Drugs, Health Technologies, Health Systems
Health Technology Review
Comparative Evidence Between Transdermal and Oral Estrogen as Part of Feminizing Hormone Therapy
Authors: Shariq Najeeb, Shannon E. Kelly, Nazmun Nahar, Melissa Brouwers, Becky Skidmore, Said Yousef Abdelrazeq, George A. Wells
This rapid review was conducted by the POst-market Drug Evaluation Team (PODET) through the Post-Market Drug Evaluation (PMDE) CoLab Network.
Key Messages
What Is the Issue?
Hormone therapy may be prescribed to support individuals identifying as transgender, nonbinary, and gender-nonconforming who would like to align their bodies or appearance with their gender identity.
For individuals seeking feminizing hormone therapy (FHT), estrogen is an established treatment.
Various administration routes (ways to take estrogen) are available for FHT with estrogen, including oral (taken as a pill) and transdermal (absorbed through the skin). However, the comparative clinical efficacy, effectiveness, and safety of transdermal and oral administration routes are unclear.
Decision-makers are interested in whether transdermal estrogen therapy should be considered for public reimbursement (funding) as a first treatment option (first-line option), as an alternative to oral estrogen therapy, in the context of gender-affirming care.
What Did We Do?
We evaluated the evidence of the clinical efficacy, effectiveness, safety, and cost-effectiveness (value from a human or health system perspective) of transdermal versus oral estrogen in gender-affirming care using a rapid review approach.
We searched for evidence-based guidelines on the use of transdermal or oral estrogen in gender-affirming care.
What Did We Find?
We identified 1 systematic review, 3 primary studies, and 4 evidence-based guidelines relevant to this review. No relevant health technology assessment (HTA) reports or cost-effectiveness studies were identified.
The systematic review reported no preidentified clinical efficacy or effectiveness outcomes and did not formally compare the safety of transdermal estrogen to oral estrogen, which limits any related conclusions regarding the safety of these products in the context of gender-affirming care.
The primary studies suggest that transdermal estrogen therapy may have lower cardiometabolic risks (conditions that affect the heart and metabolism, such as heart attack, obesity, and diabetes), have similar effects on bone health, and have similar feminizing effects (breast development, changes in body fat composition) compared to oral therapy. However, important patient outcomes such as quality of life and adverse effects remain unexamined.
Current guidelines, largely based on expert opinion, recommend transdermal estrogen therapy for patients older than either 40 or 45 years or those with a higher cardiovascular risk, starting at a lower dose and gradually increasing as necessary.
What Does This Mean?
Both transdermal and oral formulations appear safe, but it is unclear if transdermal estrogen offers the same or greater benefits in gender-affirming care.
Those making reimbursement decisions may consider individual risk for venous thromboembolism (VTE) (blood clots in the veins) or other harms that have been identified by those who are concerned about the risks associated with oral therapy.
Abbreviations
AusPATH
Australian Professional Association for Trans Health
BMD
bone mineral density
BMI
body mass index
DBP
diastolic blood pressure
FHT
feminizing hormone therapy
GAHT
gender-affirming hormone therapy
HDL
high-density lipoprotein
HTA
health technology assessment
IHD
ischemic heart disease
LDL
low-density lipoprotein
PICO
population, intervention, comparator, outcomes
RCT
randomized controlled trial
SBP
systolic blood pressure
TC
total cholesterol
TG
triglyceride
VTE
venous thromboembolism
WPATH
World Professional Association for Transgender Health
Introduction and Rationale
Background
Gender-Affirming Care
Gender-affirming hormone therapy (GAHT) mainly consists of either estrogen or testosterone.1 Transgender and nonbinary people may take hormone therapy as part of their gender transition to help their bodies and appearance align with their gender identity.2 The concept of GAHT has emerged to better address the health care needs of transgender, nonbinary, and gender-nonconforming individuals, moving beyond the traditional framework of hormone replacement therapy.3
Estrogen Therapy
Estrogen, along with antiandrogen therapy, is a key component of FHT, a form of GAHT, which involves administering hormones to suppress effects of masculinizing hormones and promote feminization. The reasons why transgender, nonbinary, and other gender-expansive people take estrogen hormone therapy varies from person to person. It can be used to reduce coarse body hair and facial hair and to develop breasts and hips.4,5 Some people choose to take estrogen because it makes them feel right in their body.6 Estrogen therapy sometimes requires the additional suppression of androgens (testosterone) in the body.7 Studies also indicate that FHT with estrogen improves bone mineral density (BMD).8
Available estrogen products used in FHT are administered by several routes of administration, each with different benefits and considerations. They are used primarily to induce and maintain feminizing physical changes in transgender women (assigned male at birth but identifying as female) and nonbinary individuals seeking feminization. The most common routes include oral tablets,9 which are convenient but carry higher risks of cardiovascular (CV) complications such as VTE, myocardial infarction, and hyperlipidemia caused by first-pass metabolism in the liver.10 Studies on individuals in postmenopause suggest that transdermal estrogen, compared to oral estrogen, may carry a reduced CV risk, particularly among smokers.11,12 Transdermal FHT involves the delivery of estrogen through patches, gels, or creams, which are applied directly to the skin and absorbed.13 There is speculation that transdermal estrogen products may have some advantages over oral products, including a potentially lower risk of CV adverse effects and VTE, and may be a more appropriate option over oral estrogen in certain patient groups, such as individuals with migraines and hypertension.14-16 Studies also suggest that, compared to oral FHT, transdermal FHT may result in lower CV risk, such as lower systolic blood pressure (SBP) and diastolic blood pressure (DBP) and healthier triglyceride (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL) lipid profiles. They may additionally lower CV risks associated with total cholesterol (TC) and body mass index (BMI).17
Policy Issue
There are 2 transdermal formulations of estrogen available in Canada: topical gels (e.g., EstroGel)18 and topical patches (e.g., Estradot).19 Multiple factors can influence the selection of an alternative hormonal therapy drug, including cost, adherence to regimen, and patient preference. Drug plans now seek more robust and current evidence comparing transdermal and oral estrogen therapy in the context of gender-affirming care. Therefore, the rationale for this rapid review arises from the need for a clear understanding of the clinical efficacy, safety, and cost-effectiveness of transdermal versus oral estrogen therapy to guide reimbursement decision-making.
Policy Question
Should transdermal estrogen be reimbursed in the first-line setting, as an alternative to oral estrogen, in the context of gender-affirming care?
Purpose
The objective of this project is to compare the efficacy, effectiveness, safety, and cost-effectiveness of transdermal estrogen therapy versus oral estrogen therapy in the context of gender-affirming care.
Research Questions
The following research questions will be used to address the policy question:
What is the clinical efficacy, effectiveness, and safety of transdermal estrogen compared to oral estrogen in the context of gender-affirming care?
What are the evidence-based guideline recommendations regarding the use of transdermal estrogen in the context of gender-affirming care?
What is the cost-effectiveness of transdermal estrogen compared to oral estrogen in the context of gender-affirming care?
Methods
We conducted a rapid review of the clinical efficacy and safety of transdermal estrogen in the context of gender-affirming care and of the evidence-based guidelines and cost-effectiveness literature.
Literature Search Methods
An experienced medical information specialist developed and tested the search strategies through an iterative process in consultation with the review team. Another senior information specialist peer-reviewed the MEDLINE strategy prior to execution using the Peer Review of Electronic Search Strategies (PRESS) Checklist.20 Using the multifile option and deduplication tool available on the Ovid platform, we searched Ovid MEDLINE® ALL, Embase Classic+Embase, APA PsycInfo, and the Cochrane Central Register of Controlled Trials. We applied a combination of controlled vocabulary (e.g., “health services for transgender persons,” “hormone replacement therapy,” “administration, topical”) and keywords (e.g., “trans female,” “HRT,” “transdermal”), adjusting the vocabulary and syntax as necessary across the databases. Robust design and economic filters were used in all databases but CENTRAL, which is prefiltered for randomized and controlled clinical trials. We did not restrict by language but did limit results to the publication years 2017 to the present and, where applicable, removed animal-only and opinion pieces. We performed all searches on August 26, 2024, and updated them on October 1, 2024.
We downloaded and deduplicated the records using EndNote version 9.3.3 (Clarivate Analytics) and uploaded them to Covidence (Veritas Health Innovation Ltd.).
We conducted a focused search for relevant HTAs by exploring potentially relevant documents published by international organizations or country-specific HTA agencies in Canada, the US, the UK, Australia, and New Zealand. This search was guided by the Canada’s Drug Agency Grey Matters Checklist, using keywords relevant to our research questions. A single reviewer systematically screened reports and publications from these HTA websites, retrieving any potentially relevant full-text documents for further evaluation to assess their eligibility for inclusion in the review.
A targeted search of various clinical guideline developers was conducted to identify guidelines for the use of FHT using transdermal estrogen published since 2017. Practice guideline developers specializing in GAHT (including FHT), both within Canada and internationally, as well as reputable generalist organizations, were selected. The targeted list of developers included the Canadian Professional Association for Transgender Health, Trans Care British Columbia, Alberta Health Services, the AIDS Committee of Toronto, Rainbow Health Ontario, Nova Scotia Health Authority, Australian Professional Association for Trans Health (AusPATH), World Professional Association for Transgender Health (WPATH), Endocrine Society, American College of Obstetricians and Gynecologists, British Medical Association, National Health Service England, Royal College of General Practitioners, and European Society for Sexual Medicine. We searched the guideline producer and organizations' websites to locate the most current version of any relevant guidelines.
FHT Marketed in Canada
In Canada, various estrogen products are available for FHT, as listed in Table 1.
Table 1: Selected Estrogen Products Marketed in Canada21
Brand | Route of administration | Active ingredient, formulation | Strength | DIN | Manufacturer |
|---|---|---|---|---|---|
Climara | Transdermal, patch | 17-beta estradiol, patch | 25 mg 50 mg 75 mg | 02247499 02231509 02247500 | Bayer Incorporated |
Lupin-Estradiol | Oral | 17-beta estradiol, tablet | 0.5 mg 1 mg 2 mg | 02449048 02449056 02449064 | Lupin Pharma Canada Limited |
Estrogel | Transdermal, gel | 17-beta estradiol, gel | 0.06% | 02238704 | Organon Canada Incorporated |
Premarin | Oral | conjugated estrogens, sustained-release tablet | 0.3 mg 0.625 mg 1.25 mg | 02414678 02414686 02414694 | Pfizer Canada |
Estradiol Derm | Transdermal, patch | 17-beta estradiol, patch | 50 mcg 75 mcg 100 mcg | 02246969 02246967 02246968 | Sandoz Canada Incorporated |
Estradot | Transdermal, patch | 17-beta estradiol, patch | 25 mcg 37.5 mcg 50 mcg 75 mcg 100 mcg | 02245676 02243999 02231509 02247500 02244002 | Sandoz Canada Incorporated |
Divigel | Transdermal, gel | 17-beta estradiol, gel | 0.1% | 02424835 02424843 02424924 | Searchlight Pharma Incorporated |
Oesclim | Transdermal, patch | 17-beta estradiol, patch | 25 mcg 50 mcg | 02243722 02243724 | Searchlight Pharma Incorporated |
DIN = Drug Identification Number.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 2.
