CADTH Health Technology Review
Radiofrequency Ablation for Chronic Knee, Hip, and Shoulder Pain
Rapid Review
Authors: Amanda Shane, Sharon Bailey
Abbreviations
ACR
American College of Rheumatology
AE
adverse event
ASPN
American Society for Pain and Neuroscience
IA-HA
intra-articular hyaluronic acid
IAS
intra-articular steroid
NICE
National Institute for Health and Care Excellence
OA
osteoarthritis
RCT
randomized controlled trial
RFA
radiofrequency ablation
SR
systematic review
VAS
visual analogue scale
WOMAC
Western Ontario and McMaster Universities Osteoarthritis Index
Key Messages
For patients with knee osteoarthritis (OA), radiofrequency ablation may reduce pain and improve function compared to other nonsurgical interventions without increasing adverse events.
There is insufficient evidence to suggest that radiofrequency ablation reduces pain or improves function among patients with chronic hip pain.
We did not find any studies or guidelines on the clinical effectiveness of radiofrequency ablation for treating chronic shoulder pain that met the inclusion criteria for this review.
Three guidelines conditionally recommend the use of radiofrequency ablation for patients with knee OA, and 1 guideline conditionally recommends the use of radiofrequency ablation for hip joint pain following diagnostic blocks.
Context and Policy Issues
Why Is Chronic Pain of the Knee, Hip and Shoulder a Concern, and How Is it Currently Treated?
Chronic pain is characterized as pain lasting longer than 3 months.1 An estimated 20% of Canadians are living with chronic pain, with 2 thirds experiencing moderate to severe pain and half experiencing long-term pain of over 10 years.2 Chronic pain is recognized as a health condition in the 11th revision of the International Classification of Diseases.3 It can be classified as chronic primary pain (pain associated with significant emotional distress and not otherwise accounted for by another diagnosis), or chronic secondary pain (pain as a symptom of another underlying health condition such as osteoarthritis).2 Current treatments for chronic pain are typically multipronged and may include pharmaceutical, psychological, physical and rehabilitation, medical devices, practitioner administered or manual therapy, and/or interventional pain procedures.2
What Is Radiofrequency Ablation and How Is it Used?
Radiofrequency ablation (RFA) was introduced in the 1970s to treat back pain (via lumbar medial branch nerves of the facet joints),4 and then its use was expanded to treat a range of conditions, including sacroiliac joint pain in the 1990s,4 and chronic knee pain in 2008.5 RFA is a procedure that disrupts the transmission of pain signals through the delivery of targeted thermal damage to nearby neural tissue.6 Ideally, the chronic pain experienced by the patient will be attenuated while the damaged nerve structure is repaired by the body.6 The duration of pain relief varies but is considered temporary and may last anywhere from 3 to 6 months,7 or up to 12 months, or more.8 A radiofrequency probe is inserted typically via fluoroscopic guidance, but sometimes via ultrasound guidance, adjacent to target nerve(s) and generates radiofrequency energy, which manifests as ionic heating (heating of the surrounding tissue, not the probe itself), causing destruction of the nerve(s).6Target temperatures typically range from 80°C to 90°C, for 90 to 120 seconds, but there is variation in procedure parametres.9
In addition to conventional (or thermal monopolar) RFA, there are other modalities available, each with the same overall objective of reducing pain through temporary damage of target nerves. Cooled RFA involves probes that allow the circulation of saline around the probe tip to help carry heat away from the tissue interface and thus reduce heat-related tissue damage. This also results in a larger heating area and lesion, because more energy can be delivered through the probe.6 Therefore, more denervation is possible. Bipolar RFA involves 2 symmetrically placed electrodes that act as a conduit, also for the purpose of creating larger lesions than conventional RFA.10 Pulsed RFA was more recently introduced (late 1990s) as an alternative to conventional RFA. This procedure involves the production of pulses of 45V lasting 20 milliseconds at a maximum tissue temperature of 42°C. Evidence suggests that pulsed RFA is less painful and causes less tissue damage compared to conventional RFA, but the duration of pain relief is generally less than RFA.9 While pulsed RFA has similar effects on neural conduction, the treatment mechanism is different than that of nonpulsed RFA.
Why Is it Important to Do This Review?
Often, a combination of conservative treatment interventions (e.g., activity modification, intra-articular steroid injections, nonsteroidal anti-inflammatory drugs, etc.) effectively reduce chronic noncancer pain in the short-term (3 to 6 months).11 Surgical interventions are an option for longer term pain relief or curative treatment, but some patients may not be eligible and those who are eligible may be faced with long surgical wait times.12 This often results in the reliance on pharmaceuticals like opioids for pain relief. Therefore, the availability of alternative nonpharmaceutical (e.g., non-opioid) treatment options are required. Indeed, there is evidence that treatment with RFA among patients with chronic axial spine pain is associated with a decrease in the proportion of patients with opioid prescriptions compared to the proportion preprocedure.13 However, whether RFA may be similarly effective for pain relief for patients with chronic knee, hip, or shoulder pain is unclear.
Objective
This report aims to summarize the evidence regarding the clinical effectiveness of, and guidelines with recommendations for, RFA for the treatment of chronic non-cancer knee, hip and shoulder pain.
Research Questions
What is the clinical effectiveness of radiofreqency ablation for adults with chronic knee pain?
What is the clinical effectiveness of radiofrequency ablation for adults with chronic hip pain?
What is the clinical effectiveness of radiofrequency ablation for adults with chronic shoulder pain?
What are the evidence-based guidelines regarding the use of radiofrequency ablation for adults with chronic knee, hip, or shoulder pain?
Methods
Literature Search Methods
An information specialist conducted a literature search on key resources including MEDLINE, Embase, the Cochrane Database of Systematic Reviews, the International HTA Database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search approach was customized to retrieve a limited set of results, balancing comprehensiveness with relevancy. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. Search concepts were developed based on the elements of the research questions and selection criteria. The main search concepts were radiofrequency ablation and chronic knee, hip, and shoulder pain. CADTH-developed search filters were applied to limit retrieval to health technology assessments, systematic reviews, meta-analyses, or indirect treatment comparisons, any types of clinical trials or observational studies, and guidelines. The search was completed on August 22, 2023 and limited to English-language documents published since January 1, 2018.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first screening level, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | Q1,4. Adults with chronic non-cancer knee pain Q2,4. Adults with chronic non-cancer hip pain Q3,4. Adults with chronic non-cancer shoulder pain |
Intervention | Radiofrequency ablationa |
Comparator | Q1-Q3: Alternative nonsurgical interventions (e.g., routine medical management, corticosteroid joint injection), placebo, or no treatment Q4. Not applicable |
Outcomes | Q1-Q3: Clinical benefits (e.g., pain relief, health-related quality of life, functional improvement [e.g., activities of daily living]) and harms (e.g., fall risk) Q4. Recommendations regarding best practices for radiofrequency ablation (e.g., indications, number of lesions needed for effective treatment, who provides the procedure, setting for procedure) |
Study designs | Health technology assessments, systematic reviews, randomized controlled trials, nonrandomized studies, evidence-based guidelines |
aExcluding pulsed radiofrequency ablation unless volume of included evidence is low.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, or were published before 2018. Systematic reviews in which all relevant studies were captured in other more recent or more comprehensive systematic reviews were excluded. Systematic reviews in which there was partial overlap of relevant studies with 1 or more recent or comprehensive systematic reviews were excluded and the primary study(ies) missing from the more recent systematic review were retained and included in this review, even if published before 2018. Primary studies retrieved by the search were excluded if they were captured in 1 or more included systematic reviews. Guidelines with unclear methodology were also excluded.
Critical Appraisal of Individual Studies
The included publications were critically appraised by 1 reviewer using the following tools as a guide: A MeaSurement Tool to Assess systematic Reviews 2 (AMSTAR 2)14 for systematic reviews, the Downs and Black checklist15 for randomized and nonrandomized studies, and the Appraisal of Guidelines for Research and Evaluation (AGREE) II instrument16 for guidelines. Summary scores were not calculated for the included studies; rather, the strengths and limitations of each included publication were described narratively.
Summary of Evidence
Quantity of Research Available
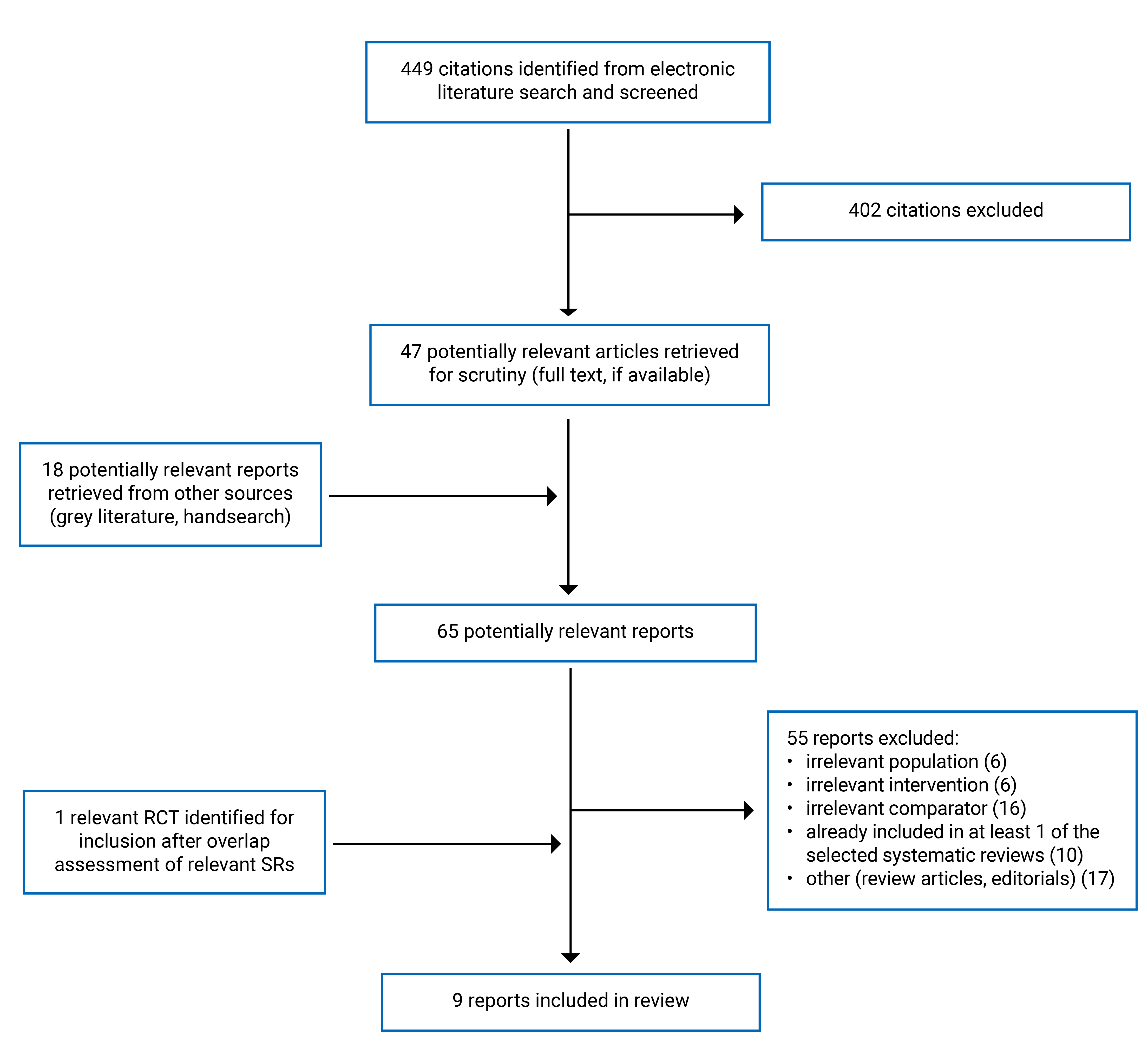
A total of 449 citations were identified in the literature search. Following screening of titles and abstracts, 402 citations were excluded and 47 potentially relevant reports from the electronic search were retrieved for full-text review. 18 potentially relevant publications were retrieved from the grey literature search for full-text review. Of these potentially relevant articles, 57 publications were excluded for various reasons. After the assessment of overlap across the SRs meeting the inclusion criteria, 1 unique RCT17 from an SR excluded for partial overlap18 was retrieved. Nine publications met the inclusion criteria and were included in this report. These comprised 4 systematic reviews, 2 primary studies, and 3 evidence-based guidelines. Appendix 1 presents the PRISMA19 flow chart of the study selection.
