Drugs, Health Technologies, Health Systems
Health Technology Review
Comparative Evidence Between Transdermal and Oral Menopausal Hormone Therapy
Authors: Said Yousef Abdelrazeq, Shu-Ching Hsieh, Shannon E. Kelly, Nazmun Nahar, Becky Skidmore, Melissa Brouwers, Shariq Najeeb, George A. Wells
This rapid review was conducted by the POst-market Drug Evaluation Team (PODET) through the Post-Market Drug Evaluation (PMDE) CoLab Network.
Key Messages
What Is the Issue?
Hormone therapy may be prescribed to support individuals experiencing symptoms of menopause due to a decline in estrogen in the body. Menopausal symptoms may vary in frequency and intensity and commonly include vasomotor symptoms (VMS) (often referred to as hot flashes or night sweats), sleep disruption, and mood changes, and they may include impacts to bone or heart health and overall quality of life.
For individuals seeking support managing these symptoms, menopausal hormone therapy (MHT) with estrogen (or a combination of estrogen and progesterone) is an established treatment.
Various routes of administration (ways to take estrogen) are available for MHT with estrogen, including oral (taken as a pill) or transdermal (absorbed through the skin), but the comparative clinical efficacy, effectiveness, and safety of these administration routes are unclear.
Decision-makers are interested in whether transdermal estrogen should be considered for public reimbursement (funding) as a first treatment option (first-line option), as an alternative to oral estrogen for MHT.
What Did We Do?
We evaluated the evidence of the clinical efficacy, effectiveness, safety, and cost-effectiveness (value from a human or health system perspective) of transdermal versus oral estrogen in MHT using a rapid review approach.
We searched for evidence-based guidelines on the use of transdermal or oral estrogen in MHT.
What Did We Find?
We identified 7 systematic reviews (SRs), 4 primary studies, and 3 clinical practice guidelines relevant to this review. No relevant health technology assessment (HTA) reports or cost-effectiveness studies were identified.
The included studies suggest that transdermal estrogen may reduce VMS, improve sleep, and be associated with a lower risk of blood clots compared to oral estrogen. Both transdermal and oral MHT may improve bone health and have similar safety risk profiles for breast and gynecological cancers. Furthermore, transdermal MHT may be a safer choice for those at risk of developing blood clots; however, there are inconsistent results related to the risk of heart disease and stroke.
The included studies suggest that oral MHT is more effective at improving cholesterol levels but may raise triglyceride levels. In contrast, transdermal MHT has mixed effects on cholesterol levels, although it also raises triglyceride levels.
Guidelines recommend considering transdermal MHT over oral MHT for addressing specific individual concerns related to sexual well-being and reducing the risk of gallstones, blood clots, stroke, and heart disease.
What Does This Mean?
There is limited evidence comparing transdermal and oral MHT, particularly for managing VMS and improving health-related quality of life, and sleep quality. However, some studies suggest that transdermal MHT may be associated with a lower risk of venous thromboembolism (VTE) (blood clots in the veins).
Given the evidence on safety, policy-makers may consider reimbursement for transdermal MHT, but additional research is needed to inform considerations of efficacy and cost-effectiveness.
Abbreviations
AACE
American Association of Clinical Endocrinology
AGREE II
Appraisal of Guidelines for Research and Evaluation II
AMSTAR 2
A MeaSurement Tool to Assess systematic Reviews
BMD
bone mineral density
CHD
coronary heart disease
CVD
cardiovascular disease
HDL
high-density lipoprotein
HTA
health technology assessment
KMI
Kupperman Menopausal Index
LDL
low-density lipoprotein
MA
meta-analysis
MHT
menopausal hormone therapy
MRS
Menopause Rating Scale
NMA
network meta-analysis
PICO
population, intervention, comparator, outcomes
PRISMA
Preferred Reporting Items for Systematic reviews and Meta-Analyses
RCT
randomized controlled trials
RoB
risk of bias
ROBINS-I
Risk Of Bias In Non-randomized Studies of Interventions
SR
systematic review
VMS
vasomotor symptoms
VTE
venous thromboembolism
Introduction and Rationale
Background
Perimenopause, Menopause, and Postmenopause
Menopause marks the permanent end of menstruation due to the cessation of ovarian follicle development. It is defined by the final menstrual period, followed by 12 consecutive months without menstruation.1,2 Perimenopause, the transitional phase leading up to menopause, is characterized by irregular cycles and the onset of menopausal symptoms.3 The initial 12-month period without cycles is termed late perimenopause, while the time after this is known as postmenopause.1-3 Menopause can occur naturally or be medically induced through interventions such as surgery, chemotherapy, or radiation. Additionally, premature ovarian insufficiency, the cessation of ovarian function before the age of 40, is another cause of menopause.3 Factors such as genetics, environment, and lifestyle influence the age at which menopause occurs. The primary result of menopause is a decline in estrogen, leading to key symptoms and health concerns, including VMS, osteoporosis, cardiovascular disease (CVD), mood disorder, genitourinary syndrome, and sexual health issues.2-4
Symptoms commonly linked to menopause include hot flashes and night sweats, which are episodes of intense heat, sweating, and flushing, often occurring around the head, neck, chest, and upper back. VMS can persist for 1 to 2 years postmenopause and, in some cases, may last 10 years or more. On average, the duration of VMS is 7.4 years. Hot flashes are a leading reason many individuals seek medical attention. Additionally, patients often report issues with concentration, mood fluctuations, sleep disturbances, and other challenges in daily activities. Untreated menopausal symptoms can significantly impact personal well-being, relationships, and work productivity.3,5,6 Estrogen products used in MHT are considered an effective treatment for the symptoms of menopause when it started in individuals under 60 years of age or within 10 years of postmenopause. The most common routes of administration include oral tablets7 or transdermal MHT formulations that are applied directly to the skin and absorbed. In Canada, various estrogen and estrogen-progestogen combination products are available, including 2 transdermal formulation options — a topical gel (e.g., EstroGel) and a patch (e.g., Estradot).8,9 Several factors influence the choice of MHT, such as patient risk, preference, and adherence and cost. For individuals without a uterus, estrogen alone, at the lowest effective dose, is typically used. In those with an intact uterus, estrogen is combined with an endometrial protective agent to reduce the risk of irregular thickening of the uterine lining.9,10 Transdermal estrogen products may offer advantages over oral dosage forms, including a potentially lower risk of VTE, and they may be more suitable for certain individuals, such as those with migraines, high blood pressure, or elevated CVD risk.9
Policy Issue
Drug plans are seeking more robust and current evidence comparing transdermal and oral MHT for managing menopausal symptoms to guide reimbursement decision-making. The rationale for this rapid review arises from the need for a clear understanding of the clinical efficacy, effectiveness, safety, and cost-effectiveness of transdermal versus oral MHT.
Policy Question
Should transdermal MHT be reimbursed in the first-line setting, as an alternative to oral MHT, for the treatment of perimenopausal, menopausal, and postmenopausal symptoms?
Purpose
The objective of this project is to compare the clinical efficacy, effectiveness, safety, and cost-effectiveness of transdermal MHT versus oral MHT in individuals being treated for perimenopausal, menopausal, or postmenopausal symptoms.
Research Questions
The following research questions will be used to address the policy question:
What is the clinical efficacy, effectiveness, and safety of transdermal MHT compared to oral MHT for the treatment of perimenopausal, menopausal, and postmenopausal symptoms?
What are the evidence-based guideline recommendations regarding the use of transdermal MHT for the treatment of perimenopausal, menopausal, and postmenopausal symptoms?
What is the cost-effectiveness of transdermal MHT compared to oral MHT for the treatment of perimenopausal, menopausal, and postmenopausal symptoms?
Methods
We conducted a rapid review of the clinical efficacy, effectiveness, and safety of MHT and of relevant evidence-based guidelines and cost-effectiveness literature.
Literature Search Methods
An experienced medical information specialist developed and tested the search strategies through an iterative process in consultation with the review team. Another senior information specialist peer-reviewed the MEDLINE strategy prior to execution using the PRESS Checklist.11
Using the multifile option and deduplication tool available on the Ovid platform, we searched Ovid MEDLINE® ALL, Embase Classic+Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL). We applied a combination of controlled vocabulary (e.g., “menopause,” “hormone replacement therapy,” “administration, topical”) and keywords (e.g., “hot flash,” “HRT,” “transdermal”), adjusting the vocabulary and syntax as necessary across the databases. We incorporated robust design and economic filters in all databases but CENTRAL, which is prefiltered for randomized and controlled clinical trials. We did not restrict searches by language but did limit results to the publication years 2017 to the present to capture the most recent evidence and relevant updates in the field. We also excluded animal-only studies and opinion pieces. We performed all searches on August 26, 2024, and updated them on October 1, 2024. We downloaded and deduplicated the records using EndNote version 9.3.3 (Clarivate Analytics) and uploaded them to Covidence (Veritas Health Innovation Ltd.).
We conducted a focused search for relevant HTAs by exploring potentially relevant documents published by international organizations or country-specific HTA agencies in Canada, the US, the UK, Australia, and New Zealand. This search was guided by the Canada’s Drug Agency Grey Matters Checklist using keywords relevant to our research questions. A single reviewer systematically screened reports and publications from these HTA websites, retrieving any potentially relevant full-text documents for further evaluation to assess their eligibility for inclusion in the review.
A targeted search of various clinical guideline developers was conducted to identify guidelines for the use of MHT published since 2017. Practice guideline developers specializing in MHT, both within Canada and internationally, as well as reputable generalist organizations, were selected. The targeted list of developers included the International Menopause Society, the Canadian Menopause Society, the Canadian Medical Association Infobase, Alberta Health Services, BC Guidelines, the Winnipeg Regional Health Authority, Nursing Best Practice Guidelines, the Institute for Clinical and Economic Review, the Agency for Healthcare Research and Quality, the US Preventive Services Task Force, the National Institute for Health and Care Excellence, Guidelines International Network, the Medical Services Advisory Committee, the Pharmaceutical Benefits Advisory Committee, the Pharmaceutical Management Agency, and the Scottish Intercollegiate Guidelines Network. We searched the guideline producers’ and organizations' websites to locate the most current version of any relevant guidelines.
MHT Marketed in Canada
In Canada, various estrogen and estrogen-progestogen combination products are available, as listed in Table 1 and Table 2, respectively. The 2 main transdermal estrogen options in Canada are topical gels (e.g., Estrogel) and patches (e.g., Estradot).8,9
Table 1: Selected Estrogen Products Marketed in Canada
Brand | Route of administration | Active ingredient, formulation | Strength | DIN | Manufacturer |
|---|---|---|---|---|---|
Climara | Transdermal, patch | 17-beta estradiol, patch | 25 mg 50 mg 75 mg | 02247499 02231509 02247500 | Bayer Incorporated |
Lupin-Estradiol | Oral | 17-beta estradiol, tablet | 0.5 mg 1 mg 2 mg | 02449048 02449056 02449064 | Lupin Pharma Canada Limited |
Estrogel | Transdermal, gel | 17-beta estradiol, gel | 0.06% | 02238704 | Organon Canada Incorporated |
Premarin | Oral | conjugated estrogens, sustained-release tablet | 0.3 mg 0.625 mg 1.25 mg | 02414678 02414686 02414694 | Pfizer Canada |
Estradiol Derm | Transdermal, patch | 17-beta estradiol, patch | 50 mcg 75 mcg 100 mcg | 02246969 02246967 02246968 | Sandoz Canada Incorporated |
Estradot | Transdermal, patch | 17-beta estradiol, patch | 25 mcg 37.5 mcg 50 mcg 75 mcg 100 mcg | 02245676 02243999 02231509 02247500 02244002 | Sandoz Canada Incorporated |
Divigel | Transdermal, gel | 17-beta estradiol, gel | 0.1% | 02424835 02424843 02424924 | Searchlight Pharma Incorporated |
Oesclim | Transdermal, patch | 17-beta estradiol, patch | 25 mcg 50 mcg | 02243722 02243724 | Searchlight Pharma Incorporated |
Table 2: Selected Estrogen-Progestogen Combination Products Marketed in Canada
Brand | Route of administration | Active ingredient, formulation | Strength | DIN | Manufacturer |
|---|---|---|---|---|---|
Angeliq | Oral | 17-beta estradio–drospirenone, tablet | 1 mg/1 mg | 02268825 | Bayer Incorporated |
Bijuva | Oral | estradiol hemihydrate–micronized progesterone, capsule | 1 mg/100 mg | 02505223 | Knight Therapeutics Incorporated |
Activelle LD, Activelle | Oral | estradiol hemihydrate– norethindrone acetate, tablet | 0.5 mg/0.5 mg 1 mg/0.5 mg | 02309009 02249405 | Novo Nordisk |
Estalis | Transdermal, patch | 17 -beta estradiol–norethindrone acetate, patch | 140 mcg/50 mcg 250 mcg/50 mcg | 02241835 02241837 | Sandoz Canada Incorporated |
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first screening level, titles and abstracts were reviewed, and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 3.
Criteria | Description |
|---|---|
Population | Individuals being treated for menopausal symptoms, including symptoms of:
Individuals were eligible whether they had an intact uterus or not. |
Intervention | Transdermal menopausal hormone therapy (includes both estrogen alone and estrogen in combination with progestogen) Differences between patch and gel are also of interest. |
Comparator | Oral menopausal hormone therapy (includes both estrogen alone and estrogen in combination with progestogen) |
Outcomes | Efficacy and effectiveness outcomes: Vasomotor symptoms (e.g., frequency and severity of hot flushes, night sweats), health-related quality of life, sleep quality Safety outcomes: Cardiovascular risk, venous thromboembolism, lipid metabolism, carbohydrate metabolism, bone mineral density, genitourinary symptoms, endometriosis, gynecological cancer, breast cancer, dementia, stroke, withdrawal due to adverse events, and withdrawal due to lack of efficacy |
Study designs | Randomized controlled trials, systematic reviews, health technology assessments, network meta-analyses, meta-analyses, comparative nonrandomized studies (e.g., cohort or case-control), evidence-based guidelines, published cost-effectiveness literature |
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 3, were duplicate publications, or were published before 2017. Abstracts, non-English articles, letters to the editor, opinion pieces, narrative reviews, books, and book chapters were excluded. Primary studies retrieved by the search were excluded if they were captured in 1 or more included SRs. Studies on chronic diseases such as hypertension were excluded if the scope of the study did not specifically align with the PICO (population, intervention, comparator, outcomes). Guidelines that did not meet the PICO criteria or did not consider the route of administration were also excluded.
Critical Appraisal of Individual Studies
All studies were critically appraised by 1 reviewer. The A MeaSurement Tool to Assess systematic Reviews (AMSTAR 2) was used to assess the included SRs.12 We used the Cochrane Risk of Bias tool (RoB v2) for the randomized controlled trials.13 The Risk Of Bias In Nonrandomized Studies of Interventions (ROBINS-I) was used to assess the nonrandomized comparative studies.14 Two domains of the Appraisal of Guidelines for Research and Evaluation II (AGREE II) instrument (rigour of development and editorial independence) were used for evidence-based guidelines appraisal.15 Each included publication’s strengths and limitations were described narratively.
Patient Engagement
Two individuals receiving transdermal or oral MHT were confidentially interviewed before the literature search to share their lived and living experiences. These individuals offered personal perspectives on treatment priorities, research questions, and outcomes outlined in the selection criteria.