Criteria | Description |
|---|---|
Population | Individuals requiring feminizing hormone therapy |
Intervention | Transdermal hormone therapy (includes estrogen alone but not estrogen in combination with progestogen) Differences between patch and gel are also of interest |
Comparator | Oral estrogen therapy (includes estrogen alone but not estrogen in combination with progestogen) Differences between oral tablet and sublingual use are also of interest |
Outcomes | Efficacy and effectiveness outcomes: Feminizing effects (hair, breast development, skin, body composition, changes to testes and prostate, sexual anatomic function, sexual desire, or libido), health-related quality of life, sleep quality, protective effect for dementia, improvement in bone mineral density Safety outcomes: Cardiovascular risk (venous thromboembolism, lipid metabolism, carbohydrate metabolism), genitourinary symptoms, endometriosis, gynecological cancer, breast cancer, stroke, withdrawal due to adverse events, withdrawal due to lack of efficacy |
Study designs | Randomized controlled trials, systematic reviews, health technology assessments, network meta-analyses, meta-analyses, comparative nonrandomized studies (e.g., cohort/case-control), evidence-based guidelines, published cost-effectiveness literature |
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 2, were duplicate publications, or were published before 2017. Abstracts, non-English articles, letters to the editor, opinion pieces, narrative reviews, books, and book chapters were excluded. Primary studies retrieved by the search were excluded if they were captured in 1 or more included systematic reviews. Guidelines that did not meet the PICO (population, intervention, comparator, outcomes) criteria or did not consider the route of administration were also excluded.
Critical Appraisal of Individual Studies
All studies were critically appraised by 1 reviewer. The included systematic review was critically assessed using the AMSTAR (A MeaSurement Tool to Assess systematic Reviews) 2 tool.22 The included primary studies were critically appraised using a modified version of the ROBINS-I (Risk Of Bias In Nonrandomized Studies of Interventions) checklist, which focused on study validity confounding, selection of participants, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes, and selection of reported results.23 Evidence-based guidelines were appraised using the AGREE II (Appraisal of Guidelines for Research and Evaluation II) instrument,24 focusing on the rigour of development and editorial independence domains. Each included publication’s strengths and limitations were described narratively.
Patient Engagement
Three individuals receiving transdermal or oral estrogen for gender-affirming care were confidentially interviewed before the literature search to share their lived and living experiences. These individuals offered personal perspectives on treatment priorities, research questions, and outcomes outlined in the selection criteria.
All individuals had several years of experience receiving hormone therapy in the context of gender-affirming care; 1 individual started oral estrogen therapy, 1 individual started on oral estrogen therapy and switched to transdermal estrogen therapy, and 1 individual started on transdermal estrogen therapy and switched to oral estrogen therapy.
Two individuals with lived and living experience reviewed the draft report to provide feedback on content and language.
A summary of the patient engagement report using the Guidance for Reporting Involvement of Patients and the Public (GRIPP2) framework is provided in Appendix 1, Table 3.
Summary of Evidence
Quantity of Research Available
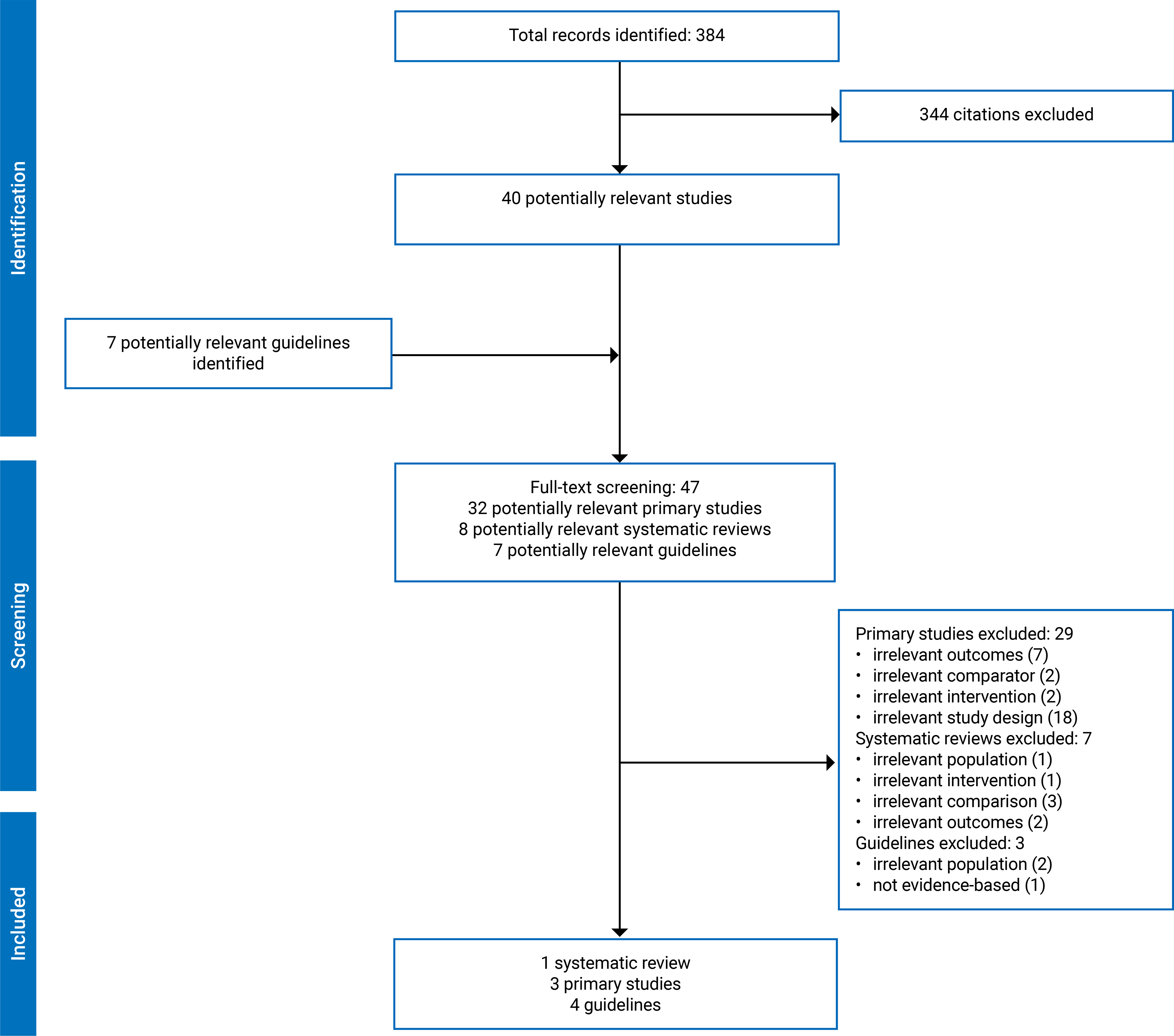
A total of 384 citations were identified in the literature search. Following the screening of titles and abstracts, 344 citations were excluded, and 40 potentially relevant reports from the electronic search were retrieved for full-text review. In addition, 7 potentially relevant guidelines were retrieved from the grey literature search for full-text review. Of these potentially relevant articles, 8 publications met the inclusion criteria and were included in this review. These comprised 1 systematic review17 and 3 primary studies (2 prospective cohort studies,25,26 1 retrospective cohort study27) and 4 evidence-based guidelines.9,28-30 No HTA reports or cost-effectiveness studies were identified. Figure 1 in Appendix 2 presents the PRISMA flow chart of the study selection.
Study Characteristics
Systematic Review
The systematic review, published in 2022, included 5 studies, of which 4 (3 prospective studies and 1 retrospective study) met the inclusion criteria of this report.17 The review provided a narrative synthesis of primary study findings, and no meta-analysis or any other statistical comparison was performed. The included studies were published up to 2016. The details of the systematic review are provided in Table 4 in Appendix 3.
Patient Population
The primary studies included in the systematic review were carried out in various countries in Europe, including the Netherlands, the UK, and Austria.17 Overall, 203 of 259 (78%) patients received transdermal or oral estradiol as FHT. The remaining patients received injectable FHT, which was not relevant to this review. None of the studies included information on ethnicity or other demographic details of the included patients.
Interventions and Comparators
The interventions in the systematic review primarily focused on the administration of hormone therapy through both non-oral (transdermal patches and intramuscular injections) and oral routes.
Across 4 studies included in the review, transdermal 17-beta estradiol was administered to more than 84 patients (the actual number of patients receiving transdermal FHT was not reported) at a dose of 100 mcg every 2 weeks for 2 to 60 months. Additionally, 23 patients received transdermal estradiol hemihydrate at a dose of 40 mcg or 50 mcg every 24 hours for 40 months. All transdermal FHTs were administered using patches. No information about the titration of doses was provided.
Comparators included various oral estrogen therapies. In 1 included study, oral ethinyl estradiol was administered to 15 patients who received 100 mcg every 24 hours for 2 to 4 months. In a second study, 57 patients received conjugated equine estrogen with doses ranging from 1.25 mg to 5 mg every 24 hours over a period of 6 to 40 months. Forty patients in the third study received oral estradiol valerate at a dose of 4 mg every 24 hours for 12 months. Lastly, in the fourth included study, 41 patients received oral 17-beta estradiol, with doses ranging from 2 mg to 4 mg every 24 hours for between 3 and 60 months.
Efficacy and Effectiveness Outcomes
The systematic review’s focus was on safety and, as such, did not report any efficacy or effectiveness outcomes of interest to the rapid review.
Safety Outcomes
The systematic review included outcomes focusing on cardiovascular risk across different routes of estrogen therapy administration. All included primary studies reported outcomes for cardiovascular events, which included LDL, HDL, TG, and TC levels. Two of the primary studies included in the review also assessed SBP and DBP measurements. However, the review did not specify or cite the studies reporting these outcomes, making it difficult to infer overall conclusions about cardiovascular risks associated with transdermal FHT versus oral FHT. None of the included primary studies reported data on all-cause or cardiovascular mortality.
Primary Studies
No RCTs were identified. Among the 3 primary studies included in this review,25-27 2 studies were described as prospective cohort studies,25,26 and 1 as a retrospective cohort study.27 None of the primary studies were included in the systematic review identified. The details of the primary studies are described in Table 5 in Appendix 3.
Countries Where the Studies Were Conducted
One study was conducted in Australia27 and 2 were conducted in the Netherlands.25,26
Patient Population
The number of patients included in the primary studies ranged from 49 to 231.25-27 One study examined the effects of estrogen administered through both transdermal and oral routes in a cohort of 212 patients.26 In 1 study, testosterone or estrogen was administered to 430 patients, with 231 patients receiving transdermal or oral estrogen.25 In 1 study, 55 patients received estrogen via transdermal or oral administration or a combination of these routes.27 After the exclusion of patients undergoing combination therapy, 49 patients who received transdermal or oral estrogen were included from this study in this review.27
All patients were adults, with median ages ranging from 28.0 to 52.8 years.25-27 Median ages of individual treatment groups were reported in 2 studies, with the transdermal group being older (median age 43.0 and 59.1 years) than those receiving oral FHT (median age 22.0 and 52.5 years).26,27 The ethnicity of the patients was reported in only 1 study, in which 97.4% of the included 430 patients were white,25 and the other 2 studies did not include ethnicity or other demographic information of patients specific to transdermal or oral FHT.26,27 In 1 study, patients were included if they had received FHT for 6 months or more,27 and in 2 studies, patients were excluded if they had previously received FHT.25,26
Interventions and Comparators
In Wiepjes et al., 99 patients received transdermal estradiol patches (50 mcg to 100 mcg twice weekly) combined with cyproterone (50 mg to 100 mg daily), and 63 patients received oral estradiol (2 mg to 4 mg daily) with cyproterone.25 The Balcerek et al. study included 32 patients on oral estradiol (median 4 mg) and 17 patients on transdermal estradiol (via patches with a median of 62.5 mcg or gels with a median dose of 1.0 mg to 1.25 mg).27 The Tebbens et al. study included 88 patients in the transdermal estradiol group (50 mcg to 100 mcg for 24 hours, twice weekly) and 124 patients in the oral estradiol group (2 mg to 6 mg daily).26 The 2 prospective studies by Wiepjes and colleagues and Tebbens and colleagues were carried out for 1 year, which was also the duration of transdermal and oral FHT.25,26 In 1 study, the median duration of transdermal FHT was 48 months,27 and the median duration of oral FHT was 78.3 months.27
None of the studies compared the efficacy of patches and gels to each other.