Two SRs7,20 and 2 primary studies10,17 were included for chronic knee pain, and 2 SRs21,22 were included for chronic hip pain. No evidence was found for chronic shoulder pain. Three guidelines23-25 were included.
Additional references of potential interest are provided in Appendix 7.
Summary of Study Characteristics
Three publications had broader inclusion criteria than the present review.
Fogarty et al. (2022)7 examined the clinical effectiveness of fluoroscopically guided genicular RFA compared to non-surgical interventions including some not relevant to the present review (e.g., other forms of RFA). The 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip and Knee24 makes recommendations for pharmacologic and nonpharmacologic management of OA, including for the use of RFA plus usual care for knee OA. Kao et al. (2018)22 conducted an SR on the clinical effectiveness of RFA for hip pain. Two of the included studies (2/10) involved patients with postoperative hip pain and patients with cancer and 2/10 studies used pulsed RFA. Only characteristics, results, and recommendations of the subset of relevant publications will be described in this report.
In addition, 1 SR did not identify any relevant studies. Chou et al. (2020)21 evaluated the clinical effectiveness of 10 intervention procedures for 10 pain conditions, including cooled RFA versus usual care, sham, placebo, or no treatment for degenerative hip pain. However, no evidence was found for these comparisons. Due to the limited amount of evidence identified on the use of RFA for hip pain (1 SR with evidence22), 2 included studies in the SR that used pulsed RFA are included and summarized separately throughout this report.
Additional details regarding the characteristics of included publications are provided in Appendix 2.
Study Design
Knee Pain
This review identified 2 SRs for RFA treatment of knee pain, 1 published in 2022,7 and 1 in 2021.20 The databases were searched from inception to October 10, 20207 and November 13, 2019.20 Fogarty et al. (2022)7 contained 5 RCTs across 8 publications, and 1 case series, of which 3 RCTs (3 publications) were relevant to the present review. The results were synthesized narratively. Chen at al. (2021)20 included 7 RCTs and summarized the results via meta-analysis. A table describing the overlap of relevant studies within these 2 SRs is provided in Appendix. 5.
This review also identified 2 primary studies on RFA treatment of chronic knee pain, a triple-blinded RCT published in 202210 and an open-label, nonrandomized controlled trial published in 2011.17
Hip Pain
This review identified 2 SRs for RFA treatment of hip pain, 1 published in 202121 and 1 published in 2018.22 The databases were searched from 1990 to April 202121 and from inception to January 20, 2017.22 Chou et al. (2021) found no evidence relevant to this review and Kao et al. (2018)22 included 6 case series or case reports on RFA and 1 nonblinded, uncontrolled, clinical trial and 1 case series using pulsed RFA.
Guidelines
This review identified 3 guidelines: 1 focuses on patients with knee osteoarthritis (OA)23 and 2 are broader but included recommendations for the use of RFA for hip and knee pain.24,25
The National Institute for Health and Care Excellence (NICE) published their guidance in 2023.23 A rapid review was conducted to gather evidence on the efficacy and safety of RFA for the treatment of patients with knee OA. All clinical study types were eligible for inclusion. Ratings of the quality of evidence and strength of the recommendations, and the methods used by the guideline development group to produce the recommendations, are not reported.
The American Society of Pain and Neuroscience (ASPN)25 published their guidelines in 2021. A literature search of 3 databases was conducted to gather evidence on the use of RFA for 7 anatomic targets including the knee and hip joints. Systematic reviews, literature reviews, RCTs, prospective and retrospective observational studies were eligible for inclusion. The US Preventive Services Task Force criteria for quality of evidence and strength of recommendations is used. The quality of evidence is rated on a 5-level scale (I, II-1, II-2, II-3 and III) where I is highest and III is lowest quality evidence. The strength of recommendations is rated from A (highest) to D (lowest), or I (insufficient evidence to make a recommendation).
The American College of Rheumatology (ACR) and the Arthritis Foundation published their guidelines in 2020.24 A systematic search of the literature was conducted to identify evidence on the benefits and harms of available interventions for OA of the hand, hip and knee, including RFA. RCTs and observational studies were eligible for inclusion. The quality of evidence and strength of recommendations was graded using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology: the certainty of the evidence is assessed from very low to high, and the strength of a recommendation for or against an intervention can be weak or strong.
Country of Origin
Knee Pain
The first authors of the SRs were from the US.7,20 The primary studies were conducted in Thailand10 and Japan.17
Hip Pain
The first author of the SR on hip pain22 was from the US.
Guidelines
The NICE guidelines23 apply to the UK and the ASPN26 and ACR guidelines24 apply to the US.
Patient Population
Knee Pain
The SRs included adult patients diagnosed with knee OA. Studies were excluded if patients underwent knee arthroscopy. A total of 392 patients were included in the Fogarty et al. (2022)7 review, while the total number of patients was not reported in Chen et al. (2021).20 No other demographic or clinical information was reported in either SR. The primary studies enrolled 6410 and 3517 patients with knee OA. The mean age of patients in the primary studies was 66.710 and 7717 years, and the majority was female.
Hip Pain
A total of 43 adult patients was included in the SR on hip pain: 26 patients received RFA (excluding pulsed RFA) and 17 patients received pulsed RFA.22
RFA (excluding pulsed RFA). Specific age was reported for 3 case reports (mean: 56 years).22 Three publications involved patients with hip pain due to OA.22 One case series involved patients with general chronic hip pain excluding metastasis to hip, a second case series involved chronic hip pain due to avascular necrosis of femoral head, and 1 case report focused on a patient with destructive coxopathy due to repeated radiation.22
Pulsed RFA: Age was not reported. Both studies involved patients with hip OA.22
Guidelines
The target population of the guidelines are patients with osteoarthritic knee pain,23 patients with pain in the knee and hip joints,25 and patients with OA of the knee or hip.24 The intended users are health care providers, but the NICE23 and ACR24 guidelines also refer to patients and caregivers as a potential audience.
Interventions and Comparators
Knee Pain
The interventions included in the SRs were fluoroscopically guided cooled RFA,7,20 conventional monopolar RFA,7,20 and bipolar RFA.7 Both SRs included medical management, intra-articular hyaluronic acid (IA-HA) injection, and intra-articular steroid (IAS) injection as comparators. Chen et al. (2021)20 included 1 study with sham-RFA as a comparator.
Among the primary studies, interventions varied in target, frequency and method. Both were fluoroscopy guided and performed sensory/motor stimulation for localization.10,17 The RCT did a prognostic nerve block before randomization10 Malaithong et al. (2022) and the nonrandomized controlled trial did a nerve block 1 day before the intervention.17 Malaithong et al. (2022)10 conducted bipolar RFA on 3 genicular nerves simultaneously and Ikeuchi et al. (2011)17 performed 2 RFA treatments 2 weeks apart on the medial retinacular nerve and the infrapatellar branch of the saphenous nerve. The lesions were made at 90° for 180s,10 and 70°C for 90s.17 Comparators were genicular nerve block at 3 target sites, local anesthetic and steroid injection plus sham-RFA,10 and local anesthetic.17
Hip Pain
The specific intervention details varied across the 8 included publications within the SR identified for this report.22
RFA (excluding pulsed; 6 studies): A pre-treatment diagnostic nerve block was performed in 3 studies. Fluoroscopy guidance was used in 4 studies, ultrasound guidance was not used, and 2 studies did not report method of guidance. Sensory/motor stimulation was used in 3 studies. Five studies used thermal RFA, at varying temperature and duration: 90°C for 180 seconds (s) per target or 90s per target, 80°C for 120 seconds per target, 75°C to 80°C for 90 seconds per target, and 75°C to 90°C for 90s per target. When reported, a 22-gauge needle was used with a 100 mm electrode and 4 mm, 5 mm, and 10 mm exposed tip.
Pulsed RFA: Two studies used pulsed RFA. Both used fluoroscopic guidance, 42°C and 20 ms generator output of 45 V (1at 120s and 1 at 180s).
Guidelines
The NICE guidelines23 describe the RFA intervention in general terms, including that it can be conducted under fluoroscopic or ultrasound guidance and that several targets have been identified including the genicular nerves. The ACR24 guidelines are similarly general, citing the intervention as “radiofrequency ablation”. The ASPN25 guideline includes a literature review and summary of the procedure techniques for both knee and hip RFA, including the general guidance on options for technique, electrode settings, target sites, and use of preprocedure diagnostic blocks. However, the recommendation statements are general: they specify the target nerves (the genicular nerve or obturator and femoral nerve branches for knee or hip pain, respectively) but do not further specify recommended treatment protocols.
Outcomes
Knee Pain
All included SRs and primary studies measured pain via the visual analogue scale (VAS), function via the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) and the frequency of adverse events (AEs).7,10,17,20 Additional function measures included the Oxford Knee Scale,7 and the patient global impression of improvement Likert scale.10,17
Hip Pain
The outcomes from the Kao et al. (2018)22 SR included pain measured via VAS, function measured via the Lower Extremity Functional Scale and the frequency of AEs.