Both individuals had more than 7 years of experience with MHT; 1 individual tried oral hormone therapy first and cycled through multiple oral and transdermal medications before finding some relief with a high dose of a transdermal patch, and 1 individual started transdermal MHT first due to their risk factors and has not tried oral medications.
One individual reviewed the draft report to provide feedback on content and language.
A summary of the patient engagement reported using the Guidance for Reporting Involvement of Patients and the Public (GRIPP2) framework is provided in Appendix 1, Table 4.
Summary of Evidence
Quantity of Research Available
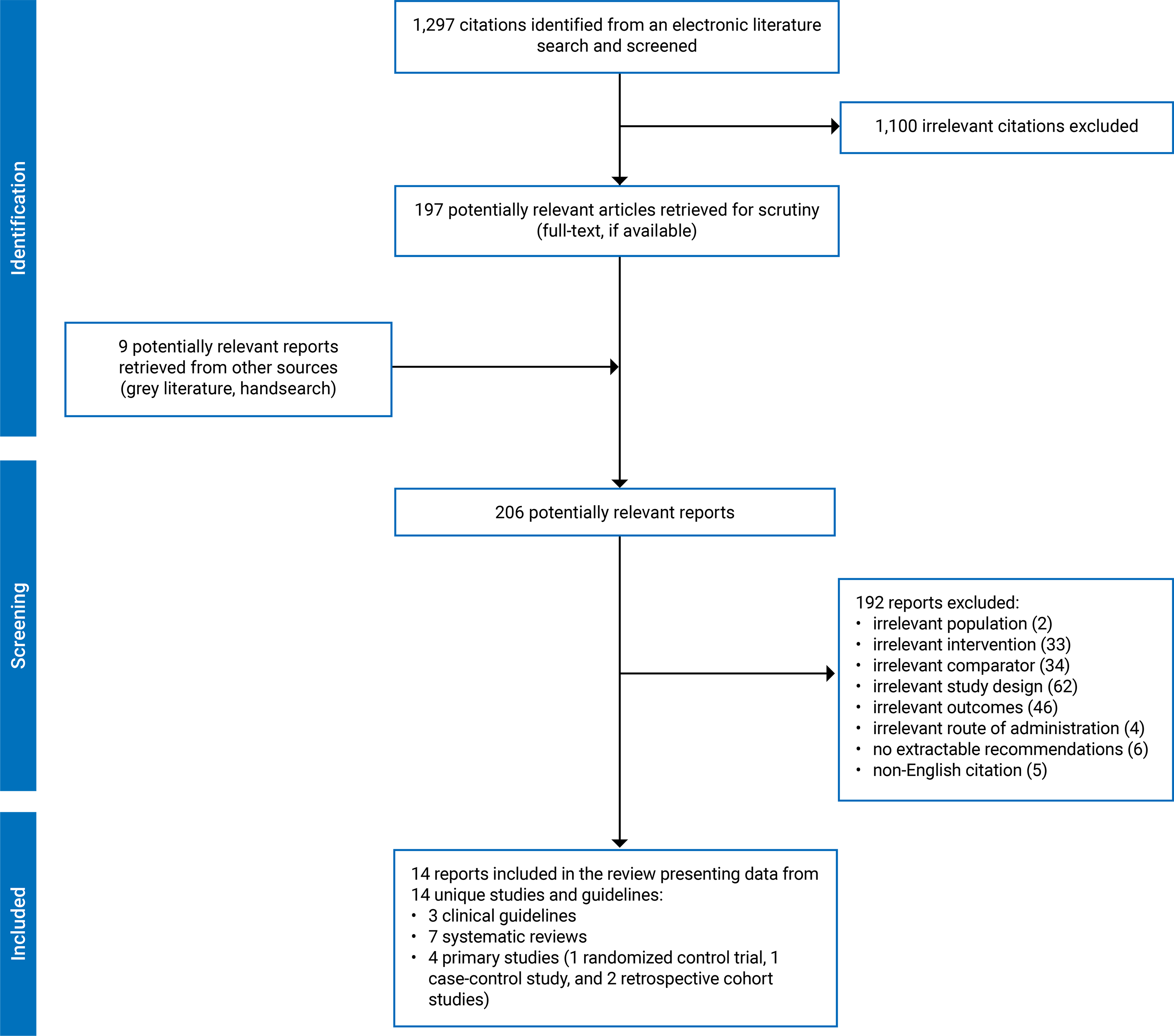
A total of 1,306 citations were identified in the literature search. Following the screening of titles and abstracts, 1,100 citations were excluded, 197 potentially relevant reports from the electronic search were retrieved for full-text review, and 9 potentially relevant guidelines were retrieved from the grey literature search for a full-text review. Of these potentially relevant articles (n = 206), 192 publications were excluded for various reasons, and 14 publications met the inclusion criteria and were included in this report. The eligible publications comprised 7 SRs,16-22 1 RCT,23 3 nonrandomized studies,24-26 and 3 evidence-based guidelines.27-29 No HTA reports or cost-effectiveness studies were identified in the search. Figure 1, Appendix 2 presents the PRISMA flow chart of the study selection.
Study Characteristics
Systematic Reviews
The 7 SRs focused on various outcomes of MHT in individuals in menopause or postmenopause.16-22 Two SRs were narrative reviews (without meta-analysis [MA]),17,19 4 included an MA,16,18,20,21 and 1 included a network meta-analysis (NMA).22 Additional details are presented in Table 5 in Appendix 3.
The Goldštajn et al. review17 narratively summarized 51 RCTs and observational studies, focusing on individuals in postmenopause and using either transdermal or oral estrogen MHT. The Oliver-Williams et al. review19 narratively summarized 33 RCTs and observational studies comparing transdermal MHT and oral MHT among individuals in postmenopause.
Four SRs with MAs were identified.16,18,20,21 The Nie et al. review18 included 73 RCTs, with pooled results from 16 studies comparing transdermal to oral MHT among individuals in postmenopause. The outcomes assessed were lipid profile changes, including total cholesterol, low-density lipoprotein (LDL), high-density lipoprotein (HDL), triglycerides, and lipoprotein (a). The follow-up durations ranged from less than 3 months to greater than 24 months. The Pan et al. review20 included 15 RCTs that are focused on individuals during menopause and that include both participants experiencing perimenopause and those in postmenopause. The studies compared transdermal to oral MHT. The primary outcome assessed was self-reported sleep quality. The study durations ranged from a minimum of 4 weeks to a maximum of 48 months. The Rovinski et al. review21 included 22 studies consisting of 4 RCTs, 9 case-control studies, and 9 cohort studies. The population comprised individuals in postmenopause with no history of VTE. The interventions compared were non-oral MHT (mostly transdermal) and oral MHT. The MA pooled results from 12 (case-control and cohort) studies. In 11 out of 12 studies, the non-oral route of administration was specified as transdermal, whereas in 1 study, the non-oral route was not defined. The primary outcome assessed was the risk of VTE events, including pulmonary embolism and deep vein thrombosis. The Anagnostis et al. study16 included 49 RCTs, 10 of which compared transdermal 17-beta estradiol to various forms of oral estrogen (such as valerate E2 and conjugated equine estrogens). The study focused on individuals in postmenopause, either with an intact uterus or a previous hysterectomy, who had been off MHT for at least 3 months. The primary outcome was the effect on lipoprotein (a) levels, with study follow-up periods ranging from 3 to 36 months.
The final eligible SR included an NMA. The Sarri et al. study22 included 47 RCTs that focused on individuals described as having natural menopause, which was defined as experiencing amenorrhea for at least 12 consecutive months. The study compared transdermal and oral MHT. The primary outcomes were the frequency of VMS, vaginal bleeding, and treatment discontinuation. While the study did not report specific results regarding the route of administration, it ranked transdermal and oral MHT based on effectiveness as an output from the NMA analysis.
Patient Population
None of the authors differentiated between sex and gender; participants were described as women in the reviews. Participants in 1 SR were described as “individuals with menopause” without reporting any further details regarding their menopausal health status.22 Individuals in postmenopause were included in 4 SRs.16-18,21 One SR19 included both individuals in menopause and in postmenopause. Individuals in menopause, including those in perimenopause and those in postmenopause, were included in 1 SR.20 None of the SRs specified whether the participants had premature ovarian insufficiency.
Interventions and Comparators
The interventions and comparators across the 7 SRs primarily focused on transdermal versus oral MHT. The transdermal MHT application was described as gel, patch, or spray in 2 reviews,17,18 whereas 1 review reported patches.22 The Rovinski et al. review21 included both gel and patches, whereas other reviews did not specify the transdermal formulations.16,19,20 The MHT interventions included estrogen monotherapy or combined estrogen and progestogen therapy. The Goldštajn et al. review17 compared transdermal estrogen MHT with oral estrogen MHT. Similarly, the Nie et al.18 and Pan et al.20 reviews also assessed transdermal versus oral MHT. The Oliver-Williams et al. review19 compared transdermal with oral MHT (including conjugated equine estrogens). The Rovinski et al. review21 focused on non-oral (primarily transdermal) versus oral MHT, and the Anagnostis et al. review16 compared transdermal 17-beta estradiol with various oral estrogen doses. The Sarri et al. review22 also compared transdermal and oral MHT.
Efficacy and Effectiveness Outcomes
Two SRs considered efficacy outcomes related to MHT. One SR assessed sleep quality20 and 1 SR with an NMA reported VMS.22 The review reporting the effectiveness of MHT for VMS ranked transdermal and oral routes based on outcomes considered in the NMA rather than providing formal quantitative comparisons for specific VMS. None of the reviews reported health-related quality of life. No quantitative results were reported for either SR.
Safety Outcomes
Five of the included SRs considered a range of safety outcomes related to MHT in individuals in menopause and postmenopause. The Goldštajn et al. review17 examined CVD, VTE, lipid and carbohydrate metabolism, bone mineral density (BMD), premalignant and malignant endometrial lesions, and breast cancer risk. CVD risk factors (e.g., risk of myocardial infarction and risk of hospitalization due to CVD) and stroke risk were reported in 2 SRs,17,19 and lipid profiles16-18 and VTE were reported in 3 SRs.17,19,21 Oliver-Williams and colleagues19 conducted an SR to assess the overall CVD risk associated with MHT. The outcomes of interest were cardiovascular events such as angina, coronary heart disease (CHD), stroke, CVD mortality, myocardial infarction (MI), heart failure, and VTE. Withdrawal due to lack of efficacy was reported in 1 SR.22
Primary Studies
Four primary studies focused on relevant outcomes for MHT for individuals during menopause, including perimenopause and postmenopause. Additional details are presented in Table 5, Table 6, and Table 7 in Appendix 3.
Tang and colleagues,23 in an RCT, included healthy individuals in perimenopause or postmenopause who were aged 40 to 55 years and randomized them to transdermal estradiol or oral estradiol valerate. The study used the Kupperman Menopausal Index (KMI) and Menopause Rating Scale (MRS) to assess menopausal symptoms.
Weller and colleagues,26 in a nested case-control study, examined commercially insured individuals with a median menopausal age of 49.6 years (ranging from 50 to 64 years) and investigated the relationship between transdermal and oral estrogen exposure and the risk of VTE. Blondon and colleagues,24 in a retrospective cohort study, included individuals in perimenopause or postmenopause who were aged 40 to 89 years and compared those who received transdermal estrogen only or with progestogen to those receiving oral estrogen only or with progestogen. The primary outcome was the risk of VTE, including pulmonary embolism and deep vein thrombosis. Laliberté and colleagues,25 in a retrospective matched cohort study, compared individuals in postmenopause who received estradiol transdermal systems to those receiving oral estrogen-only MHT. The primary outcome was the incidence of VTE and hospitalization-related VTE events.
Country of Origin
The 4 primary studies were conducted in the US,24,26 in Canada,25 and in China.23
Patient Population
None of the authors differentiated between sex and gender; participants were described as women in all studies. The studies included individuals in perimenopause and postmenopause who were aged 40 to 89 years. The Blondon et al. study24 focused on a large cohort of MHT users in the US with a mean age of 53.9 years who were mostly white (74%). The Laliberté et al. study25 focused on individuals in postmenopause with a mean age of 48.9 years. The Tang et al. study23 included individuals aged 40 to 55 years, while the Weller et al. study26 examined individuals aged 50 to 64 years.
Interventions and Comparators
The primary studies reported transdermal and oral estrogen either as monotherapy or in combination with progestogen. The transdermal MHT application was described as a gel in 1 study23 and as patches in another study,25 while in 2 studies24,26 did not specify the specific transdermal formulations. The Tang et al. study23 investigated transdermal estradiol and oral estradiol valerate. The Weller et al. study26 compared various forms of estrogen exposure combined with progesterone (oral and transdermal). Blondon and colleagues,24 in a retrospective cohort study, compared various forms of estrogen exposure (estrogen-only and estrogen plus progestogen) between transdermal estrogen and oral estradiol or oral conjugated equine estrogen in individuals in perimenopause and postmenopause without prior VTE, while the Laliberté et al. study25 compared transdermal estradiol with oral estrogen, with concomitant progestin use by 23.6% of transdermal estradiol users and 19.6% of oral HT users.
Efficacy Outcomes
One RCT, by Tang and colleagues,23 measured menopausal symptoms at 4, 12, and 24 weeks using the KMI and the MRS. KMI has been validated23 and widely used in China in clinical practice to assess 11 menopausal symptoms, including hot flashes and sweating, nervousness, sleep disorders, melancholia, vertigo, weakness, arthralgia and myalgia, headaches, palpitation and formication, depression, exhaustion, worry, frequent urination, and dysuria. The items in the KMI are rated on a 4-point scale ranging from 0 (absence of symptoms) to 3 (severe symptoms). The MRS is a reliable and valid questionnaire.23 It measures 11 symptoms using a 5-point scale ranging from 0 (absence of symptoms) to 4 (severe symptoms). This study used a Chinese version of the MRS, which shows good agreement with the original English version.
Safety Outcomes
VTE-related outcomes were the primary outcomes of interest in 3 studies. The Blondon et al.24 and Laliberté et al.25 studies targeted VTE events, hospitalization-related VTE, and VTE risk reduction. The Weller et al. study26 examined the risk of VTE. The Tang et al. sstudy23 measured withdrawal due to adverse events.
Evidence-Based Guidelines
Three guidelines met the inclusion criteria:27-29 the 2024 National Institute for Health and Care Excellence (NICE) guideline on Menopause: Diagnosis and Management,29 which is an update of the NICE 201930 and incorporates the NICE 2015 versions;31 the 2022 North American Menopause Society (NAMS) position statement on hormone therapy for menopause,27 which is an update of the 2017 version;32 and the 2017 American Association of Clinical Endocrinologists (AACE) Position Statement on Menopause,28 which introduces new recommendations to the 2011 edition.33
The NICE (2024) guideline29 targets health care professionals caring for women, trans men, and nonbinary individuals registered female at birth with menopause-associated symptoms. It addresses combined MHT (estrogen plus progestogen) and estrogen-only therapy for transdermal and oral routes, emphasizing individualized treatment based on patient needs and risks (e.g., VTE, cardiovascular risk). Key outcomes include CVD, stroke, cancer risks, bone health, dementia, type 2 diabetes, and quality of life. Recommendations, developed using SRs and the GRADE framework, are categorized as “strong” or “weak.” The guideline underwent extensive external review and advisor consultation, ensuring clarity, accuracy, and quality before approval by the NICE Guidelines Committee.29,31,34
The NAMS position statement on hormone therapy (2022) provides recommendations for individuals aged 50 and older or individuals with early or premature menopause. Developed in the US, the guideline is focused on menopausal symptoms such as vasomotor and genitourinary symptoms, sleep disturbances, osteoporosis, and quality of life.27 Recommendations were graded from level 1 to 3, with level 1 recommendations based on consistent scientific evidence, level 2 on limited or inconsistent evidence, and level 3 on expert opinion and consensus. However, no operational definition was provided for “consistent” or “inconsistent” in the guideline. Guideline validation methods were not detailed, and the authors only stated that the NAMS Board provided the necessary validation.27
The AACE (2017) guideline includes recommendations for individuals with chronic medical conditions and considers important outcomes such as CVD, osteoporosis, VMS, and breast cancer. Also developed in the US, this guideline was based on evidence gathered from MEDLINE searches and expert consensus. Quality assessment of the evidence and guideline validation methods were not detailed.28
Details are presented in Table 7 in Appendix 3. Additional references of potential interest are provided in Appendix 6 (potentially relevant but out-of-scope studies and guidelines).