Efficacy and Effectiveness Outcomes
The included primary studies reported a limited number of outcomes relevant to this review. One study each reported BMD,25 change in BMI,27 and feminizing effects (change in body fat percentage and breast development).26
Safety Outcomes
Safety-related outcomes were reported in only 1 study, which reported the differences in cardiovascular risk factors in patients receiving transdermal estradiol compared to oral estradiol.27 Specifically, the following outcomes were assessed: BMI, number of patients with ischemic heart disease (IHD), number of VTE events, number of patients with obesity (defined by BMI > 30), dyslipidemia (increased lipid levels from baseline), and new cases of diabetes.
Evidence-Based Guidelines
Four evidence-based guidelines were identified.9,28-30 These guidelines were developed by the multinational Endocrine Society (last updated in 2017),28 AusPATH in Australia (published in 2019),29 Standards of Care by WPATH (version 8, last updated in 2022),9 and Rainbow Health Ontario (last updated in 2023).30 Rainbow Health Ontario is a government-funded program dedicated to improving transgender and nonbinary patients’ health outcomes.31 Detailed characteristics of the evidence-based guidelines are provided in Table 6 in Appendix 3.
Critical Appraisal
Summaries of the critical appraisal of the systematic review, primary studies, and evidence-based guidelines are presented in Table 7, Table 8, and Table 9, respectively.
Systematic Review
The AMSTAR 2 assessment of the systematic review by Miranda and colleagues17 highlighted several strengths, including a clearly focused research question based on PICO criteria, a predefined review method registered on PROSPERO (International Prospective Register of Systematic Reviews), and a comprehensive literature search across multiple databases. Study selection and data extraction were conducted by 2 reviewers independently, and the risk of bias was assessed using the Newcastle-Ottawa Scale. The review also had limitations such as a lack of details of the study designs of the primary studies, incomplete reporting of excluded studies, and insufficient details about the populations studied. The heterogeneity of outcomes prevented a meta-analysis, which limited the ability to assess quantitatively both the risk of bias and potential publication bias. No efficacy outcomes related to FHT were included in the PICO and hence were not extracted from the primary studies.
Although the review had only 1 critical flaw (excluded studies not listed), its utility in this rapid review is limited given it did not report the overall outcomes of FHT specific to transdermal routes (the injectable and transdermal routes were combined as non-oral and compared to the oral route in the descriptive analysis) making it difficult to draw meaningful conclusions specifically about the safety of transdermal FHT.
Primary Studies
All eligible studies clearly addressed the focused research question.25-27 One study controlled for potential confounding factors such as age, vitamin D supplementation, body weight, smoking, and alcohol use,25 and another study controlled for baseline age, BMI, and estradiol concentrations.26 One study did not adjust for any confounding factors.27 Two studies did not report on patients lost to follow-up or those who discontinued due to adverse effects, making it challenging to evaluate potential attrition bias that these patients may systematically differ from those who remain in the study.26,27 Additionally, a major limitation across these studies was the lack of safety data or adverse effect reporting as well as the absence of major efficacy outcomes for FHT.25-27 One study had a small sample size of patients (49 patients) receiving FHT.27 None of the studies recorded whether patients discontinued FHT due to adverse effects, and important patient outcomes such as feminizing effects (breast development and changes in body fat) were reported by only 1 study26 and entirely omitted by the other 2 studies.25,27 Although 1 retrospective study described the treatment period as almost 6 years,27 neither of the prospective studies had a follow-up period beyond 12 months.25,26 Therefore, these limitations hinder the ability to fully assess the comparative safety, efficacy, and long-term impact of transdermal FHT versus oral FHT.
Evidence-Based Guidelines
The Endocrine Society28 guidelines referenced systematic review methods but lacked detailed information on how these were applied. It also provided limited detail on how the criteria for selecting evidence were determined, as a PICO was not clearly defined. The strengths and limitations of the evidence were discussed to some extent, with the guideline using its own grading scale. In contrast, AusPATH29 and Rainbow Health Ontario30 guidelines based their findings on pre-existing systematic reviews, expert opinion, and observational studies; did not elaborate on the PICO criteria; and failed to address the strengths and limitations of the evidence in detail. WPATH, on the other hand, showed stronger rigour by implementing a good systematic review methodology and using an elaborate PICO, though it did not explicitly mention the PICO elements in the guidelines.9 Both WPATH and AusPATH used Grading of Recommendations Assessment, Development and Evaluation (GRADE)32 to evaluate recommendations, which enhanced the clarity around the strengths and limitations of the evidence.9,29 Overall, there was variability in the transparency and depth of the methods used to formulate recommendations, with WPATH9 standing out for its thorough approach, whereas the other guidelines provide more limited explanations.
In terms of formulating recommendations, the Endocrine Society 201728 and Rainbow Health Ontario30 offered minimal details about the process. The guidelines by AusPATH,29 provided vague indications on how the recommendations were developed that lacked significant depth. In contrast, WPATH 20229 provided a more thorough description, considering the benefits, side effects, and risks of different recommendations in the formulation process.
Findings
Findings from the included systematic reviews, primary studies, and evidence-based guidelines are presented in Table 10, Table 11, and Table 12 in Appendix 5.
Systematic Review
A summary of the findings in the Miranda et al. systematic review is presented in Appendix 5, Table 10.
Efficacy and Effectiveness Outcomes
The included systematic review did not report any efficacy and efficacy outcomes.
Safety Outcomes
Cardiovascular Risk
In their 2022 systematic review, Miranda and colleagues considered cardiovascular risk among 203 of 259 patients receiving transdermal or oral FHT across 3 retrospective studies and 1 prospective study.17 The review examined changes in baseline for BMI, SBP, DBP, HDL, LDL, TC, and TG levels in patients receiving transdermal FHT and oral FHT for up to 60 months. However, there were no statistical analyses conducted to measure within- or between-group differences in the review, limiting our ability to draw firm conclusions about the relative safety of each administration route.
Primary Studies
A summary of the findings from the 3 included primary studies is presented in Appendix 5, Table 11.
Efficacy and Effectiveness Outcomes
Body Mass Index
In 1 study,27 there were no significant differences in the change from baseline in BMI between patients receiving FHT via transdermal patches (17 patients) or oral estrogen (32 patients). The median duration of therapy was 48.0 months for transdermal patches and 78.3 months for oral.27
Feminizing Effects
In 1 study, there were no statistically significant differences in breast development or in changes in fat displacement in individuals receiving transdermal FHT or oral FHT.26
Bone Mineral Density
In 1 study,25 no statistically significant difference was observed between the total BMD measured in the lumbar spine, total hip, and femoral neck regions in patients receiving transdermal estradiol FHT via patches (63 patients) compared to oral FHT (99 patients) after 1 year of therapy.
Safety Outcomes
Cardiovascular and Metabolic Risk Factors
One study reported that no differences in the proportion of patients with diabetes, hypertension, obesity, IHD, cardiovascular disease, or venous thromboembolic disease were observed regardless of the route of estrogen received as part of gender-affirming care.27 Nevertheless, the study reported that a significantly higher number of patients receiving transdermal estrogen had dyslipidemia (increased levels of lipids from baseline) compared to patients receiving oral estrogen.27
Evidence-Based Guidelines
For individuals either older than 40 years or older than 45 years, or those at higher risk of VTE or cardiovascular events, all 4 guidelines recommend transdermal estradiol over oral formulations as a safer option.9,28-30 The guidelines provide varied recommendations for the dosage and regimen of transdermal estradiol in GAHT. Estradiol patches typically range from 50 mcg to 200 mcg per day, with applications suggested either every 3 to 5 days or twice weekly.9,28-30 Estradiol gel begins at a daily dose of 2.5 g (150 mcg estradiol), with a maximum of 6.25 g (375 mcg), though application area may limit higher doses.9,28-30 The details of the regimens are provided in Table 12 in Appendix 5.
The strength of the recommendations ranged from weak (reflecting uncertainty or variability in the balance between benefits and risks, often due to lower-quality evidence, patient preferences, or differing contexts) to strong (indicating high confidence that the intervention’s benefits clearly outweigh its risk).9,28-30 The quality of evidence ranged from low — based on observational studies, case series, or case reports — to moderate — typically derived from well-conducted observational studies or RCTs with some limitations, such as small sample sizes, incomplete follow-up, or methodological flaws.9,28-30 Some recommendations were informed by studies on menopause in cisgender people in addition to individuals receiving FHT.9,29,30
Cost-Effectiveness of Transdermal FHT Compared to Oral FHT
No relevant evidence regarding the cost-effectiveness of transdermal FHT compared to oral FHT was identified in any of the included systematic reviews, primary studies, the guidelines, or other publications.
Limitations
Several limitations should be considered in this rapid review comparing transdermal versus oral estrogen for gender-affirming care. A key limitation of this report is the limited number of studies included. Only 1 systematic review and 3 primary studies met the eligibility criteria. Moreover, none of the primary studies were RCTs; they were observational studies, which are more prone to confounding, selection bias, and difficulties in establishing causality.25-27 Based on the decision to balance rigorous methodology with timeliness when conducting a rapid review, inherent methodological constraints (such as a limited search strategy, a less comprehensive risk of bias assessment, and a restricted inclusion criteria) were unavoidable.
Systematic Review
A major limitation of the systematic review included in this report is that, in reporting the individual outcomes from studies of transdermal versus oral routes, the authors combined outcomes for transdermal and intramuscular FHT as non-oral routes in their overall synthesis, diluting the specificity of the comparative evidence of interest.17 Furthermore, although outcomes from individual studies were reported, no comparisons were made between routes of adminsitration.17 Additionally, the reviewed studies lacked clear definitions for key population characteristics, such as ethnicity and prior treatment history, further limiting the generalizability and applicability of the findings to specific patient groups. Overall, it was difficult to infer meaningful conclusions from this systematic review.
Primary Studies
In 2 of the 3 primary studies included in this review, patients receiving transdermal FHT were older (aged 59.1 and 43.0 years) than those receiving oral FHT (aged 52.5 and 22.0 years),26,27 and in 1 study the median or mean age of patients receiving either transdermal or oral FHT were not reported.25 The differences in age within the treatment groups may have impacted estrogenic effects on outcomes, and this makes it challenging to inform treatment decisions for diverse age groups. One study provided the ethnicity information and included mostly participants who were white. Only 1 study was a multicentre study25 (while 1 was conducted at 1 clinic26 and, in the other study, patients were recruited from 2 clinics)27 and all studies were conducted in either the Netherlands or Australia, limiting the external validity and applicability of their results in diverse clinical settings and populations.25-27 Two of the included studies did not report if any patients were lost to follow-up or to adverse effects, which could have led to attrition bias in these studies.26,27 Two of the 3 primary studies did not exclude individuals who had received FHT before the start of the study, which may limit the applicability of the findings to inform the use of transdermal FHT as a first-line option for gender-affirming care. Although 1 study included feminizing effects such as breast development and change in body fat percentage,26 other feminizing effects such as those associated with hair, skin, testes, prostate, sexual anatomic function, sexual desire, or libido were not reported, further limiting the ability to draw comprehensive conclusions about the feminizing effects of transdermal FHT. None of the included studies reported quality of life as an outcome. The number of patients receiving FHT ranged from 49 to 231,25-27 but with only 1 study of 49 patients included in the comparison of BMI and cardiovascular risk between transdermal and oral routes, the robustness of any definitive conclusions for this outcome was limited.27 As such, random variation in the results cannot be ruled out, and additional studies with similar sample sizes may provide different findings.33 Additional studies with larger sample sizes are required to provide robust estimates for the outcomes of interest in this population. Moreover, variations in study design and bias mitigation, such as, but not limited to, adjusting for confounding variables, may have led to inconsistent or conflicting results, affecting the interpretability and comparability of outcomes. Together, selective reporting of outcomes and lack of standardized protocols likely contributed to variability in outcomes, thereby reducing the overall robustness of the evidence. Comparative RCTs are needed to better ascertain the clinical efficacy and safety of transdermal estrogen versus oral estrogen.