Guidelines
The outcomes considered by the NICE guideline panel23 were pain (VAS, the numeric rating scale), function (WOMAC, global perceived effort scale, timed up and go test and EQ-D5) and safety (pain, infection, numbness, damage to adjacent structures). ASPN25 considered improvement in pain (any scale), function, analgesic use, subsequent need for surgery, health care utilization and return to work. ACR24 considered pain, function (self-reported) and function (performance based).
Summary of Critical Appraisal
Knee
Systematic Reviews
The SRs had clear research questions, comprehensive literature reviews and study selection in duplicate. This increases the likelihood that relevant studies were not missed. With respect to the analysis, Fogarty et al. (2022)7 extracted data in duplicate, and described the included studies in some detail, but additional patient characteristics would have enhanced interpretation. An important weakness of Chen et al. (2021)20 is the lack of demographic, clinical and other information about the included patients: it’s neither in the publication nor the supplementary information. Therefore, it is difficult to interpret the results and assess the external validity of the findings.
Primary Studies
The primary studies10,17 had clearly defined objectives, main outcomes, characteristics of the included patients, and interventions. The presented results were described in the methods, the statistical tests were appropriate, compliance with the intervention was reliable and the main outcome measures were valid and reliable. Malaithong et al. (2022)10 was a blinded (both study subjects and those measuring the main outcomes), randomized trial. There was sufficient power to detect a clinically important effect in VAS at all time points except 10 and 12 months. By contrast, Ikeuchi et al. (2011)17 was an unblinded, nonrandomized trial with insufficient information provided to determine whether the study had sufficient power to detect clinically important differences between the treatment groups (a total of 35 patients were enrolled). Neither study provided sufficient information to assess external validity, or whether confounding was assessed adequately.
Hip
Both SRs21,22 had clear research questions containing the components of PICO, had comprehensive literature reviews with searches of at least 4 databases, justified their selection of included study types, conducted study selection in duplicate and reported no conflicts of interest. Neither performed data extraction in duplicate nor provided a list of excluded studies. Kao et al. (201822 provided a satisfactory explanation for, and discussion of, the heterogeneity observed but there were concerns with the risk of bias [RoB]). The method to assess RoB was not reported, but the authors stated there was a large RoB within the included studies due to lack of blinding and small sample sizes which is inherent in the design of included studies (i.e., case series/reports).22
Guidelines
The ACR guideline24 performed well across all AGREEII domains, except applicability, and has strengths that the other 2 guidelines do not. The scope and purpose of the ACR guideline24 is clear, all relevant professional groups and the target population participated in the development group, and the target users are clearly defined. The guideline was developed using rigorous methods for evidence synthesis and recommendations development, and with editorial independence, with the only area of uncertainty being whether external review occurred. The guideline’s weakness relates to its applicability: facilitators and barriers to application are not mentioned, and no advice or tools are provided to enable implementation. Conversely, both NICE23 and ASPN25 did have resources available for implementation (methods for putting evidence into practice,23 best practice summary).25 However, the rigour of development is unclear for both NICE23 and ASPN.25 Systematic methods were used to identify evidence, but the strengths and limitations of the body of evidence and the methods for formulating recommendations are not provided. While ASPN25 did have an explicit link between the evidence and the recommendation, the assigned evidence level of II-1 (defined as evidence from a “well-designed, controlled, nonrandomized clinical trial (p. 2809)”25) did not align with the stated source evidence (uncontrolled studies). NICE23 did not include an explicit link between the evidence and the recommendation. Additional concerns with the NICE guideline are uncertainty around editorial independence (there is no statement on conflict of interest, and it is unclear whether the views of the funding body have influenced the content of the guideline. This leads to difficulty in understanding the rationale for the recommendation statements. The clarity of presentation for all 3 guidelines is good: recommendations are specific and easy to find.
Additional details regarding the strengths and limitations of included publications are provided in Appendix 3.
Summary of Findings
Appendix 4 presents the main study findings.
Clinical Effectiveness of Radiofrequency Ablation for Chronic Knee Pain
Evidence from 2 SRs7,20 and 2 additional primary studies10,17 suggests that the use of RFA for chronic knee pain is clinically effective (Table 8; Table 9; Table 10; Table 11). There is general agreement across studies in favour of RFA versus nonsurgical or sham interventions for nearly all measured outcomes, follow-up time points, and treatment comparisons, with infrequent AEs.
There was some overlap in the primary studies included in the SRs; therefore, to avoid duplication of results, outcome data from an individual primary study are only reported once. The extent of overlap is summarized in Appendix 5. Due to methodologic heterogeneity, neither SR pooled results by meta-analysis.
Pain
Pain was measured by VAS at baseline and at 4, 8 and 12 weeks, and at 6 and 12 months (Table 8).
For patients with knee OA, there were inconsistent findings at 4 weeks. Namely, there was no difference between bimodal RFA and sham-RFA groups in mean change from baseline, but a statistically significant between-group difference in mean VAS among those receiving RFA compared to local anesthetic (1 nonrandomized study with 35 patients; 1 RCT with 64 patients).
From 8 weeks to 6 months, VAS measures generally favoured the intervention groups compared to the control groups (2 SRs7,20 and 2 primary studies;10,17 35 to 392 patients):
RFA was favourably associated with pain compared to intra-articular injections (IA-HA and IAS), at 12 weeks and 6 months (2 SRs;7,20 24 to 158 patients per primary study).
There was a statistically significant between-group difference in VAS 12 weeks and 6 months in favour of RFA compared to oral analgesics (2 SRs;7,20 60 patients).
When compared to sham-RFA, the evidence of treatment effectiveness was inconsistent.
There was a statistically significant between-group difference in mean VAS at 12 weeks in favour of the intervention (1 SR;20 35 patients)
There were no between-group differences at 6 and 12 months (1 RCT;10 53 to 59 patients). Note that the RCT had insufficient power to detect a clinically significant difference at 12 months.
Function
Function (WOMAC score) was measured at baseline and at 4 weeks, 8 weeks, 12 weeks, and 6 months (Table 9).
At 4 to 8 weeks, RFA had no impact on function, compared with sham-RFA or local anesthetic:
At 12 weeks, there was consistent, statistically significant, improvement in function among the RFA groups, compared to IA-HA, IAS and sham control groups, but not compared to local anesthetic.
At 6 months, there was consistent, statistically significant, improvement in function among the RFA groups compared to medical management, IA-HA and IAS, but not sham-RFA or local anesthetic.
There was a statistically significant difference between groups in mean WOMAC score change from baseline (1 SR;7 177 patients) and statistically significant between-group difference in WOMAC scores (1 SRs;7,20 96 to 125 patients).
There was no difference between groups (bimodal RFA vs. sham RFA) in mean change from baseline (1 RCT;10 64 patients) and no between-group difference in mean WOMAC score (RFA vs. local anesthetic) (1 nonrandomized trial;17 35 patients).
Other Outcomes
Additional measures were summarized in a subset of the included studies:
There was no between-group difference in mean Patient Global Impression at any time point, for bimodal RFA versus sham-RFA or RFA versus local anesthetic (2 primary studies;10,17 35 to 64 patients) (Table 10).
There was a statistically significant between-group difference in mean Oxford Knee Score at 6 months, but not 12 months (1 SR; 7 52 to 126 patients; cooled RFA versus IAS). There was a statistically significant within-group difference from baseline among the intervention arm at 24 months (1 SR;7) (Table 11).
Adverse Events
All studies reported the frequency of adverse events (AE).7,10,17,20 (Table 12).
One serious AE – significant swelling -- was reported in 1 patient receiving RFA (1 RCT,10 64 patients).
The frequency of any AE was 3.9% (n = 3/76) among those receiving cooled RFA compared to 9% (n = 7/75) in those receiving IAS (1 SR,7 392 patients).
Subcutaneous bleeding at the site of needle insertion occurred in 67% (n = 12/18) of RFA group patients, compared to 82% (n = 11/14) receiving local anesthetic (1 nonrandomized trial, 32 patients).17
Prolonged hypoesthesia at the infrapatellar branch of the saphenous nerve occurred in 78% (n = 14/18) of RFA group patients, compared to 0% receiving local anesthetic (1 nonrandomized trial, 32 patients).17
Four studies reported no AEs in either the intervention or control groups (2 SRs,7,20 total number of patients not reported).
Clinical Effectiveness of Radiofrequency Ablation for Chronic Hip Pain
We identified 2 SRs21,22 that sought evidence regarding the use of RFA, including pulsed RFA, for chronic non-cancer hip pain. Chou et al. (2021)21 did not identify evidence meeting our inclusion criteria. Kao et al. (2018)22 summarized clinical outcomes (pain, function, AE) from a small number of low-quality studies (1 nonrandomized trial, 1 prospective before-and-after study, and 6 uncontrolled case series/reports) involving 43 patients.
The quality and quantity of this identified evidence is therefore insufficient to determine the comparative effectiveness of RFA, including pulsed RFA, for patients with chronic hip pain.
Pain
For patients receiving RFA (n = 25):
There was significant within-group improvement in VAS from baseline to 6 months (1 prospective before-and-after study from 1 SR,22 Table 13).
Four case series and reports in 1 SR22 found some improvement in VAS for 8 patients at 4 weeks, 8 weeks, 12 weeks and/or 6 and 24 months of follow-up (Table 13).
For patients receiving pulsed RFA (n = 17):
There was some improvement in VAS at 1, 4, 12, and 16 weeks (1 nonrandomized trial and 1 case series in 1 SR)22 (Table 13).
Function
One patient with chronic hip pain experienced functional improvement at 6 months after the RFA procedure (1 case report reported in 1 SR,22 Table 14).
Adverse Events
AEs were reported in 4 of 25 patients who received RFA and 1 of 15 patients who received pulsed RFA (2 nonrandomized controlled trials, 2 case series in 1 SR,22 Table 15).
Clinical Effectiveness of Radiofrequency Ablation for Chronic Shoulder Pain
No relevant evidence regarding the use of RFA for chronic shoulder pain was identified; therefore, no summary can be provided.
Guidelines Regarding the Use of Radiofrequency Ablation for Chronic Knee, Hip and Shoulder Pain
Knee and Hip Pain
This review identified 3 guidelines for the use of RFA for the treatment of adults with chronic knee pain (Table 16).
The NICE guideline23 states that RFA may be used “…if standard arrangements are in place for clinical governance, consent and audit (p.2).”23 The guideline also specifies that the procedure should be done by clinicians with appropriate training. The quality of the evidence supporting this guideline was not summarized and there is no strength given to this statement.
The ACR guideline24 conditionally recommends the use of RFA for patients with knee OA, based on moderate quality evidence. The recommendation is conditional due to the methodologic heterogeneity in the supporting evidence (i.e., various intervention techniques and control procedures) and lack of long-term safety data.
The ASPN guideline25 states that RFA may be used to treat knee joint pain due to post OA and postsurgical pain (moderate to high certainty that the net benefit is moderate/substantial, based on well-designed, controlled trial).
This review identified 1 guideline for the use of RFA for the treatment of chronic hip pain (Table 16).
