Drugs, Health Technologies, Health Systems
Reimbursement Review
Brentuximab Vedotin
Requester: Public drug programs
Therapeutic area: Hodgkin lymphoma
Summary
What Is Hodgkin Lymphoma?
Hodgkin lymphoma is a B-cell lymphoid malignancy generally associated with a favourable prognosis (high cure rate and 5-year overall survival [OS] rates exceeding 90%), but it often comes with a high risk of both acute and long-term treatment-related toxicities, which have a significant impact on patients’ quality of life and ability to work, socialize, maintain mental health, and engage in daily routines.
In Canada, the estimated incidence of Hodgkin lymphoma was approximately 3 cases per 100,000 persons, corresponding to around 1,200 new diagnoses and 110 deaths in 2024.
What Are the Treatment Goals and Current Treatment Options for Hodgkin Lymphoma?
The goals for treating Hodgkin lymphoma include prolonging survival, achieving longer remission, controlling symptoms, and improving quality of life. Patients also emphasized the need to minimize side effects, maintain daily functioning and employment, and reduce caregiver burden.
OS, health-related quality of life, and adverse outcomes were identified as important outcomes in the patient group input. Other important outcomes identified through clinician input include progression-free survival and functional outcomes.
Intensive regimens such as BEACOPP (bleomycin, etoposide, doxorubicin [Adriamycin], cyclophosphamide, vincristine [Oncovin], procarbazine, and prednisone); BV + AVD (brentuximab vedotin plus doxorubicin [Adriamycin], vinblastine, and dacarbazine); and BrECADD (brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin [Adriamycin], dacarbazine, and dexamethasone) are commonly used to treat advanced or high-risk classical Hodgkin lymphoma (cHL). ABVD (Doxorubicin [Adriamycin], bleomycin, vinblastine, and dacarbazine) remains a less intensive alternative. PD-1 inhibitors like nivolumab and pembrolizumab are increasingly used in both first-line and salvage therapy settings.
What Is BrECADD and Why Did Canada’s Drug Agency Conduct This Review?
BrECADD is a combined chemotherapy regimen developed to treat advanced-stage cHL, with brentuximab vedotin (an antibody-drug conjugate targeting CD30-positive cells) as its key component. Health Canada has approved BV + AVD for previously untreated patients with advanced-stage Hodgkin lymphoma.
At the request of the participating public drug programs, Canada’s Drug Agency (CDA-AMC) reviewed BrECADD to inform a recommendation on whether it should be reimbursed for adult patients with newly diagnosed, advanced-stage, cHL.
How Did CDA-AMC Evaluate BrECADD?
CDA-AMC reviewed the clinical evidence on the beneficial and harmful effects, and compared costs, of BrECADD versus other treatments used in Canada for adult patients with newly diagnosed, advanced-stage cHL. Other combined interventions, specifically escalated doses of BEACOPP (eBEACOPP), nivolumab plus AVD (doxorubicin [Adriamycin], vinblastine, and dacarbazine), BV + AVD, and ABVD were considered relevant treatments to compare with BrECADD.
The clinical evidence was identified through systematic searches for available studies.
The review was informed by 1 patient group submission and 2 clinician group submissions in response to our call for input, and by input from the participating public drug programs around issues that may impact their ability to implement a recommendation.
Two hematologists representing Atlantic Canada and Ontario were consulted as part of the review process.
What Were the Findings?
Clinical Evidence
CDA-AMC included and reviewed a phase III, multicentre, parallel, open-label, randomized controlled trial (the HD21 trial) comparing the efficacy and safety of BrECADD with eBEACOPP in adults (≤ 60 years) with newly diagnosed, advanced-stage cHL.
BrECADD improved progression-free survival compared to eBEACOPP. Clinical experts indicated that this benefit is clinically meaningful.
The evidence showed similar OS in both groups after the 4-year follow-up and did not support an OS benefit for BrECADD in this trial.
The HD21 trial did not formally compare health-related quality of life (HRQoL) between BrECADD and eBEACOPP. However, BrECADD reduced treatment-related morbidity, which was negatively associated with HRQoL. These findings suggest that BrECADD may be associated with a better HRQoL.
Compared to eBEACOPP, BrECADD demonstrated improved tolerability, particularly in terms of hematologic toxicities such as anemia and thrombocytopenia, and lower transfusion rates. BrECADD also supported better recovery of gonadal function, which may provide meaningful advantages for patients with cHL, especially those concerned about fertility outcomes.
BrECADD was associated with a numerically high incidence of grade 3 or higher adverse events, including neutropenic disorders, fever, infections, cardiac events, and gastrointestinal and hepatobiliary disorders, highlighting the importance of close monitoring and potential dose adjustments.
Uncertainties arose from the open-label nature of the study (which might overestimate the effect of BrECADD).
The generalizability of the findings to specific populations, such as older adults and those with poor Eastern Cooperative Oncology Group Performance Status, remains uncertain due to the lack of comparative data in these subgroups. Based on a single-arm study, BrECADD may be a treatment option for older individuals with cHL. However, comparative evidence remains unavailable in this population.
We did not identify any studies comparing the efficacy and safety of BrECADD with other relevant comparators (e.g., ABVD or BV + AVD) in this patient population.
Economic Evidence
Reimbursing BrECADD for newly diagnosed, advanced-stage, cHL is expected to increase costs to the public drug programs
Abbreviations
ABVD
doxorubicin (Adriamycin), bleomycin, vinblastine, and dacarbazine
AE
adverse event
AVD
doxorubicin (Adriamycin), vinblastine, and dacarbazine
BrECADD
brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone
BV + AVD
brentuximab vedotin plus doxorubicin (Adriamycin), vinblastine, and dacarbazine
cHL
classical Hodgkin Lymphoma
CI
confidence interval
eBEACOPP
escalated doses of bleomycin, etoposide, doxorubicin (Adriamycin), cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisone
ECOG PS
Eastern Cooperative Oncology Group Performance Status
G-CSF
granulocyte colony-stimulating factor
IPS
international prognostic score
ITT
intention to treat
OS
overall survival
PFS
progression-free survival
PP
per-protocol
SAE
serious adverse event
WDAE
withdrawal due to adverse event
Background
Introduction
The objective of the clinical review is to review and critically appraise the evidence on the beneficial and harmful effects of the combined treatment of brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone — BrECADD — in the treatment of newly diagnosed, advanced-stage, classical Hodgkin lymphoma (cHL). The focus will be placed on comparing BrECADD to relevant comparators in clinical practice in Canada and identifying gaps in the current evidence. The economic review consists of a cost comparison for BrECADD compared with relevant comparators for the same population. The comparators considered relevant to the reviews were eBEACOPP (escalated doses of bleomycin, etoposide, doxorubicin [Adriamycin], cyclophosphamide, vincristine [Oncovin], procarbazine, and prednisone), nivolumab plus AVD (doxorubicin [Adriamycin], vinblastine, and dacarbazine), brentuximab vedotin plus AVD (BV + AVD), and ABVD (doxorubicin [Adriamycin], bleomycin, vinblastine, and dacarbazine).
Table 1: Information on the Drug Under Review and on the Canada’s Drug Agency Review
Item | Description |
|---|---|
Information on the drug under review | |
Drug | BrECADD; the key drug in the combination is brentuximab vedotin (IV) |
Relevant Health Canada indication | Previously untreated patients with advanced-stage Hodgkin lymphoma, in combination with doxorubicin, vinblastine, and dacarbazine |
Mechanism of action | Brentuximab vedotin is an antibody-drug conjugate that targets CD30-positive cells. |
Recommended dosage | Previously untreated stage IV Hodgkin lymphoma: 1.2 mg/kg up to 120 mg in combination with AVD, IV over 30 minutes, every 2 weeks for a maximum of 12 doses or unacceptable toxicity. No dosage adjustment is required for mild to moderate renal impairment, but avoid use in severe renal impairment. Dosage adjustments may be necessary for hepatic impairment and peripheral neuropathy, neutropenia, and thrombocytopenia. |
Data protection status | Expired in February 2021 |
Status of generic drugs or biosimilars | None available |
Information on the CDA-AMC review | |
Requester | Oncology Working Group (OWG) |
Indication under consideration for reimbursement | Adult patients with newly diagnosed, advanced-stage, classical Hodgkin lymphoma |
AVD = doxorubicin (Adriamycin), vinblastine, and dacarbazine; BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone; CDA-AMC = Canada’s Drug Agency.
Context for the Review
Brentuximab vedotin (Adcetris) has previously been reviewed by Canada’s Drug Agency (CDA-AMC) for adult patients with relapsed Hodgkin lymphoma, and it was recommended for reimbursement with clinical criteria or conditions.1 In August 2024, CDA-AMC also reviewed brentuximab vedotin for patients with previously untreated advanced-stage Hodgkin lymphoma and issued a positive recommendation for reimbursement with clinical criteria or conditions. In this setting, brentuximab vedotin should be used in combination with AVD in adult patients.2
A review of the evidence for BrECADD in patients with newly diagnosed, advanced-stage cHL was requested by the Oncology Working Group. The request was prompted by emerging evidence for a treatment regimen with potential to fulfill an unmet need in patients with newly diagnosed Hodgkin lymphoma. The data protection for brentuximab vedotin has already expired, so this drug is eligible for a nonsponsored reimbursement review as per the Procedures for Reimbursement Reviews.
Sources of Information
The contents of the clinical review are informed by studies identified through systematic literature searches, input received from interested parties (patient groups, clinician groups, and drug programs), and input from clinical experts consulted for this review.