Guidelines
Although studies on treatments for menopause and postmenopause suggest that transdermal estrogen may be a safer option compared to oral estrogen,10,34 the limited data on critical clinical outcomes in studies of individuals receiving transdermal estrogen for FHT — such as feminizing effects, cardiovascular safety, metabolic adverse effects, and quality of life — significantly impede the creation of clear, evidence-based guidelines for its use, particularly for younger and more diverse patient populations. A limitation common to all 4 included guidelines was an increased potential for bias due to their reliance on observational studies (or systematic reviews of observational studies).9,28-30 In some cases, data were derived from studies conducted on cisgender individuals to inform their recommendations.9,29,30 This may limit the applicability of the evidence to transgender populations because therapy goals, biology, and age ranges of individuals undergoing hormone therapy for the management of menopause are different from individuals who are receiving transdermal estrogen for gender-affirming care.
Patient Engagement
A notable limitation of this work relates to the patient engagement component of the rapid review. The engagement process involved 4 individuals who shared their lived and living experiences with FHT. However, the perspectives of these individuals cannot fully represent the lived and living experiences of the broader patient population. Additionally, no qualitative research or analysis of patient perspectives was conducted as part of this process.
Discussion and Implications for Decision- or Policy-Making
Conclusion
The objective of the rapid review was to summarize the evidence about the benefits and harms of transdermal estrogen therapy compared to oral estrogen therapy for gender-affirming care. We found 1 systematic review, 3 observational studies, and 4 evidence-based guidelines. No HTAs, RCTs, or pharmacoeconomic studies were identified.
Engaging with individuals who have experience with FHT was an important component of the rapid review. Through interviews, we gathered information regarding their perspective on relevant outcomes that should be considered in our review as well as their views on treatment priorities and access to treatment.
The interviewed participants identified feminization as the primary desired outcome of FHT so that their body aligns with their identity. They specifically mentioned breast development, facial features, skin, and body fat distribution as important outcomes. The evidence to date suggests that there are no statistically significant differences between the transdermal and oral formulations in changes in BMI or fat distribution or in breast development.
During the interviews, safety was raised as a main concern, and the participants indicated that transdermal FHT may have a better safety profile than oral therapy. Liver damage, cancer, and osteoporosis were all specifically mentioned as concerns. One observational study of 49 patients reported no statistically significant differences in the number of patients with diabetes, hypertension, obesity, IHD, or VTE. However, patients administered the transdermal formulation had a statistically significant increase in lipid levels. The largest observational study (231 patients receiving FHT) showed that there were no statistically significant differences in BMD.
The systematic review reported the changes from baseline for BMI, SBP, DBP, TG, TC, LDL, and HDL levels across individual studies, but no statistical analyses were conducted. It also did not conduct any statistical analyses of overall differences between transdermal and oral routes. Because of the lack of comparisons, no conclusions can be drawn from the systematic review.
Finding appropriate dosage levels of estrogen therapy was a challenge for all 3 interviewed participants. For individuals who switched from 1 formulation to another, the primary reason for switching was dosing challenges. Participants expressed a preference to start FHT with the safest, lowest-risk dosage. The guidelines offer various recommendations on the dosage regimen of transdermal estradiol in FHT.
Two observational studies included individuals receiving FHT aged 20 to 63 years, and the patients receiving transdermal estrogen were older than those receiving oral estrogen. Therefore, the results are not applicable to a younger population. One observational study reported that most patients included in the study were white, and ethnicity was not reported in the other 2 observational studies. Therefore, we cannot comment on the generalizability of the findings. Furthermore, the observational studies measured surrogate outcomes and not long-term clinical outcomes of concern to the participants.
Implications for Decision-Making
The policy question inquired as to whether transdermal estrogen should be reimbursed as first-line treatment, as an alternative to oral estrogen, in the context of gender-affirming care. The evidence considered in this review cannot adequately answer this question. While both transdermal and oral formulations appear to be safe, insofar as near-term cardiovascular risk among reasonably healthy adults, we cannot be certain if receiving transdermal estrogen for gender-affirming care confers similar or greater benefits than the oral formulation. Unlike orally administered estrogen, which undergoes first-pass metabolism in the liver, transdermal estrogen avoids this process, potentially reducing risks associated with hepatic metabolism, such as VTE. This distinction is especially critical for individuals with liver dysfunction or elevated VTE risk, who may benefit from transdermal over oral formulations. Expert guidelines recommend that the transdermal preparations be considered in certain groups of patients. All 4 evidence-based guidelines recommend transdermal FHT in those aged older than either 40 or 45 years, or those with cardiovascular or thromboembolic risk factors, and recommend starting with the lowest dosage and gradually increasing as necessary. Considering the evidence, it may be prudent to base decisions on the existing guidelines. Reimbursement policy-makers may consider individual risk for VTE or other harms among individuals who have concerns about the risks associated with oral estrogen therapy.
References
1.D'hoore L, T'Sjoen G. Gender-affirming hormone therapy: an updated literature review with an eye on the future. Journal of internal medicine. 2022;291(5):574-592. PubMed
2.Aghi K, Goetz TG, Pfau DR, Roepke TA, Guthman EM. Centering the needs of transgender, nonbinary, and gender-diverse populations in neuroendocrine models of gender-affirming hormone therapy. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging. 2022;7(12):1268-1279. PubMed
3.Randolph Jr JF. Gender-affirming hormone therapy for transgender females. Clinical obstetrics and gynecology. 2018;61(4):705-721. PubMed
4.Deutsch MB. Transgender Care & Treatment Guidelines: Overview of feminizing hormone therapy. UCSF Gender Affirming Health Program. Published June 17, 2016. Accessed November 13, 2024. https://transcare.ucsf.edu/guidelines/feminizing-hormone-therapy.
5.Klaver M, De Blok CJM, Wiepjes CM, et al. Changes in regional body fat, lean body mass and body shape in trans persons using cross-sex hormonal therapy: results from a multicenter prospective study. European Journal of Endocrinology. 2018;178(2):163-171. PubMed
6.Gender affirming hormone therapy. AIDS Committee of Toronto. Accessed June 08, 2024. https://www.actoronto.org/health-information/campaign-6-gaht/.
7.Schagen SEE, Lustenhouwer P, Cohen-Kettenis PT, Delemarre-van de Waal HA, Hannema SE. Changes in adrenal androgens during puberty suppression and gender-affirming hormone treatment in adolescents with gender dysphoria. The Journal of Sexual Medicine. 2018;15(9):1357-1363. PubMed
8.Giacomelli G, Meriggiola MC. Bone health in transgender people: a narrative review. Ther Adv Endocrinol Metab. 2022;13:20420188221099346. doi:10.1177/20420188221099346 PubMed
9.Coleman E, Radix AE, Bouman WP, et al. Standards of Care for the Health of Transgender and Gender Diverse People, Version 8. Int J Transgend Health. 2022;23(Suppl 1):S1-S259. doi:10.1080/26895269.2022.2100644 PubMed
10.Kim SE, Min JS, Lee S, Lee DY, Choi D. Different effects of menopausal hormone therapy on non-alcoholic fatty liver disease based on the route of estrogen administration. Sci Rep. Sep 19 2023;13(1):15461. doi:10.1038/s41598-023-42788-6 PubMed
11.Girdler SS, Hinderliter AL, Wells EC, Sherwood A, Grewen KM, Light KC. Transdermal versus oral estrogen therapy in postmenopausal smokers: hemodynamic and endothelial effects. Obstetrics & Gynecology. 2004;103(1):169-180. PubMed
12.Lan X, Cai S, Li G, Prabahar K, Hernández-Wolters B, Yin Y. Effects of Transdermal 17β-Estradiol+ Norethisterone Acetate on Cardiovascular Disease Risk Factors in Postmenopausal Women: A Meta-Analysis of Data From Randomized, Controlled Trials. Clinical Therapeutics. 2023; PubMed
13.Mercer J, Bandy S, Tang H, et al. Gender-affirming pharmacotherapy and additional health considerations: a contemporary review. Journal of the American Pharmacists Association. 2023; PubMed
14.Scheres LJJ, Selier NLD, Nota NM, van Diemen JJK, Cannegieter SC, den Heijer M. Effect of gender-affirming hormone use on coagulation profiles in transmen and transwomen. J Thromb Haemost. Apr 2021;19(4):1029-1037. doi:10.1111/jth.15256 PubMed
15.Smits MG, van der Meer YG, Pfeil JP, Rijnierse JJ, Vos AJ. Perimenstrual migraine: effect of Estraderm TTS and the value of contingent negative variation and exteroceptive temporalis muscle suppression test. Headache. Feb 1994;34(2):103-6. doi:10.1111/j.1526-4610.1994.hed3402103.x PubMed
16.Hranilovich JA, Kaiser EA, Pace A, Barber M, Ziplow J. Headache in transgender and gender-diverse patients: A narrative review. Headache. Jul 2021;61(7):1040-1050. doi:10.1111/head.14171 PubMed
17.Turino Miranda K, Kalenga CZ, Saad N, et al. Gender-affirming estrogen therapy route of administration and cardiovascular risk: a systematic review and narrative synthesis. Am J Physiol Heart Circ Physiol. Nov 1 2022;323(5):H861-H868. doi:10.1152/ajpheart.00299.2022 PubMed
18.CADTH. Estradiol and Progesterone (Bijuva). Canadian Journal of Health Technologies. 2022;2(4)
19.Health Canada. Product Monograph: Estradot. Accessed February 7, 2025. https://health-products.canada.ca/dpd-bdpp/info?lang=eng&code=6794.
20.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 Guideline Statement. J Clin Epidemiol. Jul 2016;75:40-6. doi:10.1016/j.jclinepi.2016.01.021 PubMed
21.Hormone replacement therapy shortage. Canadian Pharmacists Association. Published April 2024. Accessed February 07, 2025. https://www.pharmacists.ca/cpha-ca/assets/File/cpha-on-the-issues/Hormone-Replacement-Therapy-Shortage_EN.pdf.
22.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. Bmj. Sep 21 2017;358:j4008. doi:10.1136/bmj.j4008 PubMed
23.Sterne JAC, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. bmj. 2016;355.
