant comparators is unknown. The sponsor presented interim results from the ASC2ESCALATE trial, an ongoing phase II, single-arm, dose-escalation study evaluating the efficacy, safety, and tolerability of asciminib in the reimbursement-requested population.3 No evidence was provided to demonstrate comparable efficacy or safety of asciminib relative to its comparators in the second-line setting. Consequently, no formal comparative clinical or safety analysis was conducted. While outcomes such as major molecular response, health-related quality of life, and time-to-event measures were reported, these data were preliminary and incomplete and only applied to asciminib.
CDA-AMC was unable to address this limitation owing to the lack of available evidence. The CDA-AMC base case retained the sponsors’ assumption that asciminib is equally effective and safe compared to its comparators.
The type and frequency of AEs for asciminib and comparators in the second-line setting is unknown. The sponsor derived AE frequencies for asciminib and the other TKIs from the ASC4FIRST trial, including only grade 3 or 4 AEs occurring in ≥ 5% of patients.3-5 These frequencies, observed in the first-line setting, were applied to the second-line population without adjustment.2 For ponatinib, AE frequencies were sourced from a cost-effectiveness analysis conducted for a previous health technology assessment.4 Clinical experts consulted by CDA-AMC indicated that several AE frequencies were likely overestimated in the second-line setting and noted the omission of clinically relevant AEs, including cardiac toxicity, pancreatitis, elevated creatinine, pleural effusion, pulmonary hypertension, hyperglycemia, and peripheral arterial disease. Due to the lack of comparative published data reporting the frequency of these AEs, CDA-AMC was unable to incorporate them into the analysis.
CDA-AMC excluded costs associated with AE management from the base case.
The disease monitoring frequency for asciminib and comparators in the second-line setting is unknown. The sponsor estimated disease monitoring frequencies using data from a physician survey of patients receiving third-line treatment for CML and assumed these estimates were generalizable to the second-line setting.6 Unit costs were obtained from the Ontario Schedule of Benefits and adjusted to reflect the dollar amount in 2025.11 Monthly disease monitoring costs were calculated by multiplying the frequency of health care resource use by the corresponding unit cost and then extrapolated to annual estimates. The sponsor assumed equivalent disease monitoring costs across all the comparators.
CDA-AMC excluded disease monitoring costs from the base case.
The time horizon is inappropriate. The sponsor extrapolated monthly treatment costs for asciminib and its comparators to an annual estimate using a fixed 1-year time horizon.2 This time horizon was not informed by clinical data or treatment effect. CDA-AMC guidelines recommend selecting a time horizon that is adequate to capture all relevant differences in future costs and outcomes, based on the nature of the condition and the expected impact of the intervention.12 In the absence of comparative clinical data to inform an appropriate duration, the arbitrary use of a 1-year horizon introduces uncertainty in the estimated costs associated with reimbursing asciminib in this population.
CDA-AMC was unable to address this limitation owing to the lack of clinical data.
The dosages modelled for comparators may not align with clinical practice. The sponsor applied dosage assumptions from the ASC4FIRST trial, which reflect first-line treatment practices in newly diagnosed patients.2 However, clinical experts consulted by CDA-AMC noted that, in the second-line setting, dose modifications are frequently implemented to enhance tolerability while maintaining therapeutic benefit. The sponsor’s model assumed fixed daily doses and did not incorporate such modifications, potentially overestimating drug acquisition costs for comparator TKIs. This approach may bias results in favour of asciminib.
CDA-AMC did not address this issue, as the dosages used in the base case were consistent with those specified in the respective Health Canada product monographs. However, the exclusion of dose adjustment practices limits the generalizability of the results to real-world clinical settings in Canada, where individualized dosing is a standard part of care.
The drug prices are based on publicly listed prices. The prices used in the CMA for the comparators are based on publicly available list prices and do not account for potential confidential pricing agreements negotiated by public drug plans.9,10 Because the projected cost savings for asciminib compared to ponatinib and increased cost compared to all the other TKIs are based on public list prices for comparators, price reductions may be necessary for asciminib to achieve cost savings if the actual prices of the comparators are lower than their listed prices.
CDA-AMC was unable to address this limitation through reanalysis, as the existence and magnitude of negotiated prices are unknown.
CDA-AMC Cost Analysis
The CDA-AMC base case was derived by making changes in model parameter values and assumptions, in consultation with clinical experts. CDA-AMC undertook reanalyses that addressed key limitations within the submitted economic model (Table 6). All CDA-AMC reanalyses are presented deterministically.
Table 6: Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CDA-AMC value or assumption |
|---|---|---|
1. Type and frequency of AEs for asciminib and comparators | Included | Excluded |
2. Disease monitoring frequency for asciminib and comparators | Included | Excluded |
CDA-AMC base case (health care payer perspective) | ― | Reanalysis 1 + 2 |
AE = adverse event; CDA-AMC = Canada’s Drug Agency.