Calls for patient group, clinician group, and industry input are issued for each nonsponsored reimbursement review. The following submissions were received: 1 patient group submission from Lymphoma Canada, 2 clinician group submissions from Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee (DAC) and the Lymphoma Canada Clinician Group. Lymphoma Canada summarized patient input and provided evidence reflecting patients’ experiences with Hodgkin lymphoma and the BrECADD treatment regimen. Lymphoma Canada collected data through 2 anonymous online surveys. The first survey, conducted from December 16, 2024, to January 16, 2025, received 48 responses. The second survey was conducted from June 1, 2025, to June 26, 2025, and included a single response from a female patient, located in Canada, aged 45 to 54 years, who had been diagnosed with stage IV Hodgkin lymphoma within the past year and had experience using BrECADD. Clinicians from the Lymphoma Canada Clinician Group responded to the question in 1 clinician group submission based on clinical research, professional experience, and their understanding of patients’ needs and challenges. Ontario Health (Cancer Care Ontario) Hematology Cancer DAC also provided clinical group input for the review. The full submissions received are available on the project landing page in the consolidated input document.
Input from patient and clinician groups is considered throughout the review, including in the selection of outcomes to include in the clinical review and in the interpretation of the clinical evidence. Relevant patient and clinician group input is summarized in the Disease Background, Current Management, and Unmet Needs and Existing Challenges sections.
The drug programs provide input on each drug being reviewed through the reimbursement review process by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted for this review are summarized and provided to the expert committee in a separate document.
Each review team includes at least 1 clinical expert with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process. Two hematologists with expertise in the diagnosis and management of Hodgkin lymphoma participated as part of the review team with representation from Atlantic Canada and Ontario.
Disease Background
Hodgkin lymphoma is a B-cell lymphoid malignancy characterized by a low number of malignant cells, a high cure rate, and 5-year overall survival (OS) rates exceeding 90%.3 However, these excellent outcomes often come with a high risk for both acute and long-term treatment-related toxicities.3 Hodgkin lymphoma is classified into 2 main subtypes: classical Hodgkin lymphoma (cHL), which accounts for at least 90% of patients, and nodular lymphocyte-predominant Hodgkin lymphoma. cHL accounts for approximately 15% to 25% of all lymphomas, 0.5% of all cancers, and 0.2% of all cancer deaths.4 In Canada, the estimated incidence of Hodgkin lymphoma was approximately 3 cases per 100,000 persons in 2024, corresponding to approximately 1,200 new diagnoses and 110 deaths.5
The pathologic hallmark of cHL is the presence of large, multinucleated malignant cells, known as Hodgkin and Reed-Sternberg cells, which express the CD30 surface antigen.4 Most patients with cHL present between age 15 and 35 years and with asymptomatic lymphadenopathy or a mediastinal mass detected on chest imaging. Approximately 40% of patients experience constitutional “B” symptoms, including fever, night sweats, and unintentional weight loss. Other clinical symptoms may include fatigue, pruritus, alcohol-associated pain, and skin lesions. The diagnosis of cHL often requires an excisional biopsy because fine needle aspiration is considered insufficient.4
Patient group input: Patient surveys conducted by Lymphoma Canada indicated that Hodgkin lymphoma had a significant impact on patients’ quality of life, including persistent fatigue, anxiety, and stress, along with physical symptoms such as body aches and pains, indigestion, abdominal pain or bloating, itching, and night sweats. For example, among 48 patients in the survey, the impact of the disease was substantial, with 79% of patients experiencing stress, anxiety, or worry; 61% reporting a fear of disease progression; 47% reporting a fear of not being able to continue daily activities; and 42% having difficulty sleeping. These challenges affected their ability to work, socialize, and maintain mental health and daily routines, often leading to emotional distress, sleeping disorders, and financial strain.
Current Management
Treatment Goals
Patient group input: Based on the patient input submission from Lymphoma Canada, the important treatment goals for treating Hodgkin lymphoma include prolonging survival, achieving longer remission, controlling symptoms, and improving quality of life. Patients also emphasized the need to minimize side effects, maintain daily functioning and employment, and reduce caregiver burden. Many patients were willing to tolerate mild, short-term side effects in exchange for better outcomes and valued having more treatment options and personal choice in therapy decisions.
Clinician input: Based on the 2 clinician input submissions, the important treatment goals for Hodgkin lymphoma include improving progression-free survival, OS, and quality of life, reducing relapse, and ensuring better tolerability with fewer toxicities and minimizing the need for second-line therapy. The 2 clinician groups emphasized the need for better first-line therapies that not only extend survival outcomes but also maintain quality of life by reducing treatment-related side effects and preserving patients’ ability to function independently.
Current Treatment Options
Current treatment options for cHL include a combination of nonpharmacological and pharmacological interventions. Nonpharmacological interventions include radiation therapy, PET-guided treatment adaptation, and stem cell transplant (autologous or allogeneic).6 Standard pharmacological treatments involve chemotherapy regimens such as ABVD, often combined with radiation therapy for early-stage disease. For advanced or high-risk cases, more intensive regimens, such as BEACOPP or newer combinations like BV + AVD or BrECADD, are used. PD-1 inhibitors, including nivolumab and pembrolizumab, are increasingly incorporated into first-line and salvage therapies.
Key characteristics of brentuximab vedotin are summarized with other treatments available for adult patients with newly diagnosed, advanced-stage, cHL in the Key Characteristics table in Appendix 1 of the Supplemental Material document (available on the project landing page).
Unmet Needs and Existing Challenges
Patient group input: Patient input was collected from 2 patient surveys conducted by Lymphoma Canada. In the first survey, adult patients (aged 18 to 74 years, n = 48) with Hodgkin lymphoma provided the responses, with most participants diagnosed with stage III or IV cHL. The second survey provided responses regarding the experience with BrECADD of 1 female patient in Canada who was aged 45 to 54 years, was diagnosed with stage IV cHL less than a year ago, accessed treatment locally via private insurance as a first-line therapy, and is currently still receiving this therapy.
Patients frequently reported challenges related to both diagnosis and access to treatment. Delays in diagnosis were often attributed to misidentification of symptoms and prolonged wait times for biopsies. Access to treatment was inconsistent across different geographic locations. Some patients had to travel long distances to receive care, and others faced financial barriers. These financial obstacles were often due to high drug costs, the need for supplementary medications, and inadequate insurance coverage. One female patient expressed a preference for the BrECADD regimen but was unable to receive it due to a confusing and difficult access process.
Survey respondents from Lymphoma Canada were asked to report the number of treatment lines they had received for Hodgkin lymphoma. Most patients reported receiving either 1 (38%) or 2 or more (54%) lines of therapy. When asked about their satisfaction with the available treatment options, 75% of patients reported that they were satisfied or very satisfied with their first-line choices. However, 25% reported being dissatisfied or very dissatisfied with the options available to them in the first-line setting.
An important concern for patients was the side effects associated with treatment and their impacts on quality of life. Commonly reported difficult-to-tolerate AE included fatigue, hair loss, bone or muscle pain, nausea, loss of appetite or weight loss, constipation, body aches and pain, mouth sores, and diarrhea. These side effects frequently disrupted patients' daily activities, impeding their ability to work, engage in physical exercise, or fulfill familial responsibilities. The financial strain was also a significant issue, with 67% of respondents reporting workforce absences. Many patients reported additional costs for supplementary drugs to manage side effects (50%), to travel to treatment centres (50%), for the primary drug therapy (50%), and for essential medical supplies (42%).
Twelve patients provided information regarding their ability to access treatment for Hodgkin lymphoma. Six of these patients (50%) reported little to no difficulty in accessing care. The other half experienced challenges: 4 patients (33%) reported some difficulty, and 2 (17%) reported significant difficulty. Among patients who faced access barriers, the most common reasons reported were the treatment not being available at their local cancer centre (45%) or living in a community that lacked a cancer centre (27%).
Overall, patients with Hodgkin lymphoma in Canada reported several unmet needs despite available treatments. Many patients reported that currently available therapies are not effective or tolerable enough, especially for advanced or relapsed disease, and they called for more targeted, less toxic treatment options. Most patients indicated that having a choice in their treatment decisions and access to a variety of therapeutic options are of high importance. Access to newer treatments (e.g., BrECADD) is inconsistent due to financial strains, funding, geographic, or availability issues at local cancer centres, particularly affecting those in rural or remote areas. These issues underscore the need for enhanced access and more effective and tolerable treatment options, including the first-line therapy for patients with newly diagnosed, advanced-stage, cHL.
Clinician input: There is a need to seek more effective, safer, and more accessible first-line options to reduce reliance on second-line therapies. Regimens with improved tolerability and fewer toxicities, which minimize treatment interruptions, are needed. For example, clinicians may want to limit exposure to anthracyclines due to their side effects. Access to some regimens (e.g., nivolumab + AVD) remains uncertain, particularly for stage IIB patients who were not included in the pivotal trial. BrECADD has the potential to be preferred in specific patient groups (e.g., stage IIB with mediastinal bulk or extranodal disease) due to its shorter PET-directed treatment duration and improved safety profile compared to alternative options.
Considerations for Using the Drug Under Review
Contents within this section have been informed by input from the clinical experts consulted for the purpose of this review and from clinician groups. The following has been summarized by the review team.
Place in Therapy
BrECADD is a chemotherapy regimen developed for the treatment of advanced-stage cHL. The acronym stands for the combination of the following drugs:
brentuximab vedotin
etoposide
cyclophosphamide
doxorubicin (Adriamycin)
dacarbazine
dexamethasone.
Clinical experts consulted for this review indicated that BrECADD would be used as a first-line treatment for adults with newly diagnosed cHL who have advanced-stage disease. The regimen is administered as a combination chemotherapy protocol and is intended to be used with curative intent rather than to manage symptoms alone. BrECADD is not expected to be used in combination with other systemic therapies beyond its defined regimen, but it may be followed by radiotherapy depending on treatment response.