24.Brouwers MC, Kho ME, Browman GP, et al. AGREE II: advancing guideline development, reporting, and evaluation in health care. Prev Med. Nov 2010;51(5):421-4. doi:10.1016/j.ypmed.2010.08.005 PubMed
25.Wiepjes CM, Vlot MC, Klaver M, et al. Bone Mineral Density Increases in Trans Persons After 1 Year of Hormonal Treatment: A Multicenter Prospective Observational Study. J Bone Miner Res. Jun 2017;32(6):1252-1260. doi:10.1002/jbmr.3102 PubMed
26.Tebbens M, Heijboer AC, T'Sjoen G, Bisschop PH, den Heijer M. The Role of Estrone in Feminizing Hormone Treatment. J Clin Endocrinol Metab. Jan 18 2022;107(2):e458-e466. doi:10.1210/clinem/dgab741 PubMed
27.Balcerek MI, Nolan BJ, Brownhill A, et al. Feminizing Hormone Therapy Prescription Patterns and Cardiovascular Risk Factors in Aging Transgender Individuals in Australia. Front Endocrinol (Lausanne). 2021;12:667403. doi:10.3389/fendo.2021.667403 PubMed
28.Hembree WC, Cohen-Kettenis PT, Gooren L, et al. Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. Nov 1 2017;102(11):3869-3903. doi:10.1210/jc.2017-01658 PubMed
29.Cheung AS, Wynne K, Erasmus J, Murray S, Zajac JD. Position statement on the hormonal management of adult transgender and gender diverse individuals. Med J Aust. Aug 2019;211(3):127-133. doi:10.5694/mja2.50259 PubMed
30.Rainbow Health. Guidelines for gender-affirming primary care with trans and non-binary patients, 4th edition. Published 2019. Accessed February 07, 2025. https://www.rainbowhealthontario.ca/product/4th-edition-sherbournes-guidelines-for-gender-affirming-primary-care-with-trans-and-non-binary-patients/.
31.Antonio M, Lau F, Davison K, Devor A, Queen R, Courtney K. Toward an inclusive digital health system for sexual and gender minorities in Canada. Journal of the American Medical Informatics Association. 2022;29(2):379-384. PubMed
32.Guyatt G, Oxman AD, Akl EA, et al. GRADE guidelines: Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;64(4):383-94. PubMed
33.Faber J, Fonseca LM. How sample size influences research outcomes. Dental Press J Orthod. Jul-Aug 2014;19(4):27-9. doi:10.1590/2176-9451.19.4.027-029.ebo PubMed
34.Kopper NW, Gudeman J, Thompson DJ. Transdermal hormone therapy in postmenopausal women: a review of metabolic effects and drug delivery technologies. Drug design, development and therapy. 2009:193-202. PubMed
Authors and Contributors
Authors
Shariq Najeeb contributed to screening studies; extracting data; analyzing and interpreting results, figures, and tables; verifying and assessing quality; and drafting and revising the report.
Shannon E. Kelly contributed to the conceptualization, methodology, and validation; the writing of the original draft of the report and the review and editing of its contents; and supervision and project administration.
Nazmun Nahar contributed to the full-text searches.
Melissa Brouwers contributed to supervision; funding acquisition; writing, editing, and review; and validation.
Becky Skidmore developed the search strategy in consultation with the review team; responded to PRESS feedback; translated and executed final strategies; deduplicated, packaged, and uploaded records to Covidence; and tracked PRISMA, provided search documentation, and wrote methods for the final manuscript.
Said Yousef Abdelrazeq contributed to the conception and design and the revision of the final report.
George A. Wells contributed to supervision; funding acquisition; writing, editing, and review; validation; and methodology.
Contributors
We thank Kaitryn Campbell, MLIS, MSc (Hamilton, ON) for peer-reviewing the MEDLINE search strategy.
This project was selected as part of a pilot for early engagement with patients and clinicians. We thank the contributors from these impacted groups for providing feedback and expertise throughout this project.
Patient Partners
We thank Alexis Egri, Alice Holland, Laura P., and 1 other person (who wishes to remain anonymous) for providing input into the direction of the research, drawing from lived experience, and providing valuable contributions to this report.
Clinical Experts
These individuals kindly provided comments on this report:
Sheila Wijayasinghe, MD, CCFP, MSCP
Family Physician, Medical Director, Primary Care Outreach
University of Toronto
Emma Gibbons, MD CCFP
Family Physician, Gender-Affirming Care Specialist
Conflicts of Interest
Said Yousef Abdelrazeq disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
TR0016 Long-Acting Inhaled Agents for COPD
Shannon E. Kelly disclosed the following:
Involvement with projects or scientific advice
RE0048 Remdesivir for COVID-19 in the Inpatient Setting
HT0041 Remdesivir for COVID-19 in the Outpatient Setting
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
TR0016 Long-Acting Inhaled Agents for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Nazmun Nahar disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
Becky Skidmore disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
TR0016 Long-Acting Inhaled Agents for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Melissa Brouwers disclosed the following:
Involvement with projects or scientific advice
RE0048 Remdesivir for COVID-19 in the Inpatient Setting
HT0041 Remdesivir for COVID-19 in the Outpatient Setting
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
TR0016 Long-Acting Inhaled Agents for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Pagebreak
Shariq Najeeb disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
TR0016 Long-Acting Inhaled Agents for COPD
George A. Wells disclosed the following:
VBI Vaccines Inc. (2020–current): Coronavirus vaccine; Data Safety Monitoring Board, member
Thermedical (2021–current): Ablation system and catheter needle; design and analysis advice for preparation of FDA submission
VBI Vaccines Inc. (2020–current): Coronavirus vaccine; design and analysis advice for preparation of FDA submission
Involvement with projects or scientific advice
RE0048 Remdesivir for COVID-19 in the Inpatient Setting
HT0041 Remdesivir for COVID-19 in the Outpatient Setting
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
TR0016 Long-Acting Inhaled Agents for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Kaitryn Campbell disclosed the following:
Involvement with projects or scientific advice
RE0043 Omalizumab for Chronic Idiopathic Urticaria
RC1549 BRAF-MEK Inhibitors in Melanoma With Brain Metastasis
RC1552 Biologics for Inflammatory Bowel Disease
RE0050 Immuno-oncology for Mutated Non–Small Cell Lung Cancer
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
TR0016 Long-Acting Inhaled Agents for COPD
RD0076 Antiviral Drugs for the Treatment of Influenza
Sheila Wijayasinghe disclosed the following:
Payments as advisor or consultant
Sanoling AI (2024): Virtual Care
Felix Virtual Care (2024): Virtual Care
McMaster Women’s Health Lecture on Menopause
Advisory Board for National Menopause Show
Walrus Talks Menopause
Involvement with projects or scientific advice
RD0073 Transdermal Versus Oral Menopausal Hormone Therapy
No other conflicts of interest were declared.
Appendix 1: Summary of Patient Engagement
Please note that this appendix has not been copy-edited.
Table 3: Reporting of Patient Engagement Using the GRIPP2 Framework
Section and topic | Item |
|---|---|
1: Aim | Three contributors shared their experiences and perspectives of oral and/or transdermal FHT to offer insights into their lived and living experiences and priorities, and to allow the authors to contextualize the information they read in the literature. This is part of a CDA-AMC pilot of earlier engagement in Post-Market Drug Evaluation (PMDE) projects. |
2: Methods | Interviews During the scoping phase, 3 trans women with lived and living experience of FHT participated in individual semistructured interviews with CDA-AMC staff. A member of the research team participated as an observer. Their perspectives were subsequently summarized, those summaries were approved by the contributors, and then they were shared with the project team. Document review Two trans women with experience of FHT participated as partners in document review, sharing their thoughts on the draft scoping document, draft report, and the plain language summary. As this is a pilot, they will also engage in an evaluation of the engagement process. |
3: Results of engagement | The researchers were made aware of the importance of several themes. Treatment priorities Safety was a top priority, with risks of liver damage, cancer, and osteoporosis specifically mentioned as concerns. Contributors felt that starting on the safest, lowest-risk method was preferable. Efficacy and effectiveness were the primary drivers for all 3 contributors. While some preferred 1 method over another for convenience or safety reasons, all 3 wanted to find a treatment regimen that worked for them. They were willing to take a less convenient or slightly higher risk therapy to find a treatment that was effective. Appropriate dosing levels were another factor for consideration, as all 3 contributors mentioned difficulties finding appropriate and effective doses. Convenience played a role in their preferred method. Some contributors found that daily pills worked into their routine easily, while others preferred to change a patch twice a week. We heard that contributors wanted a treatment regimen that they could adhere to for the next 50 years that would not pose a mental burden or inconvenience over time. Outcomes Feminization was the top outcome priority for all 3 contributors, with breast development, facial features, skin, body fat distribution specifically mentioned as priorities. However, overall “feminization” was the most desired outcome, without a specific aspect of femininity. One contributor wanted her body to align with her facial features, another wanted to “pass” as a woman, 1 indicated that she wanted a body that aligned with her identity. Health was another major priority outcome. For 2 individuals, after trouble balancing hormone levels, they wanted to be in a body that works, that feels healthy, that feels as it should. They didn’t want crashing estrogen levels, they wanted to feel well. They also wanted to minimize risks to their health. Research questions One contributor asked about the role of the patient’s individual choice in the research questions. All 3 contributors felt that transdermal FHT should be available as a first-line therapy, as an option alongside oral medications. They felt the decision should be determined by the individual patient and their clinician, based on individual risk factors and preferences. Final thoughts Knowledge and stigma The contributors felt that FHT is portrayed negatively in the media, with little information about the benefits and an overstatement of the risks, leading to misinformation and fear. This affects both the individual considering FHT and their clinician. Some clinicians are reluctant to treat due to lack of knowledge or personal bias. One contributor hoped that there was more research and reports that could be available to the trans community and to clinicians. Accessibility Stable and consistent access to medications was another challenge, especially during drug shortages. Access to a compounding pharmacy was a barrier to injectable FHT, which presented a barrier to that method. |
4: Discussion and conclusions | While there were differing experiences and opinions on what method of FHT worked for each of the contributors, they were most concerned about safety and efficacy, with feminization as the priority outcome. One of the key comments we heard was about choice and options. Contributors wanted to have the option of starting on the method with the lowest risk profile and changing based on need. |
5: Reflections and critical perspective | Success of engagement in this project is related to several factors. First, the contributors and patient partners were briefed on the objectives of the project in an introductory call and supported in their role in the engagement process by a Patient Engagement Officer. The research team was receptive to this involvement and incorporated it in their approach to the clinical evidence. Compensation was offered for people’s time and expertise. However, there were limitations. The invitation was shared both through relevant groups and online. The outreach generated interest, but we were only able to engage with a limited number of contributors and patient partners. The perspectives of those we engaged cannot fully represent the lived and living experiences of the broader patient population. Another limitation of this engagement approach is that people need a phone or computer and reliable internet access to contribute to this work, which may exclude some voices. |
FHT = feminizing hormone therapy.
Appendix 2: Selection of Included Studies
Please note that this appendix has not been copy-edited.
Appendix 3: Characteristics of Included Publications
Please note that this appendix has not been copy-edited.