Critical Appraisal
Additional details regarding the strengths and limitations of the included SRs, primary studies, and evidence-based guidelines are presented in Table 8, Table 9, Table 10, and Table 11 in Appendix 4.
Systematic Reviews
The 7 SRs16-22 were evaluated using the AMSTAR 2 tool. All reviews had clearly defined PICOs but considered broader inclusion criteria in terms of study design, participants, interventions, and comparators compared to this current review. Five of the included reviews declared pre-established review methods, however, only 3 provided explicit protocol or registration details18,20,22 and 2 offered no further information.19,21 Two reviews made no mention of a protocol.16,17
All 7 reviews considered multiple study designs but lacked justification for their eligibility criteria. The only exception was Pan et al.20 which included only RCTs that aligned with a prior review’s scope. All but 1 review13 used comprehensive literature search strategies across at least 2 databases, with search updates within 24 months of publication. Three reviews also searched reference lists, trial registries, and grey literature or consulted experts.19,21,22 Study selection and data extraction were performed in duplicate in most reviews, but the Sarri et al. study22 provided insufficient information on data extraction. Four reviews provided clear reasons for study exclusions,18,20-22 while 3 lacked sufficient detail.16,17,19
All reviews provided adequate descriptions of the studies included. Four SRs16,18,20,22 included only RCTs, while three17,19,21 included both RCTs and nonrandomized studies, requiring different approaches for assessing RoB assessment techniques (item 9 in AMSTAR2). Among the 7 reviews with RCTs, 3 used appropriate RoB tools and were judged adequately,17,18,20 while 4 were downgraded: 1 used the inappropriate Newcastle-Ottawa Scale for RCTs,16 1 provided insufficient information,16 and 2 did not conduct a RoB assessment (1 excluded RCTs from the quantitative analysis),21 and 1 lacked information.22 All 3 reviews including nonrandomized studies17,19,21 used the Newcastle-Ottawa Scale, which was deemed appropriate.
Two narrative reviews17,19 without MA or NMA were excluded from the quantitative synthesis assessment,35 which examined the use of appropriate statistical methods (item 11 in AMSTAR2), consideration of RoB impact on quantitative results (item 12 in AMSTAR2), explanation of heterogeneity (item 14 in AMSTAR2), and investigation of publication bias (item 15 in AMSTAR2). All 5 reviews that conducted quantitative analyses assessed heterogeneity and explained it when necessary.16,18,20-22 Only 1 review acknowledged RoB as a limitation.20 Most reviews, except for the Sarri et al. study,22 adequately investigated publication bias.
Overall, all 7 SRs were assessed to have at least 1 critical flaw each, which should be considered when interpreting the findings.
Primary Studies
The included RCT23 was assessed to broadly have good internal validity based on the reported design and conduct. However, the study used an open-label study design and, therefore, treatment allocations were known to both researchers and participants. We cannot rule out, based on the details reported, that knowledge of the assigned treatment potentially introduced performance bias and influenced the assessment of outcomes. The 3 nonrandomized studies24-26 had clearly defined interventions and outcomes, low rates of missing data, and preplanned analyses using data from well-maintained databases or sources. However, there are several important limitations, including the retrospective design in 2 studies, a RoB due to residual confounding, and concerns over deviations from the intended interventions. Overall, all 3 studies were assessed to have a critical RoB.
Evidence-Based Guidelines
The assessment of 3 guidelines — NICE (2024),29 NAMS (2022),27 and AACE (2017)28 — using the AGREE II framework reveals notable differences in methodological rigour. Among the guidelines, only NICE provides comprehensive details regarding systematic evidence searches, including explicit search strategies, whereas the NAMS and AACE guidelines lack such transparency. Similarly, the NICE guideline is the sole document to clearly outline evidence selection criteria and provide descriptions of the strengths and limitations of the evidence, supported by GRADE tables. While all 3 guidelines include methods for formulating recommendations that consider health benefits, side effects, and risks, the NICE guideline does not explicitly link recommendations to the supporting evidence. Peer review processes are inconsistently reported, with limited mention of external reviews. Additionally, procedures for updating guidelines are explicitly outlined in the NICE guideline but are absent in the NAMS and AACE guidelines. Editorial independence appears to be reasonably upheld across all 3 guidelines; however, the AACE guideline does not address potential funding influences.
Findings
Findings from the included SRs, primary studies, and evidence-based guidelines are presented in Table 12, Table 13, and Table 14 in Appendix 5.
Systematic Reviews
The included SRs investigated the effects of transdermal MHT versus oral MHT. However, some of the SRs did not consistently differentiate between estrogen monotherapy and estrogen plus progesterone therapy. In these SRs, some primary studies focused on estrogen monotherapy,16,18 while others focused on combination estrogen plus progesterone therapy, but the results were generally summarized as MHT overall.17,19-22 This limitation should be considered when interpreting the findings.
Efficacy Outcomes
Vasomotor Symptoms
None of the included SRs directly compared VMS for transdermal and oral MHT.
In 1 SR with an NMA (Sarri et al.),22 which investigated RCTs only, transdermal estrogen with progesterone ranked higher than oral estrogen with progesterone for VMS (hierarchical ranking of 68.8% versus 3.7%, respectively). However, no direct comparison was conducted, and these findings should be interpreted with caution. Both groups included estrogen and progesterone together.
Sleep Quality
Sleep quality was assessed in 1 SR with an MA,20 which investigated RCTs only. The study pooled results from RCTs comparing transdermal versus oral MHT in menopausal individuals. The transdermal MHT was more effective than oral MHT in improving self-reported sleep quality (MD = −0.12, 95% CI, −0.16 to −0.07).
Safety Outcomes
Cardiovascular Risk
Two SRs17,19 assessed CVD risk, including results based on RCTs and non-RCTs. The Goldštajn et al. study17 found no difference in CVD risk between transdermal and oral MHT. The Oliver-Williams et al. study19 suggested a lower incidence of CVD events with transdermal MHT than oral MHT. Although results varied, transdermal MHT appeared to have a slight advantage, but neither route showed a clear superior safety profile for overall CVD outcomes.
Venous Thromboembolism
Three SRs17,19,21 assessed the effects of MHT on VTE, all including results based on RCTs and non-RCTs. Non-oral MHT (primarily transdermal) was associated with a lower VTE risk, especially for individuals at higher risk of VTE; therefore, most studies favoured transdermal over oral MHT. In contrast, oral MHT consistently showed an increased risk of VTE across studies. Overall findings suggest that non-oral MHT presents a safer option regarding VTE risk than oral MHT.
Lipid Metabolism
Three SRs16-18 assessed the effects of MHT on lipid metabolism, with most studies being RCTs. Transdermal MHT showed inconsistent effects on HDL and LDL but significantly reduced triglyceride levels, while oral MHT increased HDL and triglycerides and lowered LDL and total cholesterol. Overall, oral MHT was associated with more favourable changes in lipid profiles (raising HDL and lowering LDL), despite increasing triglyceride levels.
Carbohydrate Metabolism
One SR by Goldštajn and colleagues,17 assessed the effects of transdermal and oral MHT on carbohydrate metabolism, including glucose tolerance, insulin resistance, and body composition, using data from 6 RCTs and 1 cohort study. The overall conclusion, according to the published results, is that both routes of administration reduced insulin resistance, with a more notable effect observed for oral MHT in people who do not have diabetes.
Bone Mineral Density
One SR by Goldštajn and colleagues17 assessed BMD in postmenopausal individuals by comparing transdermal and oral estrogen routes. Across 5 primary studies, with most studies being RCTs, both routes effectively increased BMD, showing similar improvements in the lumbar spine and hip over time. Overall, both oral and transdermal MHT demonstrated positive effects on BMD.
Gynecological Cancer
Although rates of endometrial cancer were not reported in any review, 1 SR by Goldštajn and colleagues17 of 3 RCTs found no differences in endometrial hyperplasia risk between transdermal and oral estrogen among people in postmenopause, with both treatment routes presenting comparable risk.
Breast Cancer
In 1 SR, Goldštajn and colleagues17 assessed breast cancer risks associated with transdermal versus oral estrogen MHT in individuals in postmenopause. In 7 observational studies, no differences in breast cancer risk were found between the 2 treatment routes. Prolonged MHT use (for more than 5 years) increased the risk for both routes.
Stroke
One SR by Oliver-Williams and colleagues19 assessed stroke risk in individuals in postmenopause, including data from 8 primary studies. Of these, 2 observational studies specifically compared transdermal and oral MHT. Oral MHT may increase the risk of stroke, whereas limited evidence indicates no increased risk of stroke with the use of transdermal estrogen in formulations with less than 50 mcg of estradiol per day.
Withdrawal Due to Adverse Events
In 1 SR with an NMA,22 better patient adherence with less treatment discontinuation was reported with the transdermal preparation compared to the oral preparation.
Other Efficacy or Effectiveness and Safety Outcomes
No relevant evidence was identified in the included SRs related to health-related quality of life, genitourinary symptoms, endometriosis, or dementia. One SR and NMA22 considered the frequency of VMS symptoms, vaginal bleeding, and treatment discontinuation, but no relevant evidence was identified comparing transdermal and oral MHT directly.
Primary Studies
The included primary studies investigated the effects of transdermal MHT versus oral MHT. One study focused on estrogen monotherapy,23 while the other 3 studies focused on combination estrogen plus progesterone therapy.24-26Although the results were generally specific to the intervention used, they were often summarized collectively as MHT.
Efficacy Outcomes
Menopausal Symptoms
One RCT (Tang et al.)23 reported fewer menopausal symptoms on the MRS at 12 and 24 weeks for the oral estrogen group compared to the transdermal group. In contrast, no differences between transdermal and oral groups were found in menopausal symptoms measured by the KMI.
Safety Outcomes
Venous Thromboembolism
Three primary studies assessed VTE risk, including 2 retrospective cohort studies and 1 case-control study.24-26 Transdermal estrogen was associated with a lower VTE risk compared to oral estrogen. The Laliberté et al. study25 found a 56% risk reduction with transdermal, compared to oral, estrogen use. The Weller et al. study26 reported nearly double the VTE risk for oral estrogen (OR 1.90; 95% CI, 1.56 to 2.32) and for oral estrogen combined with progestogen (OR 1.92; 95% CI, 1.43 to 2.60) compared to transdermal routes. The Blondon et al. study24 found no significant difference in VTE risk between the 2 routes, with HRs ranging from 0.81 to 1.06. Overall, this evidence suggests a lower VTE risk for transdermal compared to oral estrogen therapy.
Withdrawal Due to Adverse Events
One RCT, by Tang and colleagues,23 assessed withdrawal due to adverse events in individuals in perimenopause or postmenopause taking transdermal versus oral estrogen therapy. The number of patients discontinuing treatment was similar between the groups (8.6% transdermal versus 9.3% oral), with only 1 participant in the transdermal group discontinuing due to breast tenderness.
Other Efficacy or Effectiveness and Safety Outcomes
No relevant evidence regarding the following outcomes was identified in the primary studies: cardiovascular risk, lipid metabolism, carbohydrate metabolism, BMD, genitourinary symptoms, endometriosis, gynecological cancer, breast cancer, dementia, stroke, and withdrawal due to lack of efficacy.
Evidence-Based Guidelines
Three updated guidelines — NICE (2024),29 NAMS (2022),27 and AACE (2017)28 — introduced new recommendations relevant to MHT.
The NICE guideline (updated in 2024) builds on the 2015 and 2019 versions,29-31 with some recommendations carried forward and cited from the earlier guidelines. The guidelines recommend transdermal MHT over oral MHT for individuals at an increased risk of VTE, including those with a BMI of more than 30 kg/m2, because transdermal MHT does not increase VTE risk at standard doses. Stroke risk is unlikely to increase with the use of combined MHT that includes transdermal estrogen. However, stroke risk increases with combined MHT containing oral estrogen (particularly at higher doses), with longer durations (e.g., more than 5 years), and when it was started after the age of 60 years, with greater risks noted in Black populations. Both oral and transdermal MHT increase the risks of endometrial and ovarian cancers, but neither affects the risk of type 2 diabetes. Additionally, oral estrogen is associated with a small increase in cardiovascular stroke risk, while transdermal estrogen is not. The strength of recommendations in the NICE guidelines is inconsistently reported. For certain outcomes, such as VTE, stroke, type 2 diabetes, and cancer risks, the strength of the recommendations is not reported in most cases. However, 1 recommendation regarding transdermal MHT for individuals at increased VTE risk is explicitly labelled as “weak.” The quality of evidence varies across outcomes, ranging from very low to high, with most recommendations based on very low- to low-quality evidence, particularly for stroke and cancer-related outcomes.
The NAMS guideline (updated in 2022) included 2 key recommendations in the most recent update. If sexual function or libido are concerns in individuals with menopause symptoms, transdermal estrogen therapy may be preferable over oral estrogen therapy because of its minimal effect on sex hormone–binding globulin and free testosterone levels. Observational studies report a lower risk of gallstones with transdermal MHT than with oral MHT, but this observation is not confirmed in evidence from an RCT. Both recommendations were based on level 2 evidence and broadly applied to users of MHT, including those with surgical menopause, early menopause, or premature ovarian insufficiency and for individuals older than 65 years. The recommendations indicate limited or inconsistent scientific support.27 The relevant recommendations carried forward from the previous (2017)32 version included a statement that lowering doses and/or changing to transdermal MHT may be appropriate as women age or for individuals with metabolic syndromes such as hypertriglyceridemia with a risk of pancreatitis or fatty liver (level 3 evidence).
The AACE Guidelines (updated in 2017) reported 1 new recommendation that builds on, but does not replace, the 2011 recommendations.28 The guidance states that “the use of transdermal estrogen preparations should be considered as less likely to produce thrombotic risk and perhaps less likely to produce the risk of stroke and coronary artery disease.” No other details are available to clarify this ambiguous language regarding the real or potential risk of these outcomes associated with hormone therapy.28 Relevant recommendations carried forward from the 2011 guideline33 state that the use of the transdermal route of estrogen administration should be considered to avoid the hepatic “first-pass effect,” which may theoretically reduce the risk of thromboembolic disease. This recommendation is reported as concordant with current FDA warnings because individuals at high risk of thromboembolic disease should not take estrogen-containing therapy, although there is evidence that transdermal estradiol may not increase this risk.