The ASPN guideline25 states that RFA may be used for the treatment of hip joint pain following diagnostic blocks. The guideline specifies that the obturator and femoral nerve branches should be targeted (recommendation with moderate to high certainty that the net benefit is moderate/substantial, based on well-designed, controlled trials).
Shoulder Pain
No relevant guidelines were found regarding the use of RFA for chronic shoulder pain.
Limitations
There was considerable methodological heterogeneity in the body of evidence gathered for this report. The 2 SRs on knee pain7,20 included studies with 2 to 3 different RFA approaches (cooled, conventional monopolar and bipolar) and between 4 and 5 different comparator treatments. Meta-analysis was therefore not justified, and results were summarized narratively. Similarly, there was inconsistency across the RCTs: 1 RCT compared bipolar RFA to IAS plus sham-RFA, whereas the other RCT compared a two-treatment RFA regimen to local anesthetic. This makes concise interpretation across the body of included evidence challenging.
The generalizability of these results is limited to patients with OA-related knee pain. The patient population from the hip pain-related evidence was more diverse (e.g., 50% of patients experience chronic hip not related to OA), but not sufficiently to be generalizable due to small numbers (n = 26 patients). Further, no demographic, clinical or other confounding variables were summarized in the SRs. Without basic information like the age and sex distribution of the patients, it is difficult to assess the generalizability of the findings to the Canadian context.
The quantity and quality of available evidence on the use of RFA for the treatment of chronic hip pain is low. Two SRs21,22 and 1 guideline25 were identified for hip pain. One well conducted SR did not find any relevant evidence.21 Several reasons for this may exist. First, the authors limited the interventions for this SR to cooled and pulsed RFA; therefore, evidence may have been missed regarding the broader clinical effectiveness of other/all RFA modalities on chronic hip pain. Second, the use of RFA for large joints is relatively new compared to its use for spinal and cranial nerves, so the volume of comparative trials may be low. This is reflected in the SR by Kao et al. (2018),22 which included 6 low-quality observational studies with a total of 26 patients across them.22 Further, the methodological heterogeneity among these studies was substantial so the results could not be pooled to increase the sample size and power to detect differences across the intervention groups.
There is discordance between this review and the ASPN guideline25 regarding the evidence assessment for the use of RFA on hip pain. The ASPN recommendation25 that RFA may be used for the treatment of hip joint pain is informed by 4 nonrandomized studies: 2 retrospective uncontrolled studies, 1 prospective uncontrolled study, and 1 case series. Two of these studies were also captured in the Kao et al. (2018)22 SR (although 1 was not summarized in this review because it included patients with cancer-related pain); 2 were excluded due to study design (i.e., uncontrolled studies). ASPN25 assigned an evidence level of II-1 to this body of evidence which is defined as “well-designed, controlled, nonrandomized clinical trial (p. 2809)”25 despite there not being a controlled trial in the included body of evidence. Due to the discordance between the included evidence and the assigned evidence level, and its unclear interpretation and application to the “best practice summary,” the resulting recommendation should be interpreted and applied with caution.
This report is also limited by the lack of relevant evidence identified on the use of RFA for the treatment of chronic shoulder pain.
Conclusions and Implications for Decision- or Policy-Making
Summary
This review identified 2 SRs7,20 and 2 primary studies10,17 on the clinical effectiveness of RFA for chronic knee pain; 2 SRs21,22 on the clinical effectiveness of RFA for chronic hip pain; and no evidence on the clinical effectiveness of chronic shoulder pain. Three guidelines were identified: 2 for the use of RFA for knee OA23,24 and 1 for the use of RFA for knee or hip OA.25
Despite the considerable methodological heterogeneity across the included studies due to variation in RFA procedures and choice of comparators, there is consistency in the direction of the effect for pain (VAS) and function (WOMAC) across several time points for the use of RFA on knee OA. This increases the certainty that the use of RFA is clinically effective in reducing pain and improving function among adult patients with chronic OA knee pain. Conversely, there is a lack of certainty around the evidence of effectiveness for the use of RFA on chronic hip pain, despite consistency in the direction of the effect for pain (VAS), due to the small number of low-quality, heterogeneous studies.
Three guidelines23-25 were identified that conditionally recommend the use of RFA for patients with knee OA. These recommendations align with the consistent clinical findings summarized in this review. One guideline25 conditionally recommends using RFA for hip pain following diagnostic blocks. This recommendation is inconsistent with the findings summarized in this review, primarily due to discordance in the interpretation of the quality of evidence, where this review concludes that the small quantity and low-quality evidence available is insufficient to suggest that RFA reduces pain and improves function among patients with chronic non-cancer hip pain.
Considerations for Future Research
A research gap exists for good-quality RCTs that evaluate the effectiveness of RFA on chronic hip and shoulder pain. While this review identified an SR focused on the use of RFA for hip pain, that SR identified 1 uncontrolled trial and 5 case series or reports relevant to this review. No RCTs of RFA versus alternative nonsurgical interventions (e.g., routine medical management, corticosteroid joint injection), placebo, or no treatment were identified – either directly through this report’s search or indirectly via included SRs. Further, no evidence was identified on the effectiveness of RFA for chronic shoulder pain going back to January 1, 2018. Decision-makers should consider that this represents a substantial gap in evidence for the use of a nonpharmacological intervention for 2 joint sites affected by OA.1
In addition, the available guidelines lacked recommendations with specificity for best practices for the RFA technique. An opportunity exists to create an evidence-informed resource to support clinicians' ability to optimally perform this procedure. Selection of key procedural parametres such as electrode size, lesioning time, and selection of the target sensory nerves have an impact on the lesion size and the likelihood of denaturing the target nerves.7
Ultrasound-guided RFA is increasingly being explored as an alternative to, or in conjunction with,25 fluoroscopy-guided RFA. As complementary to fluoroscopic guidance, the addition of ultrasound may help with needle placement, improve safety, and decrease the risk of vascular and/or nerve injury, particularly for the challenging anatomy of the hip joint.25 Suggested advantages as an alternative to fluoroscopic guidance include decreased cost, increased availability, and lack of ionizing radiation.7 This review did not identify any studies using ultrasound guidance. Well-designed and adequately powered comparative studies on the effectiveness of ultrasound guidance versus fluoroscopy may expand the opportunities for the application of this technology for knee OA.
Implications for Clinical Practice
The findings of this report suggest that RFA may be an effective treatment option for chronic knee pain due to OA as an alternative to other nonsurgical interventions such as intra-articular injections or oral analgesics in patients with postsurgical pain, those not eligible for surgery or those who are awaiting surgery. In addition, this treatment may provide an alternative nonpharmacologic (e.g., opioid) option for patients with chronic knee OA who are no longer experiencing (or have never experienced) pain relief with existing treatments.
While identified guidelines supported the use of RFA in patients with knee OA, only one25 specified parametres for electrode settings, target nerves, and use of pre-ablative diagnostic blocks and motor testing. The guideline also noted the required knowledge for application of this procedure, so there may be an opportunity for clinician training and knowledge of the anatomic innervation of the knee, the principles surrounding RFA, and experience with fluoroscopy, ultrasound, and the variety of RFA modalities.
References
1.Arthritis Society Canada. Understanding pain. 2023; https://arthritis.ca/treatment/pain-management/arthritis-pain-management-guide/understanding-pain. Accessed 2023 Sep 17.
2.The Canadian Pain Task Force. Chronic pain in Canada: Laying a foundation for action. Ottawa (ON): Health Canada; 2019: https://www.canada.ca/content/dam/hc-sc/documents/corporate/about-health-canada/public-engagement/external-advisory-bodies/canadian-pain-task-force/report-2019/canadian-pain-task-force-June-2019-report-en.pdf. Accessed 2023 Sep 17.
3.World Health Organization. Unlocking the potential of ICD-11 for chronic pain. 2023; https://www.who.int/news-room/events/detail/2023/06/14/default-calendar/who-webinar-on-unlocking-the-potential-of-icd-11-for-chronic-pain. Accessed 2023 Sep 17.
4.Russo M, Santarelli D, Wright R, Gilligan C. A history of the development of radiofrequency neurotomy. J Pain Res. 2021;14:3897-3907. PubMed
5.Jamison DE, Cohen SP. Radiofrequency techniques to treat chronic knee pain: a comprehensive review of anatomy, effectiveness, treatment parameters, and patient selection. J Pain Res. 2018;11:1879-1888. PubMed
6.Kapural L, Deering JP. A technological overview of cooled radiofrequency ablation and its effectiveness in the management of chronic knee pain. Pain Manag. 2020;10(3):133-140. PubMed
7.Fogarty AE, Burnham T, Kuo K, et al. The effectiveness of fluoroscopically guided genicular nerve radiofrequency ablation for the treatment of chronic knee pain due to osteoarthritis: a systematic review. Am J Phys Med Rehabil. 2022;101(5):482-492. PubMed
8.Kenevan M. Long-lasting pain relief with radiofrequency ablation. Rochester (MN): Mayo Clinic Health System; 2023: https://www.mayoclinichealthsystem.org/hometown-health/speaking-of-health/radiofrequency-ablation-pain-relief#:~:text=Pain%20relief%20can%20occur%20anywhere,pain%20relief%20after%20radiofrequency%20ablation. Accessed 2023 Sep 23.
9.Gupta A, Huettner DP, Dukewich M. Comparative effectiveness review of cooled versus pulsed radiofrequency ablation for the treatment of knee osteoarthritis: a systematic review. Pain Physician. 2017;20(3):155-171. PubMed
10.Malaithong W, Tontisirin N, Seangrung R, Wongsak S, Cohen SP. Bipolar radiofrequency ablation of the superomedial (SM), superolateral (SL) and inferomedial (IM) genicular nerves for chronic osteoarthritis knee pain: a randomized double-blind placebo-controlled trial with 12-month follow-up. Reg Anesth Pain Med. 2022;48(4):151-160. PubMed
11.Tran A, Gonzalez FM. Review of cooled radiofrequency ablation utilization for the treatment of symptomatic advanced knee arthritis and total knee arthroplasty. Skeletal Radiol. 2023;52(5):941-949. PubMed
12.Canadian Institute for Health Information. Wait times for priority procedures in Canada, 2022. 2022; https://www.cihi.ca/en/wait-times-for-priority-procedures-in-canada-2022. Accessed 2023 Sep 17.
13.Loh E, Reid JN, Alibrahim F, Welk B. Retrospective cohort study of healthcare utilization and opioid use following radiofrequency ablation for chronic axial spine pain in Ontario, Canada. Reg Anesth Pain Med. 2019;44(3):398-405. PubMed
14.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
15.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377-384. PubMed
16.Agree Next Steps Consortium. The AGREE II Instrument. Hamilton, ON: AGREE Enterprise; 2017: https://www.agreetrust.org/wp-content/uploads/2017/12/AGREE-II-Users-Manual-and-23-item-Instrument-2009-Update-2017.pdf. Accessed 2023 Aug 21.