Table 4: Characteristics of the Included Systematic Review
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention(s) and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Miranda et al. (2022)17 Country: Canada Funding source: University of Alberta, Canadian Institutes of Health Research | Total included: 5 studies Studies eligible for this review: 4a Study designs:a 3 prospective cohorts 1 retrospective cohort | Total: 259 FHT: 203 Age (years), range: Transdermal: 31 to 47b; Oral: 30 to 36b Ethnicities: NR | Interventionsc,d,e: Transdermal 17-beta estradiol Patients: > 84f Dose: 100 mcg every 2 weeks Duration: 2 to 60 months Transdermal estradiol hemihydrate Patients: 23 Dose: 40 mcg or 50 mcg daily Duration: 40 months Comparatorse: Oral ethinyl estradiol Patients: 15 Dose: 100 mcg daily Duration: 2 to 4 months Conjugated equine estrogen Patients: 57 Dose: 1.25 mcg to 5 mg daily Duration: 6 to 40 months Oral estradiol valerate Patients: 40 Dose: 4 mg daily Duration: 12 months Oral 17-beta estradiol Patients: 41 Dose: 2 mg to 4 mg daily Duration: 3 to 60 months | Outcomes: Cardiovascular risk factors (blood pressure, triglycerides, total cholesterol, body mass index, low- and high-density lipid levels), all-cause mortality, cardiovascular mortality. Follow-up: 0 to 60 months Duration of studies period: 4 to 12 months |
FHT = feminizing hormone therapy; NR = not reported.
aData from one study excluded from this review because outcomes were compared between intramuscular injections and oral route.
bOne study did not provide mean age.
cData was only shown for transdermal route. Intramuscular injections outcome excluded this report.
dTransdermal 17-beta estradiol reported in 4 studies and transdermal estradiol hemihydrate reported in one study.
eIn all studies, cyproterone acetate (50 to 100 mg/24 hour or 48 hour) was used an antiandrogen regardless of route
fOne study did not report number of patients undergoing transdermal therapy, so it was not possible to calculate the total number of patients undergoing transdermal therapy included in the systematic review.
Table 5: Characteristics of Included Primary Studies
Study citation, country, funding source | Study design | Population characteristics and groups | Intervention and comparator(s) | Outcomes, length of follow-up |
|---|---|---|---|---|
Wiepjes et al. (2017)25 Country: Netherlands | Prospective observational | Total: N = 430 Ethnicity (% white): 97.4 FHTa: n = 231 Age (median): Overall: 28 years; IQR 23 to 42. Transdermal and oral: NR. Inclusion criteria: Persons aged 18 years and older who started FHT between 2010 and April 2016 after a confirmed gender dysphoria diagnosis. Patients were recruited from multiple European countries. Exclusion criteria: Individuals who had previously FHT or who had protocol deviations were excluded Previous treatment: NR | Transdermal estradiol patch (n = 99) Dose, range: 50 mcg to 100 mcg twice a week with cyproterone (50 mg to 100 mg daily) Oral estradiol (n = 63) Dose, range: 2 to 4 mg daily with cyproterone (50 mg to 100 mg daily) Duration of treatment: 1 year for both groups | BMD Hormone levelsb Follow-up: 1 year. |
Balcerek et al. (2020)27 Country: Australia Funding: Australian Government National Health, Medical Research Council, Endocrine Society of Australia | Retrospective cohort (authors label as retrospective cross-sectional cohort) | Total: N = 296 (stratified by age group: ≥ 45 years [55 patients on transdermal, oral or combination therapy with estradiol] and < 45 years) Ethnicity: NR FHTa: n = 49 Age (years), median: Overall: 52.8; IQR 49.7 to 58.3 Transdermal: 59.1; IQR, 52.5 to 63.1. Oral: 52.5; IQR, 47.6 to 55.6. Inclusion criteria: Individuals on feminizing hormone therapy for ≥ 6 months Exclusion criteria: NR Previous treatment: NR | Transdermal estradiol (n = 17): Median dose: Patch: 62.5 mcg; IQR 50 to 100; Gel: Median 1 mg; IQR, 1 to 1.25 Duration (months), median: 48 ; IQR 23.8 to 87.7 Oral estradiol (n = 32): Dose, median: 4 mg, IQR 4 to 6; n = 32 Duration (months), median: 78.3; IQR, 52.2 to 168.9 months | BMI Estradiol concentrationsb Sex hormone binding globulin levelsb Prevalence of cardiometabolic risk factors in individuals ≥ 45 years BMI IHD CVD VTE Obesity Dyslipidemia Diabetes Follow-up: NR |
Tebbens et al. (2021)26 Country: The Netherlands Funding: Ghent University | Prospective cohort | Total: N = 212 Ethnicity: NR FHTa: n = 212 Age (years), median: Overall: NR. Transdermal: 43; IQR 27 to 54. Oral: 22; IQR 20 to 26. Inclusion criteria: Patients 18 years or older, diagnosed with gender dysphoria, and planned to start with hormone treatment. Exclusion criteria: Previous or current use of hormone treatment, diversion from the local treatment protocol, and inability to understand the patient information and informed consent Previous treatment: NR | Transdermal estradiol (n = 88): Range: 50 mcg to 100 mcg daily, twice weekly Oral estradiol (n = 124): Range: 2 to 6 mg daily Duration of treatment: 1 year | Body composition Fat percentage Breast volume Hormone concentrationsb Follow-up: 1 year |
BMD = bone mineral density; BMI = body mass index; CVD = cardiovascular disease; FHT = feminizing hormone therapy; IHD = ischemic heart disease; IQR = interquartile range; NR = not reported; VTE = venous thromboembolism.
aPatients receiving transdermal or oral estrogen FHT.
bExcluded from this report.
Table 6: Characteristics of Included Evidence-Based Guidelines
Intended users, target population | Intervention and practice considered | Major outcomes considered | Evidence collection, selection, and synthesis | Evidence quality assessment |
|---|---|---|---|---|
Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society Clinical Practice Guideline (Endocrine Society, US, 2017)28 | ||||
Intended users: Endocrinologists Target population: Persons with gender dysphoria or gender-incongruent persons [wordings from original source] | Intervention: Hormone therapy (oral and transdermal estrogen and testosterone), antiandrogens, puberty suppression, surgery, behavioural and psychological support. | Physical outcomes: Sexual characteristics (breast development, facial hair growth), body composition (muscle mass, fat composition), bone density and overall skeletal health, cardiovascular health. Psychological outcomes: Improvement in gender dysphoria, mental health comes, quality of life, social functioning Reproductive health outcomes: Fertility, sexual and function, reproductive organ function. Adverse effects: Thromboembolism, liver function, surgical complications, metabolic changes. Hormone levels: Estradiol and testosterone levels, adjustments needed to maintain hormone levels. | Conducting systematic reviews, clinical studies, expert consensus. | Graded based on factors like study quality, consistency of results, and the directness of the evidence in addressing the clinical questions. |
Position statement on the hormonal management of adult transgender and gender diverse individuals, Australian Professional Association for Trans Health, Australia, 201929 | ||||
Intended users: General practitioners, physicians, endocrinologists, psychiatrists, and other medical professionals providing care for transgender and gender-diverse individuals in Australia. Target population: Adult transgender and gender-diverse (TGD) individuals older than 18 years | Intervention: Oral and transdermal estradiol and testosterone therapies, antiandrogens, surgical interventions. | Physical outcomes: Changes in secondary sexual characteristics to align with affirmed gender, including body fat redistribution, muscle mass changes, and changes in hair growth. Psychological outcomes: Improvement in psychological functioning, quality of life, and reduced dysphoria, depression, and suicidal ideation. Reproductive health outcomes: Hormonal therapy may impair fertility. Sperm or oocyte cryopreservation should be discussed before starting estradiol or testosterone therapy Adverse effects: Risk of thromboembolism, hypertriglyceridemia, cardiovascular issues (especially with estradiol), polycythemia, acne, and sleep apnea (for testosterone therapy) Hormone levels: Estradiol and testosterone levels. | Based on evidence, published guidelines, expert opinion, and community feedback. Due to the absence of randomized controlled trials in this field, the recommendations were largely informed by low or very low-level evidence and expert consensus. | GRADE framework was used to classify key recommendations. |
Standards of Care for the Health of Transgender and Gender Diverse People, Version 8, World Professional Association for Transgender Health, 20229 | ||||
Intended users: Health care professionals, including multidisciplinary teams of clinicians, medical specialists, and mental health professionals who provide care for transgender and gender-diverse individuals. Target population: Transgender and gender-diverse individuals globally, across diverse cultural and sociopolitical settings, including those who identify as nonbinary or eunuch [wording from original source], intersex individuals, and those in institutional environments | Intervention: Gender-affirming medical and surgical treatments, including hormone therapy, surgery, voice and communication therapy, primary care, mental health services, reproductive and sexual health care, and psychosocial support. | Physical outcomes: Improvement in gender congruence through medical (hormonal and surgical) interventions. Psychological outcomes: Reduction in gender dysphoria and distress; mental health; psychological well-being and self-fulfillment. Reproductive health outcomes: Parenthood and reproductive health (e.g., cryopreservation of gametes). Adverse effects: Risks such as cardiovascular issues or cancer. | Based on systematic literature reviews, expert professional consensus, and data from global health perspectives. | The recommendations were graded based on available evidence, consideration of risks and harms, and feasibility. A consensus was achieved using the Delphi process, which required at least 75% agreement among committee members. |
Guidelines for Gender Affirming Health, Rainbow Health Ontario, Canada, 202330 | ||||
Intended users: Health care providers, especially primary care physicians and clinicians who provide gender-affirming care to transgender and nonbinary patients. Target population: Transgender and nonbinary individuals in need of gender-affirming health care, specifically those seeking primary care, hormone therapy, or gender-affirming surgeries | Intervention: Hormone therapies (feminizing and masculinizing) and surgical interventions for gender-affirming care; testosterone and estrogen therapy, antiandrogens, and progestins; surgeries such as chest reconstruction and genital surgeries. | Physical outcomes: Changes in secondary sex characteristics (e.g., breast development, muscle mass, fat distribution), monitoring hormone levels, and managing risks related to surgeries and hormone therapies. Psychological outcomes: Mental health outcomes, reduction in gender dysphoria, depression, and anxiety; increased quality of life and well-being. Reproductive health outcomes: Fertility preservation, and potential impacts on fertility from hormone treatments. Adverse effects: Cardiovascular issues, venous thromboembolism, liver function concerns, and bone density changes. Hormone levels: Estradiol and testosterone levels. | International standards, expert opinion, and research literature referenced. There was an emphasis on evidence-informed practice, though formal meta-analysis and graded recommendations are not consistently used throughout. | The guidelines do not specifically mention using a formal framework for assessing the quality of evidence. Authors acknowledged that much of the available evidence is limited, with small sample sizes and a reliance on expert consensus and clinical experience. |
GRADE = Grading of Recommendations Assessment, Development, and Evaluation.
Appendix 4: Critical Appraisal of Included Publications
Please note that this appendix has not been copy-edited.
Table 7: Summary of the Critical Appraisal of the Included Systematic Review Using the AMSTAR 2 Tool
Strengths | Limitations |
|---|---|
Miranda et al. (2022)17 | |
A clearly focused research question and PICO criteria defined. Predefined review methods provided in a protocol registered on PROSPERO.a A comprehensive literature search conducted using multiple databases. Reference lists of included studies also searched. Study selection and data extraction conducted in duplicate by 2 reviewers. Reported details about the interventions and comparators in the primary studies. Risk of bias assessed using an appropriate scale for the included studies (Newcastle-Ottawa Scale).a Provided an adequate discussion of the risk of bias and heterogeneity in the studies. Provided adequate information about any conflicts of interest for the authors of the review, and the funding sources. | Designs of primary studies included not justified. Excluded studies not listed.a Reported limited information about the included populations. Did not report funding information for the included studies. Outcomes of transdermal and intramuscular FHT combined. Outcomes were too heterogeneous so a meta-analysis could not be performed. Therefore, impact of the risk of bias on the meta-analysis and publication bias could not be assessed. |
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; FHT = feminizing hormone therapy; PICO = population, intervention, comparator, outcomes; PROSPERO = International Prospective Register of Systematic Reviews.
aDesignated as a critical item.