Cost-Effectiveness of Transdermal MHT Compared to Oral MHT
No relevant evidence regarding the cost-effectiveness of transdermal MHT compared to oral MHT was identified in any of the included SRs, primary studies, or evidence-based guidelines; therefore, no summary of findings can be provided.
Limitations
Several limitations should be considered in this rapid review comparing transdermal MHT versus oral MHT. First, limited data were reported in the included studies on the characteristics of individuals receiving MHT, including details related to the description of menopause, and the ethnicity or gender of study participants. This may limit the ability to generalize findings across diverse populations and may overlook important variations in perimenopausal, menopausal, and postmenopausal symptoms and treatment responses related to population characteristics. No differentiation was made between sex and gender in the included studies, which may affect the interpretation of the results presented here. Second, there is no evidence to inform any consideration of cost-effectiveness for transdermal MHT versus oral MHT, and a limited number of studies consider efficacy, effectiveness, and safety outcomes. Specifically, there is no relevant evidence on important outcomes such as genitourinary symptoms, endometriosis, and dementia, further limiting the interpretation of the evidence for efficacy and safety. As well, inconsistent reporting of previous use of MHT across studies made it difficult to evaluate treatment effects, as prior exposure could influence the outcomes of the treatments under investigation.
Third, the wide variation in MHT dosages across studies and guidelines limits the ability to draw robust conclusions regarding the most effective and safest dosing strategies. Additionally, inconsistencies in treatment duration, ranging from short-term to long-term use, further challenge the interpretation of the findings. Fourth, another limitation is that the included SRs often combined data for estrogen monotherapy and estrogen plus progesterone therapy, limiting differentiation between therapy types. Results were generally summarized as MHT overall, emphasizing the need for careful interpretation and the importance of separating outcomes in future studies to better inform clinical decision-making. Fifth, a limitation of this review is that the comparisons between oral and transdermal estrogen in some of the included SRs, such as the Sarri et al. (NMAs) and Oliver-Williams et al. reviews, were either not based on equivalent dosing or did not clearly specify whether dosage was considered when evaluating the efficacy of oral versus transdermal estrogen. In the case of the Sarri et al. review, this limitation may also be related to the NMA design of the study, which relies on indirect comparisons across studies. As a result, the conclusions about the relative efficacy or effectiveness and safety of oral versus transdermal estrogen in these reviews should be interpreted with caution because as differences in dosage may have influenced the observed outcomes. Sixth, the conclusions are based primarily on observational study designs, which may limit the strength of the evidence. Seventh, limited direct comparisons between oral and transdermal MHT, particularly on critical outcomes such as cardiovascular risk and gynecological and breast cancer risks, restricted the strength and definitiveness of the conclusions that could be drawn. The evidence-based guidelines provide inconsistent scientific support, often rated as low-quality evidence or not clearly defined, and the strength of recommendations is not explicitly specified for most of the recommendations.
Lastly, the Canadian Menopause Society guideline36 was excluded because it does not provide strong or conditional recommendations regarding the use of transdermal versus oral MHT.
As a rapid review (versus, for example, an SR), a table of overlapping primary studies between the SRs was not reported, and we did not critically appraise the NMA assumptions.
A notable limitation of this work relates to the patient engagement component of the rapid review. The engagement process involved 2 individuals who shared their lived and living experiences with MHT. However, the perspectives of these 2 individuals cannot fully represent the lived and living experiences of the broader patient population. Additionally, no qualitative research or analysis of patient perspectives was conducted as part of this process.
Discussion and Implications for Decision- or Policy-Making
Conclusion
The objective of the rapid review was to summarize the evidence on the benefits and harms of transdermal estrogen therapy compared to oral estrogen therapy for individuals in menopause. We found 7 SRs, 4 primary studies, and 3 evidence-based guidelines. No HTAs and no cost-effectiveness studies were identified in this review.
Engaging with individuals who have experience with MHT was an important component of the rapid review. Through 2 interviews, we gathered information regarding their perspectives on relevant outcomes that should be considered in our review as well as their views on treatment priorities and access to treatment.
Individuals with lived or living experience said that symptom management was their primary concern and efficacy was the primary driver of their treatment preference. Both participants said they wanted a treatment regimen that reduced their symptoms, primarily VMS (hot flashes and night sweats) alongside genitourinary symptoms, joint aches, body pain, sleep disturbances, depression, anxiety, brain fog, mood swings, and other cognitive impacts.
The limited quantity of available evidence for efficacy or effectiveness outcomes prevents any definitive conclusions from being drawn for transdermal MHT compared to oral MHT. One SR with an NMA showed that transdermal MHT ranked higher than oral MHT for managing VMS; however, no direct comparison was conducted. One SR with an MA found that transdermal MHT was more effective than oral MHT in improving self-reported sleep quality. One RCT reported fewer menopausal symptoms on the MRS at 12 and 24 weeks for the oral estrogen group than the transdermal estrogen group.
During the interviews with individuals with lived or living experience, the risk to their health was a secondary concern. Both participants felt that transdermal MHT would provide a safer initial option for patients, potentially increasing the adoption of MHT among those concerned about the risks associated with oral MHT. Specific safety concerns raised in the interviews were the risk of VTE and BMD loss.
Based on the safety evidence regarding the use of transdermal MHT versus oral MHT, the findings varied by outcome. Overall, the SR findings suggest that non-oral MHT may be associated with a lower risk of VTE compared to oral MHT. Similarly, the nonrandomized studies suggest a lower risk of VTE for transdermal MHT compared to oral MHT. Oral MHT may increase triglycerides. However, comparative effects on CVD and stroke risk itself are unclear. The SR showed no differences for BMD and risk of gynecological and breast cancer. Additionally, the RCT reported that the number of patients discontinuing treatment was similar between the transdermal MHT and oral MHT groups.
Interview participants preferred starting on the safest, lowest risk method. They also expressed concerns about finding the appropriate dose. Dose and treatment duration of MHT varied widely across studies, making it challenging to draw conclusions on which dose is effective or safe. Furthermore, 1 participant shared that they tried low doses of different medications without any adjustments, making it unclear whether the lack of effectiveness was due to the medication itself or the specific dosage. Another participant mentioned struggling with a gel formulation, noting it was difficult to determine the correct amount to apply but that they found patches easier to apply. No evidence directly compared the efficacy and safety of transdermal gel versus patches.
The 3 clinical guidelines provide different recommendations regarding the use of transdermal MHT compared to oral MHT. One guideline recommends transdermal MHT for individuals at an increased VTE risk, including those with a BMI of more than 30 kg/m2, because transdermal MHT does not increase VTE risk at standard doses. Additionally, stroke risk is unlikely to increase with transdermal estrogen but is higher with oral estrogen, especially at higher doses, for longer durations (more than 5 years), and when it started after age 60, with greater risks noted in Black populations. Both oral and transdermal MHT increase endometrial and ovarian cancer risks, but neither affects type 2 diabetes risk. Oral estrogen is also associated with a small increase in stroke risk. However, the quality of evidence is low to very low, and the strength of recommendations is often not specified. Another guideline recommends the use of transdermal MHT for individuals when sexual function or libido are concerns and to lower the risk of gallstones. However, the recommendations are based on limited or inconsistent scientific support. The last guideline recommends the use of transdermal MHT because it may be less likely to increase thrombotic risk. It states the use of transdermal MHT should be considered to avoid the hepatic “first-pass effect,” which may theoretically reduce the risk of thromboembolic disease.
There are several limitations in this rapid review comparing transdermal MHT to oral MHT. These limitations include limited data regarding the characteristics of the study participants, limited evidence regarding efficacy or effectiveness outcomes, a wide variation of MHT dosages in the included studies, and limited direct comparisons between transdermal and oral MHT. Thus, it is difficult to generalize the findings of the included studies across diverse populations. Additionally, no cost-effectiveness studies were identified. Therefore, no conclusion can be drawn regarding the cost-effectiveness of transdermal versus oral MHT.
Implications for Decision-Making
The policy question asked if transdermal estrogen should be reimbursed as first-line treatment as an alternative to oral estrogen for MHT. The evidence included in the rapid review cannot adequately answer this question. The evidence comparing the use of transdermal versus oral MHT is limited, particularly for managing VMS and improving health-related quality of life and sleep quality. The robustness of the safety evidence is quite varied depending on the outcome, and the 3 clinical guidelines provide limited recommendations regarding the use of transdermal MHT versus oral MHT.
However, 2 findings were consistent among the SRs, the primary studies, and 2 clinical guideline recommendations — compared to oral MHT, transdermal MHT carries a potentially lower risk of VTE and, when considered along with the weaker evidence for the outcome directly, a potentially lower risk of stroke. It may be prudent to consider these findings when developing reimbursement policies for individuals prescribed MHT.
References
1.Dalal PK, Agarwal M. Postmenopausal syndrome. Indian J Psychiatry. Jul 2015;57(Suppl 2):S222-32. doi:10.4103/0019-5545.161483 PubMed
2.Nappi RE. The 2022 hormone therapy position statement of The North American Menopause Society: no news is good news. The Lancet Diabetes & Endocrinology. 2022;10(12):832-834. PubMed
3.WHO. Menopause. Accessed January 22, 2025, https://www.who.int/news-room/fact-sheets/detail/menopause.
4.Dalal PK, Agarwal M. Postmenopausal syndrome. Indian journal of psychiatry. 2015;57(Suppl 2):S222-S232. PubMed
5.Thurston RC, Joffe H. Vasomotor symptoms and menopause: findings from the Study of Women's Health across the Nation. Obstet Gynecol Clin North Am. 2011;38(3):489-501. PubMed
6.Trémollieres FA, André G, Letombe B, et al. Persistent gap in menopause care 20 years after the WHI: a population-based study of menopause-related symptoms and their management. Maturitas. 2022;166:58-64. PubMed
7.Coleman E, Radix AE, Bouman WP, et al. Standards of Care for the Health of Transgender and Gender Diverse People, Version 8. Int J Transgend Health. 2022;23(Suppl 1):S1-S259. doi:10.1080/26895269.2022.2100644 PubMed
8.CADTH. CADTH Reimbursement Review- Reimbursement Recommendation - Ospemifene (Osphena). Accessed January 14, 2025, https://www.cda-amc.ca/sites/default/files/DRR/2022/SR0709%20Osphena%20-%20Draft%20CADTH%20Recommendation%20(with%20redactions)_for%20posting%20April%2014%2C%202022.pdf
9.Canadian Pharmacists Association. Hormone Replacement Therapy Shortage. Accessed November 05, 2024, https://www.pharmacists.ca/cpha-ca/assets/File/cpha-on-the-issues/Hormone-Replacement-Therapy-Shortage_EN.pdf
10.Randel A. AACE releases guidelines for menopausal hormone therapy. American Family Physician. 2012;86(9):864-868.
11.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 Guideline Statement. Journal of Clinical Epidemiology. 2016;75:40-46. doi:10.1016/j.jclinepi.2016.01.021 PubMed
12.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. bmj. 2017;358.
13.Higgins JP, Savović J, Page MJ, Elbers RG, Sterne JA. Assessing risk of bias in a randomized trial. Cochrane handbook for systematic reviews of interventions. 2019:205-228.
14.Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. bmj. 2016;355.
15.Brouwers MC, Kho ME, Browman GP, et al. AGREE II: advancing guideline development, reporting, and evaluation in health care. Prev Med. Nov 2010;51(5):421-4. doi:10.1016/j.ypmed.2010.08.005 PubMed
16.Anagnostis P, Galanis P, Chatzistergiou V, et al. The effect of hormone replacement therapy and tibolone on lipoprotein (a) concentrations in postmenopausal women: a systematic review and meta-analysis. Maturitas. 2017;99:27-36. PubMed
17.Goldštajn MŠ, Mikuš M, Ferrari FA, et al. Effects of transdermal versus oral hormone replacement therapy in postmenopause: a systematic review. Archives of gynecology and obstetrics. 2023;307(6):1727-1745. PubMed
18.Nie G, Yang X, Wang Y, et al. The effects of menopause hormone therapy on lipid profile in postmenopausal women: a systematic review and meta-analysis. Frontiers in Pharmacology. 2022;13:850815. PubMed
19.Oliver-Williams C, Glisic M, Shahzad S, et al. The route of administration, timing, duration and dose of postmenopausal hormone therapy and cardiovascular outcomes in women: a systematic review. Human reproduction update. 2019;25(2):257-271. PubMed
20.Pan Z, Wen S, Qiao X, Yang M, Shen X, Xu L. Different regimens of menopausal hormone therapy for improving sleep quality: a systematic review and meta-analysis. Menopause. 2022;29(5):627-635. PubMed
21.Rovinski D, Ramos RB, Fighera TM, Casanova GK, Spritzer PM. Risk of venous thromboembolism events in postmenopausal women using oral versus non-oral hormone therapy: a systematic review and meta-analysis. Thrombosis research. 2018;168:83-95. PubMed
22.Sarri G, Pedder H, Dias S, Guo Y, Lumsden MA. Vasomotor symptoms resulting from natural menopause: a systematic review and network meta‐analysis of treatment effects from the National Institute for Health and Care Excellence guideline on menopause. BJOG: An International Journal of Obstetrics & Gynaecology. 2017;124(10):1514-1523. PubMed
23.Tang R, Xie Z, Ruan X, et al. Changes in menopausal symptoms comparing oral estradiol versus transdermal estradiol. Climacteric. 2024;27(2):171-177. PubMed
24.Blondon M, Timmons AK, Baraff AJ, et al. Comparative venous thromboembolic safety of oral and transdermal postmenopausal hormone therapies among women Veterans. Menopause. 2021;28(10):1125-1129. PubMed
25.Laliberté F, Dea K; Duh MS; Kahler, KH; Rolli, M; Lefebvre, P. Does the route of administration for estrogen hormone therapy impact the risk of venous thromboembolism? Estradiol transdermal system versus oral estrogen-only hormone therapy. Menopause 2018;25(11):1297-1305. doi: 10.1097/GME.0000000000001232 PubMed
26.Weller SC, Davis JW, Porterfield L, Chen L, Wilkinson G. Hormone exposure and venous thromboembolism in commercially insured women aged 50 to 64 years. Research and Practice in Thrombosis and Haemostasis. 2023;7(3):100135. PubMed
27.Society N-NAM. The 2022 hormone therapy position statement of the North American Menopause Society. Menopause. 2022;29(7):767-794. doi: 10.1097/GME.0000000000002028 PubMed
28.Cobin RH, Goodman NF, Committee ARES. AACE-American Association of Clinical Endocrinologists and American College of Endocrinology position statement on menopause–2017 update. Endocrine practice. 2017;23(7):869-880. PubMed
29.NICE. Menopause: identification and management (NG23). National Institute for Health and Care Excellence. Accessed January 14, 2025, https://www.nice.org.uk/guidance/ng23/resources/menopause-identification-and-management-pdf-1837330217413
30.NICE. Menopause: identification and management (ng23). National Institute for Health and Care Excellence. Updated Nov. 2019. Accessed January 14, 2025, https://www.nice.org.uk/guidance/ng23/resources/2019-surveillance-of-menopause-diagnosis-and-management-nice-guideline-ng23-pdf-9092485277125
31.NICE. Menopause. National Institute for Health and Care Excellence. Accessed January 14, 2025, https://www.nice.org.uk/guidance/ng23/evidence/full-guideline-pdf-559549261
32.Society N-AM. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24(7):728-753. PubMed
33.Goodman NF, Cobin RH, Ginzburg SB, et al. AACE-American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for the diagnosis and treatment of menopause. Endocrine Practice. 2011;17:1-25. PubMed
34.NICE. Developing NICE guidelines: the manual NICE. National Institute for Health and Care Excellence. January 14, 2025. https://www.nice.org.uk/process/pmg20/resources/developing-nice-guidelines-the-manual-pdf-72286708700869
35.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. Sep 21 2017;358:j4008. doi:10.1136/bmj.j4008 PubMed
36.Yuksel N, Evaniuk D, Huang L, et al. Guideline No. 422a: Menopause: vasomotor symptoms, prescription therapeutic agents, complementary and alternative medicine, nutrition, and lifestyle. Journal of Obstetrics and Gynaecology Canada. 2021;43(10):1188-1204. e1.