Table 7: Summary of the Stepped Analysis
Stepped analysis | Drug | Total costs ($) |
|---|---|---|
Sponsor’s base case | Imatinib | 14,445 |
Nilotinib | 53,558 | |
Dasatinib | 56,442 | |
Bosutinib | 63,737 | |
Asciminib | 68,101 | |
Ponatinib | 138,582 | |
CDA-AMC reanalysis 1 | Imatinib | 9,933 |
Nilotinib | 47,751 | |
Dasatinib | 50,635 | |
Bosutinib | 57,930 | |
Asciminib | 64,416 | |
Ponatinib | 130,536 | |
CDA-AMC reanalysis 2 | Imatinib | 12,121 |
Nilotinib | 51,234 | |
Dasatinib | 54,118 | |
Bosutinib | 61,413 | |
Asciminib | 65,777 | |
Ponatinib | 136,258 | |
CDA-AMC base case (reanalysis 1 + 2) | Imatinib | 7,609 |
Nilotinib | 45,427 | |
Dasatinib | 48,311 | |
Bosutinib | 55,606 | |
Asciminib | 62,093 | |
Ponatinib | 128,213 |
CDA-AMC = Canada’s Drug Agency.
Note: The CDA-AMC reanalysis is based on the publicly available prices of the comparator treatments.
Results of the CDA-AMC cost comparison are presented in Table 8. At an average annual cost of $62,093 per patient, treatment with asciminib is expected to cost less compared to ponatinib (incremental savings: $66,120 per patient) and cost more than all the other TKIs (incremental costs: $6,487 (versus bosutinib) to $54,484 (versus imatinib) per patient).
Table 8: Summary of the CDA-AMC Cost Analysis
Drug | Unit drug cost ($) | Annual drug cost ($) | Incremental total costs vs. asciminib ($) |
|---|---|---|---|
Asciminib | 63.00a | 62,093 | Reference |
Imatinib | 5.21b | 7,609 | −54,484 |
Nilotinib | 24.84c | 45,427 | −16,665 |
Dasatinib | 32.88d | 48,311 | −13,782 |
Bosutinib | 38.98e | 55,606 | −6,487 |
Ponatinib | 157.08f | 128,213 | 66,120 |
CDA-AMC = Canada’s Drug Agency; vs. = versus.
aSponsor’s submitted price of $63.00 per 20 mg tablet and $85.00 per 40 mg tablet.8
bImatinib: $5.21 per 100 mg tablet and $20.83 per 400 mg tablet.10
cNilotinib: $24.84 per 150 mg tablet and $34.57 per 200 mg tablet.10
dDasatinib: $32.88 per 20 mg tablet, $66.18 per 50 mg tablet, $72.93 per 70 mg tablet, $117.36 per 80 mg tablet, and $132.27 per 100 mg tablet.10
eBosutinib: $38.98 per 100 mg tablet and $152.24 per 500 mg tablet.9
fPonatinib: $157.08 per 15 mg tablet and $351.03 per 45 mg tablet.9
Note: The CDA-AMC reanalysis is based on the publicly available prices of the comparator treatments.
Appendix 4: Budget Impact Analysis
Please note that this appendix has not been copy-edited.
Summary of the Sponsor’s BIA
The sponsor submitted a BIA that estimated the expected incremental budgetary impact of reimbursing asciminib for the treatment of adult patients with Ph+ CML in CP previously treated with 1 or more TKIs.7
The BIA was conducted from the perspective of public drug plan payers over a 3-year time horizon (2026 to 2028), with 2025 as the base year. The sponsor’s estimate reflects the aggregated results from the jurisdictional provincial budgets (excluding Quebec and the Non-Insured Health Benefits Program). The sponsor estimated the eligible population using a prevalence-based epidemiological approach.7 The sponsor’s base case included drug acquisition costs. The market uptake for asciminib was estimated using the sponsor’s market research analysis.6 The key inputs to the BIA are documented in Table 9.
The sponsor estimated the 3-year incremental budget impact associated with reimbursing asciminib for the treatment of Ph+ CML-CP would be $4,721,611 (year 1 = $754,363; year 2 = $1,557,005; year 3 = $2,410,244).