Table 8: Summary of the Critical Appraisal of Included Primary Studies Using the ROBINS-I Tool
Strengths | Limitations |
|---|---|
Wiepjes et al. (2017)25 | |
The study had a relatively large sample size of 430 patients from multiple centres. The study’s prospective design with predefined protocols allowed for systematic data collection and reduced potential biases. Controlled for age, vitamin D supplementation, body weight, smoking, and alcohol use. Low patient attrition (8.5% loss to follow-up) enhances the reliability of the findings and minimizes attrition bias. | The study lacked information on key confounding factors such, which could influence BMD outcomes. Potential unmeasured lifestyle factors, such as adherence to interventions or unreported habits, may have influenced the results. No investigation into long-term risks such as cardiovascular risks and metabolic risks, adverse effects, or patients stopping treatment due to those adverse effects, limiting the scope of reported results |
Balcerek et al. (2021)27 | |
The study effectively stratified patient s by age (≥ 45 vs. < 45 years) and hormone administration route (oral vs. transdermal), facilitating meaningful subgroup analyses. The findings highlighted key trends in cardiovascular risk factors, which are highly relevant for clinical care in this population. | Only 49 patients were included in the comparison of transdermal and oral estrogen therapy. The retrospective cohort design may have increased the risk of confounding biases. Reliance on retrospective clinical records introduced potential for inconsistencies or inaccuracies, particularly regarding adherence to therapies or detailed patient histories. Only 1 efficacy outcome was reported. The study did not control for any confounding factors. Data collected from only 2 clinics may limit generalizability to broader transgender populations. No investigation into long-term risks such as cardiovascular risks and metabolic risks, adverse effects, or patients stopping treatment due to those adverse effects, limiting the scope of reported results Not enough patient information to assess if any data were missing |
Tebbens et al. (2021)26 | |
The study described consistent treatment protocols reduced variability with a clear inclusion criterion and the study adjusted for key confounders (age, BMI, estradiol concentrations). Prospective cohort design may have reduced retrospective selection bias. The study described well-documented treatment regimens and had a relatively large sample size followed up for 12 months. Prespecified objective tools were used for outcome measurement (3D imaging for breast volume, bioelectrical impedance for fat percentage). | The study has a high probability of age bias because of patients receiving transdermal estrogen being significantly older than those receiving oral estrogen. The cohort was limited to a cohort from 1 city, reducing population diversity and external validity. Although the outcomes were adjusted for age, some key confounders such as cardiovascular risks, baseline hormone levels, and others such as smoking may have been omitted. No investigation into long-term risks such, adverse effects, or patients stopping treatment due to those adverse effects, limiting the scope of reported results. Not enough patient information to assess if any data were missing |
BMD = bone mineral density; BMI = body mass index; ROBINS-I = Risk Of Bias In Non-randomized Studies of Interventions; vs. = versus.
Table 9: Summary of the Critical Appraisal of Included Evidence-Based Guidelines Using the AGREE II Tool
AGREE II Item | Endocrine Society, 201728 | AusPATH, 201929 | WPATH, 20229 | Rainbow Health Ontario, 202330 |
|---|---|---|---|---|
Domain 3: Rigour of development | ||||
7. Systematic methods were used to search for evidence. | Systematic review mentioned but no other details given. p. 3873 Score: 3 | No systematic review conducted but uses evidence from other reviews and other observational studies. Score: 2 | Good systematic review methodology used. p. S247 Score: 7 | No systematic review conducted but uses evidence from other reviews and other observational studies. Score: 2 |
8. The criteria for selecting the evidence are clearly described. | PICO not clearly defined. Score 1 | PICO not mentioned at all. Score 1 | PICO used but not specifically mentioned. p. S249 Score: 6 | No PICO info provided Score: 1 |
9. The strengths and limitations of the body of evidence are clearly described. | Evidence graded using authors’ own scale. Fulfills most criteria for this section. Score: 5 | GRADE used for recommendations. p. 127 Score: 7 | GRADE used for recommendations. p. S250 Score: 7 | No strengths or limitations stated. Score: 1 |
10. The methods for formulating the recommendations are clearly described. | Details not provided Score: 1 | Have some indications about how the recommendations were developed but not in detail. p. 127. Score 3 | Adequately described. Score 7 | No detail Score: 1 |
11. The health benefits, side effects, and risks have been considered in formulating the recommendations. | Adequately described. Score 7 | Adequately described. Score 7 | Adequately described. Score 7 | Adequately described. Score 7 |
12. There is an explicit link between the recommendations and the supporting evidence. | Adequately described. Score 7 | Not specifically discussed but implied in some areas. Score 1 | Adequately described. Score: 7 | Not specifically discussed. Score 1 |
13. The guideline has been externally reviewed by experts before its publication. | Externally reviewed by experts Score: 7 | Guideline reviewed by experts. Score 7. p. 127. | Guideline reviewed by experts. Page: 250 Score 7 | Suggest that guidelines were developed with experts, but no other details given. Score: 5 |
14. A procedure for updating the guideline is provided. | No update information given. Score: 1 | No update information given. Score: 1 | Update details given. p. S251. Score 7 | Update details given. Page: 8. Score: 7 |
Domain 6: Editorial independence | ||||
22. The views of the funding body have not influenced the content of the guideline. | funding for the guideline was provided by the Endocrine Society, and it explicitly states that no funding or remuneration was received from commercial or other entities p. 3873. Score: 7 | Supported by the Australian Government National Health and Medical Research Council through an Early Career Fellowship awarded to Dr. Ada Cheung. Additional research support came from the Viertel Charitable Foundation and the Endocrine Society of Australia. p. 132 Score: 7 | Funding details well-documented. Not likely to influence guideline content. p. 177. Score: 7 | Funded by the province of Ontario. Page 5. Score: 7 |
23. Competing interests of guideline development group members have been recorded and addressed. | “All task force members were required to declare any potential conflicts of interest, which were reviewed and managed accordingly by the Clinical Guidelines Subcommittee.” p. 3873 Score: 7 | States ‘no relevant disclosures,’ but no other info given. p. 132. Score: 5 | No COI reported. Went through a review process. Score: 7 | No sufficient information provided. p. S247 Score: 1 |
Summary of strengths and limitations | ||||
Strengths discussed in the evidence review report | Not discussed. | Not discussed. | Not discussed. | Not discussed. |
Limitations discussed in the evidence review report | ‘Our knowledge concerning the natural history and effects of different cross-sex hormone therapies on breast development in transgender females is extremely sparse and based on the low quality of evidence.’ p. 3887. | ‘Recommendations were based on low or very low level evidence and expert opinion.’ p. 127. | ‘Many of these studies had significant limitations, such as variably adjusting for CV-related risk factors, small sample sizes—especially involving older transgender womena—and variable duration and types of GAHT.’ p. S147. | ‘Any guidelines in this area of practice—these guidelines notwithstanding—are also limited by the quality of available research on trans health.’ p. 7. |
AGREE II = Appraisal of Guidelines for Research and Evaluation II; AusPATH = Australian Professional Association for Trans Health; COI = conflict of interest; CV = cardiovascular; GAHT = gender-affirming hormone therapy; PICO = population, intervention, comparator, outcomes; WPATH = World Professional Association for Trans Health.
aThe term “women” is used by the study to define the population, and the extracted text is verbatim.
Appendix 5: Main Study Findings
Table 10: Outcomes Reported in the Systematic Review
Study citation, country, funding source | Description | Findings | Author’s conclusions |
|---|---|---|---|
Cardiovascular risk factorsa | |||
Miranda et al. (2022)17 Country: Canada Funding source: University of Alberta, CIHR | Included studies: Five primary studies (3 prospective studies, 1 retrospective study and 1 cross-sectional study) included. In 4 studies, transdermal FHT was compared with oral FHT, and in 1 study intramuscular FHT was compared with oral FHT. Outcomes included: Cardiovascular risk factors (blood pressure, triglycerides, total cholesterol, body mass index, low- and high-density lipid levels). Follow-up: Baseline to 12 months | BMI: Giltay et al. (2000): Baseline to 4 months Transdermal FHT, mean (SD): 20.9 (2.6) kg/m2 to 21.9 (2.5) kg/m2 Oral FHT, mean (SD): 22.8 (2.7) kg/m2 to 23.2 (1.8) kg/m2 Wierckx et al. (2014): Baseline to 12 months Transdermal FHT, mean (SD): 26.1 (3.5) kg/m2 to 26.1 (3.4) kg/m2 Oral FHT, mean (SD): 23.1 (4.2) kg/m2 to 23.7 (4.4) kg/m2 SBP: Wierckx et al. (2014): Baseline to 12 months Transdermal FHT, mean (SD): 131.6 (15.8) mm Hg to 128.8 (15.6) mm Hg Oral FHT, mean (SD): 125.1 (13.8) mm Hg to 118.8 (13.9) mm Hg DBP: Wierckx et al. (2014): Baseline to 12 months Transdermal FHT, mean (SD): 76.7 (9) mm Hg to 79.7 (9.3) mm Hg Oral FHT, mean (SD): 76.8 (10.8) mm Hg to 75.7 (10.6) mm Hg TG: Wierckx et al. (2014): Baseline to 12 months Transdermal FHT, median: 87; IQR, 68 to 176.5 to mg/dL to 85; IQR, 70 to 110 mg/dL Oral FHT, median: 79.5; IQR, 54.7 to 100.8 mg/dL to 70.8; IQR, 50 to 133.1 mg/dL Wilson et al. (2009): Baseline to 6 months Transdermal FHT, mean (SD): 2.0 (1.2) mmol/L to 3.3 (0.7) mmol/L Oral FHT, mean (SD): 1.3 (0.8) mmol/L to 2.0 (0.7) mmol/L Giltay et al. (2000): Baseline to 4 months Transdermal FHT, median: 0.87; IQR, 0.68 to 1.11 mmol/L to 0.76; IQR, 0.95 to 1.21 mmol/L Oral FHT, median: 1.17; IQR, 0.85 to 1.6 mmol/L to 1.57; IQR, 1.35 to 1.81) mmol/L Ott et al. (2011): Baseline to 60 months Transdermal FHT, mean (SD): 110.8 (57.3) mg/dL to 122.6 (63.6) mg/dL Oral FHT, mean (SD): 105.9 (83.4) mg/dL to 169.6 (109.9) mg/dL TC: Wilson et al. (2009): Baseline to 6 months Transdermal FHT, mean (SD): 5.3 (0.7) mmol/L to 5.2 (0.9) mmol/L Oral FHT, mean (SD): 4.7 (0.8) mmol/L to 5.7 (0.6) mmol/L Wierckx et al. (2014): Baseline to 12 months Transdermal FHT, mean (SD): 227.2 (35.6) mg/dL to 181.3 (20.6) mg/dL Oral FHT, mean (SD): 171.5 (32.7) mg/dL to 152.3 (28.3) mg/dL Ott et al. (2011): Baseline to 60 months Transdermal FHT, mean (SD): 196 (42.1) mg/dL to 198.6 (45.9) mg/dL Oral, mean (SD): 178 (48.5) mg/dL to 193.5 (42.3) mg/dL LDL: Wierckx et al. (2014): Baseline to 12 months Transdermal FHT, mean (SD): 138.3 (20.4) mg/dL to 106.3 (20.4) mg/dL Oral FHT, mean (SD): 99.4 (29) mg/dL to 92.3 (28.9) mg/dL Ott et al. (2011): Baseline to 60 months Transdermal FHT, mean (SD): 118.3 (34.2) mg/dL to 120.6 (35.2) mg/dL Oral FHT, mean (SD): 103.9 (33) mg/dL to 113.9 (36.1) mg/dL HDL: Giltay et al. (2000): Baseline to 4 months Transdermal FHT, median: 1.20; IQR, 1.05 to 1.37 mmol/L to 1.08; IQR, 0.95 to 1.21 mmol/L Oral FHT, median: 1.09; IQR, 1.00 to 1.19 mmol/L to 1.22; IQR, 1.11 to 1.34 mmol/L Wilson et al. (2009): Baseline to 6 months Transdermal FHT, mean (SD): 1.2 (0.3) mmol/L to 1.3 (0.4) mmol/L Oral FHT, mean (SD): 1.2 (0.5) mmol/L to 1.0 (0.3) mmol/L Wierckx et al. (2014): Baseline to 12 months Transdermal FHT, mean (SD): 58.2 (15.2) mg/dL to 54.8 (15.7) mg/dL Oral FHT, mean (SD): 52.9 (13.5) mg/dL to 45.7 (9.2) mg/dL Ott et al. (2011): Baseline to 60 months Transdermal FHT, mean (SD): 53.1 (15.9) mg/dL to 58.4 (19.2) mg/dL (0 to 60 months). Oral FHT, mean (SD): 53.4 (12.5) mg/dL to 54.8 (16.7) mg/dL | Safety ‘Heterogeneity of studies in reporting gender-affirming estrogen therapy formulation, dose, and duration of exposure limits quantification of the effect of gender-affirming estrogen therapy on all-cause and cardiovascular mortality, adverse cardiovascular events, and cardiovascular risk factors.’ ‘None of the studies reported data on all-cause and cardiovascular mortality.’ Risk of bias “Most studies were of moderate methodological quality. The studies rated high for selection of exposed and nonexposed, representativeness of sample, and ascertainments of exposure. However, no data were provided by any study to confirm that the outcome of interest was absent at the beginning of the study. None of the studies accounted for possible confounders or effect modifiers and were therefore not given any points for comparability.” |
BMI = body mass index; CIHR = Canadian Institutes of Health Research; DBP = diastolic blood pressure; FHT = feminizing hormone therapy; HDL = high-density lipoprotein; IQR = interquartile range; LDL = low-density lipoprotein, SBP = systolic blood pressure; TC = total cholesterol. TG = triglyceride.