37.NICE. Menopause (update) [I] Early menopause NICE guideline NG23. Accessed January 14, 2025, https://www.nice.org.uk/guidance/ng23/evidence/i-early-menopause-pdf-487915383062
38.Craciunas L, Zdoukopoulos N, Vinayagam S, Mohiyiddeen L. Hormone therapy for uterine and endometrial development in women with premature ovarian insufficiency. Cochrane Database of Systematic Reviews. 2022;(10)
39.Pickar JH, Archer DF, Goldstein SR, Kagan R, Bernick B, Mirkin S. Uterine bleeding with hormone therapies in menopausal women: a systematic review. Climacteric. 2020;23(6):550-558. PubMed
Authors and Contributors
Authors
Said Yousef Abdelrazeq contributed to the design, protocol, screening, and data extraction; the analysis and interpretation; the writing of the original draft of the report and review of its contents; and the writing and approval of the final version of the report.
Shannon E. Kelly contributed to the conceptualization, methodology, and validation; the writing of the original draft of the report and review and editing of its contents; and supervision and project administration.
Shu-Ching Hsieh extracted data from clinical guidelines; conducted RoB assessments for the included SRs, primary studies, and guidelines; and contributed to the writing and revising of the original draft in response to the review team’s feedback.
Nazmun Nahar contributed to the full-text searches.
Becky Skidmore developed the search strategy in consultation with the review team; responded to PRESS feedback; translated and executed final strategies; deduplicated, packaged, and uploaded records to Covidence; tracked PRISMA; provided search documentation; and wrote methods for the final manuscript.
Melissa Brouwers contributed to supervision; funding acquisition; writing, editing, and review; and validation.
Shariq Najeeb assisted in screening, contributed to the critical appraisal and data extraction, drafted parts of the report, proofread the report, and revised the report as needed.
George A. Wells contributed to supervision; funding acquisition; writing, editing, and review; validation; and methodology.
Contributors
We thank Kaitryn Campbell, MLIS, MSc (Hamilton, ON) for peer-reviewing the MEDLINE search strategy.
This project was selected as part of a pilot for early engagement with patients and clinicians. We thank the contributors from these impacted groups for providing feedback and expertise throughout this project.
Patient Partners
We thank 2 people (who wish to remain anonymous) for providing input into the direction of the research, drawing from lived experience, and providing valuable contributions to this report.
Pagebreak
Clinical Experts
This individual kindly provided comments on this report:
Sheila Wijayasinghe, MD, CCFP, MSCP
Family Physician, Medical Director, Primary Care Outreach
University of Toronto
Denise Black, MD, FRCSC
Assistant Professor, Department of Obstetrics and Gynecology
University of Manitoba
Conflicts of Interest
Said Yousef Abdelrazeq disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
TR0016 Long-Acting Injectables for COPD
Shu-Ching Hsieh disclosed the following:
Involvement with projects or scientific advice
RC1552 Biologics for Inflammatory Bowel Disease
RD0076 Antiviral Drugs for the Treatment of Influenza
Shannon E. Kelly disclosed the following:
Involvement with projects or scientific advice
RE0048 Remdesivir for COVID-19 in the Inpatient Setting
HT0041 Remdesivir for COVID-19 in the Outpatient Setting
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
TR0016 Long-Acting Injectables for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Nazmun Nahar disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
Becky Skidmore disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
TR0016 Long-Acting Injectables for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Melissa Brouwers disclosed the following:
Involvement with projects or scientific advice
RE0048 Remdesivir for COVID-19 in the Inpatient Setting
HT0041 Remdesivir for COVID-19 in the Outpatient Setting
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
TR0016 Long-Acting Injectables for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Shariq Najeeb disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
TR0016 Long-Acting Injectables for COPD
George A. Wells disclosed the following:
VBI Vaccines Inc. (2020–current): Coronavirus vaccine, Data Safety Monitoring Board, member
Thermedical (2021–current): Ablation system and catheter needle, design and analysis advice for preparation of FDA submission
VBI Vaccines Inc. (2020–current): Coronavirus vaccine, design and analysis advice for preparation of FDA submission
Involvement with projects or scientific advice
RE0048 Remdesivir for COVID-19 in the Inpatient Setting
HT0041 Remdesivir for COVID-19 in the Outpatient Setting
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
TR0016 Long-Acting Injectables for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Kaitryn Campbell disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
TR0016 Long-Acting Injectables for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Sheila Wijayasinghe disclosed the following:
Payments as advisor or consultant
Sanoling AI (2024): Virtual Care
Felix Virtual Care (2024): Virtual Care
McMaster Women’s Health Lecture on Menopause
Advisory Board for National Menopause Show
Walrus Talks Menopause
Involvement with projects or scientific advice
RD0074 Transdermal Versus Oral Feminizing Hormone Therapy
Denise Black disclosed the following:
Payments as advisor or consultant
Pfizer Canada (current): Menopause hormone therapy
Bayer Canada (current): Nonhormonal menopause therapy
Searchlight Canada (current): Menopause hormone therapy
Merck Canada (current): Menopause hormone therapy
BioSyent Canada (current): Menopause hormone therapy
Canadian Menopause Society: Everything related to menopause
Speaking engagements
Pfizer Canada (current): Menopause hormone therapy
Searchlight Canada (current): Menopause hormone therapy
Knight Canada (current): Menopause hormone therapy
Merck Canada (current): Menopause hormone therapy
BioSyent Canada (current): Menopause hormone therapy
Canadian Menopause Society: Everything related to menopause
No other conflicts of interest were declared.
Appendix 1: Summary of Patient Engagement
Please note that this appendix has not been copy-edited.
Table 4: Reporting of Patient Engagement Using the GRIPP2 Framework
Section and Topic | Item |
|---|---|
1: Aim | Two Patient Partners shared their thoughts and perspectives on Menopause Hormone Therapy (MHT), offering insights into their experiences and priorities. This helped the authors better understand the nuances of their lived and living experiences when reviewing the literature. This is part of a CDA-AMC pilot of earlier engagement in Post-Market Drug Evaluation (PMDE) projects. |
2: Methods | Interviews During the scoping phase, 2 women with lived experience of MHT participated in individual semistructured interviews with CDA-AMC staff. A member of the research team participated as an observer. Their perspectives were subsequently summarized, those summaries were approved by the Patient Partners and then shared with the project team. Document review One of the Patient Partners participated in document review and reviewed the draft report and the plain language summary. As this was a pilot, they also engaged in an evaluation of the engagement process. |
3: Results of engagement | The researchers were made aware of the importance of several themes. Treatment priorities Efficacy was the primary driver for both Patient Partners. They wanted a treatment regimen that reduced their symptoms, particularly hot flashes and night sweats, but other symptoms as well. Safety was a concern and felt that transdermal MHT had a better safety profile than oral hormone therapy. Clotting and general risk factors were mentioned as concerns. They preferred the opportunity to start on the lowest-risk hormone therapy. Finding appropriate and effective doses was a concern. One Patient Partner cycled through the lowest doses of multiple medications, without her clinician increasing her doses. She didn’t know whether the medication didn’t work for her, or whether she simply hadn’t found the right dosage. Outcomes Symptom management was the primary reason for starting and continuing MHT. Reducing hot flashes and night sweats were crucial, but brain fog, depression and anxiety, mood swings, sleep disturbances, joint aches, body pain, and cognitive impacts were also flagged as symptoms that need attention. Preserving health was also a notable outcome for the Patient Partners. They wanted to minimize risks, preserve health, and achieve symptom management. One Patient Partner specifically mentioned that she wanted to prevent bone density loss. Research questions Cost-effectiveness considerations were raised by both Patient Partners. One wondered whether generic medications would be considered alongside branded medications, should there not be a generic available. The other individual raised the question of individuals declining oral MHT due to the risk factors and then being treated with multiple medications to address symptoms, such as antidepressants, antianxiety medications, sleeping pills, and so forth. Both Patient Partners felt that transdermal MHT should be available as a first-line therapy, allowing for a safer first option. They suggested that it might increase the uptake of MHT for those who are concerned about the risk profile. Both Patient Partners identified knowledge and stigma were concerns, with a lack of knowledge leading to misinformation and fear among the general population and clinicians. Lack of accessible, safe treatment for older individuals (predominantly women) presented an equity issue to the Patient Partners, who suggested that it discriminates against age, gender, and socioeconomic status. |
4: Discussion and conclusions | The Patient Partners were most concerned about safety and efficacy, with symptom management as the primary outcome. One of the key comments we heard was about choice. Patient Partners wanted to have the option of starting on the method with the lowest risk profile and changing based on need. |
5: Reflections and critical perspective | Success of patient engagement in this project is related to several factors. First, the Patient Partners were briefed on the objectives of the project in an introductory call and supported in their role in the engagement process by a Patient Engagement Officer. Interviews were conducted at a time convenient to the Patient Partners. The research team was receptive to this involvement and incorporated it in their approach to the clinical evidence. Compensation was offered for Patient Partners’ time and expertise. However, there were limitations. The invitation was shared both through relevant groups and online. The outreach generated interest, but we were only able to engage with a limited number of contributors and Patient Partners. The perspectives of those we engaged in cannot fully represent the lived and living experiences of the broader patient population. Another limitation of this engagement approach is that people need a phone or computer and reliable internet access to contribute to this work, which may exclude some voices. |
Appendix 2: Selection of Included Studies
Please note that this appendix has not been copy-edited.
Appendix 3: Characteristics of Included Publications
Please note that this appendix has not been copy-edited.
Table 5: Characteristics of Included Systematic Reviews
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Goldštajn et al. (2023)17 Croatia Funded by Università degli Studi di Verona within the CRUI-CARE Agreement | Design: SRa N = 51 (RCTs, observational prospective, and retrospective studies) Search between January 1990 and December 2021 | Participants: Postmenopausal individuals Total: NR Age: NR Ethnic groups: NR Previous treatment: NR (varying between the included studies) | Intervention: Transdermal estrogen MHT Comparator: Oral estrogens MHT Comparison (Either estrogen monotherapy, combined, natural, synthetic, or CEEs) | Outcomes: CVD risk, VTE, lipid metabolism, carbohydrate metabolism, BMD, and risk of premalignant and malignant endometrial lesions (e.g., amenorrhea, hyperplasia, endometrial bleeding and thickness), or breast cancer. Follow-up: Varied, ranging between 6 months and 5 years |
Nie et al. (2022)18 China Funded by National Nature Science Foundation, Natural Science Foundation of Guangdong Province, Science and Technology Foundation of Guangzhou City, TCM Research Fund of Guangdong Provincial Hospital of Chinese Medicine, Research Fund for Bajian Talents of Guangdong Provincial Hospital of Chinese Medicine | Design: SR and MA Total 73 RCTs (Pooled results from 16 studies) Search up to December 2020 | Participants: Postmenopausal individuals Total: NR Age: NR Ethnic groups: NR Previous treatment: NR | Intervention: Transdermal MHT (50 to 100 mcg/day for estradiol) (n = 676) Comparator: Oral MHT (CEE, 0.625 mg/day; estradiol 0.5 to 2 mg/day) (n = 670) | Outcomes: Lipid profile (including total cholesterol, LDL, HDL, triglycerides, and lipoprotein [a]) Follow-up: Varied, ranging between < 3 months to > 24 months |
Pan et al. (2022)20 China Funds not reported | Design: SR and MA Total 15 RCTs (10 RCTs-9 for oral MHT and 2 for transdermal MHT) Search up to June 14, 2021 | Participants: Individuals during menopause, including perimenopausal and postmenopausal Total: (N = 27,715) Age (years), mean range (SD): 49.7 ± 4.4 to 64.1 ± 0.6 Ethnic groups: NR Previous treatment: MHT lasting at least 4 weeks | Intervention: Transdermal MHT with minimum 4 weeks Comparator: Oral MHT (with minimum 4 weeks) | Outcomes: Sleep quality (self-reported) Follow-up: Varied, ranging between 4 weeks to 48 months |
Oliver-Williams et al. (2019)19 Netherlands Funded by Metagenics Inc. | Design: SRa Total 33 studies (6 clinical trials and 27 observational studies). Only 2 observational studies compared transdermal and oral MHT. Search up to January 30, 2018 | Participants: menopausal or postmenopausal individuals Total: (N = 2 588 327) Age (years), baseline range: 30 to 94 Ethnic groups: NR Previous treatment: NR | Intervention: HT (estrogen, alone or combined with a progestogen,) Comparator: Oral MHT (estrogen, CEE alone or in combination with a progestogen) | Outcomes: Angina, CHD, stroke, CHD-stroke, CHD-stroke hospitalizations, CVD, hospitalizations and mortality due to CVD, MI, heart failure, stroke, and VTE Follow-up: Varied, ranging from 6 months to 28 years |
Rovinski et al. (2018)21 Brazil Funded by the Brazilian National Institute of Hormones and Women's Health and Funding for Research of Hospital de Clínicas de Porto Alegre | SR and MA Total 22 (4 RCTs, 9 case-control studies, and 9 cohort studies) Pooled results from 12 (case-control and cohort) comparing oral vs. non-oral routes. Search up to February 2017 | Participants: Postmenopausal individuals with no history of VTE Total: (N = 394,077) Age (years) range: 48 to 65.9 Ethnic groups: NR Previous treatment: NR | Intervention: Non-oral MHT (n = 113,059) Comparator: Oral MHT (n = 281,018) | Outcomes: Risk of VTE events (pulmonary embolism and/or deep vein thrombosis) Follow-up: Up to 20 years |
Anagnostis et al. (2017)16 Greece No funding received | Design: SR and MA Total 49 RCTs Pooled results from 10 RCTs comparing oral to transdermal Search up to February 10, 2017 | Participants: Postmenopausal individuals (either hysterectomized or nonhysterectomized) Total: NR Age: NR Ethnic groups: NR Previous treatment: Participants being at least 3 months off any MHT | Intervention: Transdermal 17-beta estradiol (50 g, twice weekly) Comparator: Oral estrogen (dose of valerate E2 (2 mg/d) in 1 study, CEE (0.625 mg/d) in 2 studies, 17-beta estradiol 2 mg/d in 6 studies, and 17-beta estradiol 1 mg/d in 1 study) | Outcomes: Lipoprotein (a) Follow-up: Varied, ranging between 3 to 36 months |
Sarri et al. (2017)22 UK Funded by NICE and additional support from the Medical Research Council | SR and NMA 47 RCTs Search up to January 13, 2015 | Participants: Individuals with a diagnosis of natural menopause (defined as amenorrhea for at least 12 consecutive months) Total: (N = 8,326) Age (years), mean: > 45 Ethnic groups: NR Previous treatment: NR | Intervention: Transdermal MHT Comparator: Oral MHT | Outcomes: Frequency of VMS and discontinuation Follow-up: Up to 26 weeks |
BMD = bone mineral density; CEE = conjugated equine estrogens; CHD = coronary heart disease; CRUI-CARE = Conference of Italian University Rectors-Care Program; CVD = cardiovascular disease; HDL = high-density lipoprotein; LDL = low-density lipoprotein; MA = meta-analysis; MHT = menopausal hormone therapy; NICE = National Institute for Health and Care Excellence; NMA = network meta-analysis; NR = not reported; RCT = randomized controlled trial; SD = standard deviation; SR = systematic review; VMS = vasomotor symptoms; vs. = versus; VTE = venous thromboembolism.