In their submission, the clinician group agreed with the clinical experts that BrECADD would be used as a first-line treatment for patients with Hodgkin lymphoma stage II with B symptoms who have either 1 or both risk factors of large mediastinal mass and extranodal involvement, stage III and stage IV. Ontario Health (Cancer Care Ontario) Hematology Cancer DAC noted that BrECADD would be an alternative to nivolumab + AVD for patients with previously untreated stage III and IV disease, and an alternative to ABVD for patients with stage IIB to IV disease. They further emphasized that BrECADD would be the preferred option over eBEACOPP if an intensive regimen is indicated, and that it fills a treatment gap for patients with stage IIB bulky disease, whose disease is often treated as if it is at an advanced stage but who currently have limited access to newer regimens. Both clinician submissions stated that BrECADD is expected to shift the current treatment paradigm in Canada due to its favourable balance of efficacy and safety in appropriately selected patients.
Patient group input: One patient from Canada, aged 45 to 54 years, who was diagnosed with stage IV cHL less than a year ago, accessed the BrECADD regimen locally via private insurance as a first-line therapy, and is currently undergoing this treatment, described the experience as “good” and indicated a willingness to recommend it to others as a first-line therapy. This positive view was maintained despite experiencing challenging side effects, such as bone pain and infections. The other main side effects reported include neutropenia, low blood pressure, irregular heartbeat or cardiac complications, and diarrhea. The psychological impact on this patient included anxiety or worry, a fear of disease progression or relapse, and challenges receiving support from friends and family.
Patient Population
Clinical experts consulted for this review indicated that BrECADD is intended for use in adults with newly diagnosed cHL who present with advanced-stage disease. This includes patients with stage IIB disease with or without mediastinal bulk or extranodal involvement, stage III, and stage IV. These patients typically require curative-intent therapy and are considered appropriate candidates for intensive chemotherapy regimens. The experts emphasized that BrECADD is best suited for patients who are “young and fit” with good performance status and no significant comorbidities. The experts noted that the median age of diagnosis for cHL is approximately 40 years, and many patients are generally healthy and active, making long-term toxicity a key consideration in treatment selection. Patients with high-risk features, such as bulky disease or extranodal involvement, may have the greatest need for an intervention like BrECADD, particularly when the goal is to maximize efficacy while minimizing long-term toxicity.
The clinical experts did not identify a companion diagnostic or biomarker requirement for initiating BrECADD. Diagnosis and staging are based on histological confirmation (via core or excisional biopsy), PET imaging, and clinical evaluation. PET scans are used both for initial staging and to guide treatment decisions, particularly through an interim PET assessment typically performed after 2 cycles of treatment (called PET2). cHL is typically diagnosed with high confidence using standard imaging and pathology tools available in routine clinical practice. The experts did not raise issues related to diagnostic uncertainty or misdiagnosis, and no genetic or molecular markers were identified to determine eligibility
In their submission, the clinician group agreed with the clinical experts regarding the primary target population. They added that, although BrECADD was studied in patients aged 60 years or younger, it may be considered for select patients aged 61 to 75 years if they have adequate performance status and manageable comorbidities. In this older population, more frequent dose adjustments may be necessary to mitigate treatment-related toxicity. The Lymphoma Canada clinician group did not recommend BrECADD for patients older than 75 years.
Neither the clinical experts nor the clinician group identified presymptomatic patients as candidates for BrECADD. Both groups emphasized that treatment should be initiated only in patients with confirmed, active disease requiring systemic therapy. Patients who are the least suitable for BrECADD include those with poor performance status, with significant comorbidities, or of an advanced age (particularly older than age 75), in which cases the risks of intensive chemotherapy may outweigh the potential benefits.
Assessing the Response to Treatment
Clinical experts consulted for this review indicated that treatment response for patients with cHL should be assessed using PET scans and clinical evaluations. PET scans included interim PET and end-of-treatment PET. Interim PET imaging, typically performed after 2 cycles of treatment (i.e., PET2), is used to guide the duration of therapy. End-of-treatment PET scans are also used to evaluate residual disease and guide further management. Besides imaging findings, clinical experts emphasized that a clinically meaningful response to treatment includes improvement of symptoms and signs of lymphoma; preservation of quality of life and personal activities; minimizing toxicities; and maximizing cure, remission, or survival. The clinical experts did not suggest other assessments.
The clinician group submission agreed with the clinical experts and provided additional detail. They confirmed that PET2 guidance is used to determine whether patients should receive 4 or 6 cycles of BrECADD and that end-of-treatment PET scans are used to assess the need for consolidative radiotherapy. They also noted that PET positivity is generally defined as a score of 4 or higher in the Deauville scale, indicating residual metabolic activity.
Discontinuing Treatment
Clinical experts consulted for this review stated that BrECADD should be discontinued in cases of disease progression, if intolerable or recurrent AE that are not manageable through dose modifications, or based on patient preference. The clinician group submission agreed with the experts and added that specific toxicities, such as significant neuropathy or cardiotoxicity, should prompt discontinuation. They also highlighted that progression of disease and considerable toxicities are key factors in treatment discontinuation. For patients with a negative PET2 scan, early discontinuation after 4 cycles is appropriate to reduce cumulative toxicity without compromising efficacy.
Prescribing Considerations
Clinical experts consulted for this review recommended that BrECADD be prescribed and monitored by hematologists or oncologists with expertise in lymphoma. Treatment is typically delivered in an outpatient setting. A multidisciplinary care team, including a hematologist or oncologist, general practitioners in oncology, nurse practitioners, and pharmacists, may support treatment delivery and toxicity management. The clinician group submissions agreed with this approach and emphasized that malignant hematologists are required to monitor patients receiving BrECADD. They agreed that BrECADD can be administered in outpatient settings, and pointed out that inpatient initiation of these regimens may be necessary in urgent cases.
Additional Considerations
Clinical experts consulted for this review noted that although BrECADD offers improved tolerability compared to other intensive regimens, it may have greater toxicity than less intensive options such as ABVD. They also highlighted that the limited evidence supports the use of BrECADD in patients aged 60 years and older or those with significant comorbidities. The experts emphasized the importance of careful patient selection and monitoring to balance treatment efficacy with long-term safety.
The Ontario Health (Cancer Care Ontario) Hematology Cancer DAC submission noted that brentuximab vedotin monotherapy should remain available as a downstream option for patients whose disease has relapsed, particularly those with a disease-free interval of 12 months or greater. This approach aligns with other first-line brentuximab-containing regimens that allow for re-treatment.
Lymphoma Canada advocates for equitable access to novel lymphoma therapies, prioritizing patient choice and the removal of barriers to care.
Clinical Review
Methods
The review team conducted a systematic review to identify evidence for BrECADD for the treatment of adult patients with newly diagnosed, advanced-stage, cHL. Studies were selected according to the eligibility criteria in Table 2. Long-term extension studies of included randomized controlled trials, indirect treatment comparisons (ITCs) that adhered to the eligibility criteria (except for the study design criteria), and studies addressing gaps that did not meet the eligibility criteria but were considered to address important gaps in the systematic review evidence were included.
Relevant comparators included treatments used in clinical practice in Canada in the patient population under review. Clinical expert input and patient and clinician group input were considered when selecting outcomes (and follow-up times) for review. Selected outcomes are those considered relevant to expert committee deliberations. Detailed methods for the literature searches, study selection, data extraction, and risk of bias appraisal are in Appendix 2 of the Supplemental Material document.
Table 2: Systematic Review Eligibility Criteria
Criteria | Description |
|---|---|
Population | Adult patients with newly diagnosed, advanced-stage, classical Hodgkin lymphoma |
Intervention | BrECADD |
Comparator | Other combined interventions
|
Outcomes | Efficacy outcomes:
Safety outcomes:
|
Study design | Phase III and IV RCTs |
ABVD = doxorubicin (Adriamycin), bleomycin, vinblastine, and dacarbazine; AE = adverse events; AVD = doxorubicin (Adriamycin), vinblastine, and dacarbazine; BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone; BV + AVD = brentuximab vedotin plus doxorubicin (Adriamycin), vinblastine, and dacarbazine; eBEACOPP = escalated doses of bleomycin, etoposide, doxorubicin (Adriamycin), cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisone; HRQoL = health-related quality of life; RCT = randomized controlled trial; SAE = serious adverse event; WDAE = withdrawal due to adverse events.
Note: Advanced-stage refers to Ann Arbor stage III or IV, stage II with B symptoms, and either 1 or both risk factors of large mediastinal mass and extranodal lesions.
Clinical Evidence
An information specialist conducted a literature search of key bibliographic databases, trial registries, and grey literature sources, using a peer-reviewed search strategy. The initial search was completed on July 3, 2025. From the search for primary studies, we identified 1,408 unique records, of which we excluded 1,401 by title and abstract. We screened 7 records by full text and included 2 reports of 1 study (the HD21 trial).7,8 We did not identify any potentially relevant records via other sources. No reports of long-term extensions of the included study or studies addressing gaps were identified.
From the search for ITCs, we identified 60 unique records via the searches of databases, of which we excluded 58 by title and abstract. We screened 2 records by full text, of which none met the eligibility criteria by full-text screening. We did not identify any potentially relevant records via other sources. Therefore, we did not include any relevant ITCs in this report.
From the regular search alert, we identified 1 report of a single-arm study aimed at addressing evidence gaps in the use of BrECADD for older adults with cHL.9
A list of excluded studies, including reasons for exclusion, is in Appendix 2 of the Supplemental Material document.