aThe review did not extract any statistically significant differences between transdermal and oral FHT from the primary studies and did not present any overall conclusions in the context of transdermal versus oral FHT. Only outcomes across individual studies were provided.
This table has not been copy-edited.
Table 11: Outcomes Reported in the Primary Studies
Outcome | Wiepjes et al. 2017a,25 | Balcerek et a. 2021b,27 | Tebbens et al. 2021c,26 |
|---|---|---|---|
Bone-related outcomes | |||
BMD (% change) | Transdermal estradiol, (n = 63): Lumbar spine, mean: + 4.00; 95% CI 2.91 to 4.87 Total hip, mean: + 1.23; 95% CI 0.53 to 2.00 Femoral neck, mean: + 2.3; 95% CI, 1.3 to 3.2 Oral estradiol valerate (n = 99): Lumbar spine, mean: + 4.0; 95% CI 3.28 to 4.55 Total hip, mean: + 1.36; 95% CI 0.90 to 1.9 Femoral neck, mean: + 2.0; 95% CI 1.3 to 2.7 ‘LS, TH, or FN BMD change did not differ between transdermal estradiol or oral estradiol valerate use in transwomen.’ Duration of treatment: 1 year | NR | NR |
BMI | |||
BMI (kg/m2) | NR | Transdermal estradiol, median (n = 17) 29.6; IQR, 24.8 to 34.1 Oral estradiol, median (n = 32): 27.7; IQR, 25.5 to 29.4 P = 0.358 Durations, median: 48 months; IQR, 23.8 to 87.7 (transdermal); Median 78.3 months; IQR, 52.2 to 168.9 (oral) | NR |
Feminizing effects | |||
Breast development (% change)d | NR | NR | Transdermal estradiol (n = 88): NR Oral estradiol valerate (n = 124): NR Duration of treatment: 1 year Mean difference: −33; 95% CI, −85 to 209f No difference between transdermal and oral estradiol. |
Change in Fat (%)e | NR | NR | Transdermal estradiol (n = 88): NR Oral estradiol valerate (n = 124): NR Duration of treatment: 1 year Mean difference: 1.1; 95% CI, −13 to 18f No difference between transdermal and oral estradiol. |
Cardiovascular and metabolic risk factors (n, %) | |||
Number of patients with dyslipidemia | NR | Transdermal estradiol: 8 of 17 (47%) Oral estradiol: 1 of 32 (3%) P = 0.0004 | NR |
Number of patients with diabetes | NR | Transdermal estradiol: 3 of 17 (18%) Oral estradiol: 2 of 32 (6%) P = 0.37 | NR |
Number of patients with hypertension | NR | Transdermal estradiol: 6 of 17 (35%) Oral estradiol: 8 of 32 (25%) P = 0.68 | NR |
Obesity (number of patients with BMI > 30 kg/m2) | NR | Transdermal estradiol: 4 of 17 (24%) Oral estradiol: 6 of 32 (19%) P = 0.89 | NR |
Number of patients with IHD | NR | Transdermal estradiol: 2 of 17 (12%) Oral estradiol: 1 of 32 (3%) P = 0.49 | NR |
Number of patients with CVD | NR | Transdermal estradiol: 1 of 17 (6%) Oral estradiol: 1 of 32 (2%) P = 0.99 | NR |
Number of patients with VTE | NR | Transdermal estradiol: 1 of 17 (6%) Oral estradiol: 0 of 32 (0%) P = 0.42 | NR |
Patients discontinuing drugs | NR | NR | NR |
Patients with AEs leading to discontinuation | NR | NR | NR |
Patients with any AE | NR | NR | NR |
Patients with AE ≥ grade 3 | NR | NR | NR |
Patients with any SAE | NR | NR | NR |
AE = adverse event; BMD = bone mineral density; BMI = body mass index; CVD = cardiovascular disease; FN = femoral neck; IHD = ischemic heart disease; IQR = interquartile range; LS = lumbar spine; NR = not reported; SAE = severe adverse event; TH = total hip; VTE = venous thromboembolism.
aAdjusted for age, vitamin D supplementation, and alcohol use.
bUnadjusted outcomes.
cChange in fat adjusted for age and BMI; breast development adjusted for age, BMI, and blood estradiol concentration. Individual changes in treatment groups were not reported in this study.
dMeasured using bioelectrical impedance analysis.
eMeasured using 3-dimensional imaging.
fMean difference between oral and transdermal estradiol users.
This table has not been copy-edited.
Table 12: Description of the Recommendations and Their Supporting Evidence for Gender-Affirming Hormone Replacement Therapy Using Transdermal Routes From Evidence-Based Guidelines
Recommendations and supporting evidence | Strength of recommendations and quality of evidence |
|---|---|
Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society Clinical Practice Guideline (Endocrine Society, 2017)28 | |
Indication: Transdermal estradiol is recommended as a safer alternative to oral estrogen, especially for individuals at higher risk of venous thromboembolism, cardiovascular events, or those with liver conditions. Dose recommendations: Estradiol transdermal patch – 0.025 mg to 0.2 mg per day. A new patch is typically placed every 3 to 5 days. Evidence: Long-term cohort studies, literature reviews | Strength: Strong Quality: Moderate |
Position statement on the hormonal management of adult transgender and gender diverse individuals, Australian Professional Association for Trans Health, Australia, 201929 | |
Indication: Transdermal estradiol is recommended over oral for individuals at higher risk of venous thromboembolism, particularly smokers, older individuals (older than 40 years of age), and those with obesity because it has a lower thrombotic risk than oral estradiol formulations. Dose recommendations: Transdermal estradiol patches — 100 mcg to 150 mcg for 24 hours, changed twice weekly. Evidence: Nonrandomized clinical studies and expert opinion. | Strength: Weak Quality: Low |
Standards of Care for the Health of Transgender and Gender Diverse People, Version 8, World Professional Association for Transgender Health, 20229 | |
Indication: Transdermal estradiol is strongly recommended over oral for patients older than 45 or those with a history of venous thromboembolism. Dose recommendations: Estradiol therapy — Initial dose (for induction of female puberty): 6.25 mcg to 12.5 mcg/24 hours by cutting a 24 g patch into one-fourth or half. Titration: Increase every 6 months by 12.5 mcg/24 hours based on estradiol levels. Adult dose: 50 mcg to 200 mcg/24 hours. Evidence: Nonrandomized clinical studies. | Strength: Strong Quality: Low |
Guidelines for Gender Affirming Health, Rainbow Health Ontario, Canada, 202330 | |
Indication: Transdermal estradiol is recommended over oral forms, especially for transgender individuals requiring FHT for feminization who are 40 years or older, or those with cardiovascular or thromboembolic risk factors. Dose recommendations: Estradiol patch: Starting dose is 50 mcg applied daily or twice weekly (maximum dose is 200 mcg daily), applied twice weekly. Estradiol gel: Starting dose is 2.5 g daily (containing 150 mcg estradiol). The maximum dose is 6.25 g daily (containing 375 mcg estradiol), although larger doses may be limited by the skin area available for application. Titration recommended based on patient response. Evidence: Observational studies on menopausal cisgender individuals identifying as women and transgender individuals receiving transdermal FHT | Strength: Strong Quality: Moderate |
FHT = feminization hormone therapy; VTE = venous thromboembolism.
This table has not been copy-edited.
Appendix 6: References of Potential Interest but Not Meeting the Inclusion Criteria
Please note that this appendix has not been copy-edited.
Primary Studies
Irrelevant Study Design
Lim HY, Leemaqz SY, Torkamani N, et al. Global Coagulation Assays in Transgender Women on Oral and Transdermal Estradiol Therapy. J Clin Endocrinol Metab. Jul 1 2020;105(7)doi:10.1210/clinem/dgaa262 PubMed
Outcomes Out of Scope
Chantrapanichkul P, Ingsathit A, Chotai P, et al. Serum hormone concentrations in transgender individuals receiving gender-affirming hormone therapy: a longitudinal retrospective cohort study. Endocr Pract. 2021;27(1):27-33. PubMed
Salakphet T, Yodkoet P, Wattanayingcharoenchai R, et al. Hormone concentrations in transgender women who self-prescribe gender-affirming hormone therapy: a retrospective study. J Sex Med. 2022;19(5):864-871. PubMed
Sumerwell C, Johnson M, Lee A, et al. Serum hormone concentrations in transgender youth receiving estradiol. Endocr Pract. 2024;30(2):155-159. PubMed
Kariyawasam NM, Wylie K, Sumerwell C, et al. Comparison of estrone/estradiol ratio and levels in transfeminine individuals on different routes of estradiol. Transgender Health. 2024. In press. Available from: https://doi.org/10.1089/trgh.2023.0138
Guidelines
Not Evidence-Based
Oliphant J, Veale J, Macdonald J, et al. Guidelines for Gender Affirming Healthcare for Gender Diverse and Transgender Children, Young People and Adults in Aotearoa New Zealand. Transgender Health Research Lab; 2018.
ISSN: 2563-6596
This work was conducted by the POst-market Drug Evaluation Team (PODET) through the Post-Market Drug Evaluation CoLab Network. It was supported by Canada’s Drug Agency (CDA-AMC) and its Post-Market Drug Evaluation Program through funding provided by Health Canada.
CDA-AMC is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
CoLab is a pan-Canadian network of experts in applied research, scientific methods, and data analysis. CoLab members work with the Post-Market Drug Evaluation Program to produce credible and timely evidence on postmarket drug safety and effectiveness.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
This document is the property of PODET. CDA-AMC has a nonexclusive, limited, royalty-free, worldwide, nontransferable, fully paid-up, and irrevocable license to use the report in support of its objects, mission, and reasonable operational requirements.