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies.
aResults not pooled.
Table 6: Characteristics of Included Primary Studies
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Tang et al. (2024)23 China Funded by National Key Research and Development Program, National High-Level Hospital Clinical Research Funding, and National Natural Science Foundation of China Project | RCT (open-label) | Inclusion criteria: Healthy perimenopausal or postmenopausal individuals, ≥ 2 months but < 36 months from the final menses, with uterus and moderate or severe menopausal symptoms (KMI score ≥ 14) Study duration: August 2017 and April 2021 Total: (N = 257) Age (years), range: 40 to 55 Ethnic groups: NR Previous treatment: Excluded if they had used exogenous reproductive hormones or other therapies for menopausal symptoms currently or in the previous 3 months | Intervention: Transdermal -E2 (n = 129) Comparator: Oral-E2V (n = 128) | Outcomes: Menopausal symptoms using both KMI and MRS at 4, 12 and 24 weeks, and adverse events Follow-up: 24-week |
Weller et al. (2023)26 US Funded by Texas Academy of the Family Physicians Foundation | Nested case-control study | Inclusion criteria: US commercially insured menopausal individuals Study duration: January 1, 2007, to December 31, 2019 Total: (N = 25,885) (n = 20,359 cases of VTE and n = 203,590 matched controls) Age (years), range: 50 to 64 Age (years), median menopause age: 49.6 Ethnic groups: NR Previous treatment: Participants not excluded based on prior exposure to estrogen or estrogen + progestogen | Intervention: Any transdermal exposure (estrogen only) Any transdermal exposure (estrogen + progestogen) Comparator: Any oral exposure (estrogen only) Any oral exposure (estrogen + progestogen) | Outcomes: VTE Follow-up: Not clear (current exposure was defined as any exposure within 60 days of the index date, and “past exposure” use was defined as any exposure in the previous 61 to 365 days before the VTE diagnosis) |
Blondon et al. (2021)24 US No funding received | Retrospective cohort study | Inclusion criteria: Peri- and postmenopausal individuals using CEE or E2, without prior VTE Study duration: 2003 and 2011 Total: (N = 51,571) MHT users at cohort entry CEE (%) 74.5% Oral E2 (%) 12.6% Transdermal E2 (%) 12.9% Age (years), range: 40 to 89 Age (years), mean (SD): All participants: 53.9 (9.5) Oral CEE: 54.7 (9.8) Oral E2: 52.4 (8.3) Transdermal E2: 51.3 (7.4) Ethnic groups: American Indian/Alaska Native 1.2%, Asian/Pacific Islander 2%, Black/African American 22.3%, and White 74.5% Previous treatment: The study distinguished between incident users (new users) and prevalent users (existing users) at cohort entry and in the analysis. | Intervention: Transdermal (E2) Transdermal E2 + progestogen Comparator: Oral estrogen (CEE) Oral estrogen (CEE) + progestogen | Outcomes: VTE, including both PE and DVT Follow-up: mean (2.2 years) |
Laliberté et al. (2018)25 Canada Funded by Novartis Pharmaceuticals Corporation | A retrospective matched cohort | Inclusion criteria: Postmenopausal individuals who received only transdermal estradiol compared with individuals only receiving oral estrogen MHT agents (sensitivity analysis) Study duration: January 2002 to October 2009 Total: (N = 54,036) Age (years), mean (SD): 48.9 (7.1) Ethnic groups: NR Previous treatment: The study excluded participants who had used other estrogen agents, such as vaginal estrogens or different types of transdermal estradiol, during the 180-day baseline period before starting the study therapy | Intervention: Estrogen-only transdermal estradiol (23.6% with progestin medication) (n = 27,018, 88.5%) Comparator: Oral estrogen-only MHT (19.6% with progestin medication) (n = 27,018) | Outcomes: VTE (risk reduction for events for DVT or PE) VTE (hospitalization-related for DVT or PE) Follow-up: Mean (median) durations transdermal estradiol 391 (264) days Oral estrogen-only MHT 401 (272) days |
CEE = conjugated equine estrogens; DVT = deep vein thrombosis; E2 = estradiol; E2V = oral estradiol valerate; KMI = Kupperman Menopausal Index; MHT = menopausal hormone Therapy; MRS = Menopause Rating Scale; NR = not reported; PE = pulmonary embolism; RCT = randomized controlled trial; SD = standard deviation; VTE = venous thromboembolism.
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies.
Table 7: Characteristics of Included Guidelines
Intended users, target population | Intervention and practice considered | Major outcomes considered | Evidence collection, selection, and synthesis | Evidence quality assessment | Recommendations for development and evaluation | Guideline validation |
|---|---|---|---|---|---|---|
NICE (2024)29 | ||||||
Intended Users: Health care professionals caring for women, trans men, and nonbinary individuals registered female at birth with menopause-associated symptoms. Target Population: Women, trans men, and nonbinary individuals registered female at birth experiencing menopause or menopause-associated symptoms, as well as their families, carers, and the public. The guideline incorporate recommendations from NICE 2015 and update the NICE 2019. | MHT including both combined MHT (estrogen + progestogen) and estrogen-only used for transdermal and oral routes. Emphasizes individualization based on patient needs, clinical conditions, and risk factors (e.g., VTE, cardiovascular risk). | 1. Cardiovascular disease and stroke 2. Cancer risks 3. Bone health 4. Dementia 5. Type 2 diabetes 6. Vasomotor symptoms and quality of life | The guideline Development Group reviewed the evidence and developed the recommendations, and undergoing extensive external review and consultation with advisor before finalizing the guideline | NICE assesses evidence using a structured process outlined in the Developing NICE Guidelines. Evidence is appraised for quality, relevance, and reliability using frameworks like GRADE. RCTs are prioritized, with observational studies included cautiously. Certainty is evaluated based on strength, bias, and consistency. | NICE develops recommendations through SRs. GRADE framework to assess the certainty of evidence and inform the strength of recommendations. Recommendations categorized as “strong” (e.g., offer) or “'weak” (e.g., “consider”). | NICE validates their guidelines through expert and advisor reviews. Feedback is used to improve accuracy and clarity, and the final version is approved by the NICE Guidelines Committee to ensure quality and relevance. |
NAMS-MHT (2022)27 | ||||||
Intended users: Although not explicitly stated, it can be inferred that “all NAMS members” Target population: Individuals aged ≥ 50 years or individuals with early or premature menopause An update of the 2017 version.32 | MHT, including varied dosing and routes of administration for the 2 formulations: estrogen, (progestogen + estrogen). No specific information regarding dose-related titrations or adjustments. | 7. Menopausal symptoms, including vasomotor symptoms, sleep disturbances, genitourinary symptoms, urinary tract symptoms (including pelvic floor disorders), sexual function. 8. Primary ovarian insufficiency 9. Skin, hair, and special senses 10. Quality of life 11. Osteoporosis and more health topics related to MHT use | “…extensive review of the pertinent literature…” | NR | “…key points identified during the review process. The resulting manuscript was submitted to and reviewed and approved by the NAMS Board of Trustees.” “When recommendations are provided, they are graded” to level 1, 2, and 3…In achieving consensus, the panel took into consideration the level of evidence (RCTs > longitudinal studies > cross-sectional studies), sample sizes, risk of bias, data from meta-analyses and SRs, and expert opinion from guidelines from other major medical societies, when appropriate.” | NR |
AACE (2017)28 | ||||||
Intended users: Endocrinologists, nonendocrinologist physicians, and interested laypersons Target population: chronic medical conditions An update of the AACE (2011) version.33 | MHT No specific information regarding dose-related titrations or adjustments. | 1. Cardiovascular outcomes (myocardial infarction, stroke, pulmonary embolism, deep vein thrombosis, 2. Osteoporosis 3. Vasomotor symptoms (hot flashes) 4. Venous thrombo-embolism /Cerebrovascular accident 5. Breast cancer 6. Cognitive functions 7. Diabetes and Glucose tolerance | “A task force convened by the American Association of Clinical Endocrinologists (AACE) reviewed all available evidence from MEDLINE searches.” “Evidence presented in these guidelines was obtained through MEDLINE searches and available references compiled by guideline chairs and task force members.” | “Each recommendation was graded and based on evidence…” “All recommendation grades were determined by unanimous consensus of the primary writers and reviewers. a” | AACE reviewed all available evidence from MEDLINE searches. Conference calls and online discussion were used to evaluate the strength of evidence. After the initial writing process, reviewers contributed their expertise to the document. In addition, expert opinion was used to evaluate the available scientific literature, which was graded for treatment recommendations by evidence-based medicine guidelines and then presented in specific references in the appended reference list.” Guidelines were developed following the AACE Protocol for Standardized Clinical Practice Guidelines, incorporating relevant subject factors when available.a | NR |
AACE = American Association of Clinical Endocrinologists; NAMS-MHT = North American Menopause Society Position Statement on Hormone Therapy; NR = not reported.
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies.
aInformation was provided in AACE (2011).33
Appendix 4: Critical Appraisal of Included Publications
Please note that this appendix has not been copy-edited.
Table 8: Strengths and Limitations of SRs and NMAs Using AMSTAR 212
Strengths | Limitations |
|---|---|
Goldštajn et al. (2023)17 | |
This review clearly defined the PICO components. The search was conducted within 24 months of completion of the review. Two reviewers independently screened and selected studies, and reviewed bibliographies. Study validity was assessed using appropriate tools, and key study characteristics were listed, though not in detail. | This review lacked quantitative analysis, a preplanned protocol, and trial registration. It included randomized and nonrandomized trials without design justification, did not search grey literature, and omitted exclusion reasons for 25 studies. Funding and conflicts of interest were disclosed. |
Oliver-Williams et al. (2019)19 | |
The review defined PICO components conducted a comprehensive search across multiple databases within 24 months of completion of the review and performed study selection and data extraction in duplicate. The RoB results for prospective observational studies (assessed by NOS), funding sources were reported, and conflicts of interest disclosed. | The review lacked quantitative analysis and provided limited protocol details. It included RCTs, non-RCTs, and observational studies without design justification, omitted exclusion details, and summarized study characteristics briefly. The Cochrane RoB tool for RCT was ambiguously cited. |
Rovinski et al. (2018)21 | |
This review defined PICO components, conducted a comprehensive search across 3 databases within 24 months of completion of the review. Study selection and data extraction were duplicated, with a third reviewer resolving discrepancies. Excluded studies with justifications and important characteristics of included studies were provided. Only case-control and cohort studies in MA were assessed with the NOS. A random-effects model addressed heterogeneity, tested for each pooled RR with explanations for observed heterogeneity. Publication bias was appropriated. Funding sources and conflicts of interest were disclosed, with no funder involvement. | The review mentions protocol but provides no further details. The choice of including clinical trials and cohort studies is not justified. It does not assess individual study RoB impact on MA results. |
Nie et al. (2022)18 | |
The review defined the PICO components and referenced a registered protocol. A thorough literature search was conducted across 4 databases and updated, and study selection and data extraction were completed in duplicate, with disagreements resolved by a third reviewer. The Cochrane RoB checklist was used. The authors justified fixed-effect modelling for low heterogeneity (I2 < 50%) and used sensitivity analyses to assess the impact from high heterogeneity (I2 > 50%). No publication bias was detected. Funding sources were disclosed, with no conflicts of interest declared. | The review included only English-language RCTs without justification, likely omitting registered trials, reference checks, and grey literature. It did not evaluate individual studies’ RoB impact on MA results or findings interpretation. |
Anagnostis et al. (2017)16 | |
The review defined clear PICO criteria, including an 8-week minimum for MHT treatment. A comprehensive search strategy was used across multiple databases and updated after submission. Abstract selection, data extraction, and RoB assessment via NOS were conducted in duplicate, with discrepancies resolved by a third reviewer. The characteristics of included studies were detailed. A random-effects model addressing study and population heterogeneity was used. Publication bias was adequately assessed. | The review included only English-language RCTs without justifying this choice and lacked searches for registered trials, references, or grey literature. The NOS was inappropriate for assessing RoB for RCTs. The potential impacts from RoB results and heterogeneity were not addressed. |
Pan et al. (2022)20 | |
The review defined PICO components and followed a prospectively registered protocol. It included double-blind RCTs, probably aligned with the scope of the previous review. A comprehensive search strategy was used across 2 databases and updated. Study selection, data extraction, and RoB assessment were performed independently. Random-effects models, publication bias evaluations, and subgroup analyses were adequately conducted. No potential conflicts of interest were reported. | The review did not search grey literature or check references. The potential impact of RoB in individual studies on the overall results was not assessed. |
Sarri et al. (2017)22 | |
The review defined PICO components and a 26-week time frame for vasomotor symptoms, limited to English RCTs without justification for the study design criteria. A thorough and up-to-date literature search spanned 3 databases and additional sources. Data extraction was independently conducted, with exclusions and baseline characteristics detailed. RoB was assessed with the NICE methodology checklist, though the RoB results impact on the results was not assessed. NMA applied fixed, random-effects, and class-effect models, and heterogeneity and inconsistency were evaluated appropriately. Publication bias analysis was not performed but acknowledged as a limitation. Funding sources were disclosed without conflicts of interest. | The publication and protocol lacked clarity on whether study selection was performed in duplicate. |
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; MA = meta-analysis; NICE = National Institute for Health and Care Excellence; NMA = network meta-analysis; NOS = Newcastle-Ottawa Scale; PICO = population, intervention, comparator, outcomes; PRISMA = Preferred Reporting Items for Systematic reviews and Meta-Analyses; PROSPERO = International prospective register of systematic reviews; RoB = risk of bias; RR = relative risk; SR = systematic review.
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies
Table 9: Strengths and Limitations of Clinical Studies Using RoB v213
Strengths | Limitations |
|---|---|
Tang et al. (2024)23 | |
Computer-generated randomization schedules were created for each centre using SAS software, reducing randomization bias. Baseline characteristics were similar between groups, and study completion rates were high: 89.1% for transdermal estradiol and 88.4% for oral estradiol. Balanced discontinuation rates minimized bias from missing data. Outcomes were reported as preregistered. | This open-label design, with treatment assignments likely known to both researchers and patients, introduces potential bias in intervention performance. Subjective outcome measures, such as the Kupperman Menopausal Index and Menopause Rating Scale, further increased the risk of bias in outcome assessment. |
ROB v2 = Cochrane Risk of Bias tool version 2; SAS = statistical software suite.