Systematic Review
Study Characteristics
Characteristics of the included study (HD21 trial)7 are summarized in the following paragraphs and in Table 3. Additional details regarding the inclusion and exclusion criteria, interventions and comparators, and relevant outcome measures are in Appendix 3 of the Supplemental Material document.
Study Design
The HD21 trial conducted by Borchmann et al. (2024)7 is a randomized, multicentre, parallel-group, open-label, phase III trial conducted by the German Hodgkin Study Group in collaboration with several international cooperative groups, including the Swiss Group for Clinical Cancer Research, Arbeitsgemeinschaft Medikamentöser Tumortherapie, the Nordic Lymphoma Group, and the Australasian Leukaemia and Lymphoma Group. The trial was funded by Takeda Oncology and conducted across 233 sites in 9 countries: Australia, Austria, Denmark, Germany, the Netherlands, New Zealand, Norway, Sweden, and Switzerland. The study aimed to evaluate the efficacy and tolerability of PET-guided BrECADD versus eBEACOPP in adult patients with newly diagnosed, advanced-stage cHL.
Population
The HD21 trial enrolled patients aged 18 to 60 years with histologically confirmed cHL at advanced stages (Ann Arbor stage III or IV or stage II with B symptoms and 1 or both risk factors of large mediastinal mass [≥ a third of the maximum thoracic diameter] or extranodal involvement).7 Eligible patients had an Eastern Cooperative Oncology Group Performance Status (ECOG PS) of 0 to 2, were HIV-negative, and had no concurrent conditions that would preclude protocol treatment. Central pathology review confirmed diagnoses postenrolment. Patients were randomly assigned in a 1:1 ratio to receive either BrECADD or eBEACOPP, with randomization stratified by recruitment region (Europe versus Australia and New Zealand), age (< 45 versus ≥ 45 years), sex (female versus male), and International Prognostic Score (IPS) (0 to 2 versus 3 to 7).
Patients and investigators were not blinded to treatment allocation. Imaging assessments were performed after cycle 2 and at the end of treatment by a multidisciplinary expert panel.
Intervention and Comparator
The HD21 trial7 investigated 2 chemotherapy regimens for patients with advanced-stage cHL: the eBEACOPP and the BrECADD. Both regimens were administered in 21-day cycles, with supportive care that included the mandatory administration of granulocyte colony-stimulating factor (G-CSF) to mitigate neutropenia. The trial employed PET-guided therapy, with patients undergoing interim PET scans after 2 cycles. Those with negative scans (Deauville score 1 to 3) received a total of 4 cycles, whereas patients who were PET-positive (Deauville score 4 or higher) received 6 cycles, allowing for individualized treatment duration based on early response.
The eBEACOPP regimen was designed for aggressive disease control but was associated with significant toxicity, including pulmonary complications, neuropathy, gonadal damage, and a high risk of secondary malignancies such as acute myeloid leukemia and myelodysplastic syndrome.
To address these concerns, the BrECADD regimen introduced several modifications. Bleomycin and vincristine were removed to reduce the risk of pulmonary toxicity and peripheral neuropathy, respectively. Procarbazine and prednisone were also excluded to minimize gonadal toxicity and long-term metabolic side effects. BrECADD incorporated dacarbazine and dexamethasone, which offer similar cytotoxic and anti-inflammatory effects with a more favourable toxicity profile than eBEACOPP.
A key difference in BrECADD was the inclusion of brentuximab vedotin, an antibody-drug conjugate targeting CD30, a marker highly expressed in Hodgkin lymphoma cells. Brentuximab vedotin was administered at a dose of 1.8 mg/kg (up to a maximum of 180 mg) on day 0 of each cycle. Other adjustments included a reduction in the dose of etoposide from 200 mg/m2 in eBEACOPP to 150 mg/m2 in BrECADD, aimed at lowering hematologic toxicity and reducing the need for transfusions. The dose of doxorubicin was slightly increased from 35 mg/m2 to 40 mg/m2 to maintain antitumour efficacy. We summarized the treatment adjustment and the differences between eBEACOPP and BrECADD in Appendix 3 of the Supplemental Material document.
Cyclophosphamide was retained at the same dose in both regimens. BrECADD and eBEACOPP were initiated at full-dose level 4, with mandatory G-CSF support and predefined dose reductions based on toxicity. Consolidative radiotherapy was recommended for PET-positive residual disease.
Outcomes
The primary end points were treatment-related morbidity (defined as investigator-assessed treatment-related morbidity from the start of therapy to 30 days after the end of chemotherapy, including Common Terminology Criteria for Adverse Events (CTCAE) grade 3 or 4 nonhematologic organ toxicity or grade 4 hematologic toxicity) and progression-free survival (PFS), defined as time from randomization to progression, relapse, or death from any cause, or censored at the date of last information on the disease status, with central review of all tumour events.7 Secondary end points relevant to this review included OS, adverse events (AEs) (including any grade, serious AEs [SAEs], withdrawals due to AEs [WDAEs], and death due to AEs), gonadal toxicity, second malignancies, and health-related quality of life (HRQoL).
Table 3: Characteristics of Study Included in the Systematic Review
Study name, design, and sample size | Key inclusion criteria | Key exclusion criteria | Interventions and comparators | Relevant end points |
|---|---|---|---|---|
Borchmann et al. (2024)7 HD21 trial Phase III open-label, parallel, multicentre RCT Setting: 233 trial sites across 9 countries: Australia, Austria, Denmark, Germany, the Netherlands, New Zealand, Norway, Sweden, and Switzerland. N = 1,500 |
|
| Two treatment arms:
| Primary end point:
Secondary end points:
|
AE = adverse event; BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone; cHL = classical Hodgkin lymphoma; eBEACOPP = escalated doses of bleomycin, etoposide, doxorubicin (Adriamycin), cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisone; ECOG PS = Eastern Cooperative Oncology Group Performance Status; HRQoL = health-related quality of life; OS = overall survival; PFS = progression-free survival; RCT = randomized controlled trial; TNM = tumour, mode, metastasis.
aDefinition of advanced stages included Ann Arbor stage III or IV as well as stage II with B symptoms and 1 or both risk factors of large mediastinal mass (≥ a third of the maximum thoracic diameter) or extranodal lesions.
bExceptions: Basal cell carcinoma, carcinoma in situ of the cervix uteri, completely resected melanoma TNM, pT1.
Source: Borchmann et al. (2024).7
Statistical Testing and Analysis Populations
The coprimary end points of the trial include treatment-related morbidity and PFS.7 The primary objective for treatment-related morbidity was to demonstrate that the BrECADD regimen reduced toxicity compared to eBEACOPP. Investigators compared treatment-related morbidity rates between treatment groups using the Cochran-Mantel-Haenszel test, stratified by recruitment area, IPS, age, and sex. A 2-sided significance level of 0.05 was applied. BrECADD was deemed superior if the test showed statistical significance and a lower treatment-related morbidity rate than eBEACOPP.
After confirming reduced treatment-related morbidity, the coprimary objective shifted to evaluating BrECADD’s noninferiority in efficacy, measured by PFS. Noninferiority was defined as an absolute difference of less than 6% in 5-year PFS, corresponding to a hazard ratio (HR) of less than 1.69 for BrECADD versus eBEACOPP. The noninferiority analysis used a 2-sided 95% confidence interval (CI) for the HR from a Cox regression model, incorporating the same stratification factors as the treatment-related morbidity analysis. This corresponded to a 1-sided significance level of 0.025.
An interim PFS analysis was conducted at a median follow-up of 36 months, applying O’Brien-Fleming boundaries with a Lan-DeMets alpha-spending function to preserve the overall type I error rate (a 2-sided significance level of 0.0108 for noninferiority). The interim analysis, completed in March 2023, confirmed the noninferiority of BrECADD. Then, the protocol was amended (version 10, dated December 8, 2023) to include a superiority test for PFS in the final analysis, adjusted for alpha spent during the interim analysis (a 2-sided significance level of 0.0392). The final analysis was set at 48 months of follow-up. The clinical experts consulted by the review team indicated that a 4-year follow-up for PFS is reasonable for patients with cHL. Secondary end points were analyzed descriptively. An unplanned secondary analysis of fertility outcomes (e.g., pregnancy and parenthood rates) was conducted to assess the recovery of gonadal function.
The authors of the trial defined several analysis populations. The intention-to-treat (ITT) set included all randomized patients whose diagnosis of cHL was confirmed by the reference pathology panel. This population served as the primary analysis set for both treatment-related morbidity and PFS. The per-protocol (PP) dataset included ITT patients who met all eligibility criteria and adhered to the trial protocol. Patients were excluded from the PP set if they switched treatment groups, received fewer than 5 or more than 7 chemotherapy cycles, were given nonprotocol antineoplastic agents, or received inappropriate irradiation based on PET findings. Deviations due to toxicity, progression, or death did not lead to exclusion. The PP analysis served as a sensitivity analysis for both primary end points. The number of patients in the PP dataset was not explicitly reported. The safety analysis set included all patients who received at least 1 documented chemotherapy cycle.
To reflect gonadal toxicity and function, the authors of the study also reported the fertility outcomes. For the unplanned fertility analysis, a specific cohort of patients of childbearing potential (POCBP) was defined. This cohort included women younger than 40 years with a baseline follicle-stimulating hormone less than 25 U/L and men younger than 50 years with a baseline follicle-stimulating hormone less than 15 U/L. Patients with baseline gonadal dysfunction were also included in pregnancy and parenthood analyses.