Parameter | Sponsor’s estimate (reported as year 1 / year 2 / year 3 if appropriate) |
|---|---|
Target population | |
Starting number of people | |
Annual population growth rate | |
Percentage of the adult population | |
Percentage receiving public payer reimbursement | 65.00%7 |
Prevalence of patients with CML | 0.0269%15 |
Percentage of Ph+ | 92.50%16 |
Percentage in CP | 95.00%17 |
Percentage of adults with health access | 85.00%7 |
Percentage of patients initiating a second-line treatment | 26.00%18 |
Number of patients eligible for the drug under review | 947 / 978 / 1,009 |
Market shares (reference scenario) | |
Asciminib | 0% / 0% / 0% |
Bosutinib | ██% / ██% / ██% |
Dasatinib | ██% / ██% / ██% |
Imatinib | ||% / ||% / ||% |
Nilotinib | ||% / ||% / ||% |
Ponatinib | ||% / ||% / ||% |
Market shares (new drug scenario) | |
Asciminib | ██% / ██% / ██% |
Bosutinib | ██% / ██% / ██% |
Dasatinib | ██% / ██% / ██% |
Imatinib | ||% / ||% / ||% |
Nilotinib | ||% / ||% / ||% |
Ponatinib | ||% / ||% / ||% |
Cost of treatment (per patient per 28 days) | |
Asciminib | $5,174 |
Bosutinib | $4,634 |
Dasatinib | $4,026 |
Imatinib | $634 |
Nilotinib | $3,786 |
Ponatinib | $10,684 |
CML = chronic myeloid leukemia; CP = chronic phase; Ph+ = Philadelphia chromosome positive.
Key Issues of the Submitted BIA
CDA-AMC identified several key issues to the sponsor’s analysis that have notable implications on the results of the BIA:
The percentage of public payer reimbursement is uncertain. The sponsor assumed that only ██% of patients in Canada would receive public payer reimbursement.19 The sponsor assumed ██% of adult patients in Ontario and the Atlantic provinces (i.e., New Brunswick, Nova Scotia, Prince Edward Island, and Newfoundland) would receive public coverage, whereas 100% of adult patients in the remaining provinces, except Quebec, would be publicly reimbursed.6 Given the younger age at diagnosis according to the ASC2ESCALATE trial, the sponsor increased private coverage to ██%. Therefore, ██% of adult patients in Canada were assumed to receive public payer reimbursement at baseline year. This approach is subject to uncertainty for several reasons. First, while the resulting estimate is used to refine the eligible population for asciminib under public drug plans, it is based on private claims data from IQVIA and reflects a subset of comparator drug claims selected by the sponsor. The extent to which this data accurately represents the proportion of public versus private claims is unclear, limiting its validity as a proxy for public drug plan eligibility. Second, the approach does not account for age-based differences in public coverage, which are recognized as key determinants of eligibility.20 Third, it does not consider jurisdictional variation in public drug plan eligibility, which further limits the generalizability of the estimate across Canadian provinces.20 As such, the estimated eligible population may not reflect actual reimbursement contexts at the jurisdictional level. Overall, CDA-AMC was unable to validate the sponsor’s approach, as the specific years of claims data and the list of drugs used for validation were not disclosed.
CDA-AMC conducted a base-case reanalysis in which public payer reimbursement was 97.2% across jurisdictions.21
The estimation of the proportion of newly diagnosed adult patients with access to health care is inappropriate. Specifically, the sponsor assumed that ██% of adult patients with newly diagnosed Ph+ CML in CP would have general access to health care across Canada.7 While access-related challenges may exist, this assumption does not reflect an epidemiological criterion and is not appropriate for determining eligibility for asciminib. Moreover, because public payer reimbursement is already incorporated as an access-related parameter, the inclusion of this additional assumption is redundant. It would be more appropriate to apply disease-related criteria that directly influence treatment eligibility.
CDA-AMC excluded this parameter due to its lack of disease specificity. In the revised base case, CDA-AMC assumed that 100% of patients with Ph+ CML in CP would have access to public health care.
The impact of subsequent therapy was excluded from the BIA. The sponsor did not include the cost of subsequent therapies in the budget impact model.2 The sponsor only included the drug acquisition costs for asciminib and comparators in the second-line setting. This is especially relevant because asciminib is indicated for patients with CML in the third-line setting. While the current reimbursement request is for second-line use, subsequent therapy costs remain relevant, as differences in treatment sequences could lead to downstream cost implications. The exclusion of subsequent therapy costs prevents a complete estimation of the budgetary impact associated with asciminib’s adoption in clinical practice.
CDA-AMC was unable to address this limitation owing to the submitted model structure.
The use of a US-based prevalence estimate to estimate adults with CML is inappropriate. The sponsor applied a constant prevalence rate of CML using US data from the Surveillance, Epidemiology, and End Results (SEER) database for adults with CML in 2021.15 These estimates were assumed to increase at the same rate as the population of people living in Canada, and be applicable across both pan-Canadian and provincial levels. While the prevalence estimate was intended to reflect the proportion of individuals currently living with CML, it is subject to several limitations. Specifically, the estimate represents a cross-sectional snapshot of the US adult population in 2021 and was applied directly to the Canadian context without adjustment for differences in demographic characteristics between the 2 populations. Evidence from the literature suggests these estimates are increasing, therefore using a constant rate may underestimate the number of eligible patients.22,23
CDA-AMC was unable to address this limitation owing to the lack of available evidence for the population of people living in Canada.