Table 10: Strengths and Limitations of Clinical Studies Using ROBINS-I14
Strengths | Limitations |
|---|---|
Weller et al. (2023)26 | |
The study clearly defined interventions and outcomes, ensuring accurate retrospective case and control identification, with low RoB in classification and outcome measurement. The RoB from missing data was low, as the big database was well-maintained. The RoB in result selection was low due to preplanned subgroup analyses and ICD code identification of VTE. | This nested case-control study used retrospective data, with incomplete control of baseline and time-varying confounders. While logistic regression adjusted for known VTE risk factors, confounding from unknown factors remained. Participant selection and deviations from intended interventions posed serious and critical risks of bias, respectively. |
Blondon et al. (2021)24 | |
The study clearly defined its interventions (oral vs. transdermal estradiol) and outcomes (VTE incidents), minimizing bias in classification and measurement. The RoB from missing data was low, assuming the database used is representative and well-maintained. The preplanned subgroup analyses were adequate, with VTE outcomes identified by ICD-9 codes. | This retrospective matched cohort study faced moderate confounding bias despite time-to-event and sensitivity analyses. The lack of randomization led to critical risk of deviations from intended interventions, and serious risk of selection bias across diverse clinical settings. |
Laliberté et al. (2018)25 | |
The study clearly defined interventions (oral vs. transdermal estradiol) and outcomes (VTE events), minimizing bias. The RoB from missing data and result selection was low, with VTE outcomes identified using ICD-9 codes and incidence rates derived from preplanned sensitivity analyses. | This claims analysis study prospectively compared VTE rates in oral vs. transdermal estradiol users. Although cohort matching reduced selection bias, the retrospective nature and varied clinical settings introduced serious risk of selection bias, and critical RoB in participants’ deviations from intended interventions. Despite adjustments, confounding bias remained serious. |
DVT = deep vein thrombosis; ICD = International Statistical Classification of Disease; PE = pulmonary embolism; RoB = risk of bias; vs. = versus; VTE = venous thromboembolism.
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies.
Table 11: Strengths and Limitations of Guidelines Using AGREE II15
Item | NICE (2024)29 | NAMS (2022)27 | AACE (2017)a,28 |
|---|---|---|---|
Domain 3: Rigour of development | |||
7. Systematic methods were used to search for evidence. | Evidence Review I: Early Menopause outlines the assessment of the following domains,37 supported by detailed methods from Developing NICE Guidelines: The Manual.34 Yes. Detailed search strategies are provided in Appendix B Literature search strategies, p. 23. | Probably Yes. Seach terms, strategies, and method were not provided. | Probably Yes. “…all available evidence from MEDLINE,” without providing search terms, strategies, and method. |
8. The criteria for selecting the evidence are clearly described. | Yes. A summary of the PICO characteristics is provided in Table 1: Summary of the protocol (PICO table) p. 7. | Not provided. | Not provided. |
9. The strengths and limitations of the body of evidence are clearly described. | Yes. GRADE tables for review questions are in Appendix F (p. 55). | Not provided. | Not provided. |
10. The methods for formulating the recommendations are clearly described. | Yes. “Methods specific to this review question are described in the review protocol in Appendix A and the methods document (Supplement 1).” | Yes | Yes. The target audience, evidence sources, and processes for compilation, evaluation, and grading were provided.a |
11. The health benefits, side effects, and risks have been considered in formulating the recommendations. | Yes. Critical and important outcomes are listed in Table 1 (PICO Summary), p. 7 | Yes | Yes |
12. There is an explicit link between the recommendations and the supporting evidence. | Probably No. All relevant evidence were documented in Appendix D Evidence tables.37 However, there seems to be no link between the recommendations and the supporting evidence. | Probably Yes. A summary of relevant evidence (with references) is presented before each group of recommendations (key points). | Yes. A summary of relevant evidence (with references) is presented before each group of recommendations. “…presented in specific references in the appended reference list… Note: Reference sources are followed by an evidence level [EL] rating of 1, 2, 3, or 4. The strongest evidence levels (EL 1 and EL 2) appear in red for easier recognition.”a |
13. The guideline has been externally reviewed by experts before its publication. | Probably Yes. The guideline development process does not mandate external peer review but encourages advisor involvement throughout. In the manual,34 Section 10, explains the validation process, noting that “Although NICE does not routinely commission peer review from external experts, members of NICE staff with responsibility for quality assurance, or the developer, may occasionally consider arranging additional external expert peer review of part or all of a guideline, or an evidence review, executable model or economic analysis.” | No information. | No information. |
14. A procedure for updating the guideline is provided. | Yes. Details are in Section 14: Updating Guideline Recommendations. | Not provided. | Not provided, but this is an update of its 2011 version. |
Domain 6: editorial independence | |||
22. The views of the funding body have not influenced the content of the guideline. | Probably Yes. The updated guideline and evidence review state: “The recommendations in this guideline represent the view of NICE, arrived at after careful consideration of the evidence available.” This statement suggests that the content of the guideline reflects an independent evaluation of the evidence and is not influenced by the views of the funding body. No additional funding information was disclosed. | Probably Yes. “The NAMS Board of Trustees conducted an independent review and revision and approved the position statement. This Position Statement was made possible by donations to the NAMS Education & Research Fund. There was no commercial support.” The views of the funding body probably did not influence the content of the guideline. | Funding sources are not provided. |
23. Competing interests of guideline development group members have been recorded and addressed. | Probably Yes. Both the guideline29 and the manual34 do not provide specific details on managing competing interests within guideline development groups. However, it requires (registered) advisor to disclose any ties to confidential information or commercial funding, past or present. | Probably Yes. “All financial relationships with ineligible companies have been mitigated.” | Yes. A disclosure reads: “The authors have no multiplicity of interest to disclose.” |
AACE = American Association of Clinical Endocrinologists; AGREE II = Appraisal of Guidelines for Research and Evaluation II; PICO = population, intervention, comparator, outcomes; NAMS-MHT = North American Menopause Society Position Statement on Hormone Therapy; NR = not reported.
Note: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
aInformation was provided in AACE (2011).33
Appendix 5: Main Study Findings
Please note that this appendix has not been copy-edited.
Table 12: Outcomes Reported in Systematic Reviews
First author, year | Description | Findings | Author’s conclusions |
|---|---|---|---|
Vasomotor symptoms (frequency and severity) | |||
Sarri et al. (2017)22 | SR and NMA aimed to identify the most clinically effective treatments for reducing VMS in menopausal individuals with a uterus. It compared oral vs. transdermal MHT on outcomes such as VMS frequency, vaginal bleeding, and treatment discontinuation rates. | The network MA suggested that transdermal treatments ranked higher in patient adherence, with fewer discontinuations than oral treatments. Transdermal estrogen plus progestogen achieved a higher effectiveness ranking (69.82%) than oral (3.73%) in relieving VMS. | “There was evidence that estrogen plus progestogen taken orally may be more effective in relieving VMSs than placebo, but this did not rank as highly as transdermal estrogen plus progestogen (69.82%) in the hierarchy of the best treatment options compared to oral (3.73%) for this outcome.” |
Sleep quality | |||
Pan et al. (2022)20 | SR and MA included pooled results from 10 RCTs comparing oral vs. transdermal MHT and assessed self-reported sleep quality in menopausal individuals. | MD = −0.12 (95% CI, −0.16, to −0.07), I2 = 33% | “The study found that transdermal MHT regimens were generally more effective than oral MHT in improving self-reported sleep quality.” |
Safety (cardiovascular risk) | |||
Goldštajn et al. (2023)17 | SRa,b evaluated the association between transdermal vs. oral MHT with CVD risks from 6 primary studies (4 case-control and 2 cohort studies) CVD risk includes coronary heart disease, hospitalization due to ischemic heart disease, MI, fatal and nonfatal outcome of MI, hospitalization due to ischemic heart disease after prolonged use period (> 3 years). | Overall, no significant advantage was seen between transdermal and oral routes overall. Corrao et al. (2007) reported a reduced hospitalization risk for ischemic heart disease with transdermal estrogen RR 0.53 (95% CI 0.34 to 0.82). In contrast, Chilvers et al. (2003) found that oral estrogen was associated with a lower risk of nonfatal OR 0.68 (95% CI 0.49 to 0.95) and fatal MI OR 0.40 (95% CI 0.26 to 0.63) compared to transdermal estrogen. | “Almost all studies agree with the beneficial effect of MHT, but none of the two routes of administration demonstrated a significant advantage, and heterogeneous results were globally reported. Moreover, none of these studies were designed to compare the two administration routes.” |
Oliver-Williams et al. (2019)19 | SRa,b evaluated the impact of the MHT administration route on CVD risks among postmenopausal individuals based on only 2 observational studies that investigated and reported the route-related association with heart disease. The CVD outcomes examined included (angina, CHD, CHD-stroke events, and hospitalizations related to CVD, MI, and heart failure). | Transdermal MHT demonstrated a favourable cardiovascular profile compared to oral MHT for the investigated outcomes. Shufelt et al. (2014) found that transdermal estrogen therapy was associated with a 19% lower incidence of CVD events than oral estrogen IRR 0.81 (95% CI 0.67 to 0.99). Similarly, Simon et al. (2016) reported reduced CVD hospitalization risk with transdermal MHT IRR 0.65(95% CI 0.41 to 1.03). However, for myocardial infarction, Simon et al. observed a higher IRR among transdermal users (IRR 2.29), indicating variability in cardiovascular outcomes by condition. | “Oral MHT administration does not increase heart disease risk and may be cardioprotective. Transdermal MHT administration is safe with regard to CHD risk.” |
Safety (venous thromboembolism) | |||
Goldštajn et al. (2023)17 | SRa,b comparing transdermal vs. oral administration and their correlated risk with VTE events (from 9 case-control or Cohort studies) | Most studies (7/9), including case-control and cohort designs, favoured transdermal administration, particularly for individuals at higher risk of VTE. Scarabin et al. (2003) found transdermal estrogen significantly safer than oral (RR 4.0), highlighting the need to consider thrombotic risk factors in route selection. In contrast, 2 studies reported no difference between the routes | “Only two studies found no difference between the two routes of administration. Conversely, other authors observed that transdermal MHT is a safer choice, especially in individuals at increased risk for VTE” |
Oliver-Williams et al. (2019)19 | SRa,b comparing route of administration of MHT risk of VTE events. Out of 8 primary studies on the route of MHT administration and VTE risk, a direct comparison between transdermal and oral estrogen was found only in 1 retrospective cohort study by Simon et al. (2016). This study utilized health insurance claims. | Transdermal MHT users had a significantly lower risk of VTE than oral MHT users. IRR = 0.42 (95% CI 0.19 to 0.96) | “Oral MHT administration increases VTE risk. Transdermal MHT is safer with regard to VTE risk than oral MHT.” |
Rovinski et al. (2018)21 | SR and MA The study compared non-oral vs. oral estrogen routes to assess VTE risk. The MA pooled results from 12 (case-control and cohort) studies. In 11 out of 12 studies, the non-oral route of administration was specified as transdermal, while in 1 study, the route was not mentioned. | OR = 1.66 (95% CI 1.39 to 1.98), I2 = 58% | “VTE risk was increased in postmenopausal individuals with no previous VTE events using oral MHT. Non-oral MHT did not significantly affect this risk.” |
Safety (lipid metabolism) | |||
Goldštajn et al. (2023)17 | SRa,b evaluated the association between transdermal vs. oral MHT with Lipid profile risks from different combinations of estrogens and progestogens. The study evaluated 13 primary studies (1 cohort study and 12 RCTs) The lipid outcomes across these studies include HDL, LDL, total cholesterol, and triglycerides. | Oral estrogen increased HDL and lowered LDL and total cholesterol but often raised triglycerides. Transdermal estrogen had mixed effects on HDL and LDL. For example, Wakatsuki et al. (2002) reported increased HDL and triglycerides with oral estrogen, while Vrablik et al. (2008) found stable triglycerides and moderate LDL reduction with transdermal estrogen. | “MHT reduced LDL values regardless of the administration route in all studies, although results provided by some of them using transdermal estrogens have not demonstrated a statistically significant decrease in LDL concentrations. Additionally, oral MHT was demonstrated to increase the HDL and triglycerides concentration. Conversely, transdermal MHT had no significant effect on HDL levels, and most studies highlighted a significant reduction in triglyceride concentration.” |
Nie et al. (2022)18 | SR and MA The study pooled results from 16 RCTs compared transdermal MHT to oral MHT. The primary outcomes were lipid profiles, including total cholesterol, LDL, HDL, triglycerides, and lipoprotein (a). | Total cholesterol: MD = −0.13 (95% CI −0.30 to 0.04), I2 = 69% No significant difference | “Oral MHT was more effective in reducing LDL-C level than transdermal MHT, while it increased TG concentration.” |
LDL-C: MD = −0.23 (−0.31, 0.14), I2 = 28% Oral reduced LDL-C level than transdermal MHT. | |||
HDL: MD = −0.02 (95% CI −0.10 to 0.06), I2 = 84% No significant difference | |||
Lipoprotein (a) MD = 5.04 (95% CI −20.32 to 30.41), I2 = 0% No significant difference | |||
TG: MD = 0.12 (95% CI 0.04 to 0.21), I2 = 50%. Oral MHT was associated with increased triglycerides | |||
Anagnostis et al. (2017)16 | SR and MA 10 RCTs comparing oral to transdermal estrogen among postmenopausal individuals on lipoprotein (a) levels. | Lipoprotein (a) reduction–mean absolute difference, (95% CI): 9.09 mg/dl (95% CI 2.98 to 15.21), I2 = 85.7%, (P = 0.004) | “Oral estrogen seems to be more effective than transdermal estradiol. The lipoprotein (a)–lowering effect of MHT does not depend on the regimen (cyclical or continuous); the dose of estrogen and the addition of progestogen do not seem to modify the lipoprotein (a)–lowering effect of MHT.” |
Lipoprotein (a) reduction–mean relative difference %, (95% CI): 37.66% (95% CI 16.84 to 58.48), I2 = 99%, (P < 0.0001) | |||
Safety (carbohydrate metabolism) | |||
Goldštajn et al. (2023)17 | SRa,b Evaluated the association between transdermal vs. oral MHT (different combinations of estrogens and progestogens) with carbohydrate metabolism risks. Six RCTs and 1 cohort study compared the impact of oral and transdermal MHT. | Studies reveal differing effects of oral and transdermal MHT on glucose tolerance, insulin resistance, and body composition. Oral MHT (e.g., o-CEE 0.625 mg in Godsland et al. 1993) was linked to worsened glucose tolerance (P = 0.05). In contrast, transdermal MHT improved insulin sensitivity and body composition; for instance, dos Reis et al. (2003) reported a 3% increase in nonfat tissue with transdermal estrogen (t-E2 patch 0.05 mg, P < 0.05), while insulin levels remained stable in Godsland et al. (1993). | “Overall, according to the published results, both oral and transdermal administration routes reduce insulin resistance, with a more evident effect of the oral administration route in non-diabetic individuals.” |
Safety (bone mineral density) | |||
Goldštajn et al. (2023)17 | SRa,b Compared transdermal and oral estrogen routes in postmenopausal individuals, evaluating BMD values across 5 primary studies (4 RCTs and 1 case-control) | Both routes were effective in increasing BMD, with minimal differences. For instance, Kim H et al. (2014) reported similar lumbar spine BMD gains over 2 years (4.8% with oral CEE vs. 4.9% with transdermal E2) and positive effects on hip BMD (3.5% with oral CEE vs. 4.2% with transdermal E2). | “Both oral and transdermal administration routes demonstrated a positive effect on BMD values.” |
Safety (gynecological cancer) | |||
Goldštajn et al. (2023)17 | SRa,b 3 RCTs used to evaluate transdermal vs. oral estrogen on rates of hyperplasia, atrophic, secretory, and proliferative endometrium, endometrial thickness ≥ 5 mm, endometrial polyps, and intermenstrual bleeding. | Three RCTs found similar endometrial health outcomes for oral and transdermal estrogen. For example, Sendag et al. (2001) reported comparable hyperplasia rates (2.9% transdermal vs. 2.7% oral) with minor differences in other markers. | “Different authors reported a risk of endometrial hyperplasia and cancer with the transdermal route comparable to or even lower than those associated with the oral administration route.” |
Safety (breast cancer) | |||
Goldštajn et al. (2023)17 | SRa,b Compared transdermal and oral estrogen routes and breast cancer risks across 7 primary studies, including cohort and case-control designs. | Similar risk of breast cancer was found between transdermal and oral MHT. Beral et al. (2003) reported slightly higher risks with oral MHT (RR 1.32) than transdermal (RR 1.24), though not statistically significant. Lyytinen et al. (2009) showed that risk increased with MHT use beyond 5 years, with oral MHT having a higher risk (RR 1.81) than transdermal (RR 1.60). | “No difference for regarding breast cancer” |
Safety (stroke) | |||
Oliver-Williams et al. (2019)19 | SRa,b Comparing transdermal vs. oral administration and stroke. Direct comparison between transdermal and oral estrogen was found in 2 observational studies. | Overall, described as oral may increase risk of stroke. Shufelt et al. found that transdermal MHT had (HR 0.87, 95% CI 0.55 to 1.38) compared to oral MHT. Similarly, Simon et al. (2016) reported (IRR 0.89, 95% CI 0.54 to 1.47) for transdermal vs. oral MHT. | “Oral MHT administration increases risk of stroke. Limited evidence indicates no increased risk of stroke associated with the use of transdermal estrogen in formulations with < 50 mcg of estradiol per day.” |
BMD = bone mineral density; CEE = oral conjugated equine estrogens; CHD = coronary heart disease; HDL = high-density lipoprotein; HR = hazard ratio; IHD = ischemic heart disease; IRR = incidence rate ratio; LDL = low-density lipoprotein; MA = meta-analysis; MD = mean difference; NMA = network meta-analysis; NR = not reported; O-O-E2 = oral E2; RCT = randomized controlled trial; RR = risk ratio; SR = systematic review; T-E2 = Transdermal E2; VMSs = vasomotor symptoms; vs. = versus; VTE = venous thromboembolism.