Patient Disposition
Between July 22, 2016, and August 27, 2020, a total of 1,500 patients with newly diagnosed advanced-stage cHL were enrolled in the HD21 trial.7 Of these, 749 patients were randomly assigned to receive eBEACOPP and 751 to BrECADD. Following central pathology review, 9 patients in each group were excluded due to revised diagnoses, resulting in an ITT population of 1,482 patients: 740 in the eBEACOPP group and 742 in the BrECADD group. Twelve patients were further excluded from the safety analysis due to missing documentation of chemotherapy administration or withdrew consent before the start of treatment: 8 from the eBEACOPP group and 4 from the BrECADD group. This left 732 patients in the eBEACOPP group and 738 in the BrECADD group for safety analyses. In the POCBP cohort for fertility analysis, a total of 1,183 patients were enrolled: 592 in the eBEACOPP group and 591 in the BrECADD group, comprising 692 men and 491 women.
Baseline Characteristics
The patients in the HD21 trial7 were adults with newly diagnosed advanced-stage cHL (IIa to IVb). The disease was characterized by high-risk features, including extensive nodal involvement, extranodal lesions, and large mediastinal masses. Most had stage III or IV disease, or stage II with systemic symptoms and additional risk factors. Prognostic assessment was based on the IPS, with a broad distribution across low, intermediate, and high-risk categories. The majority had good performance status, and comorbidities were limited by exclusion criteria to ensure treatment feasibility. The disease duration was not reported, but it was likely short, given that only newly diagnosed cases were included. Table 4 presents a summary of baseline characteristics of participants in the trial.
A subpopulation of POCBP (n = 1,183: 592 in the eBEACOPP arm and 591 in the BrECADD arm) was analyzed for fertility outcomes, which included men younger than 50 years (n = 692) and women younger than 40 years (n = 491) at enrolment with normal baseline gonadal function.8 The HD21 trial also included a nonrandomized cohort of older patients (n = 85) treated with BrECADD, analyzed separately using descriptive statistics.9
Table 4: Summary of Baseline Characteristics of the Study Included in the Systematic Review
Characteristic | eBEACOPP (N = 740) | BrECADD (N = 742) |
|---|---|---|
Age, median (IQR), years | 31 (24 to 42) | 31 (24 to 42) |
Sex, n (%) | ||
Female | 321 (44%) | 323 (44%) |
Male | 419 (56%) | 419 (56%) |
Race, n (%) | ||
Asian | 13 (2%) | 11 (1%) |
Black | 2 (<1%) | 0 |
White | 672 (91%) | 680 (92%) |
Other or unknown | 53 (7%) | 51 (7%) |
ECOG PS, n (%) | ||
0 | 517/735 (70%) | 508/739 (69%) |
1 | 200/735 (27%) | 220/739 (30%) |
2 | 18/735 (2%) | 11/739 (1%) |
International Prognostic Score | ||
0 to 1 | 175 (24%) | 195 (26%) |
2 to 3 | 395 (53%) | 390 (53%) |
4 to 7 | 170 (23%) | 157 (21%) |
Ann Arbor stage, n (%) | ||
IIa | 0 | 2 (< 1%) |
IIb | 117/739 (16%) | 115 (16%) |
IIIa | 132/739 (18%) | 129 (17%) |
IIIb | 156/739 (21%) | 164 (22%) |
IVa | 112/739 (15%) | 104 (14%) |
IVb | 222/739 (30%) | 228 (31%) |
Risk factors | ||
Large mediastinal mass | 235/735 (32%) | 253/739 (35%) |
Extranodal involvement | 175/735 (24%) | 202/739 (27%) |
Involvement of 3 or more nodal areas | 655/735 (89%) | 667/739 (90%) |
High erythrocyte sedimentation rate | 470/709 (66%) | 481/708 (68%) |
BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone; cHL = classical Hodgkin lymphoma; eBEACOPP = escalated doses of bleomycin, etoposide, doxorubicin (Adriamycin), cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisone; ECOG PS = Eastern Cooperative Oncology Group Performance Status; IQR = interquartile range.
Note: Some variables were reordered and age group data were omitted.
Source: Borchmann P, Ferdinandus J, Schneider G, et al. Assessing the efficacy and tolerability of PET-guided BrECADD versus eBEACOPP in advanced-stage, classical Hodgkin lymphoma (HD21): a randomised, multicentre, parallel, open-label, phase III trial. Lancet. 2024;404(10450):341 to 352. Copyright 2024 by the authors. Available from: https://www.sciencedirect.com/science/article/pii/S0140673624013151?via%3Dihub.7 Reprinted in accordance with Creative Commons Attribution 4.0 International License (CC BY 4.0): https://creativecommons.org/licenses/by/4.0/.
Treatment Exposure and Concomitant Treatments
Treatment adherence was high in both groups, with 708/742 (96%) of patients in the BrECADD arm and 707/740 (95%) in the eBEACOPP arm completing the scheduled number of cycles.7 PET-2-guided treatment was applied in 91% (1,346/1,482) of the ITT population across both groups. Among those, 430 (64%) of patients in each arm had a negative PRT-2 and were assigned to 4 cycles of intervention only. Full-dose treatment at cycle 4 was more frequent in the BrECADD group (561/721, 78%), compared to eBEACOPP (422/720, 59%). Among PET-2-positive patients, 191 (67%) of 284 patients in the BrECADD group received full-dose treatment at cycle 6 versus 119 (43%) of 280 patients in the eBEACOPP group. The most common reasons for dose reductions were thrombocytopenia in the BrECADD group (147/635, 23%) and leukopenia in the eBEACOPP group (241/715, 34%). Early discontinuation of key drugs was lower in the BrECADD group (2%, 18/738 for brentuximab vedotin) compared to the eBEACOPP group (18%, 132/732) for vincristine.
Consolidative radiotherapy was recommended by central review for 17% of patients in each group due to PET-positive residual disease and was administered to 15% of patients in the eBEACOPP group and 14% in the BrECADD group. The administration of G-CSF and other support treatments was not reported in the included study.
Critical Appraisal
Internal Validity
The HD21 trial7 investigators randomized participants in a 1:1 ratio using a central stratified minimization method to assign participants to either the eBEACOPP or BrECADD group. Stratification factors included country of enrolment, age (< 45 versus ≥ 45 years), sex (female versus male), and IPS (0 to 2 versus 3 to 7). The use of centralized random sequence generation and allocation concealment is likely related to a low risk of bias. Baseline characteristics were well balanced between the 2 treatment groups (eBEACOPP and BrECADD), which also indicates that the randomization process performed well and probably had a low risk of bias.
The open-label design of the HD21 trial7 may have introduced bias given that investigators and patients were aware of the treatment assignment. This lack of blinding likely overestimated the treatment effect of BrECADD, particularly for patient-reported outcomes, such as HRQoL. However, the central pathology review and validation of the primary efficacy outcomes strengthen the credibility of the study's main findings.
The HRQoL analysis presents additional concerns. The authors of the study did not report the psychometric properties of the HRQoL measures (EORTC QLQ-C30), leaving uncertainty about the reliability, validity, and responsiveness of the instrument in this context. Furthermore, the HRQoL analysis was based on the complete dataset and assumed that missing values occurred completely at random. However, this assumption may be invalid because patients with severe conditions may have been unable or unwilling to respond to HRQoL questions. Their analysis examined the impact of treatment-related morbidity on HRQoL, adjusting for age, sex, and baseline scores. However, the authors did not report the comparative effects of BrECADD versus eBEACOPP on HRQoL, which is more directly relevant to our research question.
Time-to-event analyses were conducted using HRs, with the proportional hazards assumption assessed using Schoenfeld residuals. However, the authors of the trial did not report the results for the test. We cannot fully evaluate the validity of the proportional hazards assumption underlying the HRs from the time-to-event analyses. Visual inspection of the Kaplan-Meier plots suggests that the assumption is likely held for PFS (Figure 1A), but the plots for OS show no separation at any point during follow-up (Figure 1B), raising concerns about the applicability of the assumption in this context.
The HD21 trial7 did not report the proportion of patients who discontinued the study or the reasons for censoring. We cannot determine the number of patients who were censored due to loss to follow-up or initiation of other treatments before experiencing an event. The authors of the study did not explicitly report the use of concomitant treatments such as G-CSF.7 We cannot determine how concomitant treatments may have influenced the observed effects of BrECADD. The authors of the trial used PFS as the primary end point to evaluate the efficacy of BrECADD compared to eBEACOPP. However, the validity of PFS as a surrogate for OS in this patient population remains uncertain.
The fertility outcomes analysis was stated as an “unplanned secondary analysis” in a separate report. After the interim analysis in March 2023 successfully demonstrated noninferiority of BrECADD for PFS, the protocol was further amended (10th protocol, December 8, 2023) to introduce a superiority test for PFS.7 The shift from a noninferiority hypothesis to a superiority hypothesis after observing positive noninferiority results, the “unplanned secondary analysis,” and multiple analyses (i.e., interim analyses) at different time points may be indicators of potential selective reporting bias.
External Validity
The HD21 trial7 was conducted at 233 sites across 9 countries, all of which had sufficient resources to manage potential fever and infection. However, none of these sites were in Canada. The broad geographic distribution supports generalizability to well-resourced health care settings. The absence of sites in Canada raises questions about the applicability of the findings to clinical practice in Canada. The findings may not be generalizable to medical centres with limited access to supportive management such as fever or infection management. More than 90% of the patients were white, about 2% (n = 24) were Asian, less than 1% (n = 2) were Black, and the remaining were other or unknown races. It is unclear whether the trial population is representative of the racial diversity of patients in Canada.
The trial population was predominantly composed of patients with high functional capacity, with approximately 70% having an ECOG PS of 0.7 This may be consistent with the high functional capacity expected in young patients newly diagnosed cHL. However, the trial excluded patients older than age 60 and patients with poor ECOG PS (score of 3 or higher),7 which limits the applicability of its results to older adults or patients with poorer performance status or comorbidities.