Time to treatment discontinuation was excluded from the BIA. The sponsor did not incorporate time to treatment discontinuation for asciminib or its comparators in the budget impact model, due to the prevalence-based approach used.7 Instead, treatment costs were calculated by applying monthly drug acquisition costs over a full 12-month period, multiplied by the estimated market share for each treatment. This approach assumes continuous treatment throughout the year and does not reflect real-world discontinuation patterns, which may result in an overestimation of treatment duration and drug acquisition costs.
CDA-AMC was unable to address this limitation owing to the submitted model structure.
CDA-AMC Reanalyses of the BIA
CDA-AMC revised the sponsor’s submitted analyses by making changes in model parameter values and assumptions, in consultation with clinical experts, as outlined in Table 10.
Table 10: Revisions to the Submitted BIA
Stepped analysis | Sponsor’s value or assumption | CDA-AMC value or assumption |
|---|---|---|
1. Adult population living in Canada receiving public payer reimbursement | ██% | 97% |
2. Percentage of adult patients with Ph+ CML in CP with health access | ██% | 100% |
CDA-AMC base case | ― | Reanalysis 1 + 2 |
BIA = budget impact analysis; CDA-AMC = Canada’s Drug Agency; CP = chronic phase; Ph+ CML = Philadelphia chromosome–positive chronic myeloid leukemia.
Note: CDA-AMC was unable to resolve the issues with time to treatment discontinuation and subsequent therapy.
The results of the CDA-AMC step-wise reanalysis are presented in summary format in Table 11 and a more detailed breakdown is presented in Table 12. In the CDA-AMC base case, the 3-year budget impact of reimbursing asciminib for the treatment of adult patients with Ph+ CML in CP previously treated with 1 or more TKIs was $8,306,618 (year 1 = $1,327,132; year 2 = $2,739,201; year 3 = $4,240,284).
Table 11: Summary of the Stepped Analysis of the CDA-AMC Base Case
Stepped analysis | Three-year total ($) |
|---|---|
Submitted base case | 4,721,611 |
CDA-AMC reanalysis 1 | 7,060,625 |
CDA-AMC reanalysis 2 | 5,554,837 |
CDA-AMC base case: (Reanalysis 1 + 2) | 8,306,618 |
CDA-AMC = Canada’s Drug Agency.
Note: The CDA-AMC reanalysis is based on publicly available prices of the comparator treatments.
Table 12: Disaggregated Summary of the BIA
Stepped analysis | Scenario | Year 0 (current situation) ($) | Year 1 ($) | Year 2 ($) | Year 3 ($) | Three-year total ($) |
|---|---|---|---|---|---|---|
Submitted base case | Reference total | 46,950,113 | 48,452,517 | 50,002,997 | 51,603,093 | 150,058,607 |
Asciminib | 0 | 0 | 0 | 0 | 0 | |
All other comparators | 46,950,113 | 48,452,517 | 50,002,997 | 51,603,093 | 150,058,607 | |
New drug total | 46,950,113 | 49,206,879 | 51,560,002 | 54,013,337 | 154,780,218 | |
Asciminib | 0 | 5,883,175 | 12,142,874 | 18,797,168 | 36,823,217 | |
All other comparators | 46,950,113 | 43,323,704 | 39,417,128 | 35,216,168 | 117,957,001 | |
Budget Impact | 0 | 754,363 | 1,557,005 | 2,410,244 | 4,721,611 | |
CDA-AMC base case | Reference total | 82,598,208 | 85,241,350 | 87,969,074 | 90,784,084 | 263,994,508 |
Asciminib | 0 | 0 | 0 | 0 | 0 | |
All other comparators | 82,598,208 | 85,241,350 | 87,969,074 | 90,784,084 | 263,994,508 | |
New drug total | 82,598,208 | 86,568,483 | 90,708,275 | 95,024,368 | 272,301,126 | |
Asciminib | 0 | 10,350,129 | 21,362,666 | 33,069,407 | 64,782,203 | |
All other comparators | 82,598,208 | 76,218,354 | 69,345,609 | 61,954,960 | 207,518,923 | |
Budget Impact | 0 | 1,327,132 | 2,739,201 | 4,240,284 | 8,306,618 |
BIA = budget impact analysis; CDA-AMC = Canada’s Drug Agency.
Note: The CDA-AMC reanalysis is based on the publicly available prices of the comparator treatments.
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.