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies.
aNo quantitative findings were reported.
bIn some of the primary studies, the comparison was not clearly described if it was between oral to transdermal or between any of the 2 routes to placebo.
Table 13: Outcomes Reported in Primary Studies
First author, year | Description | Findings | Author’s conclusions |
|---|---|---|---|
Menopausal symptoms-scales | |||
Tang et al. (2024)23 | An open-label RCT evaluated the effectiveness and safety of transdermal vs. oral estrogen therapy for managing menopausal symptoms among perimenopausal or postmenopausal individuals. Menopausal symptoms were assessed using the KMI and MRS. | MRS (after 12 weeks of treatment): t-E2, mean (SD) = 8.67 ± 5.66 o-E2V, mean (SD) = 6.77 ± 4.63 (P = 0.005) MRS (after 24 weeks of treatment): t-E2, mean (SD) = 7.78 ± 5.59) o-E2V, mean (SD) = 6.11 ± 4.10 (P = 0.011) | “No statistical difference was found in the mean of KMI scores at each follow-up time point between the oral and transdermal routes (p > 0.05). Mean MRS scores were significantly lower in the oral group than in the transdermal group after 12 weeks (p = 0.005) and at 24 weeks of treatment (p = 0.011).” |
Venous thromboembolism | |||
Weller et al. (2023)26 | A nested case-control study reported VTE risk by comparing Transdermal estrogen only to oral (estrogen only) | OR = 1.90 (95% CI 1.56 to 2.32)a | “Oral exposures were almost twice as high as transdermal exposures.” |
A nested case-control study reported VTE risk by comparing combined transdermal (estrogen + progestogen) to oral (estrogen + progestogen) | OR = 1.92 (95% CI, 1.43 to 2.60)a | “Oral exposures were almost twice as high as transdermal exposures.” | |
Blondon et al. (2021)24 | A retrospective matched cohort reported the risk of incident VTE by comparing oral CEE to T-E2 | Oral CEE vs. T-E2b HR = 0.85 (95% CI 0.55 to 1.33) Oral CEE vs. T-E2c HR = 0.95 (95% CI 0.60 to 1.48) Oral CEE vs. T-E2d HR 0.95 (95% CI 0.60 to 1.49) | “Compared with CEE use, there was no difference in the adjusted risk of incident VTE associated with oral E2 use or with transdermal E2 use.” |
Risk of the incident VTE by comparing oral CEE to T-E2, restricted to the incident (new) users | Oral CEE vs. T-E2b, e HR = 1.02 (95% CI 0.53 to 1.97) Oral CEE vs. T-E2c, e HR = 1.03 (95% CI 0.53 to 1.99) Oral CEE vs. T-E2d, e HR = 1.02 (95% CI 0.52 to 1.99) | “Compared with CEE use, there was no difference in the adjusted risk of incident VTE associated with oral E2 use or with transdermal E2 use.” | |
Risk of VTE by comparing (estrogen only) oral CEE to T-E2 (n = 38,800) | Oral CEE vs. T-E2b HR = 0.81 (95% CI 0.51 to 1.29) Oral CEE vs. T-E2c HR = 0.92 (95% CI 0.57 to 1.47) Oral CEE vs. T-E2d HR = 0.96 (95% CI 0.59 to 1.54) | “There was no apparent effect modification by the use of progestogen, but the number of events in the combined MHT group was low.” | |
Risk of the incident VTE by comparing combined (estrogen + progestogen) oral CEE to T-E2 (n = 12,771) | Oral CEE vs. T-E2b, e HR = 1.03 (95% CI 0.25 to 4.34) Oral CEE vs. T-E2c, e HR = 1.02 (95% CI 0.24 to 4.29) Oral CEE vs. T-E2d, e HR = 1.06 (95% CI 0.25 to 4.46) | “There was no apparent effect modification by the use of progestogen, but the number of events in the combined MHT group was low.” | |
Laliberté et al. (2018)25 | A retrospective matched cohort reported the risk reduction for VTE events for postmenopausal individuals receiving only transdermal estradiol (n = 9,264) compared with individuals only receiving oral estrogen-only (n = 9,264) | Unadjusted IRR = 0.58 (95% CI, 0.37 to 0.92) P = 0.019 Adjustedf IRR = 0.44 (95% CI, 0.25 to 0.77), P = 0.004 | “The risk reduction for VTE events associated with transdermal estradiol relative to estrogen-only MHT was even more pronounced in the sensitivity analysis in which a total of 29 transdermal estradiol users developed VTE compared with 56 individuals in the oral estrogen-only MHT cohort.” “After adjustment for confounding factors, transdermal estradiol remained independently associated with a lower risk for VTE events by 56% compared with oral.” |
Hospitalization-related VTE events for postmenopausal individuals receiving only transdermal estradiol (n = 9,264) compared with individuals only receiving oral estrogen-only (n = 9,264) | Unadjusted IRR = 0.33 (95% CI, 0.12 to 0.90), P = 0.030 Adjustedg IRR = 0.34 (95% CI, 0.10 to 1.19), P = 0.092 | “Additional exploratory analysis of hospitalization-related VTE events also corroborated the lower incidence rate associated with transdermal estradiol relative to oral estrogen only.” | |
Safety (withdrawal due to adverse events) | |||
Tang et al. (2024)23 | An open-label RCT evaluated the effectiveness and safety of transdermal vs. oral estrogen therapy for managing menopausal symptoms among perimenopausal or postmenopausal individuals. Safety assessments included monitoring adverse events, such as breast tenderness. | Overall adverse events (n (%): T-E2: 11/128 (8.6%) O-E2V: 12/129 (9.3%) (P = 0.84) | “The incidence of adverse events in the two groups seems similar, with breast tenderness being the most common side effect in both. One case of breast cancer was observed in the transdermal group, but overall, the authors noted little difference in adverse events between the two administration routes.” |
CEE = conjugated equine estrogens; HR = hazard ratio; IRR = incidence rate ratio; KMI = Kupperman Menopausal Index; MRS = Menopause Rating Scale; O-E2V = oral estradiol valerate; RCT = randomized controlled trial; T-E2 = transdermal estradiol; vs. = versus; VTE = venous thromboembolism.
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies.
aAdjusted but not specified.
bUnadjusted.
cAdjusted for age.
dAdjusted for age, race, and BMI; stratified by previous use of MHT, and progestogen use.
eRestricted to incident (new) users of MHT.
fAdjusted confounding factors (most frequent (> 10%) baseline risk factors for VTE were surgical resection of abdominal or pelvic cancer, major surgery, hyperlipidemia, hypertension, and multiple trauma).
gAdjustment in the hospitalization-related VTE models was limited to baseline health care costs.
Table 14: Summary of Recommendations in Included Guidelines
Recommendations and supporting evidence | Quality of evidence and strength of recommendations |
|---|---|
VTE risk is not increased with transdermal MHT. VTE risk is greater with oral than transdermal MHTa,29 | Quality: NR Strength: NR |
Consider transdermal rather than oral MHT for menopausal individuals who are at increased risk of VTE, including those with a BMI of more than 30 kg/m2. The risk associated with transdermal MHT given at standard therapeutic doses is no greater than baseline population risk.30 | Quality: NR Strength: Weak |
Stroke risk increases with oral estrogen-only MHT and the increase, particularly with a higher dosage of estrogen is greater if MHT is started after the age of 60. Stroke risk is unlikely to increase with transdermal estrogen-only MHT.29,30 | Quality: Very low to low-quality evidence Strength: NR |
Stroke risk is unlikely to increase with the use of combined MHT that includes transdermal estrogen.29 | Quality: Very low to low-quality evidence Strength: NR |
Stroke risk increases with combined MHT containing oral estrogen, particularly with higher dosages, longer treatment durations (e.g., more than 5 years), and increasing age at initiation of MHT. Ethnic differences are also noted, with greater risk in Black populations.29 | Quality: Very low to low-quality evidence Strength: NR |
The risk of developing type 2 diabetes does not increase with oral or transdermal MHTa,29 | Quality: NR Strength: NR |
Endometrial cancer risk increases (in people with a uterus) with both oral and transdermal estrogen-only MHTa,29 | Quality: Very low to high-quality evidence Strength: NR |
Ovarian cancer risk increases with both transdermal and oral estrogen-only MHTb,29 | Quality: Very low to low-quality evidence Strength: NR |
Explain to individuals that taking oral (but not transdermal) estrogen is associated with a small increase in the risk of cardiovascular stroke.30 | Quality: Very low to low-quality evidence Strength: NR |
NAMS (2022)27 | |
If sexual function or libidoc are concerns in individuals with menopause symptoms, transdermal ET may be preferable over oral ET because of minimal effect on sex hormone–binding globulin and free testosterone levels. (p. 773) | Quality: Level 2 Strength: NR |
Observational studies report lower risk of gallstones with transdermal MHT than with oral, and with oral estradiol compared with CEE, but neither observation is confirmed in RCTs. (p. 776) | Quality: Level 2 Strength: NR |
AACE (2017)28 | |
The use of transdermal estrogen preparations should be considered as less likely to produce thrombotic risk and perhaps the risk of stroke and coronary artery disease. (pp. 869, 874) | Quality: NR Strength: NR |
AACE = American Association of Clinical Endocrinologists; BMI = body mass index; CEE = conjugated equine estrogens; ET = estrogen therapy; MHT = menopausal hormone therapy; NAMS-MHT = North American Menopause Society Position Statement on (Menopause) Hormone Therapy; NICE = National Institute of Health and Care Excellence; NR = not reported; RCTs = randomized controlled trials; VTE = venous thromboembolism.
aThe statements from 2015 in tables 1 and 2 do not distinguish between combined and estrogen-only MHT.
bIn people with no personal history of endometrial cancer
cLibido as a menopausal symptom
Notes: MHT refers to menopausal hormone therapy, hormone therapy, or hormone replacement therapy, as used in the original studies.
Individuals refer to women, as used in the original studies.
Appendix 6: References of Potential Interest but Not Meeting the Inclusion Criteria
Please note that this appendix has not been copy-edited.
Systematic Reviews
Outcomes Out of Scope
Craciunas L, Zdoukopoulos N, Vinayagam S, Mohiyiddeen L. Hormone therapy for uterine and endometrial development in women with premature ovarian insufficiency. Cochrane Database of Systematic Reviews. 2022;(10). PubMed
Pickar J, Archer D, Goldstein S, Kagan R, Bernick B, Mirkin S. Uterine bleeding with hormone therapies in menopausal women: a systematic review. Climacteric. 2020;23(6):550-558.
Guidelines and Recommendations
No Relevant Recommendations or No Comparison of Interest Reported
Gartlehner G, Patel SV, Reddy S, Rains C, Schwimmer M, Kahwati L. Hormone therapy for the primary prevention of chronic conditions in postmenopausal persons: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2022;328(17):1747-1765. PubMed
Shoback D, Rosen CJ, Black DM, Cheung AM, Murad MH, Eastell R. Pharmacological management of osteoporosis in postmenopausal women: an Endocrine Society guideline update. J Clin Endocrinol Metab. 2020;105(3). PubMed
Academic Committee of the Korean Society of M, Lee SR, Cho MK, et al. The 2020 menopausal hormone therapy guidelines. J Menopausal Med. 2020;26(2):69-98. PubMed
A Practitioner’s Toolkit for the Management of the Menopause. 2023. https://www.menopause.org.au/images/pics/ptmm/a-practitioners-toolkit-for-managing-menopause.pdf. Accessed October 01, 2024.
AMS Guide to MHT/HRT Doses- AUSTRALIA ONLY. 2024; https://www.menopause.org.au/images/stories/infosheets/docs/AMS_Guide_to_MHT-HRT_Doses_Aus_1024_V2.pdf. Accessed October 01, 2024.
POCKET GUIDE MENOPAUSE MANAGEMENT. 2nd Edition. Canadian Menopause Society; 2023. https://www.canadianmenopausesociety.org/sites/default/files/pdf/publications/Pocket%20Guide%20-%20udpated%202023%20English%20version.pdf. Accessed October 18, 2024.
Yuksel N, Evaniuk D, Huang L, et al. Guideline No. 422a: Menopause: vasomotor symptoms, prescription therapeutic agents, complementary and alternative medicine, nutrition, and lifestyle. Journal of Obstetrics and Gynaecology Canada. 2021;43(10):1188-1204. e1.
ISSN: 2563-6596
This work was conducted by the POst-market Drug Evaluation Team (PODET) through the Post-Market Drug Evaluation CoLab Network. It was supported by Canada’s Drug Agency (CDA-AMC) and its Post-Market Drug Evaluation Program through funding provided by Health Canada.
CDA-AMC is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
CoLab is a pan-Canadian network of experts in applied research, scientific methods, and data analysis. CoLab members work with the Post-Market Drug Evaluation Program to produce credible and timely evidence on postmarket drug safety and effectiveness.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
This document is the property of PODET. CDA-AMC has a nonexclusive, limited, royalty-free, worldwide, nontransferable, fully paid-up, and irrevocable license to use the report in support of its objects, mission, and reasonable operational requirements.