In the trial, participants received regular monitoring, including PET-2 imaging and dose adjustments,7 which aligned with routine practice in Canada for managing cHL. PFS, OS, tolerability, and safety outcomes are clinically meaningful and important to most patients with cHL. The authors of the trial assessed these outcomes using methods (e.g., CTCAE for evaluation of AEs) that aligned with clinical practice for managing patients with cHL in Canada.
The HRQoL analyses were based on a subset of participants from Germany who completed follow-up assessments.7 This geographic concentration may introduce bias, because health care models and follow-up care may vary significantly between Germany and Canada. The generalizability of findings on HRQoL may be limited.
Results
Efficacy
Results for efficacy outcomes important to this review are presented in Table 5.
Key efficacy results:
PFS — BrECADD was associated with improved PFS compared to eBEACOPP.
OS — 4-year OS was similar between the BrECADD and eBEACOPP groups.
HRQoL —
Patients who experienced treatment-related morbidity reported lower global health status and reduced cognitive, physical, and social functioning at the end of treatment.
Formal longitudinal analyses were not reported.
Table 5: Summary of Key Efficacy Results
Outcome | eBEACOPP (N = 740) | BrECADD (N = 742) |
|---|---|---|
PFS | ||
Number of patients with PFS events, n (%) | 65 (9%) | 44 (6%) |
Tumour event | 58 (8%) | 37 (5%) |
Progression | 15 (2%) | 5 (1%) |
Early relapse, ≤ 1 year | 23 (3%) | 11 (1%) |
Late relapse, > 1 year | 20 (3%) | 21 (3%) |
Death without previous tumour event | 7 (1%) | 7 (1%) |
4-year PFS, % (95% CI) | 90.9% (88.7 to 93.1) | 94.3% (92.6 to 96.1) |
Comparison of BrECADD vs. eBEACOPP | ||
HR (95% CI) favouring BrECADD | 0.66 (0.45 to 0.97) | |
P value | 0.035 | |
OS | ||
Number of patients who died, n (%) | 12 (2%) | 12 (2%) |
Cause of death | ||
Hodgkin lymphoma, n (%) | 1 (< 1%) | 3 (< 1%) |
Toxicity of study treatment, n (%) | 3 (< 1%) | 0 |
Toxicity of salvage therapy, n (%) | 1 (< 1%) | 0 |
Second neoplasia, n (%) | 2 (< 1%) | 0 |
Cardiovascular, n (%) | 1 (< 1%) | 0 |
Respiratory, n (%) | 0 | 1 (< 1%) |
Infection, n (%) | 1 (< 1%) | 2 (< 1%) |
Suicide, n (%) | 0 | 1 (< 1%) |
Other disease, n (%) | 1 (< 1%) | 3 (< 1%) |
Unclear, n (%) | 2 (< 1%) | 2 (< 1%) |
Treatment-related deaths, n (%) | 1 | 3 |
4-year OS, % (95% CI) | 98.2% (97.2 to 99.3) | 98.6% (97.7 to 99.5) |
HRQoL | ||
Post hoc analysis examining the impact of treatment-related morbidity on HRQoL. | Treatment-related morbidity was statistically significantly associated with lower scores in several domains of HRQoL. Specifically, it correlated with reduced global health status (regression coefficient: –4.5, P = 0.0053, n = 619), cognitive functioning (–4.8, P = 0.0097, n = 630), physical functioning (–3.6, P = 0.024, n = 631), role functioning (–8.0, P = 0.0005, n = 630), and social functioning (–6.5, P = 0.003, n = 630). However, the association with emotional functioning was not statistically significant (–3.1, P = 0.084, n = 631). | |
BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone; cHL = classical Hodgkin lymphoma; CI = confidence interval; eBEACOPP = escalated doses of bleomycin, etoposide, doxorubicin (Adriamycin), cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisone; HR = hazard ratio; HRQoL = health-related quality of life; OS = overall survival; PFS = progression-free survival; vs. = versus.
Source: Borchmann et al. (2024).7
Figure 1: Kaplan-Meier Estimates of PFS and OS (Copied From Figure 2 in the Original Trial Publication)
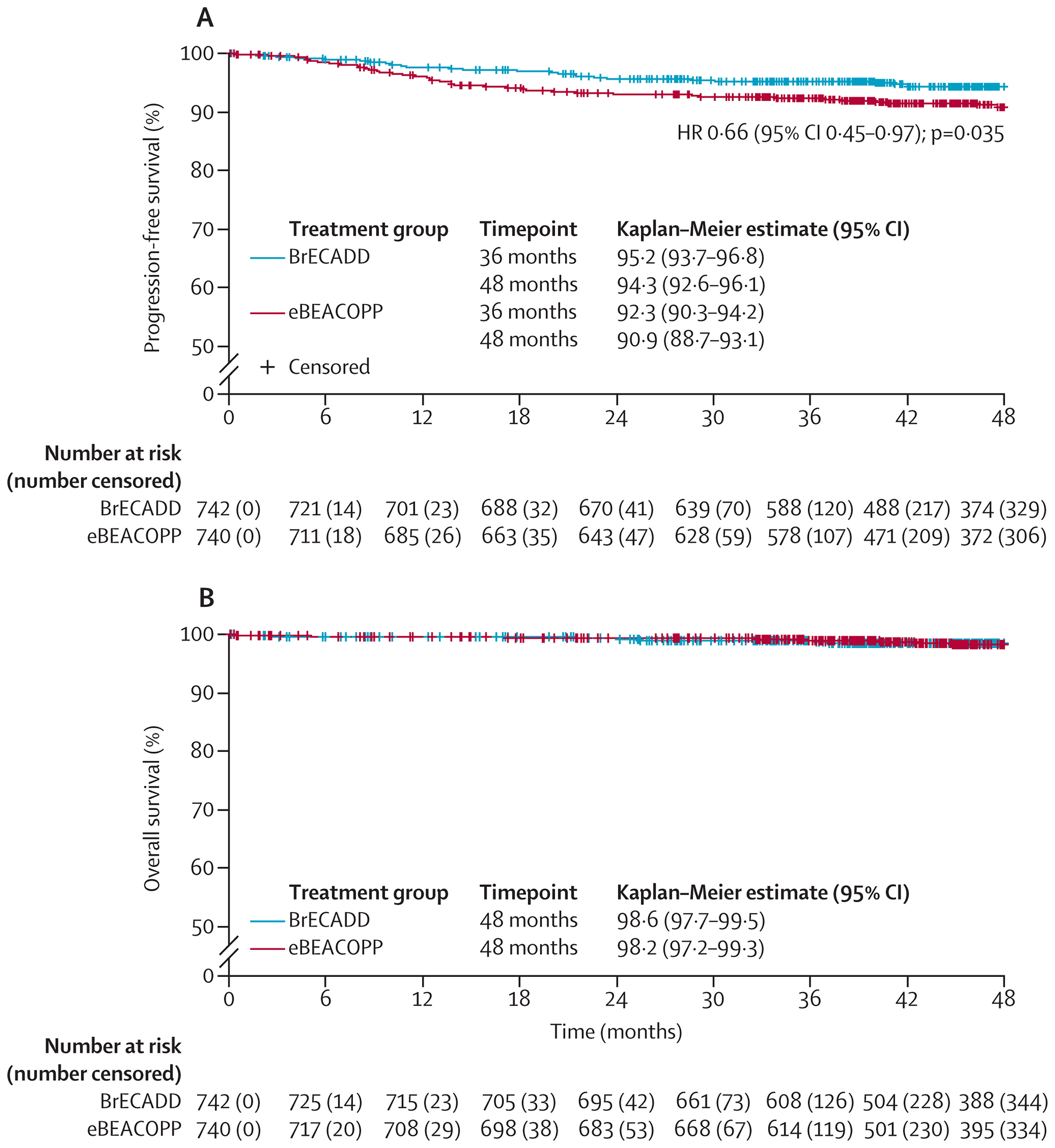
BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone; CI = confidence interval; eBEACOPP = escalated doses of bleomycin, etoposide, doxorubicin (Adriamycin), cyclophosphamide, vincristine (Oncovin), procarbazine, and prednisone; HR = hazard ratio; OS = overall survival; PFS = progression-free survival.
Notes: Graph A shows PFS, and graph B shows OS.
HR and P value obtained by Cox regression stratified by area of recruitment, International Prognostic Score, age, and sex.
Source: Borchmann P, Ferdinandus J, Schneider G, et al. Assessing the efficacy and tolerability of PET-guided BrECADD versus eBEACOPP in advanced-stage, classical Hodgkin lymphoma (HD21): a randomized, multicentre, parallel, open-label, phase III trial. Lancet. 2024;404(10450):341 to 352. Copyright 2024 by the authors. Available from: https://www.sciencedirect.com/science/article/pii/S0140673624013151?via%3Dihub.7 Reprinted in accordance with Creative Commons Attribution 4.0 International License (CC BY 4.0): https://creativecommons.org/licenses/by/4.0/.
Harms
Results for harm outcomes are presented in Appendix 4 of the Supplemental Material document.
Key harm results from the HD21 trial include the following:
Tolerability —
BrECADD showed significantly lower treatment-related morbidity (BrECADD: 42%; eBEACOPP: 59%; relative risk [RR]: 0.72 [95% CI, 0.65 to 0.80], P < 0·0001).
At 12 months post-treatment, 96% of patients in the BrECADD group had full or near-complete resolution of organ toxicities (to grade 1 or less), compared to 93% in the eBEACOPP group.
Persistent grade 2 peripheral neuropathy was reported in 2% of patients in both groups at the 1-year follow-up.