17.Ikeuchi M, Ushida T, Izumi M, Tani T. Percutaneous radiofrequency treatment for refractory anteromedial pain of osteoarthritic knees. Pain Med. 2011;12(4):546-551. PubMed
18.Ajrawat P, Radomski L, Bhatia A, Peng P, Nath N, Gandhi R. Radiofrequency procedures for the treatment of symptomatic knee osteoarthritis: a systematic review. Pain Med. 2020;21(2):333-348. PubMed
19.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
20.Chen AF, Mullen K, Casambre F, Visvabharathy V, Brown GA. Thermal nerve radiofrequency ablation for the nonsurgical treatment of knee osteoarthritis: A systematic literature review. J Am Acad Orthop Surg. 2021;29(9):387-396. PubMed
21.Chou R, Fu R, Dana T, Pappas M, Hart E, Mauer KM. Interventional treatments for acute and chronic pain: systematic review. (Comparative effectiveness review no. 247). Rockville (MD): Agency for Healthcare Research and Quality; 2021: https://www.ncbi.nlm.nih.gov/books/NBK573963/. Accessed 2023 Sep 26.
22.Kao G, Kalangara J, Woodbury A. Percutaneous, thermal,and pulsed radiofrequency for nonmalignant hip pain. Interv Pain Manag Rep. 2018;2(2):59-68.
23.National Institute for Health and Care Excellence. Radiofrequency denervation for osteoarthritic knee pain. (Interventional procedures guidance IPG767) 2023; https://www.nice.org.uk/guidance/ipg767. Accessed 2023 Sep 7.
24.Kolasinski SL, Neogi T, Hochberg MC, et al. 2019 American College of Rheumatology/Arthritis Foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Care Res (Hoboken). 2020;72(2):149-162. PubMed
25.Lee DW, Pritzlaff S, Jung MJ, et al. Latest evidence-based application for radiofrequency neurotomy (LEARN): best practice guidelines from the American Society of Pain and Neuroscience (ASPN). J Pain Res. 2021;14:2807-2831. PubMed
26.Lee A, Gupta M, Boyinepally K, Stokey PJ, Ebraheim NA. Sacroiliitis: a review on anatomy, diagnosis, and treatment. Adv Orthop. 2022;2022:3283296. PubMed
27.Jadon A, Jain P, Motaka M, Swarupa CP, Amir M. Comparative evaluation of monopolar and bipolar radiofrequency ablation of genicular nerves in chronic knee pain due to osteoarthritis. Indian J Anaesth. 2018;62(11):876-880. PubMed
28.McCormick ZL, Reddy R, Korn M, et al. A prospective randomized trial of prognostic genicular nerve blocks to determine the predictive value for the outcome of cooled radiofrequency ablation for chronic knee pain due to osteoarthritis. Pain Med. 2018;19(8):1628-1638. PubMed
29.National Institute for Health and Care Excellence. Interventional procedures programme manual. (Process and methods PMG28) 2016; https://www.nice.org.uk/process/pmg28/chapter/introduction. Accessed 2023 Sep 7.
30.National Institute for Health and Care Excellence. Radiofrequency denervation for osteoarthritic knee pain: information for the public. (Interventional procedures guidance IPG767) 2023; https://www.nice.org.uk/guidance/ipg767/informationforpublic. Accessed 2023 Sep 7.
31.National Institute for Health and Care Excellence. Principles for putting evidence-based guidance into practice. 2023; https://intopractice.nice.org.uk/principles-putting-evidence-based-guidance-into-practice/index.html. Accessed 2023 Sep 7.
32.National Institute for Health and Care Excellence. Interventional procedures guidance audit tool template. 2023; https://www.nice.org.uk/About/What-we-do/Our-Programmes/NICE-guidance/NICE-interventional-procedures-guidance. Accessed 2023 Sep 7.
33.Interventional procedure overview of radiofrequency denervation for osteoarthritic knee pain. Manchester (UK): National Institute for Health and Care Excellence; 2023: https://www.nice.org.uk/guidance/ipg767/evidence/overview-final-pdf-13074460093. Accessed 2023 Sep 7.
Appendix 1: Selection of Included Studies
Note that this appendix has not been copy-edited.
Appendix 2: Characteristics of Included Publications
Note that this appendix has not been copy-edited.
Table 2: Characteristics of Included Systematic Reviews
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Knee pain | ||||
Fogarty et al. (2022)7 US Funding source: NR | A total of 5 RCTs (across 8 publications) and 1 case series) were included; 3 RCTs relevant to this present review.a Results were synthesized mnarratively. | Patients > = 18 years with chronic knee pain due to OA. 392 patients were included in the relevant studies. Studies were excluded if patients underwent total knee arthroplasty. | Intervention: fluoroscopically guided genicular nerve RFA:
Comparator: Relevant to present review:
Not relevant:
| Outcome (measure): ≥ 50% pain reduction (VAS), function (WOMAC, OKS), and adverse events (n). Follow-up: 6, 12, 18 and 24 months. |
Chen et al. (2021)20 US Funding source: Hangzhou medical and Health Technology Planning Program | A total of 7 RCTs were included (5 high quality and 2 moderate quality). | Patients with symptomatic knee OA. No other demographic and clinical details were NR. | Intervention: Cooled RFA (1 trial) and RFA (6 trials). Comparator:
| Outcome (measure): Pain (VAS), function (WOMAC) and adverse events (n). Follow-up: 12 and 24 weeks. |
Hip pain | ||||
Chou et al. (2021)21 US Funding source: AHRQ | RCTs and cohort studies (if RCTs not available). 37 RCTs were included. No evidence on the efficacy or safety of RFA for hip pain was found. | Population: Adults with pain, of any duration, due to: vertebral compression fracture; degenerative back or hip pain; presumed discogenic back pain; radicular low back pain; trigeminal neuralgia; headache; piriformis syndrome; or ulnar, median or radial neuropathy. Relevant population: Adults ≥ 18 years of age undergoing RFA for hip pain of any duration | Intervention: vertebral augmentation procedures; intradiscal and facet joint platelet-rich plasma; intradiscal stem cells; intradiscal methylene blue; intradiscal ozone; sphenopalatine block; occipital stimulation; piriformis injection; peripheral nerve stimulation. Relevant intervention: cooled or pulsed RFA for degenerative hip pain. Comparators: usual care, sham, placebo, no treatment or conventional RFA. | Outcomes: primary outcomes were pain and function. Secondary outcomes were quality of life, emotional function, global improvement, and harms. Follow-up: 1 to 2 weeks, 2 to 4 weeks, 1 to 6 months, 6 to 12 months and ≥ 12 months. |
Kao et al. (2018)22 US Funding source: Veteran’s Affairs Rehabilitation Research and Development Service | Clinical trials, case series’ and case reports were included. Number of studies: 2 unblinded, noncontrolled clinical trials and 8 case series/reports were included and synthesized qualitatively. Number of relevant studies: 1 clinical trial and 7 case series/reports, including 2 studies using pulsed RFA which were not excluded, but reported separately, due to limited amount of evidence for the hip joint identified through the present review. | Population: Adults ≥ 18 years of age with chronic (≥ 3 months) with nonmalignant hip-related pain. Despite the exclusion of malignancy, one trial included patients with cancer so was excluded from the present review. Relevant population: patients with nonmalignant and nonsurgical hip pain. A total of 43 patients was included in the review:
| Intervention: thermal, cooled and pulsed RFA. Four studies reported pre-treatment diagnostic block. One case report involved 2 treatments (conventional single needle, then bipolar). Relevant intervention: thermal and cooled RFA Comparator: nonblinded control (standard therapy). | Outcome (measure): pain (VAS), function, (Lower Extremity Functional Scale) and adverse events (n). Follow-up: 4 weeks to 2 years. |
AHRQ = The Agency for Health care Research and Quality; CRF = cooled radiofrequency ablation; CRMRF = capacitive resistive monopolar radiofrequency; GPE = global perceived effort; HA = hyaluronic acid; NaHA = sodium hyaluronate; IA-HA = intra-articular hyaluronic acid; IAS = intra-articular steroid; NR = not reported; OA = osteoarthritis; RF = radiofrequency; RFA = radiofrequency ablation; VAS = visual analogue scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
a2 RCTs included in this SR were outside the scope of the present review. Jadon (2018)27 compared 2 RFA modalities (monopolar vs. bipolar conventional RFA); and McCormick (2018)28 examined the effectiveness of RFA on knee pain, with and without prognostic block..