Second primary malignancies — Incidence was low and comparable (BrECADD: 3%; eBEACOPP: 2%).
Gonadal toxicity —
Recovery of gonadal function was higher in the BrECADD group (women: 95.3%; men: 85.6%) than in the eBEACOPP group (women: 73.3%; men: 39.7%) after 4 years.
Compared to the eBEACOPP group, the BrECADD group was associated with a significantly higher 5-year incidence of parenthood among men, but not among women.
Any AEs — Nearly all patients in both groups experienced at least 1 AE.
SAEs —
SAEs (grade ≥ 3 AE) were generally less frequent in the BrECADD group compared to the eBEACOPP group, particularly for hematologic toxicities such as anemia, thrombocytopenia, leukopenia, and peripheral sensory neuropathy.
Patients in the BrECADD group reported numerically higher rates than those in the eBEACOPP group for certain SAEs, such as neutropenic fever, infections, cardiac disorders, gastrointestinal disorders, and hepatobiliary disorders.
WDAEs — The authors of the trial reported withdrawals due to individual agents rather than full regimens: 132 (18%) patients discontinued vincristine early in the eBEACOPP group, and 18 (2%) patients discontinued brentuximab vedotin prematurely in the BrECADD group.
Deaths due to AEs — occurred only in the eBEACOPP group (n = 3).
Studies Addressing Gaps in the Systematic Review Evidence
No randomized studies were identified that compared BrECADD to relevant comparators in older patients (i.e., 60 years or older). One single-arm phase II cohort of the German Hodgkin Study Group HD21 Trial has been summarized to provide evidence of the potential efficacy and harms of BrECADD for older patients with cHL.9
Table 6: Summary of Gaps in the Systematic Review Evidence
First author (year) | Study name | Evidence gap |
|---|---|---|
Ferdinandus et al. (2025)9 | PET-Guided Brentuximab Vedotin, Etoposide, Cyclophosphamide, Doxorubicin, Dacarbazine, and Dexamethasone in Older Patients With Advanced-Stage Classic Hodgkin Lymphoma: A Prospective, Multicenter, Single-Arm, Phase II Cohort of the German Hodgkin Study Group HD21 Trial9 | Evidence of BrECADD for older patients (i.e., aged 60 years or older) with cHL |
BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin (Adriamycin), dacarbazine, and dexamethasone; cHL = classic Hodgkin lymphoma.
Description of Study
Characteristics of the included study are summarized in Table 7.
Table 7: Characteristics of Studies Addressing Gaps in Systematic Review Evidence
First author (year), design, country, sample size | Patient population | Intervention | Relevant end points, follow-up |
|---|---|---|---|
Ferdinandus et al. (2025)9 Multicentre, prospective, noncomparative, observational study 51 trial sites in 6 countries, primarily in Europe (94%), and Australia and New Zealand (6%) Total N = 85 enrolled, 83 analyzed |
| PET-guided BrECADD regimen Dose reduction and consolidative radiation therapy were allowed; G-CSF was mandatory to reduce hematologic toxicity, and quinolone antibiotic prophylaxis during neutropenia was encouraged. | Efficacy
Harms
Follow-up: 23 months (median) |
AE = adverse event; BrECADD = brentuximab vedotin, etoposide, cyclophosphamide, doxorubicin, dacarbazine, dexamethasone; cHL = classical Hodgkin lymphoma; ECOG PS = Eastern Cooperative Oncology Group Performance Status; G-CSF = granulocyte colony-stimulating factor; HRQoL = health-related quality of life; IQR = interquartile range; OS = overall survival; PFS = progression-free survival; SAE = serious adverse event; TRMB = treatment-related morbidity; WDAE = withdrawal due to adverse event.
Source: Ferdinandus et al. (2025).9
Critical Appraisal
Ferdinandus et al. (2025)9
The single-arm, open-label design does not support causal interpretations about the efficacy of BrECADD. The absence of a comparator arm limits the ability to attribute observed outcomes solely to BrECADD. Patient selection may be subject to bias. The authors of the study provided eligibility criteria for patient selection, but the study population may represent a relatively small subset of older patients. Most patients were fit (on the Frailty index), and only 2% (n = 2) of patients had frailty. Treatment exposure was guided by PET2 response, with predefined dose adjustments for toxicity. This individualized approach enhances feasibility, but it may introduce heterogeneity in treatment intensity, which may affect the interpretation of efficacy and safety outcomes. Dose reductions were frequent, particularly in older patients and those with frailty, but the impact of these reductions on outcomes was not reported.
The open-label nature of the study introduces the risk of bias in measuring subjective outcomes, such as HRQoL. The HRQoL was assessed using EORTC QLQ-C30, only 72% (n = 60) of patients provided data, and the impact of missing HRQoL data was not fully addressed. Loss to follow-up and some concomitant treatments (e.g., G-CSF) were not explicitly reported. We cannot assess the impact of lost follow-up and concomitant treatments on the effects of BrECADD. All analyses are of a descriptive nature in this study. The lack of formal statistical comparisons and reliance on descriptive analyses limit the robustness of conclusions. The study was conducted across multiple centres in Australasia, Europe, and New Zealand, with no sites in Canada. It is unclear whether the trial population is representative of patients in Canada.
Results
Results for efficacy, HRQoL, and harm outcomes important to this review from the single-arm studies are presented in Table 8.
Table 8: Summary of Outcomes in Studies Addressing Gaps in Systematic Review Evidence
First author (year), sample size, follow-up | Efficacy outcomes | Harms outcomes |
|---|---|---|
Ferdinandus et al. (2025)9 Multicentre, prospective, noncomparative, observational study 51 trial sites in 6 countries, primarily in Europe (94%), and Australia and New Zealand (6%) Total N = 85 enrolled, 83 analyzed | PFS:
OS:
HRQoL:
| TRMB:
Second primary malignancies:
Common SAEs (Grade 3 or greater toxicities, 50% or greater)
WDAE
Death due to AE
|
AE = adverse event; CI = confidence interval; HRQoL = Health-related quality of life; OS = overall survival; PFS = progression-free survival; SAE = serious adverse event; TRMB = treatment-related morbidity; WDAE = withdrawal due to adverse event.
Source: Ferdinandus et al. (2025).9
Discussion
Efficacy
The HD21 trial7 evaluated the safety and efficacy of BrECADD compared to eBEACOPP in patients with newly diagnosed advanced-stage cHL. The trial demonstrated a statistically significant improvement in PFS with BrECADD (HR = 0.66; 95% CI, 0.45 to 0.97). The 4-year PFS rate was higher in the BrECADD group (94.3%) than in the eBEACOPP group (90.9%). The open-label design of the HD21 trial could potentially overestimate the magnitude of the treatment effect. The clinical experts consulted by the review team indicated that the follow-up time for PFS was reasonable and the observed benefit on PFS was clinically meaningful.
Advanced-stage cHL was defined as Ann Arbor stage III or IV, as well as stage II with B symptoms and 1 or both risk factors of large mediastinal mass (≥ a third of the maximum thoracic diameter) or extranodal lesions in the HD21 trial. The trial enrolled patients with stages IIb (16%), IIIa–b (39%), and IVa–b (45%). Only 2 patients (< 1%) in the BrECADD treatment group have stage IIa. A clinical expert consulted by the review team noted that the BrECADD regimen could be effective for all patients with stage IIB cHL, regardless of the presence of bulky disease or features. This suggests a slightly different and potentially broader eligible patient population than was enrolled in the HD21 trial.
Four-year OS was similar between the 2 treatment arms: 98.6% for BrECADD and 98.2% for eBEACOPP.7 No treatment-related deaths occurred in the BrECADD group, but 3 treatment-related deaths were reported in the eBEACOPP group. The clinical experts consulted by the review team noted that comorbidity during the treatment may be 1 of the causes of the difference. These OS results likely reflect the trial population’s generally favourable prognosis, characterized by good performance status and absence of significant comorbidities. OS was a secondary outcome of the HD21 trial. The study may have lacked sufficient power, and long-term follow-up may detect a meaningful difference between the groups on OS.
The authors of the trial did not report direct comparisons of BrECADD and eBEACOPP on HRQoL,7 limiting the ability to assess the impact of BrECADD on this patient-reported outcome. However, HRQoL analyses showed that patients with treatment-related morbidity had lower scores compared to those without treatment-related morbidity across several domains, including global health status, cognitive, physical, role, and social functioning.7 Given BrECADD is associated with a reduced risk of treatment-related morbidity compared to BrECADD, these findings suggest that BrECADD may be associated with better HRQoL outcomes.
The generalizability of these findings may be limited by the trial’s population and design. The HD21 trial7 was conducted in settings with well-supported treatment, including effective management of fever and infections. The trial excluded individuals older than age 60 and those with poor performance status (ECOG PS score ≥ 3), which limits the applicability of its findings to older adults, patients with poor physical performance, or those treated in hospitals or clinics with limited access to supportive care.
An open-label, single-arm study reported that BrECADD is feasible and effective in older patients with cHL.9 The authors of the study reported that the 2-year PFS rate in this cohort was comparable to that observed in younger patients. However, older patients and those with frailty more frequently require dose modifications. BrECADD may be a treatment option for older individuals with cHL. However, comparative evidence in this population remains unavailable.
Using eBEACOPP as the comparator aligns with standard practice in many jurisdictions for patients with good physical performance in Canada. However, the absence of other active comparators limits broader applicability. Additionally, reliance on post hoc HRQoL analyses introduces uncertainty in interpreting patient-reported outcomes.