Table 3: Characteristics of Included Primary Clinical Studies
Study citation, country, funding source | Study design | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Malaithong et al. (2022)10 Thailand Funding source: internal funding | RCT Patients, outcome assessors and statisticians were blind to treatment allocation | A total of 64 patients with severe OA chronic knee pain, (32 in each group) were enrolled in the trial:
| Intervention: genicular nerve bipolar RFA with fluoroscopic guidance, plus local anesthetic and steroid injection. 10cm, 18 gauge cannula with 10mm active tip. Sensory stimulation for localization. Lesioning occurred simultaneously at each of the 3 target nerves. Comparator: genicular block at 3 target sites, local anesthetic and steroid injection (same dose as intervention group) plus sham-RFA. All patients underwent a prognostic block before randomization to exclude patients with a negative response to the block (< 50% pain relief). | Outcome (measure): pain (VAS), function (WOMAC, PGI-I), adverse events (n) Follow-up: 12 months. Outcomes measured at 1, 2, 4, 6, 8, 10 and 12 months. |
Ikeuchi et al. (2011)17 Japan Funding source: NR | Prospective, nonrandomized open-label and controlled study. Recruitment through convenience sample at a clinic (clinic details not provided). Eligible patients presenting to the clinic between August and December 2005 were assigned as candidates for the intervention group. Eligible patients presenting between January and May 2006 were candidates for the control group. | A total of 35 patients with moderate or severe refractory anteromedial knee pain associated with knee OA were enrolled (n = 18 in the intervention group and n = 17 in the control group):
| Intervention: 2 RFA treatments 2 weeks apart. 50mm cannula with 5mm active tip. Sensory stimulation for localization. Control: local anesthetic Both intervention and control treatments were applied to the medial retinacular nerve and the infrapatellar branch of the saphenous nerve. 3 weeks before the first intervention, other treatments were stopped: physical therapy, acupuncture, regular use of Aspirin or NSAID, IAS or IA-HA. These were allowed to resume after 12 weeks after the first procedure. Home exercises were allowed to continue throughout. Loxoprofen sodium was allowed up to 3 tablets per day, as a rescue analgesic | Outcome (measure): pain (VAS); function (WOMAC), patient’s global assessment, adverse events (n). Follow-up: outcomes assessed at baseline, 4, 8, 12 weeks and 6 months after the first procedure. |
IA-HA = intra-articular hyaluronic acid; IAS = intra-articular steroid NR = not reported; NSAID = nonsteroidal anti-inflammatory drug; PGI-I = patient global impression of improvement Likert scale; VAS = visual analogue scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Table 4: Characteristics of Included Guidelines
Intended users, target population | Intervention and practice considered | Major outcomes considered | Evidence collection, selection, and synthesis | Evidence quality assessment | Recommendations development and evaluation | Guideline validation |
|---|---|---|---|---|---|---|
NICE 202323 | ||||||
Intended users: not explicitly reported, but accompanying tools available for both public (e.g., “questions to ask”) and health professionals (e.g., auditing and monitoring tracking spreadsheet) Target population: patients with osteoarthritic knee pain | RFA (conventional, cooled and pulsed) | Efficacy
Safety
| Rapid review on the efficacy and safety of RFA. Databases were searched from inception to 10 February 2023. A grey literature search was conducted as well (date NR). Study types: clinical studies. Abstracts were included if they reported outcomes No information on method for evidence assessment or selection Study characteristics of key included studies (n = 9) summarized in table format. N = 51 studies met the inclusion criteria, but only 9 were included in the evidence summary. | NR | Methods used by the guideline development group to produce recommendations was NR Strength of recommendation NR | No information on internal/external consultation for this guideline, but NICE’s general procedure is available on its website.29 External review: by the public, national patient organizations if applicable, medical device companies, professional organizations and other stakeholders. Internal review: NR |
ASPN 202125 | ||||||
Intended Users: clinicians Target Population: people with chronic pain in the cervical, thoracic, lumbar spine; posterior sacroiliac joint; hip and knee joints; and occipital neuralgia. | RFA | Primary:
Secondary:
| Formal literature search of 3 databases (date of search NR). Study types: Systematic reviews, literature reviews, RCTs, prospective and retrospective observational studies. A meta-analysis was not conducted. Evidence is summarized in table format by anatomic target (e.g., knee joints, hip joints). | The USPSTF criteria for quality of evidence was used (I, II-1, II-2, II-3 and III). | Recommendations were produced via consensus. Each recommendation statement was assigned a strength of recommendation as per the USPSTF criteria (A, B, C, D or I). The recommendation for use of RFA for knee pain is based on evidence from 4 studies. The recommendation for use of RFA for hip pain is based on evidence from 5 studies. | Methods for internal and external review were NR. |
ACR 202124 | ||||||
Intended Users: health care providers, caregivers and patients. Target Population: patients with osteoarthritis of hand, hip and/or knee and no specific contraindicators to the recommended therapies. | Pharmacologic and nonpharmacologic management of OA of the hand, hip and knee, including RFA plus usual care (maximally tolerable therapeutic doses of acetaminophen or nonsteroidal anti-inflammatory drugs). | Critical outcomes
| A systematic search of the literature was conducted. Databases were searched from inception to October 15, 2017 which update search on August 1, 2018. Study types: RCTs, observational studies | The quality of the evidence was assigned as per the GRADE methodology which characterizes the certainty of evidence on a four-level scale:
| Voting panel members voted on the direction and strength of reach recommendation. A threshold of 70% agreement among members was required to accept a recommendation. If not reached, additional discussions were held before re-voting. The strength of recommendations was assigned as per the GRADE methodology:
| Methods for internal and external review were NR. |
ACR = American College of Rheumatology; EQ-5D = standardized measure of health-related quality of life; GRADE = Grading of Recommendations Assessment, Development and Evaluation; NA = not applicable; NICE = National Institute for Health and Care Excellence; NR = not reported.; NRS = Numeric Rating Scale; RCT = randomized controlled trial; RFA = radiofrequency ablation; VAS = visual analogue scale; USPSTF = United States Preventive Services Task Force; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Appendix 3: Critical Appraisal of Included Publications
Note that this appendix has not been copy-edited.
Table 5: Strengths and Limitations of Systematic Reviews Using AMSTAR 214
Strengths | Limitations |
|---|---|
Knee pain | |
Fogarty (2022)7 | |
|
|
Chen (2021)20 | |
|
|
Hip pain | |
Chou (2021)21 | |
|
|
Kao (2018)22 | |
|
|
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; NA = not applicable; PICO = population, intervention, comparator, outcome, study type; RoB = risk of bias.
Table 6: Strengths and Limitations of Clinical Studies Using the Downs and Black Checklist15
Strengths | Limitations |
|---|---|
Malaithong (2022)10 | |
|
|
Ikeuchi (2011)17 | |
|
|
Table 7: Strengths and Limitations of Guidelines Using AGREE II16
Item | NICE (2023)23 | ASPN (2021)25 | ACR 202124 |
|---|---|---|---|
Domain 1: Scope and purpose | |||
1. The overall objective(s) of the guideline is (are) specifically described. | Yes | No Can be implied but not specifically described | Yes |
2. The health question(s) covered by the guideline is (are) specifically described. | No | No Can be implied but not specifically described | Yes |
3. The population (patients, public, etc.) to whom the guideline is meant to apply is specifically described. | No | No Can be implied but not specifically described | Yes |
Domain 2: Stakeholder involvement | |||
4. The guideline development group includes individuals from all relevant professional groups. | Unsure | No Development group limited to pain experts | Yes |
5. The views and preferences of the target population (patients, public, etc.) have been sought. | Unsure | Unsure | Yes Two patients were part of the guideline development group |
6. The target users of the guideline are clearly defined. | No | Yes | Yes |
Domain 3: Rigour of development | |||
7. Systematic methods were used to search for evidence. | Yes | Yes | Yes |
8. The criteria for selecting the evidence are clearly described. | No | Yes | Yes |
9. The strengths and limitations of the body of evidence are clearly described. | No | No | Yes |
10. The methods for formulating the recommendations are clearly described. | No | No | Yes |
11. The health benefits, side effects, and risks have been considered in formulating the recommendations. | Yes | No | Yes |
12. There is an explicit link between the recommendations and the supporting evidence. | No | Yes | Yes |
13. The guideline has been externally reviewed by experts before its publication. | Unsure | Unsure | Unsure |
14. A procedure for updating the guideline is provided. | Yes | No | No |
Domain 4: Clarity of presentation | |||
15. The recommendations are specific and unambiguous. | Yes | Yes | Yes |
16. The different options for management of the condition or health issue are clearly presented. | No | Yes | No |
17. Key recommendations are easily identifiable. | Yes | Yes | Yes |
Domain 5: Applicability | |||
18. The guideline describes facilitators and barriers to its application. | Yes There is a patient-focused section of the NICE website for this guideline that describes key questions to consider and how to have the discussion about the procedure with their health care professional30 | Yes Best practice summaries were provided for each anatomic target which include some clinical barriers and facilitators to treatment, if applicable. | No |
19. The guideline provides advice and/or tools on how the recommendations can be put into practice. | Yes NICE has a document describing general principles for putting evidence-based guidance into practice (i.e., not specifically for this guideline)31 | Yes The best practice summaries contain some clinical advice on implementation | No |
20. The potential resource implications of applying the recommendations have been considered. | No | No | No |
21. The guideline presents monitoring and/or auditing criteria. | Yes NICE has a generic tool (spreadsheet) to facilitate the audit of interventional procedures32 | No | No |
Domain 6: Editorial independence | |||
22. The views of the funding body have not influenced the content of the guideline. | Unsure | Yes | Yes |
23. Competing interests of guideline development group members have been recorded and addressed. | No | Unsure | Yes |
ACR = American College of Rheumatology; AGREE II = Appraisal of Guidelines for Research and Evaluation II; ASPN = American Society of Pain and Neuroscience; 0NICE = National Institute for Health and Care Excellent; NR = not reported.
Appendix 4: Main Study Findings
Table 8: Summary of Findings by Outcome for Knee Pain — Pain (VAS)
Study citation, design | N studies | Substudy (if applicable) | Measure (summary statistic) | N patients | Intervention | Comparator | Treatment group difference | P value | Direction of effect |
|---|---|---|---|---|---|---|---|---|---|
VAS at 4 weeks | |||||||||
Malaithong et al. (2022)10 RCT | 1 | NA | Mean difference from baseline (SD) | 64 | Fluoroscopy guided bimodal RFA | Sham-RFA | Intervention: 2.7 (2.3) Control: 1.9 (2.2) | 0.15 | No difference between groups |
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Mean VAS (narrative summary) | 35 | RFA | Local anesthetic | The authors concluded that mean VAS was lower among the intervention group, compared to the control group. | 0.007 | Favours intervention |
VAS at 8 weeks | |||||||||
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Mean VAS (narrative summary) | 35 | RFA | Local anesthetic | The authors concluded that mean VAS was lower among the intervention group, compared to the control group. | 0.028 | Favours intervention |
VAS at 12 weeks | |||||||||
Chen et al. (2021)20 Systematic Review | 6 | Choi et al. (2011) | Mean difference between groups (95% CI) | 35 | RFA | Sham-RFA | −3.55 (−4.84, −2.26) | NR | Favours intervention |
Shen et al. (2011) | 54 | RFA | Control | −2.09 (−2.38, −1.80) | NR | Favours intervention | |||
El-Hakeim et al. (2018) | 60 | Conventional RFA | Oral analgesic | −2.10 (−3.16, −1.04) | 0.004 | Favours intervention | |||
Ray et al. (2018) | 24 | RFA | IA-HA | −4.50 (−5.84, −3.16) | NR | Favours intervention | |||
Xiao et al. (2017) | 96 | RFA | IA-HA | −3.28 (−3.72, −2.84) | NR | Favours intervention | |||
Davis et al. (2018) | 133 | Cooled RFA | IAS | −2.40 (−3.12, −1.68) | NR | Favours intervention | |||
VAS at 6 months | |||||||||
Fogarty et al. (2022)7 Systematic review | 3 | Chen et al. (2020) | ≥ 50% pain relief (RR [95% CI]) | 158 | Cooled monopolar RFA | IA-HA | 1.88 (1.38 to 2.57) | NR | Favours intervention |
El-Hakeim et al. (2018) | Mean (SD) | 60 | Conventional RFA | Oral analgesic | Intervention: 3.13 (0.3) Control: 5.73 (0.26) | < 0.001 | Favours intervention | ||
Davis et al. (2018) | ≥ 50% pain relief (RR [95% CI]) | 151 | Cooled RFA | IAS | 4.58 (2.61 to 8.04) | NR | Favours intervention | ||
Chen et al. (2021)20 Systematic review | 1 | Xiao et al. (2017) | Mean difference (95% CI) | 96 | RFA | IA-HA | −2.72 (−3.36, −1.82) | NR | Favours intervention |
Malaithong et al. (2022)10 RCT | 1 | NA | Mean difference from baseline (SD) | 59 | Fluoroscopy guided bimodal RFA | Sham-RFA | Intervention: 2.4 (2.7) Control: 1.7 (2.7) | 0.29 | No difference between groups |
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Narrative summary | 35 | RFA | Local anesthetic | The authors concluded mean VAS was lower among the intervention group, compared to the control group. | 0.006 | Favours intervention |
VAS at 12 months | |||||||||
Malaithong et al. (2022)10 RCT | 1 | NA | Mean difference from baseline (SD) | 53 | Fluoroscopy guided bimodal RFA | Sham-RFA | Intervention: 2.3 (2.8) Control: 2.2 (2.4) | 0.73 | No difference between groups |
CI = confidence interval; HA = hyaluronic acid; IA-HA = intra-articular hyaluronic acid; IAS = intra-articular steroid injection; NA = not applicable; NR = not reported; RCT = randomized controlled trial; RFA = radiofrequency ablation; RR = relative risk; SD = standard deviation; VAS = visual analogue scale.
Note that this table has not been copy-edited.
Table 9: Summary of Findings by Outcome for Knee Pain — Function (WOMAC)
Study citation, design | N studies | Substudy (if applicable) | Measure (summary statistic) | N patients | Intervention | Comparator | Treatment group difference | P value | Direction of effect |
|---|---|---|---|---|---|---|---|---|---|
WOMAC at 4 weeks | |||||||||
Malaithong et al. (2022)10 RCT | 1 | NA | Mean difference from baseline, within-group (SD) | 64 | Fluoroscopy guided bimodal RFA | Sham-RFA | Intervention: 26.6 (37.7) Control: 27.2 (37.7) | 0.78 | No difference between groups |
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Narrative summary | 35 | RFA | Local anesthetic | The authors concluded there was no difference in WOMAC scores between the intervention and control groups. | NR | No difference |
WOMAC at 8 weeks | |||||||||
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Narrative summary | 35 | RFA | Local anesthetic | The authors concluded there was no difference in WOMAC scores between the intervention and control groups. | NR | No difference |
WOMAC at 12 weeks | |||||||||
Chen et al. (2021)20 Systematic review | 4 | Choi et al. (2011) | Mean difference between groups (95% CI) | 35 | RFA | Sham-RFA | −11.50 (−16.65, −6.15) | NR | Favours intervention |
Shen et al. (2011) | 54 | RFA | Control | −9.55 (−15.02, −4.08) | NR | Favours intervention | |||
Xiao et al. (2017) | 96 | RFA | IA-HA | −12.50 (−14.48, −10.52) | NR | Favours intervention | |||
Davis et al. (2018) | 133 | Cooled | IAS | −10.00 (−12.71, −7.29) | NR | Favours intervention | |||
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Narrative summary | 35 | RFA | Local anesthetic | The authors concluded there was no difference in WOMAC scores between the intervention and control groups. | NR | No difference |
WOMAC at 6 months | |||||||||
Fogarty et al. (2022)7 Systematic review | 2 | El-Hakeim et al. (2018) | Mean (SD) | 60 | Monoplar RFA | Medical management | Intervention: 33.13 (4.1) Control: 43.5 (2.0) | < 0.0001 | Favours intervention |
Chen et al. (2020) | Proportion improvement from baseline | 177 | Cooled RFA | IA-HA | Intervention: 48.2% Control: 22.6% | < 0.0001 | Favours intervention | ||
Chen et al. (2021)20 Systematic review | 2 | Xiao et al. (2017) | Mean difference between groups (95% CI) | 96 | RFA | IA-HA | −14.20 (−16.76, −11.64) | NR | Favours intervention |
Davis et al. (2018) | 125 | RFA | IAS | −13.30 (−10.26) | NR | Favours intervention | |||
Malaithong et al. (2022)10 RCT | 1 | NA | Mean difference from baseline, within-group (SD) | 59 | Fluoroscopy guided bimodal RFA | Sham-RFA | Intervention: 27.1 (42.7) Control: 34.7 (54.4) | 0.81 | No difference between groups |
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Narrative summary | 35 | RFA | local anesthetic | The authors concluded there was no difference in WOMAC scores between the intervention and control groups. | 0.0066 | No difference between groups |
WOMAC at 12 months | |||||||||
Malaithong et al. (2022)10 RCT | 1 | NA | Mean difference from baseline, within-group (SD) | 53 | Fluoroscopy guided bimodal RFA | Sham-RFA | Intervention: 17.7 (49.2) Control: 24.6 (38.5) | 0.7 | No difference between groups |
HA = hyaluronic acid; IA-HA = intra-articular hyaluronic acid; IAS = intra-articular steroid injection; NA = not applicable; NR = not reported; RCT = randomized controlled trial; RFA = radiofrequency ablation; RR = relative risk; SD = standard deviation; VAS = visual analogue scale.
Note that this table has not been copy-edited.
Table 10: Summary of Findings by Outcome for Knee Pain — Function (PGI-I)
Study citation, design | N studies | Substudy (if applicable) | Measure (summary statistic) | N patients | Intervention | Comparator | Treatment group difference | P value | Direction of effect |
|---|---|---|---|---|---|---|---|---|---|
Malaithong et al. (2022)10 RCT | 1 | NA | Mean (SD) | 64 | Fluoroscopy guided bimodal RFA | Sham-RFA | 4 weeks: Intervention: 2.3 (0.8) Control: 2.5 (0.8) 6 months: Intervention: 2.6 (1.0) Control: 2.4 (0.9) 12 months: Intervention: 2.8 (1.3) Control: 2.6 (1.3) | 0.65 0.46 0.56 | No difference between groups |
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Mean (SD) | 35 | RFA | Local anesthetic | 6 months: Intervention: 1.5 (0.8) Control: 1.1 (0.6) | 0.126 | No difference between groups |
PGI-I = patient global impression of improvement Likert scale; RFA = radiofrequency ablation; PGI-I = SD = standard deviation.
Note that this table has not been copy-edited.
Table 11: Summary of Findings by Outcome for Knee Pain — Function (OKS)
Study citation, design | N studies | Substudy (if applicable) | Measure (summary statistic) | N patients | Intervention | Comparator | Treatment group difference | P value | Direction of effect |
|---|---|---|---|---|---|---|---|---|---|
OKS at 6 months | |||||||||
Fogarty et al. (2022)7 Systematic review | 1 | Davis et al. (2018) | Mean (SD) | 126 | Cooled RFA | IAS | Intervention: 35.7 (8.5) Control: 22.4 (8.5) | < 0.001 | Favours intervention |
OKS at 12 months | |||||||||
Fogarty et al. (2022)7 Systematic review | 1 | Davis et al. (2019) | Mean (SD) | 52 | Cooled RFA | IAS | Intervention: 34.3 (11.1) Control: 22.0 (16.0) | 0.11 | No difference between groups |
OKS at 24 months | |||||||||
Fogarty et al. (2022)7 Systematic review | 1 | Hunter et al. (2020) | Mean difference from baseline (SD) | NR | Cooled RFA | NA | Baseline: 20.2 (7.3) 24 months: 46.8 (10.3) | < 0.001 | Significant improvement from baseline |
IAS = intra-articular steroid; OKS = Oxford Knee Score; RFA = radiofrequency ablation; NA = not applicable; SD = standard deviation.
Note that this table has not been copy-edited.
Table 12: Summary of Findings by Outcome for Knee Pain — Adverse events
Study citation, design | N studies | Substudy (if applicable) | Adverse event | Intervention group | Comparator group |
|---|---|---|---|---|---|
Fogarty et al. (2022)7 Systematic review | 2 | Davis et al. (2018) | Any | Cooled RFA: 3/76 = 3.9% | IAS: 7/75 = 9.3% |
El-Hakeim et al. (2018) | Serious AE | Conventional RFA: No serious AE | Medical management: No serious AE | ||
Chen et al. (2021)20 Systematic review | 3 | Choi et al. (2011) | Any | RFA: No AE | Sham-RFA: No AE |
Sari et al. (2018) | Any | RFA: No AE | IAS: No AE | ||
Ray et al. (2018) | Any | RFA: No AE | IA-HA: No AE | ||
Malaithong et al. (2020)10 RCT | 1 | NA | Serious AE (significant swelling) | Bimodal RFA: 1/32 = 3.1% | Sham-RFA: 0/32 = 0% |
Ikeuchi et al. (2011)17 Nonrandomized trial | 1 | NA | Serious AE | RFA: No serious AE | Local anesthetic: No serious AE |
Subcutaneous bleeding at the site of needle insertion | RFA: 12/18 = 66.7% | Local anesthetic: 11/14 = 82.4% | |||
Prolonged hypoesthesia at the IPBSN region | RFA: 14/18 = 77.8% | Local anesthetic: No AE |
AE = adverse event; IA-HA = intra-articular hyaluronic acid; IAS = intra-articular steroid; IPBSN = infrapatellar branch of the saphenous nerve; RFA = radiofrequency ablation; RCT = randomized controlled trial.
Note that this table has not been copy-edited.
Table 13: Summary of Findings by Outcome for Hip Pain — Pain (VAS)
Primary Study Within Kao (2018),22 Systematic Review | Preprocedure VAS score, mean | Postprocedure VAS score, mean | Duration of follow-up |
|---|---|---|---|
RFAa | |||
Fukui (2001) Case report | 9.5 (range: 9 to 10) | 1.5 (range: 1 to 2) | 2 years |
Malik (2003) Case series (n = 3) | 8.2 (range: 7 to 9.5) | 3 (range: 2 to 4) 3 (range: 2 to 4) 4 (range: NR) | 4 weeks 8 weeks 12 weeks |
Rivera (2012) Prospective uncontrolled study (n = 17) | 9.5 (range: 7 to 10) SD: 0.79 | 6.35 (range: 6 to 10) SD: 2.17, P < 0.05 | 6 months |
Cortinas-Saenz (2014) Case series (n = 3) | NR | Average 68.3% improvement | 4 weeks |
Gupta (2014) Case report | NR | 90% relief after first RFA procedure. 20% to 50% relief after second RFA procedure. | 6 months NR |
Pulsed RFA | |||
Chye (2015) Nonrandomized controlled trial (n = 15) | 6.7 (SD: 0.6) | 2.2 (SD: 1.5) 2.4 (SD: 1.4) 3.0 (SD: 1.8) | 1 week 4 weeks 12 weeks |
Wu (2007) Case series (n = 2) | 9 (range: 8 to 10) | 0 (range: NR) 3.5 (range: NR) | Immediately postprocedure 3 and 4 months |
NR = not reported; SD = standard deviation; VAS = visual analogue scale.
aRFA excluded pulsed RFA.
Note that this table has not been copy-edited.
Table 14: Summary of Findings by Outcome for Hip Pain — Function (Lower Extremity Function Scale)
Primary Study Within Kao (2018),22 Systematic Review | Preprocedure score | Postprocedure score | Duration of follow-up | Direction of effect |
|---|---|---|---|---|
Austria (2016) Case report | 18/80 | 49/80 | 6 months | Favours intervention |
Note that this table has not been copy-edited.