Harms
The harm results indicate a more favourable safety profile of BrECADD compared to eBEACOPP.7 BrECADD was significantly associated with a lower risk of treatment-related morbidity compared to eBEACOPP (42% versus 59%), and resolution of organ toxicities at 12 months post-treatment was slightly higher (96% versus 93%).7 Incidence of second primary malignancies (including acute myeloid leukemia, myelodysplastic syndrome, non-Hodgkin lymphoma, and solid tumour) was low and similar between BrECADD and eBEACOPP (3% versus 2%).7
Recovery of gonadal function was higher in patients treated with BrECADD compared to those treated with eBEACOPP, which may have implications for fertility outcomes.8 The 5-year incidence of parenthood was significantly higher in the BrECADD group among men (9.3% versus 3.3%), although no statistically significant difference was observed among women.8
Nearly all patients in both groups experienced at least 1 AE.7 BrECADD showed numerically lower hematological AEs, such as anemia, thrombocytopenia, or CTCAE grade 4 infection compared to eBEACOPP. Patients in the BrECADD group also reported fewer transfusions, including red cell transfusions and platelet transfusions, compared to patients in the eBEACOPP group. However, nonhematological toxicities were similar between groups. BrECADD had slightly higher rates of gastrointestinal events and cardiac toxicities compared to the eBEACOPP group.
Although BrECADD generally showed fewer grade 3 or higher AEs, certain SAEs, such as neutropenic disorders, fever, infection, cardiac disorders, gastrointestinal disorders, and hepatobiliary disorders, were numerically higher in this group.7 These findings underscore the importance of supportive treatment for patients who receive BrECADD in clinical practice.
Conclusion
BrECADD demonstrated superior PFS and a more favourable safety profile than eBEACOPP in patients with advanced-stage cHL, particularly in terms of hematologic toxicities (e.g., anemia and thrombocytopenia) and gonadal functions.7 This efficacy aligns with patient expectations for treatment that offers both improved effectiveness and tolerability for treating the disease. The improved tolerability and reduced gonadal toxicity may provide meaningful benefits for patients with cHL, particularly individuals concerned about fertility. The evidence showed similar 4-year OS between the 2 groups. The OS benefit for BrECADD remains uncertain at this stage. The potential reasons may include that the 48-month follow-up is relatively short for a highly curable disease (cHL), leading to a low number of death events (12 in each arm), which makes it difficult to detect a difference. Therefore, a longer follow-up may be needed to assess the impact of BrECADD on OS. BrECADD was associated with a numerically high incidence of grade 3 or higher AEs, including neutropenic disorders, fever, infections, cardiac events, gastrointestinal and hepatobiliary disorders, highlighting the importance of close monitoring and potential dose adjustments.
The lack of comparative evidence on specific populations, such as older adults (> age 60) and those with poor ECOG PS scores (3 or higher), leads to uncertainty in the generalizability of efficacy and safety in these subpopulations. Clinical experts may consider BrECADD to be a viable alternative to eBEACOPP, especially in patients for whom reduced hematological and gonadal toxicities are prioritized. We did not identify any studies comparing the efficacy and safety of BrECADD and other active treatments (e.g., ABVD or AVD + brentuximab vedotin).
Economic Review
Methods
The economic review consisted of a cost comparison for BrECADD compared with eBEACOPP, nivolumab + AVD, BV + AVD, and ABVD for newly diagnosed, advanced-stage, cHL.
Based on public list prices, BrECADD is expected to have a per-patient cost of $22,295 per 28 days (refer to Appendix 6 of the Supplemental Material). ABVD, eBEACOPP, nivolumab + AVD and BV + AVD are expected to have per-patient costs of $3,748, $6,779, $11,457, and $21,923 per 28 days, respectively. Therefore, the incremental costs of BrECADD compared with ABVD, eBEACOPP, nivolumab + AVD, and BV + AVD are $18,547, $15,516, $10,838, and $372 per patient per 28 days, respectively. The reimbursement of BrECADD for the treatment of newly diagnosed, advanced-stage, cHL is therefore expected to increase overall drug acquisition costs. Additional items for consideration are provided in the following bullets:
According to the clinical review, evidence from the HD21 trial7 suggests that, compared to eBEACOPP, BrECADD improves PFS and tolerability (particularly in terms of hematologic toxicities), and demonstrates better recovery of gonadal function, with no differences in OS. BrECADD generally showed fewer grade 3 or higher AEs, although certain SAEs, such as neutropenic disorders, fever, infection, cardiac disorders, gastrointestinal disorders, hepatobiliary disorders, were numerically higher in this group. No evidence was identified regarding the comparative efficacy and safety of BrECADD versus other active comparators.
No health care resource use outcomes were reported in the clinical trial.
As of September 2025, brentuximab vedotin, the key drug in the BrECADD regimen, is only available as a brand name product in Canada (Adcetris). There are no biosimilars currently under review at Health Canada.
Clinical experts consulted for this review provided the following considerations on possible impacts of the reimbursement of BrECADD on the use of health care resources: PET scans, which are part of the clinical decision-making process to define the number of BrECADD cycles, are also routinely performed with the other comparator regiments and can guide decisions to escalate, de-escalate, or switch regimens in this population; therefore, no difference in the number of PET scans is expected with the reimbursement of BrECADD. BrECADD, eBEACOPP, and BV + AVD are regimens that require primary prophylaxis for chemotherapy-induced neutropenia with granulocyte colony-stimulating factor. Given the higher risk of neutropenia with the BrECADD and eBEACOPP regimens, there might be a higher number of unscheduled visits and blood transfusions compared to other regimens, and patients on these treatments would require additional clinical visits and blood checks to assess the need for blood transfusions. Compared to eBEACOPP, BV + AVD, and ABVD, BrECADD may have lower relapse rates. Therefore cost-savings could be achieved with a reduced need for autologous stem cell transplants.
Brentuximab vedotin, as part of the BrECADD regimen, was not previously reviewed by CDA-AMC for this indication. As of September 2025, there are no drugs under review for newly diagnosed, advanced-stage, cHL. BV + AVD was previously reviewed by CDA-AMC in 2024 for adult patients with previously untreated advanced-stage Hodgkin lymphoma, with a positive recommendation for reimbursement. In 2025, CDA-AMC previously reviewed nivolumab + AVD for previously untreated stage III or IV cHL with a positive recommendation for reimbursement.
No cost-effectiveness studies conducted in Canada that included BrECADD for patients with Hodgkin lymphoma were identified based on a literature search conducted on July 7, 2025, with alerts maintained until the Formulary Management Expert Committee meeting on November 20, 2025.
Conclusion
The reimbursement of BrECADD for the treatment of newly diagnosed, advanced-stage, cHL is expected to increase overall drug acquisition costs. Based on the clinical review conclusions, compared to eBEACOPP, BrECADD may extend PFS, although there is no evidence that it improves OS. The comparative efficacy of BrECADD with other comparators is unknown, as it has not yet been assessed in the published literature.
Given that BrECADD is associated with higher drug acquisition costs relative to all comparators and is associated with incremental benefit in terms of PFS relative to eBEACOPP, a cost-effectiveness analysis would be required to determine the cost-effectiveness of BrECADD relative to its comparators. Because this was not available, its cost-effectiveness could not be determined.
References
1.CADTH. Reimbursement Review: Adcetris for Hodgkin Lymphoma. 2013. Accessed 2025-08-08. https://www.cda-amc.ca/adcetris-hodgkin-lymphoma-details
2.CADTH. Reimbursement Review: Brentuximab vedotin. 2024. Accessed 2025-08-08. https://www.cda-amc.ca/brentuximab-vedotin-0
3.Belsky JA, Hochberg J, Giulino-Roth L. Diagnosis and management of Hodgkin lymphoma in children, adolescents, and young adults. Best Practice & Research Clinical Haematology. 2023;36(1):101445. doi: 10.1016/j.beha.2023.101445 PubMed
4.Andrea K Ng MCA, PhD, Álex F Herrera. Classic Hodgkin lymphoma: Presentation, evaluation, and diagnosis in adults. In: LaCasce AS, ed. Accessed 2025-06-18. https://www.uptodate.com
5.Society CC. Hodgkin lymphoma statistics. 2024. Accessed 2025-08-01. https://cancer.ca/en/cancer-information/cancer-types/hodgkin-lymphoma/statistics
6.Ansell SM. Hodgkin lymphoma: 2025 update on diagnosis, risk-stratification, and management. Am J Hematol. 2024;99(12):2367-2378. doi: 10.1002/ajh.27470 PubMed
7.Borchmann P, Ferdinandus J, Schneider G, et al. Assessing the efficacy and tolerability of PET-guided BrECADD versus eBEACOPP in advanced-stage, classical Hodgkin lymphoma (HD21): a randomised, multicentre, parallel, open-label, phase 3 trial. Lancet. 2024;404(10450):341-352. doi: 10.1016/s0140-6736(24)01315-1 PubMed
8.Ferdinandus J, Schneider G, Moccia A, et al. Fertility in patients with advanced-stage classic Hodgkin lymphoma treated with BrECADD versus eBEACOPP: a secondary analysis of the multicentre, randomised, parallel, open-label, phase 3 HD21 trial. Lancet Oncol. 2025;10:10. doi: 10.1016/s1470-2045(25)00262-1 PubMed
9.Ferdinandus J, Kaul H, Fossa A, et al. Positron Emission Tomography-Guided Brentuximab Vedotin, Etoposide, Cyclophosphamide, Doxorubicin, Dacarbazine, and Dexamethasone in Older Patients With Advanced-Stage Classic Hodgkin Lymphoma: A Prospective, Multicenter, Single-Arm, Phase II Cohort of the German Hodgkin Study Group HD21 Trial. J Clin Oncol. 2025:JCO2500439. doi: 10.1200/jco-25-00439 PubMed
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.