Table 15: Summary of Findings by Outcome for Hip Pain — Adverse events
Primary study within Kao (2018),22 systematic review | Description of adverse event(s) |
|---|---|
RFAa | |
Malik (2003) Case series (n = 3) | Persistent lateral surface hip numbness for unknown duration (n = 1) |
Rivera (2012) Prospective uncontrolled trial (n = 17) | Transient hematoma from vessel puncture (n = 3) |
Cortinas-Saenz (2014) Case series (n = 3) | Persistent lateral surface hip numbness for unknown duration (n = 1) |
Pulsed | |
Chye (2015) Nonrandomized controlled trial (n = 15) | Subcutaneous hematoma (n = 1) |
aRFA excluding pulsed RFA.
Note that this table has not been copy-edited.
Table 16: Summary of Recommendations in Included Guidelines
Recommendations and supporting evidence | Quality of evidence and strength of recommendations |
|---|---|
NICE 202323 | |
“Radiofrequency denervation for osteoarthritic knee pain may be used if standard arrangements are in place for clinical governance, consent and audit (p. 2).”23 Efficacy outcomes Pain relief (measured by VAS): Evidence from an NMA of 16 RCTs (at 3 months follow-up) and 10 RCTs (at 6 months follow-up), 2 meta-analyses, 1 RCT, and 2 cohort studies showed that radiofrequency ablation was associated with a statistically significant decrease in pain compared to control at all time points (varied by study, but includes 1 week, and 1, 3, 6, 12, 18 and 24 months). Composite knee function and pain measures (WOMAC): Evidence from 1 NMA, 1 meta-analysis, 1 RCT and 1 cohort study showed that radiofrequency ablation was associated with a statistically significant decrease in WOMAC compared to control at all time points (varied by study, but includes 1, 3, 6, 18 and 24 months). Functional outcomes
Quality of life (measured by EQ-5D-5L): Evidence from 1 cohort study showed that total EQ-5D-5L among patients who received cooled radiofrequency ablation significantly increased from baseline at 18 and 24 months. Efficacy by treatment modality: Evidence from 1 NMA “found that whilst all modalities of RF denervation were typically more effective than placebo or exercise, patients responded better to the cooled modality than the conventional and pulsed modalities, and bipolar is more effective than monopolar for pain function in conventional and pulsed modalities…However, the authors caution that the number of studies, including for cooled modality, is insufficient (p. 28).”33 Safety outcomes Complications and major complications: Evidence from 1 NMA found evidence of adverse events reported in 6 studies: 8.4% of those in the treatment group with any adverse events, and 3.9% with major adverse events. Evidence from 1 meta-analysis found no significant difference in risk of adverse events between treatment and control groups. Other:
| NR |
“The procedure should only be done by clinicians with specific training and experience in this procedure (p. 2).”23 Supporting evidence NR | NR |
“For auditing the outcomes of this procedure, the main efficacy and safety outcomes identified in this guidance can be entered into NICE’s interventional procedure outcomes and audit took (for use at local discretion) (p. 2).”23 Supporting evidence NA | NA |
ASPN (2021)25 | |
“Genicular nerve radiofrequency neurotomy may be used for the treatment of osteoarthritis related and post-surgical knee joint paint. GRADE II-I B (p. 2821).”25 Evidence overview This recommendation was informed by 4 studies: a meta-analysis of 12 RCTs, a systematic review of 13 publications, 1 RCT (which was included in the systematic review) and 1 literature review. Efficacy outcomes Pain relief: The body of evidence included in this guideline (n = 4 studies) showed reduction in pain scores among those who received RFA compared to the control group at various time points (1, 3 and 12 months). Function: Evidence from the systematic review and RCT (which was included in the systematic review) showed improved function about the treatment groups compared to the controls. Safety outcomes Adverse events: The systematic review did not report any adverse events associated with RFA treatment. | Quality of evidence: Well-designed, controlled, nonrandomized clinical trial. Strength of recommendation: “…high certainty that the net benefit is moderate or there is a moderate certainty that the net benefit is moderate to substantial (p. 2810).”25 |
Best practices summary:
Evidence overview The link between the evidence and the best practice summary is not clearly defined: there is no evidence quality or strength assessment assigned to each summary point. No additional publications are cited to inform this practice summary. | NR |
“Hip joint radiofrequency neurotomy targeting the obturator and femoral nerve branches may be used for the treatment of hip joint pain following diagnostic blocks. GRADE II-I-B (p. 2824).”25 Evidence overview The efficacy of RFA for hip pain was informed by evidence from 4 studies: 2 retrospective uncontrolled studies, 1 prospective uncontrolled study, and 1 case series. Efficacy outcomes Pain relief: Evidence from the 4 studies showed improvement in pain scores from baseline. | Quality of evidence: Well-designed, controlled, nonrandomized clinical trial. Strength of recommendation: “…high certainty that the net benefit is moderate or there is a moderate certainty that the net benefit is moderate to substantial (p. 2810).”25 |
Best practices summary:
Evidence overview The link between the evidence and the best practice summary is not clearly defined: there is no evidence quality or strength assessment assigned to each summary point. In addition to the 4 studies on the hip joint referenced above, one additional anatomic study may have been used to inform this practice summary (it is summarized in a table with the other 4 studies). The authors assessed the contribution of the femoral, obturator and accessory obturator nerves and found that the femoral and obturator nerves contributed to the capsular innervation in all specimens (n = 3). | NR |
ACR 202124 | |
“Radiofrequency ablation is conditionally recommended for patients with knee OA. “A number of studies have demonstrated potential analgesic benefits with various ablation techniques but, because of the heterogeneity of techniques and controls used and lack of long-term safety data, this recommendation is conditional (p. 156).”24 Efficacy outcomes Pain relief: Evidence from 1 moderate quality and 2 high-quality RCTs found RFA was associated with decreased pain compared to sham ablation for OA knee. Evidence from 4 low-quality RCTs found that RFA was associated with decreased pain compared to intra-articular injections for OA knee. Function: Evidence from 1 low-quality RCT found that RFA was associated with improved SF-36 physical function score compared to intra-articular injections for OA knee. Evidence from 1 low-quality RCT found that RFA was associated with a higher rate of walking at 3 months compared to intra-articular injections for OA knee. Evidence from 1 low-quality RCT found that RFA was associated with improved stair climbing at 3 months compared to intra-articular injections for OA knee. Safety outcomes NR for knee or hip. | Quality of evidence: “Quality of evidence across all critical outcomes: moderate (for direct comparison data) (p. 322).”24 Strength of recommendation: Conditional |
ACR = American College of Rheumatology; ASPN = American Society of Pain and Neuroscience; NA = not applicable; NICE = National Institute for Health and Care Excellence; NMA = network meta-analysis; NR = not reported; OA = osteoarthritis; RCT = randomized controlled trial; RFA = radiofrequency ablation; VAS = visual analogue scale; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Note that this table has not been copy-edited.
Appendix 5: Overlap Between Included Systematic Reviews
Note that this appendix has not been copy-edited.
Table 17: Overlap in Relevant Primary Studies Between Included Systematic Reviews
Primary study citation | Fogarty (2022)7 | Chen (2021)20 |
|---|---|---|
Chen AF, et al. J Bone Joint Surg Am. 2020;102:1501 to 10. | Yes | — |
Chen AF, et al. BMC Musculoskelet Disord. 2020;21:363. | Yes | — |
Hunter C, et al. Pain Pract. 2020;20:238 to 46. | Yes | — |
Davis T, et al. Reg Anesth Pain Med. 2019;44:499 to 506. | Yes | — |
Davis T, et al. Reg Anesth Pain Med. 2018;43:84 to 91. | Yes | Yes |
El-Hakeim EH, et al. Pain Physician. 2018;21:169 to 77. | Yes | Yes |
Reddy RD, et al. Anesth Pain Med. 2016;6:e39696. | — | Yes |
Xiao L, et al. Exp Ther Med. 2018;5:3973 to 77. | — | Yes |
Sari S, et al. Int J Rheum Dis. 2018;21:1772 to 78. | — | Yes |
Ray D, et al. Indian J Pain. 2018;1:36 to 39 | — | Yes |
Shen WS, et al. Am J Ther. 2017;6:e693-e700. | Yes | — |
Choi WJ, et al. Pain. 2011;3:481 to 87. | — | Yes |
Appendix 6: References of Potential Interest
Note that this appendix has not been copy-edited.
Systematic Reviews
Network Meta-Analysis With Methodological or Administrative Issues
Wu L, Li Y, Si H, et al. Radiofrequency ablation in cooled monopolar or conventional bipolar modality yields more beneficial short-term clinical outcomes versus other treatments for knee osteoarthritis: a systematic review and network meta-analysis of randomized controlled trials. Arthroscopy. 2022 07;38(7):2287-2302. PubMed
Note: This SR with network meta-analysis was identified in the search and reviewed at full text. It was also a key piece of evidence used to inform the NICE guideline.23 It was excluded from the present review due to issues with critical citations: there were numerous instances where the references for the included studies were inaccurate in-text (i.e., they cross-referenced to a different citation in the reference list). Due to the presence of multiple publications from the same author, and one reference missing completely from the study’s reference list (i.e., Ahmed et al. was the first included study in-text, but not listed in the reference list). It was difficult to assess overlap between other SRs included in this review, and to interpret findings. Therefore, it was excluded from this review but shared here as it has relevant findings from direct and indirect comparisons of RFA effectiveness on knee pain.
Systematic Reviews That Met Inclusion Criteria but Were Excluded Due to Complete Overlap With Included SRs
Ajrawat P, Radomski L, Bhatia A, Peng P, Nath N, Gandhi R. Radiofrequency procedures for the treatment of symptomatic knee osteoarthritis: a systematic review. Pain Med. 2020 02 01;21(2):333-348. PubMed
Li G, Zhang Y, Tian L, Pan J. Radiofrequency ablation reduces pain for knee osteoarthritis: a meta-analysis of randomized controlled trials. Int J Surg. 2021 Jul;91:105951. PubMed
Zhang H, Wang B, He J, Du Z. Efficacy and safety of radiofrequency ablation for treatment of knee osteoarthritis: a meta-analysis of randomized controlled trials. J Int Med Res. 2021 Apr;49(4):3000605211006647. PubMed
Additional References
Case Report or Series Identified Through the Literature Search
Belba A, Vanneste T, Jerjir A, et al. Complex regional pain syndrome of the knee after conventional radiofrequency ablation of the genicular nerves treated successfully with dorsal root ganglion stimulation: a case report. Pain Pract. 2022;22(5):541-546. PubMed
Koshi E, Cheney CW, Sperry BP, Conger A, McCormick ZL. Genicular nerve radiofrequency ablation for chronic knee pain using a three-tined electrode: a technical description and case series. Pain Med. 2020;21(12):3344-3349. PubMed
Contributors: Kendra Brett, Elizabeth Carson
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for noncommercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca.