Drugs, Health Technologies, Health Systems
Reimbursement Review
Clindamycin Plus Benzoyl Peroxide and Adapalene (Cabtreo)
Sponsor: Bausch Health, Canada Inc.
Therapeutic area: Acne vulgaris
This multi-part report includes:
Clinical Review
Pharmacoeconomic eview
Clinical Review
Abbreviations
Acne-QoL
acne-specific quality of life
AE
adverse event
ANCOVA
analysis of covariance
ARSC
Acne and Rosacea Society of Canada
BAM
bias adjustment model
BPO
benzoyl peroxide
CI
confidence interval
CrI
credible interval
CSPA
Canadian Skin Patient Alliance
DIC
deviance information criterion
EGSS
Evaluator’s Global Severity Score
GRADE
Grading of Recommendations Assessment, Development and Evaluation
HRQoL
health-related quality of life
IDP-126 gel
clindamycin 1.2% plus BPO 3.1% and adapalene 0.15% topical gel
IGA
Investigator’s Global Assessment
ISGA
Investigator’s Static Global Assessment
ITC
indirect treatment comparison
ITT
intention to treat
MCMC
Markov chain Monte Carlo
MD
mean difference
MID
minimal important difference
NMA
network meta-analysis
OR
odds ratio
PP
per protocol
RCT
randomized controlled trial
SAE
serious adverse event
SD
standard deviation
TEAE
treatment-emergent adverse event
TSS
Truncal Severity Score
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Table 1: Background Information for the Application Submitted for Review
Item | Description |
|---|---|
Drug product | Clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel (Cabtreo) |
Sponsor | Bausch Health, Canada Inc. |
Indication | For the topical treatment of acne vulgaris in patients 12 years of age and older |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Standard review |
NOC date | August 14, 2024 |
Recommended dose | Apply a thin layer to the affected area once daily |
NOC = Notice of Compliance.
Introduction
Acne vulgaris (hereafter referred to as acne) is a chronic inflammatory skin condition of the pilosebaceous glands that typically begins at puberty and may continue through adulthood with flares that are associated with an increase in androgen levels.1 Acne is characterized by noninflammatory lesions (open or closed comedones) and inflammatory lesions (papules, pustules, and nodules) that develop primarily on the face, neck, upper back, and chest.2,3 Acne affects 5.6 million individuals living in Canada, nearly 20% of the population.4
According to the 2016 Canadian clinical practice guideline in the management of acne,5 topical therapies, including topical retinoids, benzoyl peroxide (BPO), and fixed-dose combinations of retinoids with BPO or clindamycin are a reasonable first-line treatment option for comedonal and mild papulopustular acne. For more extensive moderate papulopustular acne, or acne in areas not amenable to topical therapy, systemic therapies, including oral antibiotics and combined oral contraceptives in female patients, in addition to topical therapies, are a reasonable treatment option.5 For severe acne, systemic therapies, including oral isotretinoin and oral antibiotics in combination with BPO, with or without topical retinoids, are a reasonable treatment option.5
Patient group input provided for this review identified an unmet need for early, effective treatment to prevent acne scarring and hyperpigmentation. The clinical expert identified the following challenges in the treatment of patients with acne: not all patients’ acne responds to current topical therapies and adherence to acne therapies is generally low, often due to associated side effects (particularly with retinoids), treatment modality, and complicated treatment regimens (i.e., requiring ≥ 2 different products with the potential for active ingredient cross-reactivity and reduced efficacy).
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects of clindamycin 1.2% plus BPO 3.1% and adapalene 0.15% topical gel (hereafter referred to as IDP-126 gel) in the treatment of acne in patients aged 12 years and older. Of note, this Reimbursement Review was conducted before the Health Canada Notice of Compliance was issued; the scope of this review was based on the anticipated indication.
Perspectives of Patients, Clinicians, and Drug Programs
The information in this section is a summary of input provided by the patient groups that responded to the call for input and by the clinical expert consulted for the purpose of this review.
Patient Input
The Acne and Rosacea Society of Canada (ARSC) and the Canadian Skin Patient Alliance (CSPA) submitted a joint input on the current review of IDP-126 gel. Patient input was gathered using an online survey that was conducted from June 7 to 30, 2022. A total of 154 responses were collected from patients with acne (either diagnosed by a dermatologist or other health care provider or self-diagnosed) and their caregivers living in Canada. Most respondents (68%) were female, 30% were male, and 2% identified as nonbinary. The distribution of respondents by age range was 20 to 29 years (55% of respondents), 30 to 39 years (23%), and 16 to 19 years (12%). Additionally, CSPA and ARSC created a survey targeting participants in clinical trials of IDP-126 gel and received a total of 3 responses.
Almost half (47%) of the survey respondents reported moderate acne, while 16% reported severe acne. Almost half (42%) of the survey respondents indicated they had undergone 2 to 5 health care visits before receiving their diagnosis and treatment for acne. Nearly 30% of survey respondents reported visiting a health care provider more than 5 times. Almost half of survey respondents reported feeling self-conscious either often or always due to acne. Most respondents (87%) reported using a strategy to hide their acne, with 63% using makeup and 59% avoiding social gatherings altogether. Most respondents reported acne scarring (87%) and changes in skin pigmentation due to acne (90%).
Overall, survey respondents reported experience with various treatments with varying degrees of improvement (or sometimes worsening) in their acne and experience with associated side effects. The majority of respondents (89%) reported prior use of prescription gels or creams for acne. Of these, 21% reported no change in their condition, 43% reported a little improvement, 13% reported a big improvement, and 12% reported a worsening of their condition. Most survey respondents (59%) reported experience with isotretinoin therapy, of which 28% reported significant improvement and 43% reported slight improvement. Similarly, 59% of respondents reported experience with hormone therapy, including birth control and spironolactone, of which 23% reported significant improvement and 36% reported minor improvement. Most respondents (95%) reported experiencing adverse effects associated with their treatment regimen for acne in the last year, with the most common adverse effects being skin irritation (64%), dry skin (62%), and skin flaking (55%). More specifically, 85% of respondents reported experiencing side effects associated with their current topical treatment regimen (nonprescription and prescription); most side effects (70%) were reported as minor. Most survey respondents indicated they were willing to accept these side effects because they thought the treatment was effective. Additionally, facials and peels were used by more than half of all respondents (53%), while 65% reported undergoing light or laser therapy.
All 3 patients with experience with IDP-126 gel reported manageable side effects. Two of the 3 patient respondents indicated treatment with IDP-126 gel was easier to use versus their previous therapies. Further, 2 of the 3 patient respondents indicated the value of IDP-126 gel is treatment effectiveness and time to improvement.
According to the survey respondents, common challenges in the management of acne include hiding their acne, identifying triggers, and coping with high out-of-pocket expenses on nonprescription acne products and treatments. Survey respondents identified the following goals for improved outcomes: ability to enjoy personal relationships, to have less scarring, and to have fewer changes in skin pigmentation. Other goals include clearer skin, improved mental health, increased self-confidence, and improved overall daily life.
Clinician Input
Input From Clinical Expert Consulted
The clinical expert stated they expect that a triple therapy, such as IDP-126 gel, would become widely adopted as first-line therapy in the treatment of acne. The clinical expert anticipated that the drug under review could cause a shift in the current treatment paradigm away from topical (dual) combination therapies (e.g., adapalene and BPO topical gel, clindamycin phosphate and tretinoin gel) that have been widely adopted in clinical practice. The clinical expert does not expect IDP-126 gel to be used in combination with other therapies. The clinical expert advised that patients with acne should not be required to try other therapies before initiating treatment with IDP-126 gel due to its overlap with the other therapies currently available and due to patients becoming exhausted and frustrated with topical products. More specifically, the clinical expert indicated that if there were a requirement for prior treatment failure with other topical therapies before being able to access IDP-126 gel, patients would be more likely to request a step up to oral therapies.
The clinical expert suggested that patients with moderate acne (i.e., numerous inflammatory papules and noninflammatory lesions) are most likely to respond to treatment with IDP-126 gel. In contrast, the clinical expert suggested that patients with nodulocystic acne or severe acne and patients with scarring are less likely to respond to treatment with IDP-126 gel (nodulocystic acne, a severe form of acne, tends to require systemic medication). As such, the clinical expert would likely use IDP-126 gel only in patients who do not have nodules or cysts.
The clinical expert indicated that family physicians, nurse practitioners, and other primary care providers can prescribe and monitor treatment with IDP-126 gel in any setting. In the assessment of treatment response in clinical practice, the clinical expert indicated that, in general, clinicians estimate (i.e., do not count) the number of inflammatory and noninflammatory lesions and conduct a global assessment (no acne; mild, moderate, or severe acne). The clinical expert indicated that patients using topical therapies are generally reassessed every 3 to 6 months. The clinical expert indicated the following would be considerations for discontinuation of IDP-126 gel: lack of response or minimal response (i.e., no improvement or minimal improvement from baseline as determined by examining the patient), severity of side effects, and disease progression (i.e., increase in the number of nodules or scar formation).
Clinician Group Input
No input from clinician groups was submitted for the present review of IDP-126 gel.
Drug Program Input
Input was obtained from the drug programs that participate in the Reimbursement Review process. The following items were identified as key factors that could potentially impact the implementation of a recommendation for IDP-126 gel: relevant comparators, considerations for the initiation of therapy, and considerations for the discontinuation of therapy. The clinical expert consulted provided advice on the potential implementation issues raised by the drug programs (Table 4).
Clinical Evidence
Systematic Review
Description of Studies
Two phase III, multicentre, double-blind, randomized controlled trials (RCTs) (Study 301, N = 183; Study 302, N = 180) assessed whether there is a difference in the proportion of patients with treatment success (defined by ≥ 2-grade reduction from baseline in the Evaluator’s Global Severity Score [EGSS] and an EGSS of clear or almost clear) and change from baseline in inflammatory and noninflammatory lesion counts in patients aged 9 years and older with moderate to severe acne applying IDP-126 topical gel once daily for 12 weeks when compared with its vehicle gel. Other outcomes of interest include change in health-related quality of life (HRQoL) measured by the self-perception and symptom subscales of the Acne-Specific Quality of Life (Acne-QoL) questionnaire. Notable harms include general disorders and administration site conditions, skin and subcutaneous tissue disorders, and serious adverse events (SAEs). The mean age of patients randomized to each study drug group was similar, approximately 20 years, and ranged from 10 to 48 years across studies. The majority of patients in each study drug group had moderate acne, defined as a baseline EGSS of 3, ranging from 87.7% to 95.1% of patients across studies. The remainder of patients in each study drug group had severe acne, defined as a baseline EGSS of 4, ranging from 4.9% to 12.3% of patients across studies.
Efficacy Results
Treatment Success Based on the EGSS
Study 301: The treatment difference in treatment success based on the EGSS at week 12 between IDP-126 gel and its vehicle gel was 24.7% (95% confidence interval [CI], 10.7% to 38.7%; P value = 0.003) in favour of IDP-126 gel.
Study 302: The treatment difference in treatment success based on the EGSS at week 12 between IDP-126 gel and its vehicle gel was 30.0% (95% CI, 16.4% to 43.6%; P value = 0.001), also in favour of IDP-126 gel.
Inflammatory Lesion Count
Study 301: The treatment difference in the mean absolute change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −5.94 (95% CI, −8.73 to −3.14; P value < 0.001) in favour of IDP-126 gel. The treatment difference in the mean percent change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −16.08% (95% CI, −23.72% to −8.44%; P value < 0.001), also in favour of IDP-126 gel.
Study 302: The treatment difference in the mean absolute change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −9.30 (95% CI, −12.38 to −6.23; P value < 0.001), also in favour of IDP-126 gel. The treatment difference in the mean percent change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −23.95% (95% CI, −31.73% to −16.16%; P value < 0.001), also in favour of IDP-126 gel.
Noninflammatory Lesion Count
Study 301: The treatment difference in the mean absolute change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −11.85 (95% CI, −16.56 to −7.14; P value < 0.001), in favour of IDP-126 gel. The treatment difference in the mean percent change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −25.09% (95% CI, −34.96% to −15.22%; P value < 0.001), also in favour of IPD-126 gel.
Study 302: The treatment difference in the mean absolute change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −13.27 (95% CI, −17.74 to −8.80; P value < 0.001), also in favour of IDP-126 gel. The treatment difference in the mean percent change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −24.27% (95% CI, −32.86% to −15.68%; P value < 0.001), also in favour of IPD-126 gel.
Self-Perception and Symptom Domain Score on the Acne-QoL Questionnaire
█████ ███ █ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ █████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ███████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ ███████████ ███ █ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ █████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ █████
Harms Results
The following summary of harms results from studies 301 and 302 are based on pooled data. There were no reports of patients with SAEs and no reports of deaths in either study.
Adverse Events
The proportion of patients with at least 1 treatment-emergent adverse event (TEAE) was 21.9% of patients (53 of 242 patients) in the IDP-126 gel group and 7.4% of patients (9 of 121 patients) in its vehicle gel group. The most common TEAE reported was application site pain in 9.1% of patients (22 of 242 patients) in the IDP-126 gel group and 0.8% of patients (1 of 121 patients) in its vehicle gel group.
Withdrawal Due to Adverse Events
The proportion of patients who stopped their study drug and/or withdrew from the study due to any TEAE was 2.9% of patients (7 of 242 patients) in the IDP-126 gel group and no patients in its vehicle gel group. The most common TEAEs reported to have led to discontinuation of the study drug and/or from the study was application site pain and erythema; each TEAE was reported in 0.8% of patients (2 of 242 patients) in the IDP-126 gel group.
Notable Harms
A total of 9.1% of patients (22 of 242 patients) in the IDP-126 gel group and 0.8% of patients (1 of 121 patients) in its vehicle gel group were reported with a TEAE categorized as a general disorder and administrative site condition.
A total of 2.9% of patients (7 of 242 patients) in the IDP-126 gel group and 0.8% of patients (1 of 121 patients) in its vehicle gel group were reported with a TEAE categorized as a skin and subcutaneous tissue disorder.
Critical Appraisal
Internal Validity
Studies 301 and 302 were generally appropriately designed and powered to evaluate the efficacy of IDP-126 gel relative to vehicle gel. Type I error was controlled in each study by requiring all 3 coprimary efficacy end points to be statistically significant to be able to draw a conclusion of superiority for IDP-126 gel relative to its vehicle gel and by testing the secondary efficacy end points using a gated, sequential process. No inferential statistics were conducted in the subgroup analyses and HRQoL outcomes; therefore, these results are considered as supportive evidence only.
The 2018 FDA guidance6 suggested treatment success, defined by a score of 0 (clear) or 1 (almost clear) and at least a 2-grade improvement from baseline on the Investigator’s Static Global Assessment (ISGA) scale (an ordinal scale of 5 severity grades, each defined by a distinct and clinically relevant morphologic description), is a clinically meaningful outcome in the treatment of acne. Recognizing that there is no standardized grading system for disease severity, the FDA guidance6 suggests considering both changes in lesion counts and treatment success in the assessment of treatment effect; this is reflected in studies 301 and 302. Additionally, there is evidence in the literature to support the validity, reliability, and responsiveness of the Acne-QoL questionnaire as a measure of HRQoL in patients with acne.7,8 Therefore, bias in the measurement of important outcomes is unlikely.
In consultation with the clinical expert, age, sex, and ethnicity or race were identified as possible effect modifiers in the treatment of acne. Although randomization was not stratified, the relevant patient demographic and disease characteristics at baseline were generally well balanced between the study drug groups in each study. As such, it was concluded that any possible impact on the interpretation of the efficacy results due to baseline differences between the study drug groups is unlikely.
External Validity
The inclusion criteria used in studies 301 and 302 — patients aged 9 years and older with moderate to severe acne — include the population of interest identified in the indication for IDP-126 gel, which is for the topical treatment of acne in patients 12 years of age and older. In consultation with the clinical expert, it was concluded that the inclusion criteria adequately capture (and, consequently, the study population from both studies is representative of) the patients seen in practice who would be candidates for IDP-126 gel.
In consideration of the goal to minimize confounders using exclusion criteria, it was concluded that no patient who would be a candidate for IDP-126 gel was missed as a result of any exclusion criterion. However, the clinical expert highlighted that patients with these exclusion criteria seen in practice may still have an indication for topical therapy and be considered for IDP-126 gel. The clinical expert provided examples of patients meeting such criteria, including patients with polycystic ovarian disease, clinically significant menstrual irregularities, or secondary acne, and patients taking birth control pills.
In consultation with the clinical expert, it was concluded that the outcome measures of acne severity and lesion counts used in the trials are applicable to Canadian clinical practice. It was also concluded that a follow-up at 12 weeks after starting a topical therapy is appropriate for an assessment of effect in this therapeutic area.
GRADE Summary of Findings and Certainty of the Evidence
Methods for Assessing the Certainty of the Evidence
For pivotal studies and RCTs identified in the sponsor’s systematic review, Grading of Recommendations Assessment, Development and Evaluation (GRADE) was used to assess the certainty of the evidence for outcomes considered most relevant to inform the expert committee deliberations, and a final certainty rating was determined as outlined by the GRADE Working Group.9,10
Following the GRADE approach, evidence from RCTs started as high-certainty evidence and could be rated down for concerns related to study limitations (which refer to internal validity or risk of bias), inconsistency across studies, indirectness, imprecision of effects, and publication bias.
When possible, certainty was rated in the context of the presence of an important (nontrivial) treatment effect based on thresholds identified in the literature and/or informed by the clinical expert consulted for this review; if this was not possible, certainty was rated in the context of the presence of any treatment effect (i.e., the clinical importance is unclear). In all cases, the target of the certainty-of-evidence assessment was based on the point estimate and where it was located relative to the threshold for a clinically important effect (when a threshold was available) or to the null.
For the GRADE assessments, the findings from Study 301 and Study 302 were considered together and summarized narratively per outcome because the 2 studies were similar in population, interventions, design, and outcome measures.
The selection of outcomes for GRADE assessment was based on the sponsor’s summary of clinical evidence, consultation with the clinical expert, and the input received from patient groups and public drug plans. The following list of outcomes was finalized in consultation with expert committee members:
acne severity (treatment success defined using the EGSS)
lesion counts (inflammatory and noninflammatory lesions)
HRQoL (Acne-QoL self-perception and acne symptom subscale scores)
notable harms (general disorders and administration site conditions, skin and subcutaneous tissue disorders, and SAEs).
Results of GRADE Assessments
Table 2 presents the GRADE summary of the findings for IDP-126 gel versus IDP-126 vehicle gel.
Table 2: Summary of Findings for IDP-126 Gel Versus IDP-126 Vehicle Gel for Patients With Acne
Outcome and follow-up | Patients (studies), N | Effect | Certainty | What happens |
|---|---|---|---|---|
Acne severity | ||||
Treatment success: Proportion of patients with ≥ 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (95% CI) Follow-up: Week 12 |
| Study 301
Study 302
| Higha | Once-daily topical application of IDP-126 gel results in a clinically meaningful increase in the proportion of patients with treatment success when compared with its vehicle gel. |
Lesion count | ||||
Inflammatory lesion count, LS mean absolute change from baseline (95% CI) Follow-up: Week 12 |
| Study 301
Study 302
| Highb | Once-daily topical application of IPD-126 gel results in little to no clinically meaningful difference in inflammatory lesion count when compared with its vehicle gel. |
Inflammatory lesion count, LS mean percent change from baseline (95% CI) Follow-up: Week 12 |
| Study 301
Study 302
| Highc | Once-daily topical application of IDP-126 gel results in a clinically meaningful reduction in inflammatory lesion count when compared with its vehicle gel. |
Noninflammatory lesion count, LS mean absolute change from baseline (95% CI) Follow-up: Week 12 |
| Study 301
Study 302
| Moderated | Once-daily topical application of IDP-126 gel likely results in a clinically meaningful reduction in noninflammatory lesion count when compared with its vehicle gel. |
Noninflammatory lesion count, LS mean percent change from baseline (95% CI) Follow-up: Week 12 |
| Study 301
Study 302
| Highe | Once-daily topical application of IDP-126 gel results in a clinically meaningful reduction in noninflammatory lesion count when compared with its vehicle gel. |
HRQoL | ||||
Acne-QoL self-perception domain score, mean absolute change from baseline (95% CI) Follow-up: Week 12 |
| Study 301 ███████ ███| ███ ████████████ ███████ ███| ███ ███████████████| ███ ████ ██ █████ Study 302 ███████ ███| ███ ████████████ ███████ ███| ███ ███████████████| ███ ████ ██ █████ | Moderatef | Once-daily topical application of IDP-126 gel likely results in little to no clinically meaningful difference in Acne-QoL self-perception domain score when compared with its vehicle gel. |
Acne-QoL acne symptom domain score, mean absolute change from baseline (95% CI) Follow-up: Week 12 |
| Study 301 ███████ ███| ███ ████████████ ███████ ███| ███ ███████████████| ███ ████ ██ █████ Study 302 ███████ ███| ███ ████████████ ███████ ███| ███ ███████████████| ███ ████ ██ █████ | Highg | Once-daily topical application of IDP-126 gel results in little to no clinically meaningful difference in Acne-QoL acne symptom domain score when compared with its vehicle gel. |
Harms | ||||
Proportion of general disorders and administration site conditions, n (95% CI) Follow-up: Week 12 | 363 (2 RCTs) | Study 301 and Study 302 pooled
| Moderateh | Once-daily topical application of IDP-126 gel likely results in an increase in general disorders and administration site conditions, and skin and subcutaneous tissue disorders, when compared with its vehicle gel. |
Proportion of skin and subcutaneous tissue disorders, n (95% CI) Follow-up: Week 12 | 363 (2 RCTs) | Study 301 and Study 302 pooled
| Moderateh | |
Proportion of SAEs, n (95% CI) Follow-up: Week 12 | 363 (2 RCTs) | Study 301 and Study 302 pooled
| Moderateh | Once-daily topical application of IDP-126 gel likely results in little to no difference in SAEs when compared with its vehicle gel. |
Acne-QoL = Acne-Specific Quality of Life; CI = confidence interval; EGSS = Evaluator’s Global Severity Score; HRQoL = health-related quality of life; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; LS = least squares; MID = minimal important difference; NA = not available; RCT = randomized controlled trial; SAE = serious adverse event.
Note: Study limitations (which refer to internal validity or risk of bias), inconsistency across studies, indirectness, imprecision of effects, and publication bias were considered when assessing the certainty of the evidence. All serious concerns in these domains that led to the rating down of the level of certainty are documented in the following table footnotes.
Overall, no serious risk-of-bias concern and no serious concern about the generalizability of results to the population of interest were identified in the review and appraisal of studies 301 and 302.
In consultation with 1 clinical expert consulted for the purpose of this review, the thresholds of importance (i.e., a clinically meaningful difference) shown in the following footnotes were determined for the assessment of outcomes on acne severity and lesion counts. The thresholds of importance (MID) used in the assessment of HRQoL outcomes are based on findings in the literature.
aData from both trials show IDP-126 gel may provide a benefit based on a clinically meaningful difference of at least 100 more patients with treatment success per 1,000 patients.
bData from the trials show IDP-126 gel may provide little to no benefit based on a clinically meaningful difference of at least 10 lesions.
cData from the trials show IDP-126 gel may provide a benefit based on a clinically meaningful difference of at least 10% of lesions.
dRated down 1 level for serious imprecision; data from both trials show IDP-126 may provide either a benefit or little to no benefit based on a clinically meaningful difference of at least 10 lesions.
eData from both trials show IDP-126 gel may provide a benefit based on a clinically meaningful difference of at least 10% of lesions.
fAnalysis of this HRQoL outcome was not adjusted for multiplicity and, as such, results are considered supportive evidence. Rated down 1 level for serious imprecision; data from both trials show IDP-126 gel may provide either a benefit or little to no benefit based on a clinically meaningful difference of 5.15 points.
gAnalysis of this HRQoL outcome was not adjusted for multiplicity and, as such, results are considered supportive evidence. Data from the trials show IDP-126 gel may provide little to no benefit based on a clinically meaningful difference of 4.62 points.
hRated down 1 level for serious imprecision; the total sample size and number of events did not reach the optimal information size.11
Sources: Study V01-126A-301 Clinical Study Report,12 Study V01-126A-302 Clinical Study Report,13 and Common Technical Document section 2.7.4: Summary of Clinical Safety.14 Details included in the table are from the sponsor’s summary of clinical evidence15 and sponsor’s response to an April 8, 2024, request for additional information regarding the review of IDP-126 gel.16
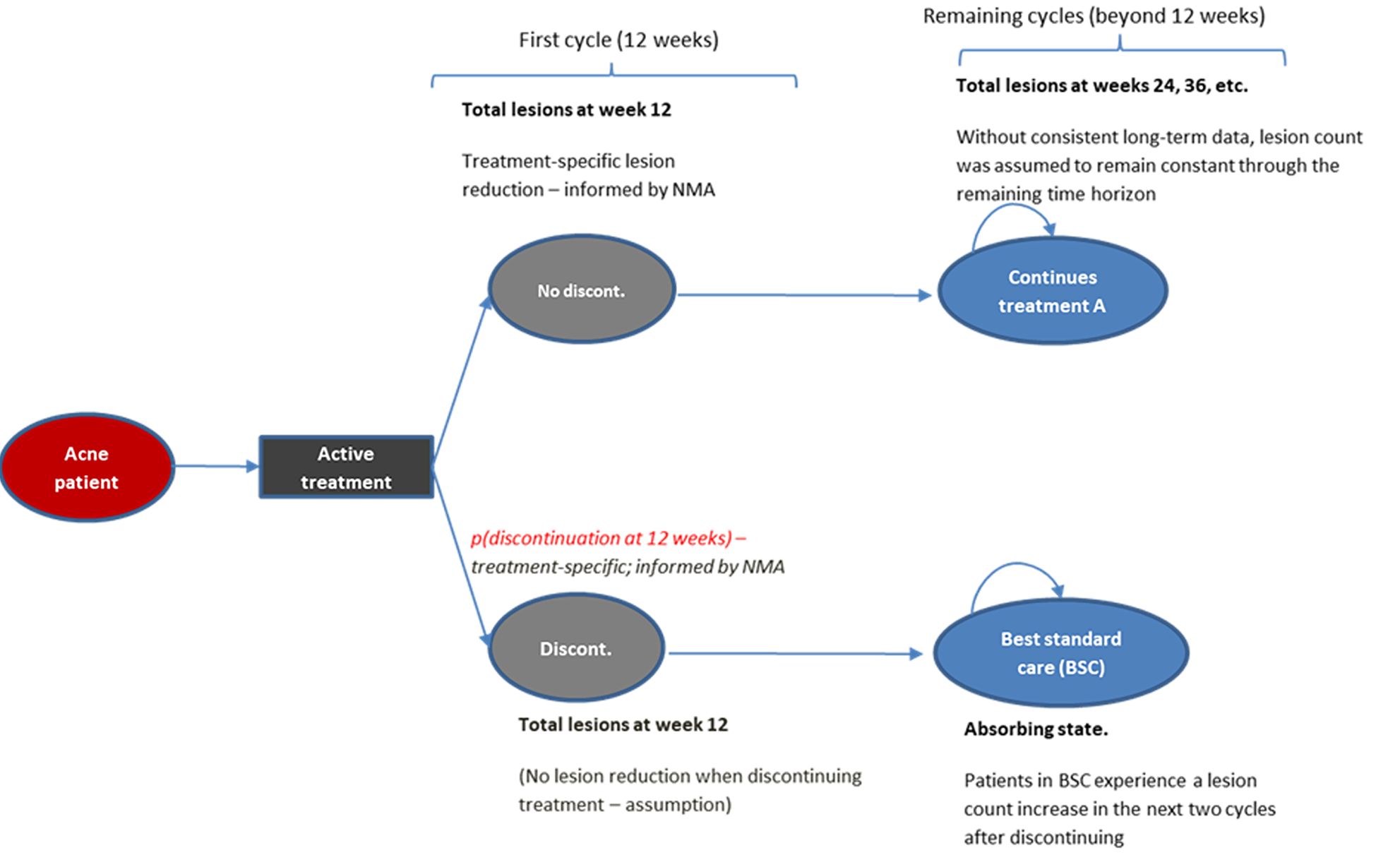
Long-Term Extension Studies
No long-term extension study was submitted by the sponsor.
Indirect Comparisons
Description of Indirect Treatment Comparisons
The sponsor conducted an indirect treatment comparison (ITC), designed to assess the efficacy of IDP-126 gel compared with other treatments available in Canada for patients with moderate to severe acne. The analyses included network meta-analyses (NMAs) of 85 RCTs identified from a systematic literature search that reported on the percentage of patients with at least a 2-grade reduction from baseline and a score of clear or almost clear on the Investigator’s Global Assessment (IGA) or equivalent scales (treatment success) and changes in inflammatory lesion and noninflammatory lesion counts. The NMA incorporated the following 14 treatment groups: fixed-dose combinations of topical antibiotic plus BPO and retinoid, topical retinoid plus BPO, topical antibiotic plus BPO, and topical antibiotic plus retinoid; 4 topical monotherapy groups (antibiotic, retinoid, BPO, other); combinations of topical treatments that include an oral antibiotic; oral antibiotic monotherapy; combinations of physical treatments with an oral antibiotic; physical treatment only; other treatments; and vehicle (or placebo). A Bayesian framework was used, employing random-study, fixed class–effect models to estimate treatment effects for each outcome as the primary analyses.
One published NMA of 221 trials conducted by Huang et al.17 was also submitted by the sponsor for this review. The analysis adopted a frequentist approach to assess the effect of different treatments for acne on outcomes of interest, including reductions in total, inflammatory, and noninflammatory lesions; treatment success defined using the IGA; and discontinuation due to any adverse events (AEs).
Efficacy Results
The NMA on the proportion of patients experiencing treatment success included 46 trials and 12 treatment groups. According to the estimated odds ratio (ORs), IDP-126 gel demonstrated higher efficacy compared with the vehicle or placebo comparison group (OR = 6.30; 95% credible interval [CrI], 3.90 to 9.87). Moreover, IDP-126 gel was favoured in comparisons with other active treatments (oral antibiotic monotherapy, topical monotherapies, topical fixed-dose combinations of antibiotic plus retinoid and antibiotic plus BPO).
The NMAs for changes in inflammatory and noninflammatory lesion counts consisted of 50 and 46 trials, respectively. There were 12 treatment nodes available in the networks for both outcomes. The number of patients ranged from 107 to 2,813 per study for the 2 networks. Findings regarding lesion counts revealed IDP-126 gel to be associated with a greater impact on the reduction of lesions compared with placebo, with a change of −8.21 (95% CrI, −10.33 to −6.13) in the inflammatory lesion count and −13.41 (95% CrI, −16.69 to −10.32) in the noninflammatory lesion count. Comparisons with other active treatments (i.e., oral antibiotic, topical monotherapies, and topical antibiotic plus retinoid fixed-dose combinations) showed that IDP-126 gel was favoured for reducing both inflammatory and noninflammatory lesion counts.
Findings from the published NMA reporting efficacy outcomes on the comparison of triple therapy (i.e., topical antibiotic, a topical retinoid, and BPO) with placebo17 were aligned with the sponsor-conducted ITC.
Harms Results
███████ ██ ███ █████████ ███ █████████ ████ ███████ ███████████████████████ ███████ █████████ █ ██████ █████████ ██ ███████████████ ████████ ██ ███████ ███ █ █████ ███ ███ ████ ██ ██████17
Critical Appraisal
The sponsor-conducted NMA used recommended methods for the conducting and reporting of NMAs and demonstrated favourable benefits relative to other available treatments, though important limitations were noted. Restrictive exclusion criteria were applied in the literature review, prohibiting the inclusion of single RCTs and studies with small sample sizes. The NMA appeared to include study populations that varied greatly in terms of their disease severity and sex distributions, which raises concerns for heterogeneity across studies in the network that may bias the comparison. Even though a literature review and meta-regression were performed to identify and assess the influence of effect-modifying variables (i.e., duration of treatment, severity of acne, diversity of treatments), their impact on the overall NMA estimates could not be properly addressed due to limited reporting by the included trials. Input from the clinical expert suggested that certain treatments of interest for Canadian clinical practice (oral isotretinoin, azelaic acid, topical dapsone) were missing in the NMA network. Moreover, treatment group nodes incorporated some monotherapies and combination therapies that are unavailable in Canada, limiting the generalizability of the included treatments. Considering all of the preceding, it is likely that the NMA estimates are subject to an unknown amount and direction of bias.
Limitations of the published ITC included the lack of prespecification of study methods through a review protocol and notable heterogeneity in prognostic and effect-modifying factors across the studies included in the network. The exploration of between-study differences and potential biases was further limited by missing information on patient and study characteristics. A notable generalizability issue was that the NMA estimates came from comparisons with placebo only and included treatments in the network that do not have market approval in Canada. This further limited the applicability of this analysis to the Canadian clinical context.
Studies Addressing Gaps in the Evidence From the Systematic Review
Study 201
Description of Study
One phase II, multicentre, double-blind, RCT, Study 201 (N = 741), was submitted by the sponsor to further address the evidence gap in terms of the direct comparative evidence of IDP-126 gel versus other relevant comparators. Study 201 also assessed whether there is a difference in the proportion of patients with treatment success (the same definition of success used in studies 301 and 302) and change from baseline in inflammatory and noninflammatory lesion counts in patients aged 9 years and older with moderate to severe acne following the application of IDP-126 topical gel once daily for 12 weeks. The other efficacy and safety outcomes assessed are similar to those assessed in studies 301 and 302. The 4 comparators are IDP-126 component A (BPO 3.1% plus adapalene 0.15% gel), IDP-126 component B (clindamycin phosphate 1.2% plus BPO 3.1% gel), IDP-126 component C (clindamycin phosphate 1.2% plus adapalene 0.15% gel), and IDP-126 vehicle gel. The mean age of patients randomized to each study drug group was similar, approximately 20 years, and ranged from age 10 to 60 years. The majority of patients in each study drug group had moderate acne, ranging from 79.3% to 86.0% of patients in a group. The remainder of patients had severe acne, ranging from 14.0% to 20.7% of patients in a group.
Efficacy Results
Acne Severity
Treatment Success Based on the EGSS
The percentage of patients with at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 was 52.5% in the IDP-126 gel group, 27.8% in the IDP-126 gel component A group, 30.5% in the IDP-126 gel component B group, 30.3% in the IDP-126 gel component C group, and 8.1% in the vehicle gel group. The treatment difference in treatment success based on the EGSS was not reported.
Lesion Count
Inflammatory Lesion Count
The LS mean change from baseline in inflammatory lesion count at week 12 was −29.9 (standard deviation [SD] = 11.86) in the IDP-126 gel group, −26.7 (SD = 11.74) in the IDP-126 component A group, −24.8 (SD = 11.71) in the IDP-126 component B group, −26.8 (SD = 11.69) in the IDP-126 component C group, and −19.6 (SD = 12.12) in the vehicle gel group. The treatment difference in the mean absolute change from baseline in inflammatory lesion count at week 12 was not reported.
███ ████ ███████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ████ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ███ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ███ ██ ████ ███████ ██████ ███ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ████ ███ █████████
Noninflammatory Lesion Count
The LS mean change from baseline in noninflammatory lesion count at week 12 was −35.5 (SD = 16.25) in the IDP-126 gel group, −29.9 (SD = 16.40) in the IDP-126 component A group, −27.8 (SD = 15.97) in the IDP-126 component B group, −30.0 (SD = 16.40) in the IDP-126 component C group, and −21.8 (SD = 16.58) in the vehicle gel group. The treatment difference in the mean absolute change from baseline in noninflammatory lesion count at week 12 was not reported.
███ ████ ███████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ████ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ███ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ███ ██ ████ ███████ ██████ ███ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ████ ███ █████████
Quality of Life
Self-Perception and Symptom Domain Score on the Acne-QoL Questionnaire
The mean change from baseline in the Acne-QoL self-perception domain score at week 12 was 9.8 (SD = 8.80) in the IDP-126 gel group, 7.3 (SD = 8.34) in the IDP-126 component A group, 7.5 (SD = 7.22) in the IDP-126 component B group, 8.5 (SD = 8.22) in the IDP-126 component C group, and 5.9 (SD = 7.99) in the vehicle gel group. The treatment difference in the absolute change from baseline in the Acne-QoL self-perception domain score at week 12 was not reported.
The mean change from baseline in the Acne-QoL symptoms domain score at week 12 was 7.4 (SD = 6.19) in the IDP-126 gel group, 7.3 (SD = 6.52) in the IDP-126 gel component A group, 6.9 (SD = 5.63) in the IDP-126 gel component B group, 6.6 (SD = 6.07) in the IDP-126 gel component C group, and 4.9 (SD = 5.53) in the vehicle gel group. The treatment difference in the absolute change from baseline in the Acne-QoL symptoms domain score at week 12 was not reported.
Harms Results
Adverse Events
The proportion of patients who reported at least 1 TEAE was similar in the IDP-126 gel and IDP-126 component A groups (36.2% and 35.6%, respectively), while the proportion of patients who reported a TEAE in the IDP-126 component B, IDP-126 component C, and IDP-126 vehicle gel groups was 18.1%, 27.0%, and 15.1%, respectively. The most common TEAEs reported were application site pain (7.8% of patients in the IDP-126 gel group, 11.0% in the IDP-126 component A group, 0.7% in the IDP-126 component B group, 3.4% in the IDP-126 component C group, and 0.7% in its vehicle gel group), application site dryness (6.4% of patients in the IDP-126 gel group, 5.5% in the IDP-126 component A group, 1.4% in the IDP-126 component B group, 6.1% in the IDP-126 component C group, and 0.7% in its vehicle gel group), and application site exfoliation (3.5% of patients in the IDP-126 gel group, 2.1% in the IDP-126 component A group, 0.0% in the IDP-126 component B group, 1.4% in the IDP-126 component C group, and 0.7% in its vehicle gel group).
Serious Adverse Events
A total of 4 patients were reported with SAEs: 1 patient in the IDP-126 gel group experienced sickle cell anemia with crisis, and 3 patients in the IDP-126 component C group (hyperbilirubinemia, enteritis, and abortion induced; n = 1 each).
Withdrawal Due to Adverse Events
A total of 17 patients, 4 in the IDP-126 gel group, 8 in the IDP-126 component A group, 3 in the IDP-126 component C group, and 2 in the IDP-126 vehicle gel group, had their study drug withdrawn due to TEAEs. A total of 16 patients (4 in the IDP-126 gel group, 8 in the IDP-126 component A group, 3 in the IDP-126 component C group, and 1 in the IDP-126 vehicle gel group) discontinued the study due to TEAEs.
Mortality
There were no reports of patients who died in Study 201.
Notable Harms
| █████ ██ █████ ██ ████████ ██ ███ ███████ ███ ██████ ████ █ ██ █████████ █ ██████ ████ ██ █████████ █ ██████ █████ ██ █████████ █ ██████ ███ ████ ████████ ██ ███ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ███████ ████████ ███ ██████████████ ████ ████████████ █████ ██ ████ ██ ████████ ██ ███ ███████ ███ ██████ ███ █ ██ █████████ █ ██████ ████ ██ █████████ █ ██████ ████ ██ █████████ █ ██████ ███ ████ ██ ███ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ████ ███ ████████████ ██████ █████████
Critical Appraisal
The randomization and masking procedures in Study 201 were considered appropriate. Since it was a phase II trial aiming to provide preliminary evidence about the efficacy and harms of the study drug, the results cannot be considered confirmatory. The relevant patient demographic characteristics at baseline appeared to be well balanced between the study drug groups. No notable differences between the study drug groups in the baseline EGSS or in lesion counts were identified. Similar to the pivotal trial, the washout periods used in the studies were considered adequate and the list of prohibited treatments for acne was considered comprehensive by the clinical expert. No adjustments were made for multiple comparisons for primary and secondary outcomes; therefore, there is a greater likelihood of type I error. For the outcomes on quality of life (Acne-QoL self-perception and symptom domains), no inferential analyses or multiplicity adjustments were conducted, as per the statistical analysis plan, so these data are considered supportive, and no definitive conclusions could be drawn. Study discontinuation rates were similar between the pivotal trials and Study 201 (i.e., not high in the context of this patient population and rates were similar between groups in terms of the overall rates of study discontinuation and in the reasons for study discontinuation). Similar to the pivotal trials, in consultation with the clinical expert, it was concluded that the study discontinuation rates are reasonable in the context of the therapeutic area and, as such, the risks of attrition bias and possible unblinding are unlikely. Overall, no major concerns regarding bias in the results were identified; however, the results cannot be interpreted as conclusive evidence due to the phase II trial design.
The inclusion criteria used in Study 201 include the population of interest identified in the indication for IDP-126 gel, that is, patients aged 9 years and older with a clinical diagnosis of moderate to severe acne vulgaris, defined as a baseline EGSS of 3 (moderate) or 4 (severe) for facial acne. In consultation with the clinical expert, it was concluded that the inclusion criteria adequately capture (and, consequently, the study population is representative of) the patients seen in practice who would be candidates for IDP-126 gel. Similar to the pivotal trials, the majority of patients in each study (approximately ≥ 79.3% of the patients in each study drug group) had moderate acne. In consultation with the clinical expert, it was concluded that topical fixed-dose combination therapies (i.e., retinoid plus BPO, antibiotic plus BPO, and retinoid plus antibiotic) are relevant comparators for IDP-126 gel in the Canadian practice setting. Similar to the pivotal trials, in consultation with the clinical expert, it was concluded that the outcome measures of acne severity and lesion counts used in Study 201 are applicable to Canadian clinical practice. Overall, no notable concerns on the generalizability of the results to the population of interest in the Canadian setting were identified in the appraisal of Study 201.
Study 202
Description of Study
One additional phase II, multicentre, double-blind RCT, Study 202 (N = 686), was submitted by the sponsor to further address the gap in the direct comparative evidence of IDP-126 gel versus other relevant comparators. Study 202 also assessed whether there is a difference in the proportion of patients with treatment success (the same definition of success used in studies 301 and 302) and change from baseline in inflammatory and noninflammatory lesion counts in patients aged 12 years and older with moderate to severe acne applying IDP-126 topical gel once daily for 12 weeks when compared with adapalene 0.3% plus BPO 2.5% gel. The other efficacy and safety outcomes assessed are similar to those assessed in studies 301 and 302. ███ ████ ███ ██ ████████ ██████████ ██ ████ █████ ████ █████ ███ ████████ █████████████ ██ █████ ███ ██████ ██ ██ ██ ██████ ███ ████████ ██ ████████ ██ ████ █████ ████ █████ ███ ████████ ████ █ ███████ ████ █████ ██ ██████ ███ █████████ ██ ████████ ███ ██████ ████ █ ███████ ████ █████ ██ ██████
Efficacy Results
████ ██████████████████ ███████ █████ ██ ███ ███████████ ██████ ████████ █████████ █████████ ██████████ ██ █████████ ███████ █████ ██ ███ ████ ██ ████ ██ ███████ ███████ ███ ███ █████████ ████████ ████ ███ ███ ██████ ████ ███ █████ ██ ████████ ███████ ██████████████████ ██████ █████████ ██ ████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███ █████ ███ █ ██████ ██ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ █████████ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ █████ ███ ██████ ████ ███ ██████ ██ ███████ ███ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ █████████ ████████ ████ ███ █████ ███ █████ ████ ███ █ ████ ██ ██████ ████ ██ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███ ██████ ███ █ █████████ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ █████████ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ ███ ███████ ████ ███ ███████ ██ █████████ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ █████████ ████████ ████ ███ █████ ███ ██████ ████ ███ ██████ ██ ██████████████████████ ██████ █████████ ██ ████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███ █████ ███ █ ██████ ██ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ █████████ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ █████ ███ ██████ ████ ███ ██████ ██ ████████ ███ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ █████████ ████████ ████ ███ █████ ███ █████ ████ ███ █ ████ ██ ██████ ████ ██ ████ ███████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███ ██████ ███ █ ███████ ██ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ █████████ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ ███ ███████ ████ ███ ███████ ██ █████████ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ █████████ ████████ ████ ███ █████ ███ ██████ ████ ███ ██████ ██ ██████████████ ██ ████████████████████ ███ ███████ ██████ █████ ██ ███ █████████████ ███████ ██ ████ █████████████████ ████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ ███ █ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ███ ███ █ █████ ██ ███ ████████ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ████ ███ █████████████ ████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ ███ █ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ███ ███ █ █████ ██ ███ ████████ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ████ ███ ███████████████ ███████████████ ██████████ ██████████ ██ ████████ ████ ████████ ██ █████ ███ ████ ████ ███████ ██ ███ ███████ ███ ███ █████████ ████████ ████ ███ ██████ ██████ ███ ██████ ██████████████ █████ ███ ██████████ ██ ████████ ████ ████████ █ ████ ██ ███ ████████ ███████ ███ █████ ███ ██████ ███ ████ ██████ █████ ████████ ████ ███████████ ████ ████ ██████ ██ ████████ ██ ███ ███████ ███ ██████ ████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███ ███████ ███ ███████████ ████ ███████ █████ ██ ████████ ██ ███ ███████ ███ ██████ ████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███████ ███ ███████████████ ███████ ██████████ ███████ ███ ████████ ████ ███ ██ ███ ███████ ███ █████ ███ ████ ███████ █████████ ████████████ ███ ██ ███████ ████████ █████ ██ ██ █████████ █ ██ ███ ███████ ███ █████ ███ █ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ █████ ████ █████████ ███ ██ ██████ ███ ██ ██ █████ ████████ ██ ██ ███ ███████ ███ █████ ███ █ ██ ███ █████████ ████████ ████ ███ ███████ ███ █████ ████ ███ ██ █████ ████ ██████████ ████ ███ ██ █████ ████████████████ ████████████████ ████ ██ ███████ ██ ████████ ███ ████ ██ █████ ████████████ ███████ █████ ██ █████ ██ ████████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ███████ ████████ ███ ██████████████ ████ ████████████ █████ ██ ████ ██ ████████ ██ ███ ███████ ███ ██████ ████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ████ ███ ████████████ ██████ █████████
Critical Appraisal
The randomization and masking procedures in Study 202 were considered appropriate. The coprimary and secondary end points were controlled for multiplicity. Type I error was controlled by requiring that all coprimary efficacy end points be statistically significant; the failure of any 1 coprimary efficacy end point invalidated the statistical significance of all secondary efficacy end points. Since it was a phase II trial aiming to provide preliminary evidence about the efficacy and harms of the study drug, the results cannot be considered confirmatory. The relevant patient demographic characteristics at baseline appeared to be balanced among the study drug groups. No notable differences in the baseline EGSS and lesion counts among the study drug groups were identified. Similar to the pivotal trial, the washout periods used in the studies were considered adequate and the list of prohibited treatments for acne was considered comprehensive by the clinical expert. A total of 7 patients had protocol deviations regarding the specified washout period for prior medications. For the outcomes on quality of life (Acne-QoL self-perception and symptom domains), no inferential analyses were conducted, as per the statistical analysis plan, so no conclusions could be drawn from these data. Of note, while all data were summarized in listings presented by patient, the data collected at early discontinuation and unscheduled visits that occurred before study day 8 were not included in the analyses of efficacy and safety, except for baseline values.
The inclusion criteria used in Study 202 included the population of interest identified in the indication for IDP-126 gel. In consultation with the clinical expert, it was concluded that the inclusion criteria adequately capture (and, consequently, the study population is representative of) the patients seen in practice who would be candidates for IDP-126 gel. In consultation with the clinical expert, it was concluded that topical fixed-dose combination therapies, including retinoid plus BPO, are relevant comparators for IDP-126 gel in the Canadian practice setting. Similar to the pivotal trial and Study 201, in consultation with the clinical expert, it was concluded that the outcome measures of acne severity and lesion counts used in Study 202 are applicable to Canadian clinical practice. Similar to the pivotal trial, the majority of patients from each study (approximately ≥ 87.4% of patients in each study drug group) had moderate acne.
Overall, no notable concerns on the generalizability of results to the population of interest in the Canadian setting were identified in the appraisal of Study 202.
Conclusion
Studies 301 and 302 demonstrated that 12 weeks of treatment with IDP-126 gel applied once daily results in a clinically meaningful improvement in acne, when compared with its vehicle gel alone, in patients aged 10 years and older with moderate to severe acne. Treatment effect was measured by the proportion of patients with treatment success, defined as at least a 2-grade reduction from baseline in the EGSS, an EGSS of clear or almost clear, and a reduction in inflammatory and noninflammatory lesion counts. While a statistically significant reduction in the absolute change in inflammatory lesion count was demonstrated in the trials, the GRADE assessment for this end point concluded there is little to no clinically meaningful difference, as the effect estimates for the absolute difference did not exceed the suggested threshold of importance. In contrast, the trials showed IDP-126 gel results in a clinically meaningful reduction in the inflammatory lesion count based on the percent change from baseline when compared with its vehicle gel. The sponsor-conducted NMA results showed a favourable effect on treatment success with IDP-126 gel versus vehicle or placebo, oral antibiotic, and topical monotherapies and change in lesion counts. While the effect estimates are suggestive of a possible favourable treatment effect with IDP-126 gel versus topical fixed-dose combination dual therapies, there is some uncertainty, as the 95% CrIs included the null or were close to the null for certain dual-combination therapies. Due to the possible exclusion of relevant studies, heterogeneity across trials in the networks, and the omission of relevant comparators in the Canadian setting that suggest concerns for bias in the NMA estimates, the magnitude of effect associated with IDP-126 gel is uncertain. Findings from the NMA by Huang et al. aligned with the sponsor-conducted NMA results; however, these findings are of limited applicability to the Canadian context, as IDP-126 gel was combined with other triple therapies in the analyses and only comparisons with placebo were reported. Results from the phase II trials (studies 201 and 202) are supportive evidence suggesting a possible favourable treatment effect with IDP-126 gel versus vehicle gel and topical dual-combination therapies (fixed-dose combination of retinoid plus BPO, antibiotic plus BPO, and retinoid plus antibiotic), based on treatment success and change in lesion counts at week 12. No concerns with the safety profile of IDP-126 gel were identified based on the safety results from all submitted trials.
Introduction
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects of IDP-126 gel in the treatment of acne in patients aged 12 years and older. Of note, this Reimbursement Review was conducted before the Health Canada Notice of Compliance was issued; the scope was based on the anticipated indication.
Disease Background
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following has been summarized and validated by the review team.
Acne is a common chronic inflammatory skin condition of pilosebaceous glands that typically begins at puberty and may continue through adulthood with flares that are associated with an increase in serum levels of androgen.1 Endogenous androgens mediate excess sebum production in the skin and stimulate abnormal keratinization and desquamation. These events lead to obstruction of the pilosebaceous duct (clogged hair follicle) that allows Cutibacterium acnes to proliferate. In response to the proliferation of bacteria, proinflammatory mediators are released that trigger localized inflammation and the exacerbation of lesions.18-20
Acne is characterized by noninflammatory and inflammatory lesions that primarily develop on the face, neck, upper back, and chest.2,3 Noninflammatory lesions are comedones that can be open or closed by the skin (i.e., blackheads or whiteheads). Inflammatory lesions consist of papules that are red and/or tender bumps, pustules that are filled with purulent material, and nodules that are large red bumps on the skin.2,3 Diagnosis is a visual assessment completed by the clinician who considers the distribution of acne, type and number of lesions, and the presence of scarring.18,21
Acne affects 5.6 million individuals living in Canada, nearly 20% of the population.4 Although acne predominantly affects the adolescent population (approximately 90%), it can also affect preadolescents (aged 7 to 12 years) and postadolescents.4,22-24 Severe acne is associated with depression, anxiety, and poor self-esteem.25 Scarring negatively affects HRQoL independent of acne, and is associated with frustration, sadness, and anxiety.26
Standards of Therapy
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following has been summarized and validated by the review team.
The 2016 Canadian clinical practice guideline in the management of acne advises the following:5
Comedonal acne: Topical therapies are a reasonable first-line treatment option for comedonal acne, including topical retinoids (tretinoin, adapalene, and tazarotene are commonly associated with irritation), BPO (available over the counter), and fixed-dose combinations of retinoids with BPO or clindamycin.
Localized mild to moderate papulopustular acne: The topical therapies mentioned earlier are again a reasonable treatment option for mild papulopustular acne. For more extensive moderate papulopustular acne and acne in areas not amenable to topical therapy (e.g., the back), systemic therapies in addition to topical therapies are a reasonable treatment regimen, including oral antibiotics (tetracycline, doxycycline, or minocycline, the use of which is discouraged due to concerns with the selection of antibiotic-resistant bacteria) and combined oral contraceptives in female patients (ethinyl estradiol and levonorgestrel, ethinyl estradiol and drospirenone, and ethinyl estradiol and norgestimate).
Severe acne: Systemic therapies are a reasonable treatment option for severe acne, including oral isotretinoin (a retinoid; its use is limited by the potential for AEs and teratogenicity); oral antibiotics in combination with BPO (to limit the emergence of antibiotic-resistant bacteria), with or without topical retinoids, and combined oral contraceptives in female patients.
In addition to the treatment options advised by the 2016 Canadian guideline in the management of acne, the clinical expert indicated the following are also current treatment options: alpha hydroxy acid (glycolic acid), beta hydroxy acid (salicylic acid), azelaic acid, antiandrogen (clascoterone), and other topical therapies, including sulphur or sulfacetamide sodium and resorcinol.
The following factors are considerations for selecting the most appropriate first-line therapy in the management of acne, according to the guidelines:5 patient experience with prior therapies (i.e., efficacy and tolerance), skin type and type of vehicle (e.g., gel, a less greasy formula, may be preferred over cream and lotion in individuals with oily skin), practicality (e.g., convenience of combination therapy requiring a once-daily application versus separate therapies requiring multiple daily applications), and cost and access.
The clinical expert indicated that combination therapies tend to improve patient adherence, while treatment regimens that are complicated (i.e., requiring 2 to 3 different products) tend to reduce adherence. Further, the clinical expert advised that combining monotherapies introduces the potential for active ingredient cross-reactivity and reduced efficacy.
The goal of treatment, as identified by the clinical expert, is to reduce the severity of acne symptoms and prevent disease progression and scar formation. Additionally, the clinical expert indicated that improvement in HRQoL is important to patients.
Drug Under Review
Key characteristics of IDP-126 gel are summarized in Table 3 along with other treatments available in Canada for acne.
IDP-126 gel is indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.27 The sponsor’s reimbursement request aligns with the Health Canada indication. IDP-126 gel is supplied in a 20 g and 50 g pump. A thin layer of IDP-126 gel is recommended to be applied to the affected area once daily.27
The active ingredients of IDP-126 gel — clindamycin, adapalene, and BPO (an antibiotic, a retinoid, and an antibacterial, respectively) — have complementary mechanisms of action that target the 3 pathophysiologic factors known to contribute to acne: altered follicular growth and differentiation (comedogenesis), colonization of the pilosebaceous unit with Propionibacterium acnes, and inflammation.27
Table 3: Key Characteristics of IDP-126 Gel and Relevant Comparators for the Treatment of Acne
Characteristic | IDP-126 (clindamycin 1.2%, adapalene 0.15%, and BPO 3.1%) | Topical retinoids | Topical antibiotics | Topical retinoid and BPO fixed-dose combinations | Topical retinoid and antibiotics fixed-dose combinations | Topical antibiotic and BPO fixed-dose combinations |
|---|---|---|---|---|---|---|
Drugs under the same class | NA | Tazarotene 0.045% (Arazlo); adapalene 0.1% (Differin) or 0.3% (Differin XP); tretinoin (Retin-A) 0.025% gel or 0.05% cream; tretinoin 0.1% (Retin-A Micro) gel. | Clindamycin phosphate (generic) 1% solution. | Adapalene 0.1% plus BPO 2.5% (TactuPump); adapalene 0.3% plus BPO 2.5% (TactuPump Forte). | Clindamycin phosphate 1.2% plus tretinoin 0.025% gel (Biacna). | Clindamycin 1% plus BPO 5% gel (Clindoxyl); clindamycin 1% as phosphate and BPO 5% topical gel (Benzaclin and generics); clindamycin 1% and BPO 3% gel (Clindoxyl Adv); erythromycin plus BPO topical gel (Benzamycin). |
Mechanism of action | The active ingredients have an effect on 3 pathophysiologic factors known to contribute to acne vulgaris: altered follicular growth and differentiation (comedogenesis), colonization of the pilosebaceous unit with P. acnes, and inflammation. | Vitamin A (retinol) derivatives that modify gene expression, subsequent protein synthesis, and epithelial cell growth and differentiation by activating retinoic acid nuclear receptors. | Inhibits P. acnes from producing free fatty acids in the sebum of the skin. Erythromycin also reduces inflammation. BPO prevents growth of drug-resistant bacteria. | The active ingredients have an effect on 3 pathophysiologic factors known to contribute to acne vulgaris: altered follicular growth and differentiation (comedogenesis), colonization of the pilosebaceous unit with P. acnes, and inflammation. | Clindamycin demonstrated in vitro activity against P. acnes; however, the clinical significance of this activity was not examined with clindamycin. The exact mode of action of tretinoin is unknown. Current evidence suggests a decrease in the cohesiveness of follicular epithelial cells resulting in decreased microcomedone formation. | Clindamycin demonstrated in vitro activity against P. acnes. BPO demonstrates antibacterial activity by releasing active or free-radical oxygen capable of oxidizing bacterial proteins. |
Indicationa | For the topical treatment of acne vulgaris in patients 12 years of age and older. | Treatment of acne vulgaris (in patients 12 years of age and older). | Treatment of acne vulgaris. | Adapalene 0.1% plus BPO 2.5% (TactuPump): Treatment of mild and moderate acne vulgaris, characterized by comedones and inflammatory papules or pustules in patients 9 years of age and older. Adapalene 0.3% plus BPO 2.5% (TactuPump Forte): Treatment of moderate and severe acne vulgaris, characterized by comedones, inflammatory papules/pustules with or without occasional nodules in patients 12 years of age and older. | For the topical treatment of acne vulgaris characterized by comedones and inflammatory papules or pustules, with or without an occasional nodule, in adults and children 12 years or older. | Clindamycin 1% plus BPO 5% (Clindoxyl) or BPO 3% (Clindoxyl Adv): The topical treatment of moderate acne vulgaris characterized by the presence of comedones, papules, and pustules. Clindamycin, as phosphate, 1% and BPO 5% topical gel (Benzaclin and generics): The topical treatment of moderate acne vulgaris characterized by comedones and inflammatory papules and/or pustules, with or without an occasional cyst or nodule (grade 2 to 3). |
Route of administration | Topical | Topical | Topical | Topical | Topical | Topical |
Recommended dose | A thin layer applied to affected area once a day. | A thin layer applied to affected area once a day in the evening. | A thin layer applied to affected area twice daily. | Applied to affected areas once daily in the evening. | A thin layer applied to affected area once a day. | A thin layer applied to affected area twice daily. |
Serious adverse effects or safety issues | Orally and parenterally administered clindamycin have been associated with severe colitis, which may result in patient death. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of topical and systemic clindamycin. Prolonged use may result in overgrowth of nonsusceptible organisms including fungi. | Increased susceptibility to burning, such as sunburn including sunlamps, extreme weathers, such as wind or cold. Caution when co-administered with photosensitizers (e.g., tetracyclines, fluoroquinolones, sulfonamides) Application site reactions including irritation, scaling, dryness, erythema, burning, stinging. | Dry skin, peeling, itching, burning sensation, erythema, pruritus, edema, irritation of the eyes, skin discoloration, sunburn. | Certain cutaneous signs and symptoms such as erythema, dryness, scaling, burning, or pruritus are associated with the topical application of retinoids. | Occasional gram-negative folliculitis has been reported during treatment with clindamycin phosphate 1% topical products. CDAD has been reported with the use of topical clindamycin. CDAD may range in severity from mild diarrhea to fatal colitis. | Gram-negative folliculitis has been reported in association with the long-term use of clindamycin. CDAD has been reported with the use of topical clindamycin. CDAD may range in severity from mild diarrhea to fatal colitis. |
Other | Contraindicated in patients with a history of regional enteritis (Crohn disease), ulcerative colitis, or antibiotic-associated colitis. Exposure to excessive sunlight, including sunlamps, should be avoided. | Contraindicated in pregnancy and women of childbearing potential. Avoid breast-feeding while using topical retinoids. | Resistance develops when erythromycin or clindamycin is used as monotherapy. | Contraindicated in pregnancy and for women planning a pregnancy; contraindicated in application to areas of skin affected by eczema or seborrhoeic dermatitis. | Contraindicated in patients with regional enteritis, ulcerative colitis, or history of antibiotic-associated colitis. Exposure to sunlight or unnecessary UV light should be minimized. | Contraindicated in patients with regional enteritis, ulcerative colitis, or history of antibiotic-associated colitis. Exposure to sunlight or unnecessary UV light should be minimized. |
BPO = benzoyl peroxide; CDAD = Clostridium difficile–associated disease; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; NA = not applicable; P. acnes = Propionibacterium acnes.
aHealth Canada–approved indication.
Perspectives of Patients, Clinicians and Drug Programs
Patient Group Input
This section was prepared by the review team based on the input provided by patient groups. The full original patient input received has been included in the patients, clinicians, and drug programs input section of this report.
ARSC and CSPA submitted a joint input on the current review of IDP-126 gel. Patient input was gathered using an online survey that was conducted from June 7 to 30, 2022. A total of 154 responses were collected from patients with acne (either diagnosed by a dermatologist or other health care provider or self-diagnosed) and their caregivers living in Canada. Most respondents (68%) were female, 30% were male, and 2% identified as nonbinary. The distribution of respondents by age range was 20 to 29 years (55% of respondents), 30 to 39 years (23%), and 16 to 19 years (12%). Additionally, CSPA and ARSC created a survey targeting participants in clinical trials of IDP-126 gel and received a total of 3 responses.
Almost half (47%) of the survey respondents reported moderate acne, while 16% reported severe acne. Almost half (42%) of the survey respondents indicated they had undergone 2 to 5 health care visits before receiving their diagnosis and treatment for acne. Nearly 30% of survey respondents reported visiting a health care provider more than 5 times. Almost half of survey respondents reported feeling self-conscious either often or always due to acne. Most respondents (87%) reported using a strategy to hide their acne, with 63% using makeup and 59% avoiding social gatherings altogether. Most respondents reported acne scarring (87%) and changes in skin pigmentation due to acne (90%).
Overall, survey respondents reported experience with various treatments with varying degrees of improvement (or sometimes worsening) in their acne and experience with associated side effects. The majority of respondents (89%) reported prior use of prescription gels or creams for acne. Of these, 21% reported no change in their condition, 43% reported a little improvement, 13% reported a big improvement, and 12% reported a worsening of their condition. Most survey respondents (59%) reported experience with isotretinoin therapy, of which 28% reported significant improvement and 43% reported slight improvement. Similarly, 59% of respondents reported experience with hormone therapy, including birth control and spironolactone, of which 23% reported significant improvement and 36% reported minor improvement. Most respondents (95%) reported experiencing adverse effects associated with their treatment regimen for acne in the last year, with the most common adverse effects being skin irritation (64%), dry skin (62%), and skin flaking (55%). More specifically, 85% of respondents reported experiencing side effects associated with their current topical treatment regimen (nonprescription and prescription), and most side effects (70%) were reported as minor. Most survey respondents indicated they were willing to accept these side effects because they thought the treatment was effective. Additionally, facials and peels were used by more than half of all respondents (53%), while 65% reported undergoing light or laser therapy.
All 3 patients with experience with IDP-126 gel reported manageable side effects. Two of the 3 patient respondents indicated treatment with IDP-126 gel was easier to use versus their previous therapies. Further, 2 of the 3 patient respondents indicated the value of IDP-126 gel is treatment effectiveness and time to improvement.
According to the survey respondents, common challenges in the management of acne include hiding their acne, identifying triggers, and coping with high out-of-pocket expenses on nonprescription acne products and treatments.
Survey respondents identified the following goals for improved outcomes: ability to enjoy personal relationships, to have less scarring, and to have fewer changes in skin pigmentation. Other goals include clearer skin, improved mental health, increased self-confidence, and improved overall daily life.
Clinician Input
Input From Clinical Expert Consulted
All review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 1 clinical specialist with expertise in the diagnosis and management of acne.
Unmet Needs
An unmet need identified by the clinical expert is the current lack of a topical therapy that can demonstrate long-term disease remission. When considering this unmet need in the context of the drug under review, the clinical expert advised that continued treatment effect is not expected if the topical therapy is stopped (i.e., IDP-126 gel is not expected to meet this unmet need for long-term disease remission).
The second unmet need identified by the clinical expert is that not all patients’ acne responds to current topical therapies. The third unmet need identified by the clinical expert relates to the generally low adherence to acne therapies observed in practice (i.e., the treatment discontinuation rate in this patient population is expected to be high), often due to associated side effects (e.g., application site dryness, irritation, and pain, especially with retinoids), treatment modality, and complicated treatment regimens (i.e., requiring ≥ 2 different products with the potential for active ingredient cross-reactivity and reduced efficacy).
Place in Therapy
The clinical expert expected a triple therapy, such as IDP-126 gel, to become widely adopted as first-line therapy in the treatment of acne. The clinical expert anticipated IDP-126 gel may cause a shift in the current treatment paradigm away from topical dual-combination therapies (e.g., adapalene and BPO topical gel, clindamycin phosphate and tretinoin gel) that have been widely adopted in clinical practice. The clinical expert anticipated that monotherapy will still be used at times due to concerns about tolerability or when specific treatment outcomes are targeted (i.e., for improvement of comedonal or hormonal acne).
The clinical expert did not expect IDP-126 gel to be used in combination with other therapies. The clinical expert advised that patients should not have to be required to try other therapies before initiating treatment with IDP-126 gel due to its overlap with other current treatment options. Moreover, the clinical expert indicated that many patients become exhausted and frustrated with topical products. More specifically, the clinical expert advised that if there were a requirement for prior treatment failure with other topical therapies before being able to access IDP-126 gel, patients would be more likely to request a step up to oral therapies.
Patient Population
The clinical expert indicated that the following patients are most in need of an intervention: patients who are experiencing a major negative impact on their HRQoL and psychological well-being from acne, and patients who are at risk of scar development or life-long disfiguration from acne.
The clinical expert suggested that patients with moderate acne (i.e., numerous noninflammatory lesions and inflammatory papules) are most likely to respond to treatment with IDP-126 gel. In contrast, the clinical expert suggested that patients with nodulocystic acne or severe acne and patients with scarring are less likely to respond to treatment with IDP-126 gel; nodulocystic acne, a severe form of acne, tends to require systemic medication. As such, the clinical expert indicated they would likely use IDP-126 gel only in patients who do not have nodules or cysts.
The clinical expert indicated they would avoid the use of IDP-126 gel in female patients of childbearing age who are not willing to use contraceptives or are currently pregnant. Additionally, the clinical expert indicated they would avoid the use of IDP-126 gel in patients who have had previous issues with the side effects associated with topical retinoids.
Assessing Treatment Response
The clinical expert indicated that the assessment of treatment response in practice includes an estimate of the total number of inflammatory and noninflammatory lesions (i.e., not counted), which contributes to their global assessment (most assess according to no acne, mild acne, moderate acne, or severe acne). The clinical expert advised that patients using topical therapies are typically reassessed every 3 to 6 months in practice.
The clinical expert suggested an EGSS of 0 or 1 is a clinically meaningful measure of treatment response in patients with acne, as this represents clear or almost clear skin. Based on their literature search, the clinical expert acknowledged the limited data on Acne-QoL for a minimal important difference (MID) of 0.5 to 10.0 points and the lack of an estimated MID in lesion counts to date.
Discontinuing Treatment
The clinical expert identified the following factors for consideration when deciding to discontinue IDP-126 gel: lack of response or minimal response (defined by no improvement or minimal improvement from baseline, as determined by examining the patient), severity of side effects, and disease progression (defined by an increase in the number of nodules or scar formation).
Prescribing Considerations
The clinical expert suggested that family physicians, nurse practitioners, and other primary care providers can prescribe and monitor treatment with IDP-126 gel in any setting, including community, outpatient hospital, and specialty dermatology clinic.
Clinician Group Input
No input from clinician groups was submitted for the present review of IDP-126 gel.
Drug Program Input
The drug programs provide input on each drug being reviewed through the Reimbursement Review processes by identifying issues that may impact their ability to implement a recommendation for the drug under review. The implementation questions and corresponding responses from the clinical expert consulted are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Relevant comparators | |
In studies 301 and 302, many patients appeared to respond to the vehicle gel. Is the magnitude of response to vehicle gel similar to other studies for topical treatments of acne? If so, what is the rationale for high response rates to vehicle (e.g., do patients just require the use of a moisturizer)? If not, what study design characteristics could explain this? | The clinical expert advised that placebo (or vehicle) response rates in acne trials generally range from 20% to 25%. The clinical expert indicated that moisturizers alone have a clinically meaningful, beneficial effect on acne; the mechanism of action is thought to be related to improved water balance, decreased inflammation, and improved keratinocyte differentiation and sloughing of corneocytes. |
In clinical practice, what type of patients significantly benefit from the use of combination topical therapies compared with the use of its active ingredients separately? | The clinical expert anticipated that monotherapy will still be used at times due to tolerability concerns or when specific treatment outcomes are targeted (i.e., comedonal acne or hormonal acne). The clinical expert suggested that combination therapies tend to improve patient adherence, while treatment regimens that are complicated (i.e., requiring ≥ 2 different products) tend to reduce adherence. Further, the clinical expert advised that combining monotherapies introduces the potential for active ingredient cross-reactivity and reduced efficacy. |
Most public drug plans in Canada do not cover any or most of the combination products for the treatment of acne. Specifying the failure of a dual-combination product before being eligible for a triple-combination product would not allow this triple-therapy product to be listed in jurisdictions that do not cover dual-combination products for the treatment of acne. | This is a comment from the drug plans to inform CDEC deliberations. |
Considerations for initiation of therapy | |
Are the coprimary efficacy end points assessed in studies 301 and 302 applicable to clinical practice? Could they be used as criteria for assessing continued reimbursement of the drug under review? | The clinical expert indicated that the assessment of treatment response in practice includes an estimate of the total number of inflammatory and noninflammatory lesions, which contributes to their global assessment (no acne; mild, moderate, or severe acne). Thus, requiring lesion counts as part of the initiation and renewal criteria would not be reflective of current practice. |
According to the adapalene monograph, the safety and effectiveness of adapalene have not been established in children younger than the age of 12 years. In studies 301 and 302, the mean age of patients was approximately 20 years with a range of 10 to 48 years. Are there any concerns with using the drug under review in patients who are 9 years of age? | The clinical expert expressed no major concerns with the use of adapalene in patients as young as 9 years. According to the clinical expert, several guidelines suggest topical retinoid use for childhood acne, as systemic absorption of adapalene is extremely low. |
In studies 301 and 302, patients did not have to experience the failure of 1 or 2 topical products for acne before being enrolled. Are there situations where you would start a patient on triple topical therapy for acne without trying mono and/or combination therapy before? What is considered the minimum trial length of monotherapy and combination therapy? How is a lack of response determined? | The clinical expert expected a triple therapy, such as IDP-126 gel, to become widely adopted as first-line therapy in the treatment of acne. The clinical expert advised that patients with acne should not have to be required to try other treatments before initiating treatment with IDP-126 gel due to its major overlap with other current treatment options. In general, the clinical expert indicated that any topical therapy should be used for 3 to 6 months before considering treatment failure. The clinical expert defined lack of response as no improvement or minimal improvement from baseline, as determined by examining the patient. |
Patients with secondary acne were excluded from studies 301 and 302. Could the drug under review be used to treat patients with secondary acne? | The clinical expert suggested it is possible to consider using IDP-126 gel in secondary acne. However, the clinical expert advised that secondary acne would likely be more difficult to treat and may require other therapeutic manipulations (e.g., stopping a medication that is causing the acne, treating a congenital hormonal condition). |
Considerations for discontinuation of therapy | |
What parameters would you consider before discontinuing the drug under review due to ineffectiveness? | The clinical expert indicated the following are considerations for discontinuation of IDP-126 gel: lack of response or minimal response, severity of side effects, and disease progression (i.e., increase in the number of nodules or scar formation). |
CDEC = Canadian Drug Expert Committee; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel.
Clinical Evidence
The objective of this Clinical Review Report is to review and critically appraise the clinical evidence submitted by the sponsor on the beneficial and harmful effects of IDP-126 gel in the treatment of acne in patients aged 12 years and older. The focus is placed on comparing IDP-126 gel with relevant comparators and identifying gaps in the current evidence. Of note, this Reimbursement Review was conducted before the Health Canada Notice of Compliance was issued; the scope was based on the anticipated indication.
A summary of the clinical evidence included by the sponsor in the review of IDP-126 gel is presented in 3 sections, with the critical appraisal of the evidence included at the end of each section. The first section, the systematic review, includes pivotal studies and RCTs that were selected according to the sponsor’s systematic review protocol. The assessment of the certainty of the evidence in this first section using the GRADE approach follows the critical appraisal of the evidence. The second section includes indirect evidence from the sponsor. The third section includes additional studies that were considered by the sponsor to address important gaps in the systematic review evidence.
Included Studies
Clinical evidence from the following is included in the review and appraised in this document:
2 pivotal studies identified in systematic review
2 indirect treatment comparisons
2 additional studies addressing gaps in evidence.
Systematic Review
Contents within this section have been informed by materials submitted by the sponsor. The following has been summarized and validated by the review team.
Description of Studies
Characteristics of the included pivotal studies, studies 301 and 302, are summarized in Table 5.
Table 5: Details of Studies Included in the Systematic Review
Detail | Study 301 | Study 302 |
|---|---|---|
Designs and populations | ||
Study design | Phase III, multicentre, randomized, double-blind, vehicle-controlled, parallel-group efficacy and safety study | |
Locations | A total of 15 study centres in North America: 13 in the US and 2 in Canada | A total of 15 study centres in North America: 12 in the US and 3 in Canada |
Study period | First patient screening: January 17, 2020 Last patient exit: January 18, 2021 | First patient screening: January 27, 2020 Last patient exit: March 25, 2021 |
Randomized (N) | 183 patients, including 122 patients in the IDP-126 gel group and 61 patients in the IDP-126 vehicle gel group | 180 patients, including 120 patients in the IDP-126 gel group and 60 patients in the IDP-126 vehicle gel group |
Inclusion criteria |
| |
Exclusion criteria |
| |
Drugs | ||
Intervention | IDP-126 gel (fixed combination of clindamycin phosphate 1.2%, benzoyl peroxide 3.1%, and adapalene 0.15%) applied topically once daily for 12 weeks | |
Comparator | IDP-126 vehicle gel, applied topically once daily for 12 weeks | |
Study duration | ||
Screening phase | 5 weeks | |
Treatment phase | 12 weeks | |
Outcomes | ||
Coprimary end points |
| |
Secondary, exploratory, and other end points | Secondary (evaluated in the stated order):
Exploratory (evaluated in the stated order):
Other:
Safety:
| |
Publication status | ||
ClinicalTrials.gov ID | NCT04214639 | NCT04214652 |
Publications | Stein Gold L, Lain E, Del Rosso JQ, et al. Clindamycin phosphate 1.2% plus adapalene 0.15% benzoyl peroxide 3.1% gel for moderate-to-severe acne: Efficacy and safety results from randomized phase 3 trials. J Am Acad Dermatol. 2023;89(5):927 to 935.38 | |
Acne-QoL = Acne-Specific Quality of Life; AE = adverse event; EGSS = Evaluator’s Global Severity Score; ADP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; SAE = severe adverse event; TSS = Truncal Severity Score.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Studies 301 and 302 are phase III, multicentre, randomized, double-blinded, vehicle-controlled, parallel-group (identically designed) clinical trials (N = 183 and N = 180, respectively). The primary objective of both studies was to assess the efficacy, safety, and tolerability of a once-daily topical application of IDP-126 gel, compared with its vehicle gel, in patients aged 9 years and older with moderate to severe acne. A total of 15 study centres in North America were included in each study, including 2 study centres in Canada from Study 301 and 3 study centres in Canada from Study 302. Patients who met all inclusion criteria and none of the exclusion criteria were randomized in a 2:1 ratio, using an interactive web response system, to receive either IDP-126 gel or IDP-126 vehicle gel for 12 weeks. Randomization was not stratified.
At each study visit, the evaluator assigned an EGSS based on their assessment of the patient’s face, followed by counting all observed inflammatory and noninflammatory lesions. Although not a requirement for study inclusion, patients may have been assessed for truncal acne, involving the neck, upper chest, upper back, and shoulders. If the patient chose to apply their assigned study drug to the truncal area, a separate assessment was performed to assign a Truncal Severity Score (TSS) at baseline and all postbaseline study visits thereafter. Of note, lesion counts were only applicable to facial acne and not to truncal acne. Effort was made to ensure the same evaluator assessed the same patient at all postscreening study visits (on efficacy outcomes). If this was not possible, then effort was made to ensure the same evaluator assessed the same patient at both the baseline and week 12 visits. Patients who discontinued the study drug and/or study early were asked to complete all week 12 assessments before starting any alternative therapy for acne.
Populations
Inclusion and Exclusion Criteria
A summary of the inclusion and exclusion criteria used in studies 301 and 302 is presented in Table 5.
Patients aged 9 years and older with a clinical diagnosis of moderate or severe facial acne, defined as an EGSS of 3 or 4 at baseline, were included in the studies. Other key inclusion criteria relating to facial acne included an inflammatory lesion (papules, pustules, and nodules) count of 30 to 100, inclusive; a noninflammatory lesion (open and closed comedones) count of 35 to 150, inclusive; and 2 or fewer facial nodules. Key exclusion criteria included patients with greater than 2 facial nodules or any dermatological condition affecting the face that could have interfered with clinical evaluation, such as secondary acne or a history of significant burning or stinging on application of any facial treatment.
Interventions
Study Drug
Study drugs were packaged and labelled identically, numbered sequentially, and dispensed randomly (using an Oracle randomization and supplies system) to patients entering the study within each study centre.
The study drug (IDP-126 gel or IDP-126 vehicle gel with no active ingredient) was topically applied to the face (chin, cheeks, nose, and forehead) evenly once daily in the evening for 12 weeks. For the subset of patients who chose to also treat their truncal acne, the study drug was applied in the same manner (as described for the face) to the neck, upper chest, upper back, and shoulders. The initial application of the assigned study drug was made at the study centre with instruction from the study coordinator or designee. Thereafter, the study drug was applied by the patient at the study centre on study visit days or at home on all subsequent days.
Adherence to the study drug was assessed at each applicable postbaseline study visit. Each pump containing the study drug was weighed before dispensing and after returning at each applicable study visit. Any missed applications of the study drug were captured in a diary calendar that was completed by the patients.
Concomitant and Prior Therapies
Patients were permitted to use only nonmedicated cleansers, moisturizers, and sunscreens approved in the study protocol. Regarding makeup and shaving products, patients were required to use noncomedogenic products. No other topical and/or physical treatment other than the study drug was permitted for acne.
Patients were excluded from study enrolment if they required the concomitant use of medications and/or vitamins that were reported to exacerbate acne, including azathioprine, haloperidol, vitamin D, vitamin B12, halogens, lithium, systemic or topical mid- to super high–potency corticosteroids on the treatment area, phenytoin, or phenobarbital. However, multivitamins, including vitamin A at the recommended daily dose and vitamin D at a sTable dose, were permitted.
Patients were also excluded from study enrolment if they required the concomitant use of potentially irritating over-the-counter products that contained ingredients such as BPO, alpha hydroxy acid, salicylic acid, retinol, or glycolic acids.
Patients who had not undergone the specified washout period, or patients who required the concurrent use of specified topical preparations or physical treatments on the face or systemic therapies, were also excluded from study enrolment. A summary of the washout periods used in studies 301 and 302 is presented in Table 6.
Table 6: Summary of Washout Periods for Topical Preparations, Physical Treatments, and Systemic Therapies in Studies 301 and 302
Treatment | Washout period |
|---|---|
Topical preparations or physical treatments used on the face | |
Topical astringents and abrasives | 1 week |
Disallowed moisturizers or sunscreens | 1 week |
Antibiotics | 2 weeks |
Soaps containing antimicrobials | 2 weeks |
Anti-inflammatory drugs and corticosteroids | 4 weeks |
Retinoids, including retinol | 4 weeks |
Chemical peel, microdermabrasion, light (light-emitting diode, photodynamic therapy) and laser therapy | 4 weeks |
Acne surgery | 4 weeks |
Other topical acne treatments | 2 weeks |
Systemic therapies | |
Corticosteroids, including intramuscular injectionsa | 4 weeks |
Antibiotics | 4 weeks |
Systemic retinoids | 6 months |
Other systemic acne treatments | 4 weeks |
aInhaled corticosteroids were permitted.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Outcomes
A list of efficacy end points assessed in this Clinical Review Report is provided in Table 7, followed by descriptions of the outcome measures. Summarized end points are based on outcomes included in the sponsor’s summary of clinical evidence as well as any outcomes identified as important to this review, according to the clinical expert consulted and the stakeholder input from patient groups and public drug plans. Using the same considerations, the review team selected end points that were considered to be most relevant to inform the expert committee deliberations and finalized this list of end points in consultation with members of the expert committee. All summarized efficacy end points were assessed using GRADE. Select notable harms outcomes considered important for informing the expert committee deliberations were also assessed using GRADE.
Acne Severity
EGSS and Truncal Severity Score
The EGSS and TSS are static assessments (i.e., the score was independently determined at each study visit) of the severity of facial and truncal acne, respectively, performed by the evaluator. The grade ranges from clear (score of 0) to severe (score of 4); the corresponding descriptions for each grade are presented in Table 8. At each study visit, the EGSS was applied to all patients following an evaluation of the face and the TSS was applied to patients who had chosen to also treat their truncal acne. Treatment success was defined as at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear at week 12.
Table 7: Outcomes Summarized From Studies 301 and 302
Outcome measure | Time point | Studies 301 and 302 |
|---|---|---|
Acne severity | ||
Percentage of patients with ≥ 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) | At week 12 | Coprimarya |
Lesion count | ||
Absolute change from baseline in the inflammatory lesion count | At week 12 | Coprimarya |
Percent change from baseline in the inflammatory lesion count | At week 12 | Secondarya |
Absolute change from baseline in the noninflammatory lesion count | At week 12 | Coprimarya |
Percent change from baseline in the noninflammatory lesion count | At week 12 | Secondarya |
HRQoL | ||
Absolute change from baseline in the Acne-QoL self-perception domain score | At week 12 | Other |
Absolute change from baseline in Acne-QoL acne symptom domain score | At week 12 | Other |
Notable harms | ||
General disorders and administration site conditions | At week 12 | Safety |
Skin and subcutaneous tissue disorders | At week 12 | Safety |
SAEs | At week 12 | Safety |
Acne-QoL = Acne-Specific Quality of Life; EGSS = Evaluator’s Global Severity Score; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; GRADE = Grading of Recommendations Assessment, Development and Evaluation; HRQoL = health-related quality of life; SAE = serious adverse event.
Note: Based on input from the patient groups and the clinical expert, the following outcomes are important but were not assessed in studies 301 and 302: treatment failure defined by an increase in scar formation despite a reduction in lesion counts, changes in skin pigmentation, and/or change in mental health.
The select notable harms for assessment using GRADE were based on the product monograph, which indicated that local skin irritation may occur with treatment with IDP-126, including erythema, scaling, dryness, stinging, and/or burning, as well as irritant and allergic contact dermatitis.
aStatistical testing for these end points was adjusted for multiple comparisons.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Table 8: EGSS and TSS in Studies 301 and 302
Score | Grade | Description |
|---|---|---|
0 | Clear | Normal, clear skin with no evidence of acne |
1 | Almost clear | Rare noninflammatory lesions are present, with rare noninflamed papules (papules must be resolving and may be hyperpigmented, though not pink-red) |
2 | Mild | Some noninflammatory lesions are present, with few inflammatory lesions (papules and/or pustules only; no nodulocystic lesions) |
3 | Moderate | Noninflammatory lesions predominate, with multiple inflammatory lesions evident; several to many comedones and papules and/or pustules, and there may or may not be 1 small nodulocystic lesion |
4 | Severe | Inflammatory lesions are more apparent, there are many comedones and papules and/or pustules, there may or may not be up to 2 nodulocystic lesions |
EGSS = Evaluator’s Global Severity Score; TSS = Truncal Severity Score.
Note: The EGSS and TSS used the same grading scale with the same definitions and are static assessments (i.e., all postbaseline determinations were independent of the baseline score).
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Lesion Count
Inflammatory lesions were counted in the following manner: pustules and papules were counted and recorded together, while nodular lesions were counted and recorded separately (but were included in the total inflammatory lesion count and in the statistical analysis of inflammatory lesion counts). Noninflammatory lesions (open and closed comedones) were counted and recorded together. At each study visit, the evaluator counted the total number of lesions on the face based on the description of inflammatory and noninflammatory lesions presented in Table 9.
Table 9: Description of Inflammatory and Noninflammatory Lesions in Studies 301 and 302
Lesion | Description |
|---|---|
Inflammatory lesions | |
Papule | An erythematous, raised, palpable lesion < 5 mm in diameter |
Pustule | An erythematous, raised, likely palpable lesion containing white exudate or pus < 5 mm in diameter |
Nodule | A deep-seated, erythematous, firm lesion > 5 mm in diameter |
Noninflammatory lesions | |
Open comedone (blackhead) | A widely dilated sebaceous follicle plugged with darkly pigmented sebum |
Closed comedone (whitehead) | A small, closed sebaceous follicle distended with sebum, with a white appearance |
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Health-Related Quality of Life
Acne-Specific Quality of Life
The Acne-QoL is a patient-reported, disease-specific, 19-item questionnaire comprising 4 subscales: self-perception, role-emotional, role-social, and acne symptoms. The domain structures of the Acne-QoL are as follows:7
Self-perception: Feeling unattractive, embarrassed, self-conscious; dissatisfied with appearance; and self-confidence that is negatively affected.
Role-emotional: Feeling upset about having facial acne, annoyed about time spent cleaning and treating the face, concerned about not looking their best, concerned about acne medication not working fast enough, and bothered by the need to have medication and cover-up.
Role-social: Feeling concerned about meeting new people and going out in public; socializing is a problem and interacting with others is a problem.
Symptoms: Bumps on the face, bumps full of pus on the face, scabbing from facial acne, concerned about scarring from facial acne, and oily facial skin.
The response options can range from extremely to not at all or extensive to none based on a recall period of 1 week. Responses are summed within each subscale to yield 4 domain scores, where higher scores are indicative of favourable HRQoL.7 The Acne-QoL was completed by patients at baseline and week 12. A summary of this outcome measure and its measurement properties and MID is presented in Table 10.
Harms
AEs included any unfavourable and unintended illness, sign, symptom, clinically significant laboratory test or vital sign abnormality (i.e., symptomatic, required corrective treatment, or led to discontinuation), or disease temporally associated with the use of a drug and appeared or worsened during the study, regardless of a causal relationship to the study drug. Cutaneous safety and/or tolerability signs and symptoms (application site scaling, erythema, hypo- and hyperpigmentation, itching, burning, and stinging) that resulted in a patient requiring concomitant therapy, interruption of study drug use, or discontinuation from the study were reported as AEs.
SAEs were defined as any untoward medical occurrence that, at any dose, was immediately life-threatening; a congenital anomaly, birth defect, or a spontaneous abortion; events that resulted in death or persistent or significant disability or incapacity; events that required hospitalization or prolongation of an existing hospitalization; or a medically important event that may have jeopardized the patient and may have required medical or surgical intervention to prevent any of the aforementioned events.
The collection of AEs was conducted at each study visit; AEs were recorded and classified using the Medical Dictionary for Regulatory Activities, version 22.0. Events that occurred on or after the date of the first application of the study drug were considered TEAEs.
Notable Harms
Based on the product monograph,28 local skin irritation, including erythema, scaling, dryness, and stinging and/or burning, as well as irritant and allergic contact dermatitis, may occur with IDP-126 gel treatment. Thus, general disorders and administration site conditions as well as skin and subcutaneous tissue disorders reported in studies 301 and 302, along with SAEs, were assessed using GRADE.
Table 10: Summary of Outcome Measures in Studies 301 and 302 and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | Minimal important difference |
|---|---|---|---|
Acne-QoL | A patient-completed questionnaire developed to measure the impact of facial acne across 4 dimensions of patient quality of life.7,8 The questionnaire has a 1-week recall period composed of 19 items in 4 subscales: self-perception, role-emotional, role-social, and acne. The domain scores are calculated by adding all items within a specific domain. Responses for all items are numbered on a scale from 0 (extremely or extensive) to 6 (not at all or none), where each item within a domain is weighted equally. Higher scores indicate better HRQoL for all domains.39 | Validity: In a study combining data from 2 randomized, double-blind, placebo-controlled, parallel-group trials for patients with facial acne, an association was found between acne severity as reported by clinicians with acne-specific QoL as reported by patients, demonstrating convergent validity of the Acne-QoL. The discriminant validity was also supported by the very low correlations observed between the Acne-QoL subscale scores and some clinical measures assumed to be unrelated to acne-specific QoL (e.g., blood pressure, heart rate, and height).7 Reliability: In the same study combining data from 2 randomized trials for patients with facial acne, internal consistency (measured by Cronbach alpha) ranged from 0.87 to 0.96 for the self-perception, role-emotional, and role-social subscales of the Acne-QoL, whereas, for the acne symptoms subscale, the range was from 0.77 to 0.86. All 4 subscales demonstrated reliability (alpha > 0.70).7 The reliability has also been established in other study.8 Responsiveness: In the same study combining data from 2 randomized trials for patients with facial acne, responsiveness of the Acne-QoL was demonstrated by its ability to determine both small (baseline to midstudy) and moderate (baseline to study end) treatment advantages for trial patients.7 Results from another study suggested responsiveness of the Acne-QoL to changes in acne severity following 3 to 4 months of usual care treatment.8 | In a study combining data from 2 randomized, double-blind, placebo-controlled, parallel-group trials for patients with moderate facial acne vulgaris, MCID was established using 3 approaches:40 Global patient-reported method: The MCID estimates for each domain were 5.15 for self-perception, 4.73 for role-emotional, 3.08 for role-social, and 4.62 for acne symptoms, which was based on the classification of somewhat improved (those who had experienced a minimal amount of improvement in acne symptoms). This method was the most strenuous among the 3 methods that were used to define the MCID for the Acne-QoL, with a mean change of between 0.77 and 1.03 per item. Clinician-reported measure: The MCID estimates for each domain were 3.36 for self-perception, 3.86 for role-emotional, 2.07 for role-social, and 3.59 for acne symptoms. This method demonstrated a mean change of between 0.52 and 0.77 per item. Rule for Likert-type items: The self-perception, role-emotional, and acne symptoms domains had an MCID value of 2.5 (5 items multiplied by 0.5), whereas the role-social MCID was 2.0 (4 items multiplied by 0.5). These estimates were the least conservative based on the MCIDs generated from the previous 2 methods. A mean change of 0.5 per item was used to define an MCID under this method. Based on these 3 approaches and the domain, an MCID estimated mean change per item ranging from 0.50 to 10.3 was determined for this population.40 |
Acne-QoL = Acne-Specific Quality of Life; HRQoL = health-related quality of life; MCID = minimal clinically important difference; QoL = quality of life.
Statistical Analysis
The statistical analysis plan for studies 301 and 302 was approved on December 17, 2020, based on protocol amendment 1 (June 17, 2020), before the unblinding of the study drug assignments. No changes were made to the prespecified analyses after database lock.
Of note, while all data were summarized in the listings presented by patient, data collected at early discontinuation, and unscheduled visits occurring before study day 8, these data were not included in the analyses of efficacy and safety, except for baseline values. Baseline was defined as the last nonmissing assessment before the first application of the study drug.
Sample Size and Power Calculation
The power calculations were based on the observed results at week 12 from Study 201, summarized in the studies addressing gaps in the systematic review evidence section of this report. A sample size of 120 patients in the IDP-126 gel group and 60 patients in the IDP-126 vehicle gel group had a greater than 99% power to detect the following:
A significant difference in inflammatory lesions at an alpha level of 0.05. The estimated mean absolute changes from baseline in inflammatory lesion counts were 29.9 in the IDP-126 gel group and 19.6 in its vehicle gel group, with an SD of 12.0.
A significant difference in noninflammatory lesions at an alpha level of 0.05. The estimated mean absolute changes from baseline in noninflammatory lesion counts were 35.5 in the IDP-126 gel group and 21.8 in its vehicle gel group, with an SD of 16.4.
The same sample size also had at least 99% power to detect a significant difference in the percentage of patients with at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear at week 12 (i.e., treatment success) at an alpha level of 0.05. The estimated percentages of patients with treatment success based on the EGSS were 52.5% in the IDP-126 gel group and 8.1% in its vehicle gel group.
No interim analysis was planned for the studies.
Definition and Evaluation of Analysis Centres
Each study was conducted under a common protocol for each study centre with the intention of pooling the data for analysis. A minimum of 15 patients, 10 in the IDP-126 gel group and 5 in its vehicle gel group, at each study centre was planned to be included in each study. If the desired minimum sample size for each study drug group was not met at a specific study centre, the data from that study centre were combined with the data from a different study centre within the same geographic region. Specifically, the data from the lowest-enrolling study centre were combined with the data from the highest-enrolling study centre in the same country, and so on (if there was a further need to combine data). The process of combining data from paired study centres redefined the groups of study centres as analysis centres for the purpose of the statistical analysis.
Before evaluating the treatment by analysis centre interaction effect, the magnitude of the study centre effect was assessed to determine whether variability between the study centres could have masked the analysis centre effects. The consistency of the study drug response was also assessed across analysis centres after combining data to form the analysis centres. Statistical tests were conducted to identify any extreme analysis centres that could have impacted the interpretation of the common statistical and clinical conclusions. An evaluation of the treatment by analysis centre interaction effect was included in the primary variable analyses to test for a parallel treatment effect at an alpha level of 0.10.
Statistical Test or Model
The 3 coprimary efficacy end points were as follows:
absolute change from baseline in the inflammatory lesion count at week 12
absolute change from baseline in the noninflammatory lesion count at week 12
percentage of patients with at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear at week 12 (i.e., treatment success).
A summary of the statistical analysis of the coprimary efficacy end points in studies 301 and 302 is presented in Table 11.
Tests of superiority for the absolute change from baseline in mean inflammatory and noninflammatory lesion counts were based on either parametric or nonparametric methods consistent with the statistical assumptions required to support the analyses. Specifically, the tests were based on an analysis of covariance (ANCOVA) or on ranked data submitted to an ANCOVA, with treatment group and analysis centre as factors, and the respective baseline lesion count as a covariate. If a treatment by analysis centre interaction effect was significant at an alpha of less than 0.10, the effect was included in the model; otherwise, it was removed.
A skewness test was applied to the residuals resulting from an ANCOVA. A 2-sided P value for the skewness test that was significant at an alpha of 0.01 implied the use of the nonparametric method. If a parametric analysis was indicated, the results of the parametric analysis were considered the primary analysis results. If a nonparametric analysis was indicated, the absolute changes in noninflammatory and inflammatory lesion counts were rank transformed before being submitted to the ANCOVA. Subsequently, the results of the rank-transformed analyses were considered the primary analysis; regardless, the results of the nonrank-transformed analyses were also presented.
The analysis of treatment success according to EGSS results was based on a logistic regression test with treatment group and analysis centre as factors. Similar to the analysis of lesion counts, if a treatment by analysis centre interaction effect was significant at an alpha of less than 0.10, the effect was included in the model; otherwise, it was removed. If there was a quasi-complete separation of the data that prevented the model from converging, Firth’s penalized likelihood was applied to allow the model to converge.
Multiple Testing Procedure
Type I error was controlled by requiring the coprimary efficacy end points to be statistically significant. To draw a conclusion of superiority for IDP-126 gel relative to its vehicle gel on the coprimary efficacy end points, the analyses of all 3 end points had to yield significant results at an alpha level of 0.05.
To control for the overall type I error, failure of any coprimary efficacy end point invalidated the statistical significance of all secondary efficacy end points. Failure of any secondary efficacy end point invalidated the statistical significance of all supportive efficacy end points. To control for multiplicity, testing of the secondary and supportive efficacy end points followed a gated, sequential testing process in which the testing stopped if a nonsignificant result was obtained, and all subsequent tests for the remaining steps were considered not significant. The order of testing followed the order in which the secondary and supportive efficacy end points are listed in Table 5.
Data Imputation Methods
In the efficacy analyses conducted with the intention-to-treat (ITT) population, missing data on lesion count and EGSS at week 12 (from which treatment success was derived) were derived using the Markov chain Monte Carlo (MCMC) multiple imputation method; imputation was conducted independent of the study drug group. In the efficacy analyses conducted with the per-protocol (PP) population, missing data were imputed using the last observation carried forward approach.
Subgroup Analyses
Subgroup analyses were conducted on the coprimary efficacy end points using the ITT population for the following subgroups: baseline EGSS, sex, age (< median age and ≥ median age; aged < 18 years and aged ≥ 18 years), ethnicity or race, and geographic region.
Of the subgroups prespecified in the studies, the subgroup of patients according to their baseline acne severity (i.e., baseline EGSS) was considered to be most relevant to inform the expert committee deliberations and was therefore included in this report. No inferential statistics were used in the subgroup analyses.
Sensitivity Analyses
Four sensitivity analyses were conducted using the ITT population to evaluate the coprimary efficacy end points: a model-based multiple imputation method, repeated measures modelling, tipping-point analysis, and assessment of outcomes in a subset of patients from the ITT population who were not affected by COVID-19 disruptions.
Secondary Outcomes of the Studies
The secondary efficacy end points were as follows (and were evaluated in the stated order using only the ITT population):
percent change from baseline in noninflammatory lesion count at week 12
percent change from baseline in inflammatory lesion count at weeks 12
percentage of patients with at least a 2-grade reduction from baseline in EGSS at week 12
percent change from baseline in noninflammatory lesion count at week 8
percent change from baseline in inflammatory lesion count at week 8
percent change from baseline in noninflammatory lesion count at week 4
percent change from baseline in inflammatory lesion count at week 4.
Each secondary efficacy end point was analyzed in a manner consistent with the relevant method used to analyze the coprimary efficacy end points. The treatment by analysis centre interaction effect was not tested for the secondary efficacy end points; therefore, it was not included in the model.
Other Outcomes of the Studies
Other efficacy end points based on the Acne-QoL questionnaire were as follows:
self-perception domain score (calculated from questions 1, 2, 3, 6, and 10) at baseline and week 12
role-emotional domain score (calculated from questions 4, 5, 7, 8, and 9) at baseline and week 12
role-social domain score (calculated from questions 11, 12, 13, and 14) at baseline and week 12
acne symptoms domain score (calculated from questions 15, 16, 17, 18, and 19) at baseline and week 12.
Each domain was the sum of the values from the questions it comprised, provided at least 3 of these questions had nonmissing results. For domains that had 3 or more questions with nonmissing results, any missing values were imputed through mean substitution before calculating the domain score. Descriptive statistics were used to summarize the data reported for each Acne-QoL domain. No inferential analyses were conducted, as per the statistical analysis plan.
Table 11: Statistical Analysis of the Coprimary Efficacy End Points in Studies 301 and 302
End point | Statistical model | Adjustment factors | Handling of missing data | Sensitivity analyses |
|---|---|---|---|---|
Absolute change from baseline in inflammatory lesion count at week 12 | ANCOVA, or based on ranked data submitted to an ANCOVA | Factors: Treatment group and analysis centre Covariate: Baseline lesion count | ITT population: MCMC multiple imputation PP population: LOCF method |
|
Absolute change from baseline in noninflammatory lesion count at week 12 | As per row 1 | As per row 1 | As per row 1 | As per row 1 |
Percentage of patients with at least a 2-grade reduction from baseline in the EGSS and with an EGSS of clear or almost clear at week 12 (i.e., treatment success) | Logistic regression test | Factors: Treatment group and analysis centre | As per row 1 | As per row 1 |
ANCOVA = analysis of covariance; EGSS = Evaluator’s Global Severity Score; ITT = intention to treat; LOCF = last observation carried forward; MCMC = Markov chain Monte Carlo; PP = per-protocol.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the table are from the sponsor’s summary of clinical evidence.15
Analysis Populations
A summary of the analysis populations used in studies 301 and 302 is presented in Table 12.
The primary efficacy analyses were performed using the ITT population, defined as all randomized patients who received the study drug. The supportive efficacy analyses were performed using the PP population, defined as all patients who completed the 12-week evaluation without any noteworthy study protocol violations (i.e., any patient or investigator activity that could have possibly interfered with the therapeutic application of the study drug or the evaluation of treatment efficacy).
The safety analyses were performed using the safety population, defined as all randomized patients who were confirmed to have used the study drug at least once.
A separate analysis set, referred to as the patients impacted by COVID-19 disruption, was used to provide listings of data for relevant patients and to identify patients to exclude for sensitivity analyses of the primary and secondary efficacy end points.
Table 12: Analysis Populations in Studies 301 and 302
Population | Definition | Application |
|---|---|---|
ITT population | All randomized patients who received the study drug | Primary efficacy analyses |
PP population | All patients who completed the 12-week evaluation without noteworthy study protocol violationsa | Supportive efficacy analyses |
Safety population | All randomized patients who were confirmed to have used the study drug at least once | Safety analyses |
Patients impacted by COVID-19 disruption | A subset of patients in the ITT population who had ≥ 1 assessment missed, ≥ 1 visit out of the visit window, or discontinued from the study due to COVID-19 disruptions, or had an AE of COVID-19 | Used to provide listings of data for relevant patients and to identify patients to exclude from sensitivity analyses of the primary and secondary efficacy end points |
AE = adverse event; ITT = intention to treat; PP = per-protocol.
aA noteworthy study protocol violation included any patient or investigator activity that could have possibly interfered with the therapeutic application of the study drug or the precise evaluation of treatment efficacy. Before breaking the blind, other criteria could have been added to the list to accommodate for unforeseen events that occurred during the study and resulted in noteworthy study protocol violations. Patients who discontinued from the study due to an AE that was considered by the investigator to be related to the study drug or who experienced a documented lack of treatment effect and/or worsening of their condition were included in the PP population.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Results
Patient Disposition
A summary of patient disposition from studies 301 and 302 is presented in Table 13. A total of 208 patients in Study 301 and 227 patients in Study 302 were screened; the reasons for screening failure were not available. Sample size was achieved in each study; in Study 301, 122 patients were randomized to receive IDP-126 gel, and 61 patients were randomized to receive its vehicle gel and, in Study 302, 120 patients were randomized to receive IDP-126 gel and 60 patients were randomized to receive its vehicle gel.
In Study 301, 12.3% of patients (15 of 122 patients) randomized to receive IDP-126 gel and 9.8% of patients (6 of 61 patients) randomized to receive its vehicle gel discontinued from the study. Similarly, in Study 302, 10.8% of patients (13 of 120 patients) randomized to receive IDP-126 gel and 6.7% of patients (4 of 60 patients) randomized to receive its vehicle gel discontinued from the study. In both studies, any 1 reason for study discontinuation was reported in less than 6.0% of patients randomized to each study drug group. In Study 301, the most common reason for study discontinuation was patient request, reported for 5.7% of patients (7 of 122 patients) in the IDP-126 gel group and 4.9% of patients (3 of 61 patients) in its vehicle gel group. In Study 302, the most common reason was lost to follow-up, reported for 5.0% of patients (6 of 120 patients) in the IDP-126 gel group and 3.3% of patients (2 of 60 patients) in its vehicle gel group.
Table 13: Summary of Patient Disposition From Studies 301 and 302 (All Randomized Patients)
Patient disposition | Study 301 | Study 302 | ||
|---|---|---|---|---|
IDP-126 gel | IDP-126 vehicle gel | IDP-126 gel | IDP-126 | |
Screened, N | 208 | 227 | ||
Reason for screening failure, n (%) | NR | NR | ||
Randomized, N | 122 | 61 | 120 | 60 |
Discontinued from study, n (%) | 15 (12.3) | 6 (9.8) | 13 (10.8) | 4 (6.7) |
Reason for study discontinuation, n (%) | — | — | — | — |
Patient request | 7 (5.7) | 3 (4.9) | 1 (0.8) | 1 (1.7) |
Lost to follow-up | 5 (4.1) | 1 (1.6) | 6 (5.0) | 2 (3.3) |
AEs | 2 (1.6) | 0 | 3 (2.5) | 0 |
Disruption caused by COVID-19 | 1 (0.8) | 0 | 3 (2.5) | 0 |
Withdrawal by parent or guardian | 0 | 1 (1.6) | 0 | 0 |
Progressive disease | 0 | 1 (1.6) | 0 | 0 |
Lack of efficacy | 0 | 0 | 0 | 1 (1.7) |
ITT, N | 122 | 61 | 120 | 60 |
PP, N | 97 | 51 | 89 | 44 |
Safety, N | 122 | 61 | 120 | 60 |
AE = adverse event; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; NR = not reported; PP = per-protocol.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Protocol Deviations
A summary of protocol deviations from studies 301 and 302 is presented in Table 14. ██ █████ ████ █████ ██ ████████ ███ ██ ███ █████████ ██████████ ██ ███████ ███████ ███ ███ █████ ██ ████████ ███ ██ ██ █████████ ██████████ ██ ███████ ███ ███████ ███ ████ ████████ ████ ███ ██ ███████████ ██████████ ██ █████ ████ █████ ██ ████████ ███ ██ ███ █████████ ██████████ ██ ███████ ███████ ███ ███ █████ ██ ████████ ███ ██ ██ █████████ ██████████ ██ ███████ ███ ███████ ███ ████ ████████ ████ ███ ██ ███████████ ████ ███ █████████ ██ █████ ██ ████████ ███ ██ ███ █████████ ██ ███ ███████ ███ █████ ███ █████ ██ ████████ ██ ██ ██ █████████ ██ ███ ███████ ███ █████ ██████ ███ █ ████ ██ █████ ███████ ███ █████████ █████ ██████ ██ █████ ████ ███ ██████ ███ █████████ ████ ███ ██ ██████████ ███ ████████ ██ ████ ████ █████ ██ ████████ ██ ██████████ ██ ████ █████ ████ █████ ████ ████ ███████
Table 14: Summary of Protocol Deviations From Studies 301 and 302 (All Randomized Patients)
Patient disposition | Study 301 | Study 302 | ||
|---|---|---|---|---|
IDP-126 gel | IDP-126 vehicle gel | IDP-126 gel | IDP-126 | |
████████ ████████ ████ ███ ██ ███████████ █ ███ | ██ █████ | ██ █████ | ██ █████ | ██ █████ |
██████ ███ █████████ ████ ███ ██ ███████████ █ ███ | — | — | — | — |
███ ███ ████ █████████ ██████ █████████ ████████ | | █████| | ||| | | █████| | | █████| |
████ ██ ███████████ ███████████ ██████████ | | █████ | | █████ | ||| | | █████ |
███ ███ ██████ ███ ████ ██ █████ | ██ █████ | | █████ | ██ █████ | | █████ |
███ ███ ████████ ██████ ██████ ███ ████ ██ ████ ██ | ||| | ||| | | █████ | | █████ |
███ ███ ████████ ██ ███████████ ███████ | | █████ | | █████ | | █████ | | █████ |
███ █ ████ ██ █████ ███ ██ ██████ ███ ███ ██ █████| | | █████ | | █████ | ██ █████ | | ██████ |
██ █ █████████████
██████ ███ █████ █████ ████████ ███ ███████ ██████ █████████ ██ ███ █████ █████████ ██ ███ ██████ ███ █████ █ ███████ ███ ████████ ████ ███ ██ ███████████ ███████ ███ █████████ ███ ███████ ██ ██ ███████ ██ ██████ █████ ████ █████ ███ ████████ ███ ███ ████████ ███████ ██████ ██████ ██ █████ ██ ██ ██████ █ ███ ██ ████████ ██████████ ████ ███ ████████ ██ ██████ ██████ ███ ███ ███████ ██ █████████ ████████████ ██ ███ ██ ████████████| ███ ███████ ███ ███ █████ ███████ ███ █████████ ███████ ██████ ███ █████ █████████████| ██ ███ ███████ ███ ██████ █ ███████ ███ ███ █████ ███████ ███ █████████ ███████ ██████ ███ █████ ███████████ ███ █ ███████ ███ ███ ████ ███ █████████████ ██████████████ ██ ███ ███████ ███████ ███ ██████ █ ███████ ███ ███ █████ ███████ ███ █████████ ███████ ██████ ███ █████ ███████████ ███ █ ███████ ████ ██ ████████ ███████████ ████████████| ████████ ████ ██ █████████ ████ ███ ██████████| ███ ████ ██ █████ ██████ ███ ████████ ██ █ ████ ███████ ███ ██ ████ █████ ███ ████████ ███ ████ ████████ ██ █ ████████ ███████████
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13
Baseline Characteristics
A summary of baseline characteristics from studies 301 and 302 is presented in Table 15. The baseline characteristics outlined in this Table are limited to those that are most relevant to this review or were felt to affect the outcomes or interpretation of the study results.
The mean age of patients randomized to each study drug group was similar: 20.2 years (SD = 7.11 years) in the IDP-126 gel group and 19.8 years (SD = 6.28 years) in its vehicle gel group in Study 301, and 20.2 years (SD = 7.50 years) in the IDP-126 gel group and 21.4 years (SD = 7.49 years) in its vehicle gel group in Study 302. The age range of patients was similar within and between studies, ranging from 10 to 44 years in Study 301 and 10 to 48 years in Study 302.
Of those randomized to each study drug group, 61.5% of patients (75 of 122 patients) in the IDP-126 gel group and 50.8% of patients (31 of 61 patients) in its vehicle gel group from Study 301, and 57.5% of patients (69 of 120 patients) in the IDP-126 gel group and 61.7% of patients (37 of 60 patients) in its vehicle gel group from Study 302 were female. The remainder of patients were male: 38.5% of patients (47 of 122 patients) in the IDP-126 gel group and 49.2% (30 of 61 patients) in its vehicle gel group from Study 301, and 42.5% of patients (51 of 120 patients) in the IDP-126 gel group and 38.3% of patients (23 of 60 patients) in its vehicle gel group from Study 302.
Most patients randomized to each study drug group were white: 61.5% of patients (75 of 122 patients) in the IDP-126 gel group and 73.8% of patients (45 of 61 patients) in its vehicle gel group from Study 301, and 78.3% of patients (94 of 120 patients) in the IDP-126 gel group and 88.3% of patients (53 of 60 patients) in its vehicle gel group from Study 302. Other races included in the studies were Black or African American and Asian. Study 301 included 1 patient who was native Hawaiian or other Pacific Islander in each study drug group, and neither study included patients who were American Indian or Alaska Native.
The majority of patients in each study drug group had a clinical diagnosis of moderate acne, defined as a baseline EGSS of 3: 87.7% of patients (107 of 122 patients) in the IDP-126 gel group and 95.1% of patients (58 of 61 patients) in its vehicle gel group from Study 301, and 90.8% of patients (109 of 120 patients) in the IDP-126 gel group and 95.0% of patients (57 of 60 patients) in its vehicle gel group from Study 302. The remainder of patients in each study drug group had a clinical diagnosis of severe acne, defined as a baseline EGSS of 4: 12.3% of patients (15 of 122 patients) in the IDP-126 gel group and 4.9% of patients (3 of 61 patients) in its vehicle gel group from Study 301, and 9.2% of patients (11 of 120 patients) in the IDP-126 gel group and 5.0% of patients (3 of 60 patients) in its vehicle gel group from Study 302.
The mean inflammatory lesion count at baseline was similar between the study drug groups: 36.4 (SD = 7.52) in the IDP-126 gel group and 37.1 (SD = 9.22) in its vehicle gel group from Study 301, and 37.4 (SD = 7.94) in the IDP-126 gel group and 37.7 (SD = 9.43) in its vehicle gel group from Study 302. The inflammatory lesion count range was also similar within and between studies, ranging from 30 to 82 in Study 301 and 30 to 79 in Study 302. The mean noninflammatory lesion count at baseline was similar between the study drug groups: 50.7 (SD = 19.38) in the IDP-126 gel group and 45.9 (SD = 14.80) in its vehicle gel group from Study 301, and 48.2 (SD = 14.92) in the IDP-126 gel group and 49.3 (SD = 15.94) in its vehicle gel group from Study 302. The inflammatory lesion count range was also similar within and between studies, ranging from 35 to 144 in Study 301 and 35 to 108 in Study 302.
Table 15: Summary of Baseline Characteristics From Studies 301 and 302 (ITT Population)
Characteristic | Study 301 | Study 302 | ||
|---|---|---|---|---|
IDP-126 gel | IDP-126 | IDP-126 gel | IDP-126 | |
Patient demographic characteristics | ||||
Age (years) | — | — | — | — |
Mean (SD) | 20.2 (7.11) | 19.8 (6.28) | 20.2 (7.50) | 21.4 (7.49) |
Median (range) | 17.0 (10 to 42) | 18.0 (12 to 44) | 17.0 (10 to 48) | 19.5 (11 to 43) |
Sex, n (%) | — | — | — | — |
Male | 47 (38.5) | 30 (49.2) | 51 (42.5) | 23 (38.3) |
Female | 75 (61.5) | 31 (50.8) | 69 (57.5) | 37 (61.7) |
Race, n (%) | — | — | — | — |
White | 75 (61.5) | 45 (73.8) | 94 (78.3) | 53 (88.3) |
Black or African American | 28 (23.0) | 9 (14.8) | 12 (10.0) | 5 (8.3) |
Asian | 13 (10.7) | 4 (6.6) | 8 (6.7) | 1 (1.7) |
Native Hawaiian or other Pacific Islander | 1 (0.8) | 1 (1.6) | 0 | 0 |
American Indian or Alaska Native | 0 | 0 | 0 | 0 |
Not reported or multiple | 5 (4.1) | 2 (3.3) | 6 (5.0) | 1 (1.7) |
Patient characteristics relating to acne | ||||
EGSS, n (%) | — | — | — | — |
0 (clear) | 0 | 0 | 0 | 0 |
1 (almost clear) | 0 | 0 | 0 | 0 |
2 (mild) | 0 | 0 | 0 | 0 |
3 (moderate) | 107 (87.7) | 58 (95.1) | 109 (90.8) | 57 (95.0) |
4 (severe) | 15 (12.3) | 3 (4.9) | 11 (9.2) | 3 (5.0) |
Inflammatory lesion count | — | — | — | — |
Mean (SD) | 36.4 (7.52) | 37.1 (9.22) | 37.4 (7.94) | 37.7 (9.43) |
Median (range) | 34.0 (30 to 78) | 34.0 (30 to 82) | 34.5 (30 to 77) | 34.0 (30 to 79) |
Noninflammatory lesion count | ||||
Mean (SD) | 50.7 (19.38) | 45.9 (14.80) | 48.2 (14.92) | 49.3 (15.94) |
Median (range) | 42.0 (35 to 144) | 41.0 (35 to 120) | 42.0 (35 to 108) | 42.0 (35 to 107) |
EGSS = Evaluator’s Global Severity Score; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; SD = standard deviation.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Exposure to Study Treatments
A summary of patient exposure from Study 301 and Study 302 is presented in Table 16. The mean total number of days of exposure to the study drug was similar across studies: 81.1 days (SD = 14.85 days) in the IDP-126 gel group and 83.6 days (SD = 7.99 days) in its vehicle gel group from Study 301, and 82.0 days (SD = 14.26 days) in the IDP-126 gel and 82.8 days (SD = 12.69 days) in its vehicle gel group from Study 302.
The mean total number of applications administered was similar across studies | ████ ███ █ ██████ ██ ███ ███████ ███ █████ ███ ████ ███ █ █████ ██ ███ ███████ ███ █████ ████ █████ ███ ███ ████ ███ █ ██████ ██ ███ ███████ ███ █████ ███ ████ ███ █ ██████ ██ ███ ███████ ███ █████ ████ █████ ████ ███ ████████ ██ ████████ ██ ████ █████ ████ █████ ███ ███ ████ ████ ████ █ ███████████ ████ ██ █████████ ███ ███████ ███ ██ ████ ██ ███ ████████ ████████████ █████ █████████████ ██ ███ █████ ██████ ████████ ██ ███ ██████ █████████ █████████████ █████ ██ ████████ ████ ██ ███ █████████ ██ ███ ███████ ███ █████ ███ █████ ██ ████████ ███ ██ ██ █████████ ██ ███ ███████ ███ █████ ████ █████ ███ ███ █████ ██ ████████ ████ ██ ███ █████████ ██ ███ ███████ ███ █████ ███ █████ ██ ████████ ███ ██ ██ █████████ ██ ███ ███████ ███ █████ ████ █████ ███ ████ ████████ ██ █████ ██████ ████████
Table 16: Summary of Patient Exposure From Studies 301 and 302 (Safety Population)
Study drug exposure | Study 301 | Study 302 | ||
|---|---|---|---|---|
IDP-126 gel | IDP-126 | IDP-126 gel | IDP-126 | |
Number of days of exposure | — | — | — | — |
n | 116 | 58 | 114 | 58 |
Mean (SD) | 81.1 (14.85) | 83.6 (7.99) | 82.0 (14.26) | 82.8 (12.69) |
██████ ███████ | ███ █ █ ███ | ██ ███ ██ ███ | ███ ██ ██ ██ | ███ █ ██ ███ |
Amount of study drug used (g) | — | — | — | — |
||| | ███ | ██ | ███ | ██ |
████ ████ | █████ ████████ | █████ ██████ | █████ ████████ | █████ ████████ |
██████ ███████ | ██████ ██ ████ | █████ █████ ██ ██████ | █████ ████ ██ ██████ | █████ ████ ██ ██████ |
Total number of applications | — | — | — | — |
||| | ███ | ██ | ███ | ██ |
████ ████ | ████ █████ | ████ ██████ | ████ █████ | ████ ██████ |
██████ ███████ | ██ █ ██ ███ | ██ ███ ██ ████ | ██ ██ ██ ███ | ███ ██ ██ ███ |
Adherencea | — | — | — | — |
||| | ███ | ██ | ███ | ██ |
████ █ ███ | ███ █████ | ██ ██████ | ███ ██████ | ██ ██████ |
███ █ ███ | ██ █████ | | █████ | ██ █████ | | █████ |
IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; SD = standard deviation.
aA patient was considered adherent to the dosing regimen if the patient did not miss more than 5 consecutive days of dosing and applied 80% to 120% of the expected applications while participating in the study.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Efficacy
A summary of the key primary efficacy analysis results from Study 301 and Study 302 is presented in Table 17. For the primary analysis of efficacy, the ITT population included 122 patients in the IDP-126 gel group and 61 patients in its vehicle gel group from Study 301, and 120 patients in the IDP-126 gel group and 60 patients in its vehicle gel group from Study 302.
Four sensitivity analyses, which are described in the preceding statistical analysis section, were conducted to assess the robustness of the coprimary efficacy results. Overall, the results of the sensitivity analyses as well as the supportive efficacy analyses conducted with the PP population were similar to and supportive of the coprimary efficacy analysis results in both Study 301 and Study 302.
Acne Severity
Treatment Success Based on the EGSS
Study 301: The percentage of patients with at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 was 49.6% (95% CI, 40.3% to 58.1%) in the IDP-126 gel group versus 24.9% (95% CI, 13.8% to 35.4%) in its vehicle gel group. The treatment difference in treatment success based on the EGSS at week 12 between IDP-126 gel and its vehicle gel was 24.7% (95% CI, 10.7% to 38.7%; P value = 0.003), in favour of IDP-126 gel.
Study 302: The percentage of patients who experienced treatment success at week 12 was 50.5% (95% CI, 41.1% to 58.9%) in the IDP-126 gel group versus 20.5% (95% CI, 9.9% to 30.1%) in its vehicle gel group. The treatment difference in treatment success based on the EGSS at week 12 between IDP-126 gel and its vehicle gel was 30.0% (95% CI, 16.4% to 43.6%; P value = 0.001), also in favour of IDP-126 gel.
Lesion Count
Inflammatory Lesion Count
Study 301: The LS mean change from baseline in inflammatory lesion count at week 12 was −27.7 (SD = 9.55; 95% CI, −29.4 to −26.0) in the IDP-126 gel group versus −21.7 (SD = 8.79; 95% CI, −23.9 to −19.5) in its vehicle gel group. The treatment difference in the mean absolute change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −5.94 (95% CI, −8.73 to −3.14; P value < 0.001), in favour of IDP-126 gel.
The LS mean percent change from baseline in inflammatory lesion count at week 12 was −75.70% (SD = 26.663%; 95% CI not reported) in the IDP-126 gel group versus −59.62% (SD = 24.348%; 95% CI not reported) in its vehicle gel group. The treatment difference in the mean percent change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −16.08% (95% CI, −23.72% to −8.44%; P value < 0.001), also in favour of IDP-126 gel.
Study 302: The LS mean change from baseline in inflammatory lesion count at week 12 was −30.1 (SD = 9.64; 95% CI, −31.8 to 28.4) in the IDP-126 gel group versus −20.8 (SD = 9.90; 95% CI, −23.3 to −18.3) in its vehicle gel group. The treatment difference in the mean absolute change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −9.30 (95% CI, −12.38 to −6.23; P value < 0.001), also in favour of IDP-126 gel.
The LS mean percent change from baseline in inflammatory lesion count at week 12 was −80.13% (SD = 25.274%; 95% CI not reported) in the IDP-126 gel group versus −56.18% (SD = 25.149%; 95% CI not reported) in its vehicle gel group. The treatment difference in the mean percent change from baseline in inflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −23.95% (95% CI, −31.73% to −16.16%; P value < 0.001), also in favour of IDP-126 gel.
Noninflammatory Lesion Count
Study 301: The LS mean change from baseline in noninflammatory lesion count at week 12 was −35.4 (SD = 15.52; 95% CI, −38.2 to −32.6) in the IDP-126 gel group versus −23.5 (SD = 14.93; 95% CI, −27.2 to −19.8) in its vehicle gel group. The treatment difference in the mean absolute change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −11.85 (95% CI, −16.56 to −7.14; P value < 0.001), in favour of IDP-126 gel.
The LS mean percent change from baseline in noninflammatory lesion count at week 12 was −72.70% (SD = 32.364%; 95% CI not reported) in the IDP-126 gel group versus −47.61% (SD = 31.069%; 95% CI not reported) in its vehicle gel group. The treatment difference in the mean percent change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −25.09% (95% CI, −34.96% to −15.22%; P value < 0.001), also in favour of IPD-126 gel.
Study 302: The LS mean change from baseline in noninflammatory lesion count at week 12 was −35.2 (SD = 14.48; 95% CI, −37.8 to −32.6) in the IDP-126 gel group versus −22.0 (SD = 14.27; 95% CI, −25.6 to −18.4) in its vehicle gel group. The treatment difference in the mean absolute change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −13.27 (95% CI, −17.74 to −8.80; P value < 0.001), also in favour of IDP-126 gel.
The LS mean percent change from baseline in noninflammatory lesion count at week 12 was −73.26% (SD = 27.640%; 95% CI not reported) in the IDP-126 gel group versus −48.99% (SD = 27.345%; 95% CI not reported) in its vehicle gel group. The treatment difference in the mean percent change from baseline in noninflammatory lesion count at week 12 between IDP-126 gel and its vehicle gel was −24.27% (95% CI, −32.86% to −15.68%; P value < 0.001), also in favour of IPD-126 gel.
Health-Related Quality of Life
Self-Perception Domain Score in the Acne-Specific Quality of Life Questionnaire
█████ ███ █ ███ ████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ███ ███ ███ █ █████ ██ ███ ███████ ███ █████ ██████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ ███████████ ███ █ ███ ████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ███ ███ ███ █ █████ ██ ███ ███████ ███ █████ ██████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ █████
Symptoms Domain Score in the Acne-Specific Quality of Life Questionnaire
█████ ███ █ ███ ████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ███ ███ ███ █ █████ ██ ███ ███████ ███ █████ ██████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ ███████████ ███ █ ███ ████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ███ ███ ███ █ █████ ██ ███ ███████ ███ █████ ██████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ ███████ ███ ███ ███ ████ ███ ███ ██ █████
Table 17: Summary of Key Efficacy Results From Studies 301 and 302 (ITT Population)
Efficacy outcome | Study 301 | Study 302 | ||
|---|---|---|---|---|
IDP-126 gel | IDP-126 | IDP-126 gel | IDP-126 | |
Acne severity | ||||
Percentage of patients with ≥ 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 | ||||
Treatment success,a % (95% CI) | 49.6 (40.3 to 58.1) | 24.9 (13.8 to 35.4) | 50.5 (41.1 to 58.9) | 20.5 (9.9 to 30.1) |
Treatment difference, % (95% CI) | 24.7 (10.7 to 38.7) | 30.0 (16.4 to 43.6) | ||
P valuea | 0.003 | 0.001 | ||
Lesion count | ||||
Absolute change from baseline in the inflammatory lesion count at week 12 | ||||
Baseline lesion count, mean (SD) | 36.4 (7.52) | 37.1 (9.22) | 37.4 (7.94) | 37.7 (9.43) |
Week 12 lesion count, mean (SD) | 8.6 (8.17) | 15.4 (11.49) | 7.6 (7.55) | 17.4 (13.17) |
LS change from baseline, mean (SD)b | −27.7 (9.55) | −21.7 (8.79) | −30.1 (9.64) | −20.8 (9.90) |
95% CI | −29.4 to −26.0 | −23.9 to −19.5 | −31.8 to 28.4 | −23.3 to −18.3 |
Treatment difference, mean (95% CI) | −5.94 (−8.73 to −3.14) | −9.30 (−12.38 to −6.23) | ||
P value | < 0.001b < 0.001c | < 0.001b < 0.001c | ||
Percent change from baseline in the inflammatory lesion count at week 12 | ||||
LS change from baseline, mean (SD)b | −75.70 (26.663) | −59.62 (24.348) | −80.13 (25.274) | −56.18 (25.149) |
Treatment difference, mean (95% CI) | −16.08 (−23.72 to −8.44) | −23.95 (−31.73 to −16.16) | ||
P value | < 0.001b < 0.001c | < 0.001b < 0.001c | ||
Absolute change from baseline in the noninflammatory lesion count at week 12 | ||||
Baseline lesion count, mean (SD) | 50.7 (19.38) | 45.9 (14.80) | 48.2 (14.92) | 49.3 (15.94) |
Week 12 lesion count, mean (SD) | 14.5 (14.19) | 24.4 (20.34) | 13.5 (12.22) | 27.5 (22.76) |
LS change from baseline, mean (SD)b | −35.4 (15.52) | −23.5 (14.93) | −35.2 (14.48) | −22.0 (14.27) |
95% CI | −38.2 to −32.6 | −27.2 to −19.8 | −37.8 to −32.6 | −25.6 to −18.4 |
Treatment difference, mean (95% CI) | −11.85 (−16.56 to −7.14) | −13.27 (−17.74 to −8.80) | ||
P value | < 0.001b < 0.001c | < 0.001b < 0.001c | ||
Percent change from baseline in the noninflammatory lesion count at week 12 | ||||
LS change from baseline, mean (SD)b | −72.70 (32.364) | −47.61 (31.069) | −73.26 (27.640) | −48.99 (27.345) |
Treatment difference, mean (95% CI) | −25.09 (−34.96 to −15.22) | −24.27 (−32.86 to −15.68) | ||
P value | < 0.001b < 0.001c | < 0.001b < 0.001c | ||
HRQoL | ||||
Absolute change from baseline in Acne-QoL self-perception domain score at week 12d | ||||
|| | ███ | ██ | ███ | ██ |
█████ ██████ ████ ████ | ████ █████ | ████ █████ | ████ █████ | ████ █████ |
███ ██ ████ ████ ████ | ████ █████ | ████ █████ | ████ █████ | ████ █████ |
██████ ████ █████████ ████ ████ | ███ ██████ | ███ ██████ | ███ ██████ | ███ ██████ |
██████ ████████ ████ ████ ███ | ███ ████ ██ ████ | ███ ████ ██ ████ | ||
Absolute change from baseline in Acne-QoL acne symptoms domain score at week 12d | ||||
|| | ███ | ██ | ███ | ██ |
█████ █████ ████ ████ | ████ █████ | ████ █████ | ████ █████ | ████ █████ |
███ ██ █████ ████ ████ | ████ █████ | ████ █████ | ████ █████ | ████ █████ |
██████ ████ █████████ ████ ████ | ███ █████ | ███ ██████ | ███ ██████ | ███ ██████ |
█████████ ████████ ████ ████ ███ | ███ ████ ██ ████ | ███ ████ ██ ████ | ||
Acne-QoL = Acne-Specific Quality of Life; ANCOVA = analysis of covariance; CI = confidence interval; EGSS = Evaluator’s Global Severity Score; HRQoL = health-related quality of life; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; LS = least squares; SD = standard deviation.
Note: Type I error was controlled by requiring the coprimary efficacy end points to be statistically significant and failure of any coprimary efficacy end points invalidated the statistical significance of all secondary efficacy end points. Multiple imputation (MCMC) was used to impute missing values in the efficacy analyses conducted with the ITT population, missing data on lesion count, and EGSS at week 12 (from which treatment success was derived).
aEstimates and P value are from a logistic regression (using Firth’s penalized likelihood) with treatment group and analysis centre as factors.
bLS means, SDs, difference in LS means and associated 95% CIs, and treatment P values were from an ANCOVA, with treatment group and analysis centre as factors and the respective baseline lesion count as a covariate. For the inflammatory lesion count ANCOVA (coprimary end point), the treatment by analysis centre interaction effects yielded P values of less than 0.10 and were therefore included in the model. Specifically, 1 analysis centre was considered an extreme analysis centre for the evaluation of this particular end point; however, it was observed that its exclusion did not meaningfully affect the results of the absolute change from baseline in inflammatory lesion counts at week 12. Negative LS mean values represent reduction from baseline.
cIn the primary analyses of the absolute and percent changes from baseline in inflammatory and noninflammatory lesion counts, the skewness P value was less than 0.01; this indicated the use of a nonparametric method in which the changes in lesion counts were rank transformed before being submitted to the ANCOVA. The P value was from a ranked ANCOVA with treatment group and analysis centre as factors and the respective baseline lesion count as a covariate. For the inflammatory lesion count ranked ANCOVA (coprimary end point), the treatment by analysis centre interaction effects yielded P values of less than 0.10 and were therefore included in the model.
dChange from baseline was calculated as the baseline score subtracted from the score at week 12; a positive mean change indicates a favourable result. There was no imputation of missing values. Multiplicity of the other efficacy end points, including Acne-QoL outcomes, was not controlled for.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13 Details included in the Table are from the sponsor’s summary of clinical evidence15 and sponsor response to the April 8, 2024, request for additional information regarding the review of IDP-126 gel.16
Subgroup Analysis Results
A summary of the coprimary efficacy results by baseline acne severity from Study 301 and Study 302 is presented in Table 18. The coprimary efficacy results by the subgroup of patients who had a clinical diagnosis of moderate acne, defined as a baseline EGSS of 3, were similar to the primary analysis results in both Study 301 and Study 302.
Of note, the sample size of the subgroup of patients who had a clinical diagnosis of severe acne, defined as a baseline EGSS of 4, was 15 patients in the IDP-126 gel group and 3 patients in its vehicle gel group from Study 301, and 11 patients in the IDP-126 gel group and 3 patients in its vehicle gel group from Study 302.
Table 18: Summary of Coprimary Efficacy Results by Baseline Acne Severity From Studies 301 and 302 (ITT Population)
Subgroup and efficacy outcome | Study 301 | Study 302 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Baseline EGSS of moderate | Baseline EGSS of severe | Baseline EGSS of moderate | Baseline EGSS of severe | ||||||||
IDP-126 gel | IDP-126 | IDP-126 gel | IDP-126 | IDP-126 gel | IDP-126 | IDP-126 gel | IDP-126 | ||||
Acne severity | |||||||||||
Percentage of patients with ≥ 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 | |||||||||||
███████ ██████ █ ████ ███ | █████ ████ | █████ ████ | █████ ████ | || | ████ ████ | █████ ████ | █████ ████ | || | |||
Lesion count | |||||||||||
Absolute change from baseline in the inflammatory lesion count at week 12 | |||||||||||
█████ ██████ | █████ ██████ | █████ █████ | █████ █████ | █████ █████ | ████ ████ | █████ █████ | █████ █████ | █████ █████ | |||
Absolute change from baseline in the noninflammatory lesion count at week 12 | |||||||||||
██████ ████████ | █████ ██████ | █████ █████ | █████ █████ | █████ █████ | ████ ████ | █████ █████ | █████ █████ | █████ █████ | |||
EGSS = Evaluator’s Global Severity Score; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; NA = not available; SD = standard deviation.
Notes: Multiple imputation (MCMC) was used to impute missing values. Summary statistics represent average values, obtained from averaging the summary statistics generated from each imputed dataset. Negative absolute change values represent a reduction from baseline.
Sources: Study V01-126A-301 Clinical Study Report12 and Study V01-126A-302 Clinical Study Report.13
Harms
A summary of harms results from Study 301 and Study 302 (pooled data) is presented in Table 19.
Per the study protocols for Study 301 and Study 302, cutaneous safety and tolerability assessments were to be reported as AEs only if they resulted in the need for concomitant therapy, interruption of study drug administration, or patient discontinuation. However, this protocol specification was not consistently followed, and some cutaneous safety and tolerability assessments were captured as AEs, despite not meeting the preceding reporting definition. A total of 18 patients in the pooled IDP-126 gel group and 1 patient in its pooled vehicle gel group had 1 or more AEs associated with cutaneous safety or tolerability assessments that did not qualify to be reported as AEs and, as such, were excluded in the summary.
Adverse Events
The proportion of patients who were confirmed to have applied their assigned study drug at least once and experienced at least 1 TEAE was 21.9% of patients (53 of 242 patients) in the IDP-126 gel group and 7.4% of patients (9 of 121 patients) in its vehicle gel group. The most common TEAE reported was application site pain in 9.1% of patients (22 of 242 patients) in the IDP-126 gel group and 0.8% of patients (1 of 121 patients) in its vehicle gel group.
Serious Adverse Events
There were no reports of patients with SAEs in either study.
Withdrawal Due to Adverse Events
The proportion of patients who were confirmed to have applied their assigned study drug at least once and who stopped the study drug and/or withdrew from the study due to a TEAE was 2.9% of patients (7 of 242 patients) in the IDP-126 gel group and no patients in its vehicle gel group. The most common TEAEs reported to have led to discontinuation of the study drug and/or from the study was application site pain and erythema; each TEAE was reported in 0.8% of patients (2 of 242 patients) in the IDP-126 gel group.
Mortality
There were no reports of patients who died in either study.
Notable Harms
A total of 9.1% of patients (22 of 242 patients) in the IDP-126 gel group and 0.8% of patients (1 of 121 patients) in its vehicle gel group were reported with a TEAE categorized as a general disorder and administrative site condition.
A total of 2.9% of patients (7 of 242 patients) in the IDP-126 gel group and 0.8% of patients (1 of 121 patients) in its vehicle gel group were reported with a TEAE categorized as a skin and subcutaneous tissue disorder.
Table 19: Summary of Harms Results From Studies 301 and 302 (Safety Population)
AEs | Study 301 and 302 (pooled) | |
|---|---|---|
IDP-126 gel | IDP-126 vehicle gel | |
AEs, n (%) | ||
Patients with ≥ 1 TEAE | 53 (21.9) | 9 (7.4) |
Most common TEAEsa | — | — |
General disorders and administration site conditions | 33 (13.6) | 1 (0.8) |
Application site pain | 22 (9.1) | 1 (0.8) |
Application site dryness | 5 (2.1) | 0 |
Application site exfoliation | 3 (1.2) | 0 |
Application site irritation | 3 (1.2) | 0 |
Application site dermatitis | 2 (0.8) | 0 |
Skin and subcutaneous tissue disorders | 7 (2.9) | 1 (0.8) |
Erythema | 3 (1.2) | 0 |
Dermatitis contact | 2 (0.8) | 0 |
██████████████ | | █████ | | █████ |
██████████ ████ ██████ | | █████ | || |
█████ ████████ █████████ | || | | █████ |
███████████ █████████ | | █████ | || |
██████████ | | █████ | || |
███████ ███████ ███████ █ ███ | ||
████████ ████ | | ███ | || | || |
████████ ███ ███████ █████ ████ ██████ █████ ███ ██ █ █████ █ ███ | ||
██████ ███ █████ ███ ██████ | | █████ | || |
█████ ███████ ███ ██████ ████ ██████████ | | █████ | || |
███████████ ████ ████ | | █████ | || |
██████████ ████ ████████ | | █████ | || |
█████████ ████ ████████ | | █████ | || |
█████████ ████ ████████ | | █████ | || |
███████████ ████ ████ | | █████ | || |
█████████ ████ ███████ | | █████ | || |
████ ███ ████████████ ███████ █████████ | | █████ | || |
████████ | | █████ | || |
████████ ████ | | █████ | || |
Deaths, n (%) | ||
Patients who died | 0 | 0 |
AE = adverse event; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; SAE = serious adverse event; TEAE = treatment-emergent adverse event.
Note: AEs were coded using the Medical Dictionary for Regulatory Activities, version 22.0. TEAEs are those with an onset on or after the date of the first application of the study drug.
The preceding Table excludes cutaneous safety and tolerability assessments that did not result in the patient requiring a concomitant therapy, interruption of the study drug, or discontinuation from the study. Per the study protocols for Study 301 and Study 302, cutaneous safety and tolerability assessments were to be reported as AEs only if they resulted in the need for concomitant therapy, interruption of study drug administration, or patient discontinuation. This protocol specification was not consistently followed, and some cutaneous safety and tolerability assessments were captured as AEs, despite not meeting the preceding reporting definition. As such, AE summary tables were generated that included all reported AEs and, separately, that excluded AEs related to cutaneous safety and tolerability assessments that did not qualify to be reported as AEs. Overall, 18 patients in the pooled IDP-126 gel group and 1 patient in its pooled vehicle gel group had 1 or more AEs associated with cutaneous safety or tolerability assessments that did not qualify to be reported as AEs and, as such, were subsequently excluded from the appropriate AE summary table.
For notable harms, refer to preceding data on general disorders and administration site conditions and skin and subcutaneous tissues disorders (TEAE categories) and SAEs.
aTEAEs experienced by more than 1 patient in either study drug group by system organ class and preferred term. At each level of system organ class or preferred term, patients reporting more than 1 TEAE were counted only once.
Sources: Common Technical Document section 2.7.4: Summary of Clinical Safety.14 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Critical Appraisal
Internal Validity
Studies 301 and 302 were generally appropriately designed and powered to evaluate the efficacy of IDP-126 gel relative to its vehicle gel. Although randomization was not stratified, the analyses of the coprimary end points included adjustment factors (treatment group, analysis centre, and respective baseline lesion count). The double-blind was maintained by dispensing identically packaged and labelled study drugs. As such, it was concluded that the risk of bias arising from the randomization process is unlikely.
In consultation with the clinical expert, it was concluded that the prespecified washout periods were adequate for each class of prohibited product or treatment for acne. Further, the expert indicated the list includes the therapies that are most used for acne in practice. Thus, any impact on the interpretation of the results due to their prior use is unlikely a result of completing the washout period.
The 2018 FDA guidance6 suggests that a clinically meaningful outcome in the treatment of acne is treatment success, defined by a score of 0 (clear) or 1 (almost clear) and at least a 2-grade improvement from baseline on the ISGA scale (an ordinal scale of 5 severity grades, each defined by a distinct and clinically relevant morphologic description). Recognizing that there is no standardized grading system for disease severity, the FDA guidance suggests considering both changes in lesion counts and treatment success in the assessment of treatment effect; this is reflected in studies 301 and 302. Additionally, there is evidence in the literature to support the validity, reliability, and responsiveness of the Acne-QoL questionnaire as a measure of HRQoL in patients with acne.7,8 Therefore, bias in the measurement of important outcomes is unlikely.
Interobserver variation in lesion counts was possible if the same evaluator was not available for each site visit; moreover, central adjudication was not performed. In consideration of the double-blind trial design, it was concluded that any possible impact on the interpretation of the efficacy results due to possible interobserver variation in lesion counts is unlikely.
Type I error was controlled in each study by requiring all 3 coprimary efficacy end points to be statistically significant to be able to draw a conclusion of superiority for IDP-126 gel relative to its vehicle gel and by testing the secondary efficacy end points using a gated, sequential process. No inferential statistics were conducted in the subgroup analyses and HRQoL outcomes; therefore, these results are considered as supportive evidence only.
Study discontinuation rates were 12.0% or less in each study drug group from each study. Any reason for study discontinuation (including lost to follow-up, AEs, progressive disease, and lack of efficacy) was reported in less than 6.0% of patients in each study drug group. In consultation with the clinical expert, it was concluded that the study discontinuation rates are reasonable in the context of the therapeutic area and, as such, the risks of attrition bias and possible unblinding are unlikely.
Protocol deviations were reported in at least 20.0% of patients in each study drug group from each study, with the exception of the vehicle gel group in Study 301 (16.4% of patients). The proportion of patients with any protocol deviation was generally similar between the study drug groups (approximately ≤ 10%), including use of an interfering concomitant medication, nonattendance at the week 12 visit, and incomplete lesion counts and EGSS at week 12. Thus, it was concluded that the risk of bias due to deviations from the intended intervention is low. Moreover, the overall adherence to the study drug was 90.0% or greater in both studies.
In consultation with the clinical expert, age, sex, and ethnicity or race were identified as possible effect modifiers in the treatment of acne. Although randomization was not stratified, the relevant patient demographic and disease characteristics at baseline were generally well balanced between the study drug groups in each study. As such, it was concluded that any possible impact on the interpretation of the efficacy results due to baseline differences between the study drug groups is unlikely.
The results of the sensitivity analyses in which missing data were handled differently from the primary method suggest the primary imputation method is robust; therefore, bias due to missing outcome data is unlikely.
Overall, no serious risk-of-bias concern was identified in the appraisal of studies 301 and 302.
External Validity
The inclusion criteria used in studies 301 and 302 — patients aged 9 years and older with moderate to severe acne — include the population of interest identified in the indication for IDP-126 gel, which is for the topical treatment of acne in patients aged 12 years and older. In consultation with the clinical expert, it was concluded that the inclusion criteria adequately capture (and, consequently, the study population from both studies is representative of) the patients seen in practice who would be candidates for IDP-126 gel. Of note, the majority of patients in each study drug group (ranging from 87.7% to 95.1% of patients across studies) had moderate acne. The age range of patients was similar within and between studies, ranging from 10 to 48 years across studies.
In consideration of the goal to minimize confounders using exclusion criteria, it was concluded that no patient who would be a candidate for IDP-126 gel was missed as a result of any exclusion criterion. However, the clinical expert highlighted that patients with these exclusion criteria seen in practice may still have an indication for topical therapy and be considered for IDP-126 gel. The clinical expert provided examples of patients meeting such criteria, including patients with polycystic ovarian disease, clinically significant menstrual irregularities, or secondary acne, and patients taking birth control pills.
In consultation with the clinical expert, it was concluded that the outcome measures of acne severity and lesion counts used in the trials are applicable to Canadian clinical practice. In general, the clinical expert indicated the assessment of treatment response includes an estimate of the total number of lesions, which contributes to their global assessment (no acne; mild, moderate, or severe acne). Important outcomes identified by patients with acne include self-confidence and scarring, which overlap with the domain structures of self-perception and symptom subscales in the Acne-QoL questionnaire. In consultation with the clinical expert, it was concluded that a follow-up at 12 weeks after initiation of a topical therapy is appropriate for an assessment of effect in this therapeutic area (i.e., an observable treatment difference is anticipated by week 12 of a trial).
Overall, no serious concern about the generalizability of the results to the population of interest in the Canadian setting was identified in the appraisal of studies 301 and 302.
GRADE Summary of Findings and Certainty of the Evidence
Methods for Assessing the Certainty of the Evidence
For pivotal studies and RCTs identified in the sponsor’s systematic review, GRADE was used to assess the certainty of the evidence for outcomes considered most relevant to inform the expert committee deliberations, and a final certainty rating was determined as outlined by the GRADE Working Group:9,10
High certainty: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: We are moderately confident in the effect estimate. The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. We use the word “likely” for evidence of moderate certainty (e.g., “X intervention likely results in Y outcome”).
Low certainty: Our confidence in the effect estimate is limited. The true effect may be substantially different from the estimate of the effect. We use the word “may” for evidence of low certainty (e.g., “X intervention may result in Y outcome”).
Very low certainty: We have very little confidence in the effect estimate. The true effect is likely to be substantially different from the estimate of effect. We describe evidence of very low certainty as “very uncertain.”
Following the GRADE approach, evidence from RCTs started as high-certainty evidence and could be rated down for concerns related to study limitations (which refer to internal validity or risk of bias), inconsistency across studies, indirectness, imprecision of effects, and publication bias.
When possible, certainty was rated in the context of the presence of an important (nontrivial) treatment effect based on thresholds identified in the literature and/or informed by the clinical expert consulted for this review; if this was not possible, certainty was rated in the context of the presence of any treatment effect (i.e., the clinical importance is unclear). In all cases, the target of the certainty-of-evidence assessment was based on the point estimate and where it was located relative to the threshold for a clinically important effect (when a threshold was available) or to the null.
For the GRADE assessments, the findings from studies 301 and 302 were considered together and summarized narratively per outcome because the 2 studies were similar in population, interventions, design, and outcome measures.
Results of GRADE Assessments
Table 2 presents the GRADE summary of findings for IDP-126 gel versus IDP-126 vehicle gel.
Indirect Evidence
Contents within this section have been informed by materials submitted by the sponsor. The following has been summarized and validated by the review team.
Objectives for the Summary of Indirect Evidence
Two pivotal trials (studies 301 and 302) provided a comparison between IDP-126 gel and vehicle gel among patients aged 9 years and older with moderate or severe acne. Still, there is a lack of direct evidence comparing the effectiveness of IDP-126 gel with other available treatments in this population. Hence, an ITC is warranted to address this evidence gap.
Description of Indirect Comparisons
The sponsor submitted 1 NMA estimating the relative effectiveness of IDP-126 gel versus other therapies for the treatment of patients with acne vulgaris. In addition, the sponsor provided 1 published NMA by Huang et al.17 that evaluated the efficacy and safety of diverse pharmacological therapies for acne vulgaris. A description of study selection and methods of literature review for the 2 ITCs is available in Table 20.
Table 20: Study Selection Criteria and Methods for ITCs Submitted by the Sponsor
Characteristics | Bausch Health Companies Inc. (2023) | Huang et al. (2023) |
|---|---|---|
Population |
|
|
Intervention | Monotherapies:
Treatment combinations:
| Single or combination therapies:
|
Comparator |
| Single or combination therapies:
|
Outcome | Clinician-rated improvement in:
|
|
Study designs | RCTs | |
Publication characteristics | Published and unpublished studies | Published studies |
Exclusion criteria |
|
|
Databases searched | Ovid MEDLINE, Ovid Embase, Cochrane Central Register of Controlled Trials (CENTRAL), PubMed (MEDLINE), Pediatric Economic Database Evaluation, and National Health Service Economic Evaluation Database. | PubMed, Embase |
Selection process | Title or abstract and full text screening were conducted by 2 independent reviewers and reconciled by a third senior reviewer. | Two researchers independently assessed all trials according to the predefined selection criteria. Any disagreement was resolved through discussion with a third researcher. |
Data extraction process | Data extraction was performed in DistillerSR. The extracted information included:
Additional data extracted included concentration and vehicle used, therapies associated with topical treatments, systemic treatments, physical treatments, posology, and duration of treatment (weeks) | The following data were extracted: trial design, trial size, details of intervention (including route, dose, frequency, and treatment duration), patient characteristics (e.g., mean age, sex, and baseline lesion counts), and outcome data for each time point. For crossover trials, only data from the first period were extracted to avoid possible carryover effects. For split-face studies, the lesion counts were multiplied by 2. Outcome data were approximated from the figure (e.g., data presented in graphs or forest plots) when no precise numerical data were provided. Standard deviations were calculated or imputed from standard errors, 95% CIs, or P values when necessary, according to the Cochrane handbook. |
Quality assessment | Cochrane risk-of-bias tool v. 2.0.41 | Slightly adapted version of the Cochrane Collaboration risk-of-bias approach, which was assessed by the 2 investigators.41 |
CI = confidence interval; EGSS = Evaluator's Global Severity Score; FDC = fixed-dose combination; IGA = Investigator’s Global Assessment; IL = inflammatory lesion; ISGA = Investigator’s Static Global Assessment; ITC = indirect treatment comparison; RCT = randomized controlled trial.
Source: Sponsor’s summary of clinical evidence.15
Sponsor-Conducted ITC Design
Objectives
The sponsor-conducted ITC aimed to understand the relative efficacy of IDP-126 gel in relation to other competing interventions among patients with moderate to severe acne vulgaris.
Study Selection Methods
A systematic literature search was conducted in February 2023 to identify RCTs assessing treatments among patients with moderate or severe acne that are currently approved or under review by the FDA. The literature search included the following efficacy outcomes of interest for this review: proportion of patients with at least a 2-grade reduction from baseline in the IGA or equivalent scales (EGSS or ISGA), a score that equated to clear or almost clear after treatment duration, and absolute change in the counts of inflammatory lesions and noninflammatory lesions from baseline.
The following academic electronic databases were screened: Ovid MEDLINE, Ovid Embase, Cochrane Central Register of Controlled Trials (CENTRAL), PubMed (MEDLINE), Pediatric Economic Database Evaluation, and National Health Service Economic Evaluation Database. Grey literature sources included nonacademic databases (Google Scholar, health technology assessment databases, and trial registries) as well as conference proceedings from the American Academy of Dermatology, International Society of Dermatology, ISPOR (research) Presentations Database, and Northern Light Life Sciences Conference Abstracts. The search strategy was limited to RCTs published in English. The systematic review protocol was published in PROSPERO, an international prospective register of systematic reviews (CRD42023430668).42
According to the exclusion criteria prespecified by the sponsor, treatment groups with only a single RCT demonstrating comparative efficacy were omitted. This was in accordance with FDA process guidelines,43 which require that 2 well-designed clinical trials be included in the drug development and approval process. Moreover, trials with fewer than 50 participants in each arm were also excluded.
Screening for potentially eligible research articles was conducted by 2 separate reviewers, and any discrepancies were resolved through discussion with a third reviewer until consensus was reached. Data extraction was conducted in DistillerSR. A quality assessment of the included studies was conducted using the Cochrane Collaboration risk-of-bias tool (version 2).41
Sponsor-Conducted ITC Analysis Methods
Before conducting an NMA, a 3-step feasibility assessment was undertaken in line with the published guidelines:44,45
assessment of the evidence networks (i.e., assessing whether the treatments formed a coherent network for each outcome)
assessment of clinical heterogeneity (studies, treatments, outcomes, and patient characteristics)
assessment of differences within and between direct pairwise comparisons (in terms of baseline risk and observed effects).
For the direct treatment comparisons conducted during the feasibility assessment, forest plots were developed using random-effects models, while heterogeneity was assessed using the I2 statistic.
The NMAs for the outcomes of interest were conducted using a Bayesian framework. Two types of models were used for the base-case analysis: a fixed-study, fixed class–effect model, assuming equal effectiveness of treatments within the same class and no variability among trials when estimating the treatment effects for a particular class, and a random-study, fixed class–effect model, assuming the same effectiveness of treatments within the same class, and between-study variability that accounts for differences between trial-specific estimates. Point estimates (log-ORs for binary outcomes and mean differences [MDs] for continuous outcomes) and 95% CrIs were constructed using the MCMC method. The model parameters were estimated by running 4 Markov chains with different initial values for the parameters. Each chain involved 2,000 simulations, with an initial burn-in period of 1,000 and a thinning parameter of 50. Assessment of model fit was based on the deviance information criterion (DIC) and posterior residual deviance. To specify the prior distribution for the intercept and treatment effects, noninformative priors were used, with a half-normal (0, 25) prior being assigned for the between-study SD in random effects.
The nodes in the network were initially made up of individual treatment regimens with different doses (i.e., disconnected networks). However, the sponsor combined the similar drugs with different doses into treatment groups. This grouping was further validated by clinical experts that the sponsor engaged in their research process. After the initial grouping of diverse treatments into groups, 2 disconnected networks were formed, 1 using a vehicle (such as a gel or cream without the active ingredient) as a comparator and 1 using a placebo pill as a comparator. A random-effects meta-analysis revealed significant variability in effects within both the vehicle and the placebo arms (I2 ranging from 84% to 91% and from 79% to 83% for vehicle and placebo groups across all outcomes, respectively). However, there were no significant subgroup effect observed, which allowed the sponsor to create 1 single class (vehicle or placebo) rather than considering them as separate classes in the NMAs.
The following 14 treatment groups were incorporated in the NMAs: TFDCABR3 (topical antibiotic plus BPO and retinoid fixed-dose combination), TFDCRB2 (topical retinoid plus BPO fixed-dose combinations), TFDCAB2 (topical antibiotic plus BPO fixed-dose combinations), TFDCAR2 (topical antibiotic plus retinoid fixed-dose combinations), TMA1 (topical antibiotic monotherapy), TMR1 (topical retinoid monotherapy), TMB1 (topical BPO monotherapy), TMO1 (other topical prescription monotherapy), TOA3 (combinations of topical treatments that include an oral antibiotic), OA1 (oral antibiotic monotherapy), POA (combinations of physical treatments with oral antibiotic), PH1 (physical treatment only), ORx (other treatments), and V/P (vehicle or placebo).
The NMA included trials with different follow-up times (i.e., week 12, week 16, and week 24), and this variation was omitted when defining the outcomes. However, treatment duration was considered as a covariate in all of the NMA models. Due to the lack of complete data for certain study-level outcomes, the sponsor used diverse imputation methods to estimate the required summary statistics; the majority of these approaches relied primarily on the assumption that the outcome data were normally distributed.
Potential treatment-effect modifiers were identified through a rapid literature review. Moreover, a random-effects meta-regression was conducted to identify which variables impacted efficacy across all measured outcomes, based on the data available across the included studies.
To verify the consistency between direct and indirect comparisons, a comparison was made between a base-case model that assumed consistency and a global inconsistency model that assumed unrelated mean effects. This comparison was based on the models’ posterior residual deviance and DIC. Moreover, visual assessments were performed to identify data points contributing to inconsistency.
Regarding the bias of the included studies, the sponsor created bias adjustment models (BAMs) to mitigate the potential impact of bias on the overall results. Moreover, a threshold analysis was performed to assess the robustness of the decision regarding the effectiveness of TFDCABR3 for treating the patients from the NMA with moderate to severe acne.
Table 21: Sponsor-Conducted ITC Analysis Methods
Methods | Description |
|---|---|
Analysis methods | A Bayesian framework was used to conduct an NMA to assess the relative effectiveness of IDP-126 gel compared with other competing treatments for patients with moderate to severe acne vulgaris. The relative treatment effects in the pairwise analyses were presented as log-odds ratios with 95% CrI for binary outcomes and mean difference (95% CrI) for continuous outcomes. The parameters of the different models for all scenarios were estimated using the MCMC method using Stan programming language implemented through R statistical software version 4.3.0 (https://www.r-project.org/) by “multinma.”46 A threshold analysis was conducted using the “nmathresh” package.47 |
Priors | Noninformative priors were used to specify the prior distribution for the intercept as well as for treatment effects. A half-normal (0, 25) prior was assigned for the between-study SD in random effects and was sufficiently wide so that the posterior distribution was not constrained. The model parameters were estimated by running 4 Markov chains with different initial values for the parameters. Each chain involved 2,000 simulations, with an initial burn-in period of 1,000 and a thinning parameter of 50. |
Assessment of model fit | Deviance information criterion and posterior residual deviance were used to identify the best-fitted model. |
Assessment of consistency | To assess the presence of inconsistency in the data, a comparison was made between a base-case model assuming consistency and a global inconsistency model, which assumed unrelated mean effects. |
Assessment of convergence | The |
Outcomes |
|
Follow-up time points | The NMA included studies that reported efficacy outcomes at various time points, such as week 12, week 16, and week 24. Consequently, a specific time duration is not mentioned when defining the outcomes, allowing for flexibility in capturing the diverse time frames considered in the included studies. |
Construction of nodes | Network diagrams were constructed to depict network structures visually and to highlight the differences in outcome definitions. Due to the availability of various treatment regimens with different doses, disconnected networks were formed first. To address this challenge, similar drugs with different doses were combined to create treatment groups. |
Sensitivity analyses | None |
Subgroup analysis | None |
Methods for pairwise meta-analysis | Conducted |
Crl = credible interval; DIC = deviance information criterion; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; IGA = Investigator’s Global Assessment; IL = inflammatory lesion; ITC = indirect treatment comparison; MCMC = Markov chain Monte Carlo; NIL = noninflammatory lesion; NMA = network meta-analysis; SD = standard deviation.
Source: Details included in the Table are from the sponsor’s summary of clinical evidence.15
Results of Sponsor-Conducted ITC
Summary of Included Studies
A total of 107 publications from 85 studies were found eligible for inclusion in the NMA. A Table of baseline study characteristics presented within the sponsor’s NMA is provided in Appendix 1. A feasibility assessment was conducted for all 3 outcomes of interest for the NMA (proportion of patients with treatment success and reduction in inflammatory and noninflammatory lesion counts). The majority of the trials were phase III (more than 54% across all the outcomes) and double-blind (more than 83% across the 3 outcomes (Table 22). More than 90% of the trials were conducted as multicentre studies and more than two-thirds of them were performed in North America, specifically, Canada and the US. Regarding the risk-of-bias assessment conducted with the Cochrane risk-of-bias tool (version 2.0), the proportion of studies with a high risk, some concerns, or a low risk of bias is reported in Table 22.
Assessment of the patients’ baseline characteristics revealed there was minimal variation in the age of the participants across the diverse trials of the network, with the majority of them reporting a mean age of 20 years. There were notable differences in sex distributions across the trials, with studies reporting a proportion of female patients as low as 21% and as high as 100% across all 3 outcomes of interest. Although acne severity was scarcely reported in some of the trials (about 31% of studies lacked this information), the variation in the proportion of patients with moderate acne severity was high, ranging from 0 to 100%. Of note, the sponsor reported there was significant heterogeneity in terms of reported baseline characteristics both across and within the treatment groups.
A number of potential effect modifiers were identified through a rapid literature review and assessed in the homogeneity assessment. The rapid literature review revealed the following prognostic factors of importance: age, sex, body mass index, severity of disease, and family history. Moreover, a random-effects meta-regression was conducted to assess the impact of various variables (i.e., treatment group, mean age, proportion of female patients, proportion of patients with moderate disease, study’s risk-of-bias assessment, type of scale used to assess disease severity at baseline, and duration of treatment) on treatment efficacy (Table 23). The meta-regression revealed that different treatment groups, the proportion of patients with moderate disease at baseline, and the duration of treatment showed statistically significant effects on efficacy across all measured outcomes, based on the data available across the trials in the network (N = 33 trials for acne severity; N = 35 trials for change in lesion counts). Of note, variations in treatment efficacy were observed within treatment groups. Despite this, the base-case analysis assumed no significant differences in efficacy within the same treatment group with different treatment dosing, except for sampling fluctuation. Moreover, outcome measures that were assessed at different time points (i.e., weeks 12, 16, or 24) were grouped together; however, treatment duration was considered a covariate in all NMA models to account for its potential impact on the outcomes of interest.
Table 22: Summary Description of Study and Participant Baseline Characteristics of Trials included in the Sponsor-Conducted NMA
Characteristics | NMA outcome | ||
|---|---|---|---|
Proportion of patients experiencing treatment successa (N = 48 trials) | Absolute change in ILs from baseline (N = 50 trials) | Absolute change in NILs from baseline (N = 46 trials) | |
Trial characteristics | |||
Phase | 58% phase III (28/48) 23% phase II (11/48) 19% NR (9/48) | 54% phase III (27/50) 32% phase II (16/50) 14% NR (7/50) | 59% phase III (27/46) 28% phase II (13/46) 13% NR (6/46) |
Blinding | 88% double-blinded (42/48) 10% quadruple-blinded (5/48) 2% triple-blinded (1/48) | 84% double-blinded (42/50) 14% quadruple-blinded (7/50) 2% triple-blinded (1/50) | 83% double-blinded (38/46) 15% quadruple-blinded (7/46) 2% triple-blinded (1/46) |
Centre | 4% single centre (2/48) 96% multicentre (46/48) | 6% single centre (3/50) 94% multicentre (47/50) | 6% single centre (3/46) 94% multicentre (43/46) |
Country or continent | 69% North America (US and Canada) (33/48) 19% North America, Europe, Australia, and Russia (9/48) 4% Europe (2/48) 4% Asia (2/48) 2% US and Taiwan (1/48) 2% NR (1/48) | 80% North America (US and Canada) (40/50) 16% North America, Europe, Australia, and Russia (8/50) 2% Europe (1/50) 2% Asia (1/50) | 78% North America (US and Canada) (36/46) 18% North America, Europe, Australia, and Russia (8/46) 2% Europe (1/46) 2% Asia (1/46) |
Risk of bias | 12% high risk (6/48) 65% some concerns (31/48) 23% low risk (11/48) | 20% high risk (10/50) 56% some concerns (28/50) 24% low risk (12/50) | 13% high risk (6/46) 67% some concerns (31/46) 20% low risk (9/46) |
Participant baseline characteristics | |||
Age | Minimal variation Mean age = 20 years in most of the studies | Minimal variation Mean age = 20 years in most of the studies | Minimal variation Mean age = 20 years in most of the studies |
Proportion of female patients | A total of 47 trials reported sex distribution Observed variation, with range from 21% to 100% Pooled estimateb = 0.61 (95% CI, 54 to 67; I2 = 93%; P < 0.01) | A total of 50 trials reported sex distribution Observed variation, with range from 21% to 75% Pooled estimateb = 0.56 (95% CI, 0.54 to 0.58; I2 = 92%; P < 0.01) | A total of 46 trials reported sex distribution Observed variation, with range from 21% to 88% Pooled estimateb = 0.56 (95% CI, 0.53 to 0.58; I2 = 92%; P < 0.01) |
Proportion of patients with moderate disease | A total of 34 trials reported acne severity Observed variation, with range from 0% to 100% Pooled estimateb = 0.92 (95% CI, 0.83 to 0.97; I2 = 93%; P < 0.01) | A total of 36 trials reported acne severity Observed variation, with range from 0% to 100% Pooled estimateb = 0.87 (95% CI, 0.80 to 0.92; I2 = 94%; P < 0.01) | A total of 36 trials reported acne severity Observed variation, with range from 0% to 100% Pooled estimateb = 0.87 (95% CI, 0.80 to 0.92; I2 = 94%; P < 0.01) |
CI = confidence interval; IL = inflammatory lesion; ITC = indirect treatment comparison; NIL = noninflammatory lesion; NMA = network meta-analysis; NR = not reported.
Note: The assessment of the risk of bias was conducted using the Cochrane Collaboration risk-of-bias tool (version 2).41
aOut of the 48 trials, 9 used the EGSS, 6 used the ISGA, and the remaining trials used the IGA to assess severity at baseline.
bTo assess heterogeneity in terms of the proportion of female patients and the proportion of patients with moderate disease at baseline, a random-effects meta-analysis was conducted.
Source: Details included in the Table represent summary information that the review team produced based on ITC technical report.48
Table 23: Assessment of Homogeneity for the Sponsor-Conducted ITC
Characteristics | Description and handling of potential effect modifiers |
|---|---|
Age | Assessed. |
Proportion of female patients | Assessed. |
Different treatment groups | Assessed. This variable exhibited statistically significant effects on efficacy across all measured outcomes. |
Proportion of patients with moderate disease | Assessed. This variable exhibited statistically significant effects on efficacy across all measured outcomes. |
Risk-of-bias assessment of the study | Assessed. |
Type of scale used to assess disease severity at baseline | Assessed. |
Duration of treatment | Assessed. This variable exhibited statistically significant effects on efficacy across all measured outcomes. |
ITC = indirect treatment comparison.
Source: Sponsor summary of clinical evidence.15
Evidence Networks
Proportion of Patients Experiencing Treatment Success
Forty-eight out of 85 trials reported the outcome of acne disease severity based on the IGA, EGSS, or ISGA. The network consisted of 12 treatment groups, with the number of patients ranging from 108 to 2,813 per study (Figure 1).
Figure 1: Network Diagram for Proportion of Patients Experiencing Treatment Success (N = 48 Trials)
BPO = benzoyl peroxide; OA1 = oral antibiotic monotherapy; ORx = other treatment; TFDCAB2 = topical antibiotic and BPO fixed-dose combination; TFDCABR3 = topical antibiotic plus BPO and retinoid fixed-dose combination; TFDCAR2 = topical antibiotic plus retinoid fixed-dose combination; TFDCRB2 = topical retinoid and BPO fixed-dose combination; TMA1 = topical antibiotic monotherapy; TMB1 = topical BPO monotherapy; TMO1 = other topical (prescription) monotherapies; TMR1 = topical retinoid monotherapy; TOA3 = combinations of dual-drug fixed-dose topical treatments with an oral antibiotic; V/P = vehicle or placebo.
Source: ITC technical report.48
Inflammatory Lesions Reduction
Fifty out of 85 trials reported the outcome of mean reduction in inflammatory lesion counts. The network consisted of 12 treatment groups, with the number of patients ranging from 107 to 2,813 per study (Figure 2).
Figure 2: Network Plot For Inflammatory Lesion Count Reduction Outcome (N = 50 Trials)
BPO = benzoyl peroxide; OA1 = oral antibiotic monotherapy; PH1 = physical therapies only; TFDCAB2 = topical antibiotic and BPO fixed-dose combination; TFDCABR3 = topical antibiotic plus BPO and retinoid fixed-dose combination; TFDCAR2 = topical antibiotic plus retinoid fixed-dose combination; TFDCRB2 = topical retinoid and BPO fixed-dose combination; TMA1 = topical antibiotic monotherapy; TMB1 = topical BPO monotherapy; TMO1 = other topical (prescription) monotherapies; TMR1 = topical retinoid monotherapy; TOA3 = combinations of dual-drug fixed-dose topical treatments with an oral antibiotic; V/P = vehicle or placebo.
Source: ITC technical report.48
Noninflammatory Lesions Reduction
Forty-six out of 85 trials reported the outcome mean reduction in noninflammatory lesion counts. The network consisted of 12 treatment groups, with the number of patients ranging from 107 to 2,813 per study (Figure 3).
Figure 3: Network Plot For Noninflammatory Lesion Count Reduction Outcome (N = 46 Trials)
BPO = benzoyl peroxide; OA1 = oral antibiotic monotherapy; PH1 = physical therapies only; TFDCAB2 = topical antibiotic and BPO fixed-dose combination; TFDCABR3 = topical antibiotic plus BPO and retinoid fixed-dose combination; TFDCAR2 = topical antibiotic plus retinoid fixed-dose combination; TFDCRB2 = topical retinoid and BPO fixed-dose combination; TMA1 = topical antibiotic monotherapy; TMB1 = topical BPO monotherapy; TMO1 = other topical (prescription) monotherapies; TMR1 = topical retinoid monotherapy; TOA3 = combinations of dual-drug fixed-dose topical treatments with an oral antibiotic; V/P = vehicle or placebo.
Source: ITC technical report.48
Efficacy
The base-case NMA results for all outcomes are summarized in Table 24. Only comparisons between IDP-126 gel (i.e., the TFDCABR3 group, topical antibiotic plus BPO and retinoid fixed-dose combinations) and other available treatments are presented. League tables summarizing the remaining pairwise indirect comparisons of the different outcome networks are presented in Appendix 1.
For all treatment outcomes assessed, the best-fitting model for the NMA networks was the random-study, fixed class–effect model, based on the favourable posterior residual deviance and DIC value (Table 24). Regarding inconsistency testing, there were no differences observed between the fit statistics and between-study SD of the random-effects consistency and inconsistency models, suggesting no evidence of inconsistency across all the 3 networks. Similarly, there were no statistical differences between the fit of the random-effects base case and BAMs. Threshold analyses showed robustness of the recommendation for IDP-126 gel for all 3 outcomes.
Proportion of Patients Experiencing Treatment Success
The results for the comparison of IDP-126 gel versus different treatments regarding the proportion of patients experiencing treatment success are reported in Table 24. According to the estimated ORs, IDP-126 gel demonstrated higher efficacy compared with the vehicle or placebo comparison group (OR = 6.30; 95% CrI, 3.90 to 9.87). Moreover, IDP-126 gel was favoured in comparisons with other active treatments (oral antibiotic monotherapy, topical antibiotic plus BPO fixed-dose combinations, topical antibiotic plus retinoid fixed-dose combinations, topical antibiotic monotherapy, topical BPO monotherapy, topical retinoid monotherapy, and other topical [prescription] monotherapies), based on the outcome estimates presented in Table 24.
Inflammatory Lesions
Results for the comparison of IDP-126 gel versus different treatments on the reduction in inflammatory lesion counts are reported in Table 24. IDP-126 gel was favoured in comparisons with vehicle or placebo based on estimated treatment effects (MD = −8.21; 95% CrI, −10.33 to −6.13). Moreover, IDP-126 gel demonstrated higher efficacy when compared with oral antibiotic therapy, topical antibiotic plus retinoid fixed-dose combinations, topical retinoid plus BPO fixed-dose combinations, topical antibiotic monotherapy, topical BPO monotherapy, topical retinoid monotherapy, and other topical (prescription) monotherapies.
Noninflammatory Lesions
Results for the comparison of IDP-126 gel with different treatments regarding the outcome of reduction in noninflammatory lesion counts are reported in Table 24. IDP-126 gel was favoured in comparisons with vehicle or placebo based on estimated treatment effects (MD = −13.41; 95% CrI, −16.69 to −10.32). Moreover, IDP-126 gel was favoured in the remaining comparisons with other active treatments (oral antibiotic therapy, topical antibiotic plus BPO fixed-dose combinations, topical antibiotic plus retinoid fixed-dose combinations, topical antibiotic monotherapy, topical BPO monotherapy, topical retinoid monotherapy, and other topical [prescription] monotherapies) and physical therapy.
Table 24: Summary of IDP-126 Gel Comparisons From the Random-Effects NMA Provided by the Sponsor, Base-Case Analyses
Detail | Proportion of patients experiencing treatment successa | Inflammatory lesions | Noninflammatory lesions |
|---|---|---|---|
Number of studies in the network | ██ | ██ | ██ |
Model | ████ | ████ | ████ |
Posterior total residual deviance | █████ | █████ | █████ |
DIC | █████ | █████ | █████ |
IDP-126 gel (TFDCABR3 group) versus comparator | |||
Comparator | OR (95% CrI) | MD (95% CrI) | MD (95% CrI) |
V/P | ████ █████ ██ █████ | −8.21 (−10.33 to −6.13) | −13.41 (−16.69 to −10.32) |
OA1 | ████ █████ ██ █████ | −4.50 (−7.00 to −2.05) | −10.83 (−15.17 to −6.71) |
ORx | ████ █████ ██ █████ | NA | NA |
TFDCAB2 | ████ █████ ██ █████ | −1.60 (−4.22 to 0.87) | −5.10 (−9.01 to −1.22) |
PH1 | ██ | −3.55 (−9.32 to 2.03) | −13.62 (−20.43 to −6.40) |
TFDCAR2 | ████ █████ ██ █████ | █████ ██████ ██ ██████ | ████ ███ ██ ██████ |
TFDCRB2 | ████ █████ ██ █████ | █████ ██████ ██ ██████ | ████ ███ ██ █████ |
TMA1 | ████ █████ ██ █████ | █████ ██████ ██ ██████ | ████ ███ ██ ██████ |
TMB1 | ████ █████ ██ █████ | █████ ██████ ██ ██████ | ████ ███ ██ ██████ |
TMO1 | ████ █████ ██ █████ | █████ ██████ ██ ██████ | ████ ███ ██ ██████ |
TMR1 | ████ █████ ██ █████ | █████ ██████ ██ ██████ | ████ ██ ██ ██████ |
TOA3 | ████ █████ ██ █████ | ████ ██████ ██ █████ | ███ ███ ██ ██████ |
BPO = benzoyl peroxide; CrI = credible interval; DIC = deviance information criteria; EGSS = Evaluator’s Global Severity Score; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; IGA = Investigator’s Global Assessment; ISGA = Investigator’s Static Global Assessment; ITC = indirect treatment comparison; MD = mean difference; NA = not applicable; NMA = network meta-analysis; OA1 = oral antibiotic monotherapy; OR = odds ratio; ORx = other treatment; PH1 = physical therapy only; TFDCABR3 = topical antibiotic plus benzoyl peroxide and retinoid fixed-dose combination; TFDCAR2 = topical antibiotic plus retinoid fixed-dose combination; TFDCRB2 = topical retinoid plus benzoyl peroxide fixed-dose combination; TFDCAB2 = topical antibiotic plus benzoyl peroxide fixed-dose combination; TMR1 = topical retinoid monotherapy; TMA1 = topical antibiotic monotherapy; TMB1 = topical benzoyl peroxide monotherapy; TMO1 = other topical prescription monotherapy; TOA3 = combination of double-drug fixed-dose topical treatment with oral antibiotic; V/P = vehicle or placebo.
aTreatment success was defined as a percentage of patients with at least a 2-grade reduction from baseline in the IGA or an equivalent scale (EGSS or ISGA) and with a score that equated to clear or almost clear after treatment duration.
Note: Statistically significant results are shown in bold font.
Sources: ITC technical report48 and sponsor response to the May 13, 2024, request for additional information regarding the review of IDP-126 gel.49
Critical Appraisal of Sponsor-Conducted ITC
The sponsor-conducted ITC followed standard methods for the conducting and reporting of reviews, including the following: defining the research question according to PICO (patient or population, intervention, comparison, and outcomes) criteria, searching through multiple database sources, comprehensive literature searches, and involvement of multiple reviewers for study selection and data extraction. Of note, the systematic review protocol was prespecified and registered on a public platform,42 leading to increased internal validity of the literature review that was used to inform the NMA. Exclusion criteria for the review covered single studies as well as studies with fewer than 50 participants in each arm. Even though justification was provided for these exclusions,43 the review team deemed the exclusion criteria to be restrictive, leading to the possible exclusion of relevant studies from the network.
A comprehensive and structured feasibility assessment was performed to assess the variability in study and patient characteristics across the 3 outcomes of interest for the ITC. Some differences were observed in the trial characteristics (phase, blinding procedures, and geographical locations). Moreover, variability in patients’ baseline characteristics included the disease severity (range from 0 to 100%) and sex variables (proportion of female participants varied from 21% to 100%). The identification of effect-modifying variables was conducted through a literature review, while the meta-regression analysis was adopted to explore the effects of various variables on estimates of treatment effect. Different treatment groups, acne severity, and treatment duration were identified as effect-modifying variables, based on meta-regression. However, due to limited reporting in the studies included in the network, only the treatment duration variable was included as a covariate and accounted for in the NMA analysis. According to the clinical expert consulted, notable differences in baseline characteristics, such as baseline disease severity, could potentially have an impact on relative treatment effects, especially absolute changes in acne lesions. Hence, it remains highly possible that the heterogeneity in the baseline characteristics across the studies in the network will bias the results of the NMA.
Outcomes included in the NMA (i.e., proportion of patients with treatment success and inflammatory and noninflammatory lesion counts) were relevant to the treatment of patients with acne. However, acne scaring, an important outcome according to the clinical expert consulted, was not reported in the sponsor’s technical report despite being specified in the literature review protocol as an outcome of interest, and the reasons for the exclusion were not specified. Different scales were incorporated in the definition of the treatment success outcome (i.e., EGSS, IGA, and ISGA), but there was no variability in the outcome according to the scale used, based on the subgroup analyses conducted by the sponsor. The clinical expert validated this assumption and confirmed that these scales are considered equivalent in clinical practice. Moreover, outcomes that were assessed at different time points, such as weeks 12, 16, or 24, were included in the NMA analysis. Despite the variability in time frames, the median duration of treatment assessment was 12 weeks, and treatment duration was included as a covariate in the NMA analyses. Considering this, the inclusion of various assessment time points would likely exert minimal impact on the NMA estimates, according to the clinical expert consulted for this review.
In reference to the comparators included in the sponsor’s NMA, different treatments were pooled together within individual nodes. As such, the efficacy of specific formulations and specific dosing schedules could not be assessed. Considerations regarding the representation of included treatments as well as the generalizability constraints of the included therapies were discussed with the clinical expert consulted for this review. The treatment groupings presented in the network were generally validated by the clinical expert. The expert reported that most of the treatments used in Canadian clinical practice are represented in the network; however, 1 of the key oral monotherapies (i.e., oral isotretinoin) and 2 topical monotherapies (i.e., azelaic acid and topical dapsone) were not included, and their omission might be a result of the exclusion criteria adopted in the sponsor’s review. For instance, the sponsor specified that 1 study evaluating the efficacy of adapalene plus BPO and doxycycline versus oral isotretinoin was ultimately excluded from the literature review, as it exceeded the prespecified review threshold of 5% of patients with a very severe acne condition at baseline. Moreover, the clinical expert indicated that within the same nodes, monotherapies and combination therapies that are unavailable in Canada (e.g., oral lymecycline, oral sarecycline, topical olumacostat glasaretil, and BPO plus adapalene in combination with lymecycline) are grouped together with the treatments that are currently available in Canada. As such, the effect estimates for only those treatments that are used in Canada could not be presented separately, and the NMA estimates are reflective of comparators that are both relevant and irrelevant to the Canadian practice setting. The treatment node of other therapies, which included oral contraceptives and metformin, has limited alignment with the indication, as these drugs are considered for patients with polycystic ovarian disease (oral contraceptives) or diabetes (metformin), according to the clinical expert. Finally, the creation of a unique reference category (vehicle or placebo) was justified by the lack of subgroup effects observed in the sponsor’s statistical analysis, and the appropriateness of combining placebo pill and vehicle gel was further validated by the clinical expert consulted.
The selection of the models (random-study, fixed class–effect model with noninformative priors) was based on the lowest DIC and posterior residual deviance. Checks of the consistency assumption, both quantitative and visual, did not raise concerns regarding consistency in the evidence base. Risk-of-bias assessments of the included trials identified that only around 20% of the trials were at low risk of bias across the 3 outcomes of interest, while most of the remaining studies were associated with some concerns. However, BAMs, which were applied to account for the impact of bias in each domain of the tool on the overall results, revealed no concerns when comparing the base-case analysis with a BAM. Threshold analyses further confirmed the robustness of the recommendation regarding the effectiveness of TFDCABR3 for all 3 outcomes.
Beyond the outcome- and comparator-related representation issues raised earlier, the clinical expert reported no major generalizability concerns regarding the applicability of the NMA findings in the Canadian context. According to the expert, assessment of variation in efficacy outcomes across additional variables, such as ethnicity, would be favourable; however, the expert noted that the representation of individuals with diverse ethnic backgrounds is often limited across acne clinical trials.
Design of Published ITC (by Huang et al.)
Objectives
The NMA conducted by Huang et al.17 aimed to provide a comparison of the common pharmacological treatments for acne, including oral and topical medications as single or combined treatments.
Study Selection Methods
A systematic literature search was conducted to identify RCTs assessing the efficacy of therapies among patients with acne of any age or sex who had undergone treatment for at least 2 weeks. The review included the following efficacy outcomes: percentage or absolute decrease in the number of total, inflammatory, or noninflammatory lesions, and the proportion of participants experiencing treatment success, which was defined by an improvement of 2 grades from baseline and/or reaching clear or almost clear on the IGA of acne severity Table 20. The safety end point of interest in the NMA was discontinuation due to AEs.
The PubMed and Embase electronic databases were screened for studies from inception to February 2022. No language restrictions were applied.
Screening for potentially eligible articles was conducted by 2 separate reviewers, and any discrepancies were resolved through discussion with a third reviewer until consensus was reached. A quality assessment of the included studies was conducted using a modified version of the Cochrane Collaboration risk-of-bias tool (version 2.0).41
Published ITC Analysis Methods
Details on the ITC methods used by Huang et al.17 are reported in Table 25.
The NMAs for the outcomes of interest were conducted using a frequentist approach, with random-effects models being adopted in the analyses. Odds ratios with 95% CIs were computed for binary outcomes (treatment success and discontinuation due to AEs). MDs were computed using a restricted maximum likelihood estimation for continuous outcomes (percentage and absolute reductions in total, inflammatory, and noninflammatory lesion counts).
Global heterogeneity was assessed by I2 statistics. Inconsistency between direct and indirect comparisons was assessed using a node-splitting method and by visually inspecting heat plots.
A total of 37 treatment nodes (including 6 oral antibiotics, 5 topical antibiotics, oral isotretinoin, 5 topical retinoids, 6 combination oral contraceptives, topical clascoterone, 10 combination therapies, BPO, azelaic acid, and placebo) were included in the base-case analysis. Nodes were grouped by therapeutic intervention categories. An additional, separate analysis was conducted to evaluate the efficacy of more general treatment types by pooling the treatment nodes with similar mechanisms (e.g., oral antibiotics, topical antibiotics, topical retinoids).
Approaches adopted for the sensitivity analysis are reported in Table 25.
Table 25: Published ITC Analysis Methods
Methods | Description |
|---|---|
Analysis methods | The network meta-analysis used frequentist methods. All computations were conducted in the R software, version 3.1.1 (R Foundation for Statistical Computing) with the package netmeta. |
Priors | Frequentist methods were used (no priors). |
Assessment of model fit | A random-effects model was used. |
Assessment of consistency | Node-splitting method. |
Assessment of convergence | NA |
Outcomes | Mean percentage reduction in total, inflammatory, and noninflammatory lesions. Mean absolute reduction in total, inflammatory, and noninflammatory lesions. Odds ratio of patients with treatment success, defined by an improvement of 2 grades from baseline and/or reaching clear or almost clear on the IGA of acne severity. |
Follow-up time points | Not specified. |
Construction of nodes | There were 37 treatment nodes included in the primary analysis, which included the 1 reference treatment (i.e., placebo). Nodes were grouped by therapeutic intervention categories. A separate analysis evaluating the efficacy of more general treatment types by pooling the treatment nodes with similar mechanisms (e.g., oral antibiotics, topical antibiotics, topical retinoids) was performed. |
Sensitivity analyses | Exclusion of studies performed before 1985 and exclusion of studies with low-quality scores (1 or 2 as per a modified version of the Cochrane Collaboration risk-of-bias tool v. 2.0). |
Subgroup analysis | None |
Methods for pairwise meta-analysis | NA |
IGA = Investigator’s Global Assessment; ITC = indirect treatment comparison; NA = not applicable.
Source: Sponsor’s summary of clinical evidence.15
Results of the Published ITC
Summary of Included Studies
A total of 210 publications reporting on 221 trials were included in the analyses.
Regarding baseline characteristics, across all studies, the mean age of participants was 20 years (range, 10 to 38 years) and the median duration of treatment was 12 weeks (range, 2 to 48 weeks). Proportion of female participants showed great variation across the studies that reported these data, ranging from 0 to 100%. The median total and inflammatory and noninflammatory baseline lesion counts were 71.5, 27, and 44, respectively.
In reference to the quality assessment scale items, 194 (88%) of the studies were investigator-blinded and 130 (58%) were double-blinded. Seventy-four trials (34%) reported appropriate random-sequence generation, and 136 trials (62%) reported reasons for withdrawals, with similar proportions of missing data between groups.
Evidence Networks
Figure 4 reports on the evidence network for percentage reduction in total lesion counts, which displays all available treatment nodes (N = 37). In total, there were 65,601 patients in the primary analysis of percentage reduction in total lesion counts.
The treatment group of interest for this review (topical antibiotic, retinoid, and BPO node) included the following combination of therapies: topical antibiotic (either topical clindamycin or topical erythromycin, or topical nadifloxacin or topical minocycline) plus retinoid (either topical isotretinoin or topical tretinoin, or topical tazarotene, topical adapalene, or topical trifarotene) plus topical BPO. Specifically, the category of interest included IDP-126,50 topical tretinoin or clindamycin plus BPO 0.025%, 1%, or 5%;51,52 topical adapalene 0.1% plus clindamycin 1% and BPO 5%;53 and topical tazarotene 0.1% plus clindamycin 1% and BPO 5%.54 Of note, none of these drugs are marketed in Canada.
Figure 4: Network Diagram for Percentage Reduction in Total Lesion Count (N = 37 Treatment Groups)
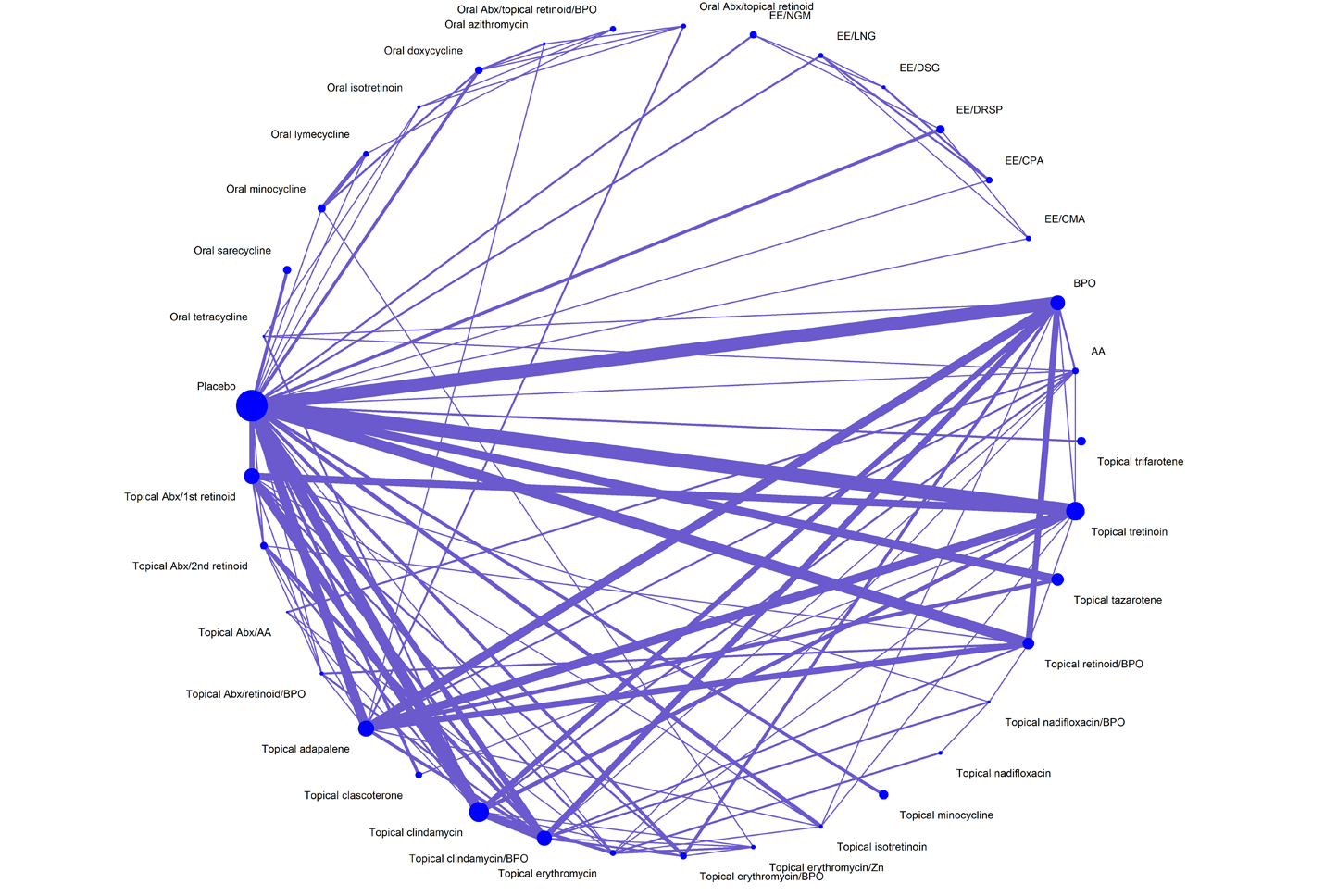
AA = azelaic acid; Abx = antibiotic; BPO = benzoyl peroxide; CMA = chlormadinone acetate; CPA = cyproterone acetate; DRSP = drospirenone; DSG = desogestrel; EE = ethinyl estradiol; LNG = levonorgestrel; NGM = norgestimate; Zn = zinc.
Source: Network meta-analysis conducted by Huang et al.17
Efficacy
The results of the NMAs of all outcomes of interest are summarized in Table 26. Only comparisons between triple therapy (i.e., a topical antibiotic, a topical retinoid, and BPO) and placebo were available in the published articles; therefore, these are the comparisons presented subsequently.
Proportion of Patients Experiencing Treatment Success
Findings on the proportion of patients with the outcome of treatment success, based on 69 trials, demonstrated that triple therapy was favoured in comparison with placebo (OR = 6.76; 95% CI, 4.33 to 10.55) (Table 26).
Total Lesion and Inflammatory and Noninflammatory Lesion Count
Results for the comparison of triple therapy versus placebo regarding percentage and absolute reductions in lesion counts are reported in Table 26. According to the estimated MDs, triple therapy demonstrated higher efficacy in the percentage reduction of total lesions (MD = 38.15; 95% CI, 27.40 to 48.90) and inflammatory (MD = 33.04; 95% CI, 25.73 to 40.34) and noninflammatory (MD = 32.65; 95% CI, 24.69 to 40.60) lesions compared with the placebo group. Similarly, absolute reductions in total, inflammatory, and noninflammatory lesions were observed for the comparison of triple therapy with placebo, with MDs of 24.97 (95% CI, 17.55 to 32.39), 8.90 (95% CI, 5.36 to 12.43), and 15.46 (95% CI, 10.13 to 20.78) in total, inflammatory, and noninflammatory lesions, respectively.
Statistical heterogeneity, assessed with the I2 statistic, was reported for the networks of percentage reduction of lesion counts: 79% for total lesions, 67% for inflammatory lesions, and 61% for noninflammatory lesions.
Limited inconsistency was observed, based on the node-splitting analysis and net heat plot assessment. There were 9 out of 111, 9 out of 118, and 9 out of 110 pairwise comparisons in the networks of percentage reduction in total, inflammatory, and noninflammatory lesion counts, respectively, showing significant inconsistency between direct and indirect estimates.
There was no indication of asymmetry suggesting publication bias, based on the visual assessment of funnel plots for all lesion count outcomes.
Two sensitivity analyses excluding studies published before 1985 and studies of low quality suggested consistent findings with the base-case analyses.
Discontinuation Due to Adverse Events
The results for the comparison of triple therapy with placebo regarding AEs are reported in Table 26. Topical antibiotic plus retinoid and BPO therapy was found to be associated with a higher risk of discontinuation when compared with placebo (OR = 3.08; 95% CI, 1.02 to 9.36).
Table 26: Summary of Triple-Therapy Comparisons From the Random-Effects NMA Published by Huang et al., Base-Case Analyses
NMA comparison Outcome | Topical antibiotic plus retinoid and BPOa versus placebo | ||
|---|---|---|---|
Number of studies in the network | Point estimate | 95% CI | |
Proportion of patients with treatment successb,c | 69 | 6.76 | 4.33 to 10.55 |
Lesion counts (percentage reduction) | |||
Total lesionsd | 190 | 38.15 | 27.40 to 48.90 |
Inflammatory lesionsd | 204 | 33.04 | 25.73 to 40.34 |
Noninflammatory lesionsd | 187 | 32.65 | 24.69 to 40.60 |
Lesion counts (absolute reduction) | |||
Total lesionsd | 176 | 24.97 | 17.55 to 32.39 |
Inflammatory lesionsd | 186 | 8.90 | 5.36 to 12.43 |
Noninflammatory lesionsd | 169 | 15.46 | 10.13 to 20.78 |
Discontinuation due to AEsb | 132 | 3.08 | 1.02 to 9.36 |
AE = adverse event; BPO = benzoyl peroxide; CI = confidence interval; IGA = Investigator’s Global Assessment; NMA = network meta-analysis.
aThe topical antibiotic plus retinoid and BPO category included IDP-126 (clindamycin 1.2% plus adapalene 0.15% and BPO 3.1%),50 topical tretinoin 0.025% plus clindamycin 1% and BPO 5%,51,52 topical adapalene 0.1% plus clindamycin 1% and BPO 5%,53 and topical tazarotene 0.1% plus clindamycin 1% and BPO 5%.54
bPoint estimate = odds ratio.
cTreatment success was defined as the proportion of participants experiencing treatment success, defined as an improvement of at least 2 grades from baseline and/or reaching clear or almost clear on the IGA of acne severity.
dPoint estimate = mean difference.
Source: NMA conducted by Huang et al.17
Critical Appraisal of the Published ITC (Huang et al.)
The published NMA conducted by Huang et al. followed appropriate methodology for conducting systematic literature reviews. However, details regarding the review protocol were not available in the publication, limiting the ability to assess the performance of the review. Evidence from the small studies (fewer than 200 participants) was not incorporated in the network, as per the review’s exclusion criteria, leading to possible bias in the NMA estimates.
In this ITC, differences were observed in the study and patients characteristics, mainly treatment duration (median = 12 weeks; range, 2 to 48 weeks) and disease severity at baseline (median inflammatory lesion count = 27; range, 4 to 83; median noninflammatory lesion count = 44; range, 10 to 145), which may have influenced the overall NMA estimates.
The results of the 2 sensitivity analyses were consistent with the base case. No evidence of publication bias was observed, according to the assessment of the funnel plots.
Regarding generalizability, the published article provided efficacy estimates for active treatments in reference to placebo only. Moreover, the treatment node of interest for this review (topical antibiotic, retinoid, and BPO) included IDP-126 gel and other active combination treatments, none of which are marketed for use in Canada. As such, the findings from the analyses are of limited applicability to the Canadian clinical context.
Studies Addressing Gaps in the Systematic Review Evidence
Contents within this section have been informed by materials submitted by the sponsor. The following has been summarized and validated by the review team.
In the absence of direct comparative evidence of IDP-126 gel versus other relevant comparators in the treatment of acne, the sponsor submitted 2 phase II trials. This section summarizes 2 studies — Study 201, which assessed IDP-126 gel versus each of its component products and vehicle, and Study 202, which assessed IDP-126 gel versus adapalene 0.3% plus BPO 2.5% topical gel — to address this evidence gap in the pivotal trials, Study 301 and Study 302.
Table 27: Summary of Gaps in the Systematic Review Evidence
Evidence gap identified by sponsor | Studies that address gaps | |
|---|---|---|
Study description | Summary of key results | |
In studies 301 and 302, IDP-126 gel was compared with its vehicle gel only and not with the dyad components. | Study 201 is a phase II, multicentre, randomized, double-blind, vehicle-controlled, parallel-group study to assess the safety, tolerability, and efficacy of IDP-126 gel in comparison with each of its component products and vehicle to establish the contribution of the elements. | The percentage of patients with at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 was 52.5% in the IDP-126 gel group, 27.8% in the component Aa group, 30.5% in the component Bb group, 30.3% in the component Cc group, and 8.1% in the vehicle gel group. The LS mean change from baseline in inflammatory lesion count at week 12 was −29.9 (SD = 11.86) in the IDP-126 gel group, −26.7 (SD = 11.74) in the component A group, −24.8 (SD = 11.71) in the component B group, −26.8 (SD = 11.69) in the component C group, and −19.6 (SD = 12.12) in its vehicle gel group. The LS mean change from baseline in noninflammatory lesion count at week 12 was −35.5 (SD = 16.25) in the IDP-126 gel group, −29.9 (SD = 16.40) in the component A group, −27.8 (SD = 15.97) in the component B group, −30.0 (SD = 16.40) in the component C group, and −21.8 (SD = 16.58) in its vehicle gel group. |
In studies 301 and 302, IDP-126 gel was not compared with an active comparator used for the treatment of acne. | Study 202 is a phase II, multicentre, randomized, double-blind, vehicle-controlled study to compare the safety and efficacy of IDP-126 gel versus adapalene 0.3% plus BPO 2.5% in the treatment of acne vulgaris. | ███ ████████ ██ ████████ █████████ █████████ ███████ ███ ██ ███ ████ ██ ████ ██ ███ █████ ███ █████ ██ ███ ███████ ███ █████ ███ ███████ ████████ ████ ██████ █████████████ █████████ ██████████ ███ ██████ ████ ███ █████ ████████ █ █████ █ ███████ ███ █████████ ████████ ██ ███ ████ ████████ ████ ████ ████████ ████████████ ██████ ███ ██ ████ ██ ███████ ███████ ███ ███ ████████ █████ ███ █████ ███ ██████ ████ ███ ██████ ██ ████ █ █████ █ ███████ ██ ██████ ██ ███████ ████ ███ ███████ ██████████ ██ ███ ████ ████████ ██████ ████ ██████ ██ ███████████████ ██████ █████ ██ ██ ██ ███████ ███████ ███ ███ ██████ █████ ███ █████ ███ ██████ ████ ███ ██████ ██ ███████ █ ███ █ ███████ ██ ██████ ██ ███████ ████ █████████ ██ ███████ ███ ██ █████ ██████ ████ ███ ███ █████████ █████ ██ ███ ████████ ██ █████ ████ ██████ ██████ ████ ██ ██ ███ |
BPO = benzoyl peroxide; CI = confidence interval; EGSS = Evaluator’s Global Severity Score; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; SD = standard deviation.
aComponent A: BPO 3.1% plus adapalene 0.15% topical gel.
bComponent B: Clindamycin phosphate 1.2% plus BPO 3.1% topical gel.
cComponent C: Clindamycin phosphate 1.2% plus adapalene 0.15% topical gel.
Source: Details included in the Table are from the sponsor’s summary of clinical evidence.15
Description of Study 201
Study 201 is a phase II, multicentre, randomized, double-blind, vehicle-controlled, parallel-group study that assessed the safety, tolerability, and efficacy of IDP-126 gel in comparison with each of its component products and its vehicle gel in patients aged 9 years and older with a clinical diagnosis of moderate to severe acne (N = 741):
Component A: BPO 3.1% plus adapalene 0.15% topical gel
Component B: Clindamycin phosphate 1.2% plus BPO 3.1% topical gel
Component C: Clindamycin phosphate 1.2% plus adapalene 0.15% topical gel.
Patients were enrolled at a total of 35 study centres across North America, including 4 centres in Canada. The duration of the study was 12 weeks and included the following study visits: screening (day −35 to 0), baseline (day 0), and weeks 2, 4, 8, and 12. Patients who met the inclusion criteria and none of the exclusion criteria were randomized at the baseline visit to receive treatment with either IDP-126 gel, component A, component B, component C, or its vehicle gel in a 1:1:1:1:1 ratio using the Oracle Interactive Response Technology system. A study drug kit was dispensed to patients at baseline and at weeks 4 and 8 by the study centre staff. The patients were required to return their kits, including all used and unused study drug, at weeks 4, 8, and 12.
Populations
The inclusion and exclusion criteria for Study 201 were identical to the criteria used in studies 301 and 302, which are presented in Table 5.
Interventions
Patients applied their assigned study drug topically once daily in the evening for 12 weeks. More specifically, study drug application began at the baseline visit and continued through the evening before the week 12 visit. The study drug was applied to the face in the same manner as in studies 301 and 302, described in the intervention subsection of the systematic review section in this report.
Outcomes
Coprimary Efficacy Outcomes
The coprimary outcomes assessed in Study 201 were similar to those in studies 301 and 302. These outcomes included:
absolute change in inflammatory lesion count from baseline at week 12
absolute change in noninflammatory lesion count from baseline at week 12
percentage of patients with at least a 2-grade reduction in the EGSS from baseline and an EGSS that equated to clear or almost clear at week 12 (i.e., treatment success).
The secondary outcomes of interest to this review included:
absolute changes in inflammatory and noninflammatory lesion counts from baseline at weeks 2, 4, and 8
percentage of patients with treatment success defined using the EGSS at weeks 2, 4, and 8
percentage of patients with at least a 2-grade reduction in the EGSS from baseline at weeks 2, 4, 8, and 12
percent changes in inflammatory and noninflammatory lesion counts from baseline at weeks 2, 4, 8, and 12.
The Acne-QoL questionnaire was included as part of other assessments in this study.
Harms
AEs and SAEs were assessed as part of the safety assessments in this study. Information on the notable harms selected for the purpose of this review is described in the Outcomes subsection of the systematic review section in this report.
Statistical Analysis
Coprimary End Points
The coprimary efficacy end points were summarized using descriptive and inferential statistics. No adjustments were made for multiple comparisons.
The absolute changes in mean inflammatory and noninflammatory lesion counts from baseline to week 12 were analyzed using an ANCOVA with treatment group and analysis centre as factors and the respective baseline lesion count as a covariate. Four pairwise tests were conducted to compare IDP-126 gel with its vehicle gel and with each of its combination component products. For the primary efficacy end point, if a treatment by analysis centre interaction effect was significant at an alpha of less than 0.10, then the effect was included in the model; otherwise, it was removed.
A skewness test was applied to the residuals resulting from the ANCOVA. A 2-sided P value for the skewness test that was significant at an alpha of 0.01 implied the use of a nonparametric method. If a parametric analysis was indicated, the results of the parametric analysis were considered the primary analysis. If a nonparametric analysis was indicated, the absolute changes in inflammatory and noninflammatory lesion counts were rank transformed before being submitted to the ANCOVA. The results of the rank-transformed analyses were then considered the primary analysis; however, the results of the nonrank-transformed analyses were also presented.
The percent of patients with treatment success at week 12 was analyzed using a logistic regression test with treatment group and analysis centre as factors. If there was quasi-complete separation of the data that prevented the model from converging, the Firth option was used to apply Firth’s penalized likelihood to allow the model to converge. Four pairwise tests were conducted to compare IDP-126 gel with its vehicle gel and each of its combination component products.
Secondary End Points
All secondary efficacy end points were summarized using descriptive statistics. No inferential analyses were conducted.
Other End Points
Descriptive statistics were used to summarize the data reported for each domain of the Acne-QoL questionnaire. According to the statistical analysis plan, no inferential analyses were conducted. Each domain was the sum of the values from the questions it comprised, provided at least 3 of its questions had nonmissing results. For domains that met this criterion, any missing values were imputed through mean substitution before calculating the domain score.
Analysis Populations
Efficacy was analyzed using the ITT and PP populations. The ITT population consisted of all randomized patients who were dispensed the study drug. The PP population consisted of all patients in the safety population who completed the 12-week evaluation without any noteworthy protocol deviations.
Safety was analyzed using the safety population, which consisted of all randomized patients who were presumed to have used the study drug at least once and who had at least 1 postbaseline evaluation. For the efficacy analyses conducted with the ITT population, imputations for missing data were based on estimations using the MCMC imputation method. For the efficacy analyses conducted with the PP population, the last observation carried forward method was used to impute missing data. No imputations were made for missing safety data.
Results
Patient Disposition
A summary of the patient disposition from Study 201 is presented in Table 28. A total of 741 patients were randomized and 649 patients (87.6%) completed the study. Study discontinuation rates were higher in the IDP-126 gel group (15.0%) and IDP-126 vehicle gel group (14.2%) compared with the IDP-126 component products groups (component A group, 12.0%; component B group, 8.9%; component C group, 12.0%). The most common reasons for study discontinuation were lost to follow-up (6.1% in the IDP-126 gel group, 4.7% in the IDP-126 component A group and IDP-126 vehicle gel group, 4.0% in the IDP-126 component C group, and 3.4% in the IDP-126 component B group) and patient request (6.1% in the IDP-126 vehicle gel group, 4.7% in the IDP-126 component C group, 3.4% in the IDP-126 vehicle gel group, 2.7% in the IDP-126 component B group, and 0.7% in the IDP-126 component A group).
Table 28: Summary of Patient Disposition From Study 201 (All Randomized Patients)
Patient disposition | IDP-126 gel | IDP-126 component A | IDP-126 component B | IDP-126 component C | IDP-126 vehicle gel |
|---|---|---|---|---|---|
Screened, N | 908 | ||||
Randomized, N (%) | 147 (100) | 150 (100) | 146 (100) | 150 (100) | 148 (100) |
Discontinued from study, n (%) | 22 (15.0) | 18 (12.0) | 13 (8.9) | 18 (12.0) | 21 (14.2) |
Reason for discontinuation, n (%)a | — | — | — | — | — |
Lost to follow-up | 9 (6.1) | 7 (4.7) | 5 (3.4) | 6 (4.0) | 7 (4.7) |
Patient request | 5 (3.4) | 1 (0.7) | 4 (2.7) | 7 (4.7) | 9 (6.1) |
AEs | 4 (2.7) | 8 (5.3) | 0 | 3 (2.0) | 1 (0.7) |
Withdrawal by parent or guardianb | 1 (0.7) | 2 (1.3) | 3 (2.1) | 1 (0.7) | 1 (0.7) |
Pregnancy | 1 (0.7) | 0 | 0 | 1 (0.7) | 0 |
Protocol violation | 1 (0.7) | 0 | 0 | 0 | 1 (0.7) |
Other | 1 (0.7) | 0 | 0 | 0 | 1 (0.7) |
Lack of efficacy | 0 | 0 | 1 (0.7) | 0 | 0 |
Worsening condition | 0 | 0 | 0 | 0 | 1 (0.7) |
ITT, N | 146 (99.3) | 150 (100.0) | 146 (100.0) | 150 (100.0) | 148 (100.0) |
PP, N | 113 (76.9) | 115 (76.7) | 114 (78.1) | 116 (77.3) | 111 (75.0) |
Safety, N | 141 (95.9) | 146 (97.3) | 144 (98.6) | 148 (98.7) | 146 (98.6) |
AE = adverse event; IDP-126 component A = benzoyl peroxide plus adapalene; IDP-126 component B = clindamycin phosphate plus benzoyl peroxide; IDP-126 component C = clindamycin phosphate plus adapalene; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; PP = per-protocol.
aOnly those reasons for which at least 1 patient in any study drug group discontinued are included in the table. The reasons are presented in descending order based on total frequency.
bRefers to the parent or legal guardian.
Sources: Study V01-Study 126A-201 Clinical Study Report (2019)55 and Patients by Site Study 201 (2022).56 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Baseline Characteristics
A summary of the baseline characteristics of the patients from Study 201 is presented in Table 29. The baseline characteristics outlined in this Table are limited to those that are most relevant to this review or that were felt to affect the outcomes or interpretation of the study results.
In Study 201, the mean age of patients randomized to each study drug group was similar: 19.9 years (SD = 7.0 years) in the IDP-126 gel group, 19.2 years (SD = 8.0 years) in the component A group, 19.6 years (SD = 6.7 years) in the component B group, 19.4 years (SD = 6.5 years) in the component C group, and 19.6 years (SD = 7.1 years) in the vehicle gel group. The age range of patients was similar across the study drug groups, ranging from 10 to 60 years.
In each study drug group, slightly more patients were female: 64.4% in the IDP-126 gel group, 57.3% in the component A group, 62.3% in the component B group, 62.0% in the component C group, and 60.1% in the vehicle gel group. The remainder of the patients were male: 35.6% in the IDP-126 gel group, 42.7% in the component A group, 37.7% in the component B group, 38.0% in the component C group, and 39.9% in the vehicle gel group.
Most patients randomized to each study drug group were white: 67.1% in the IDP-126 gel group, 72.7% in the component A group, 69.2% in the component B group, 72.7% in the component C group, and 64.2% in the vehicle gel group. Other races included in the study were Black or African American, Asian, native Hawaiian or other Pacific Islander, and American Indian or Alaska Native.
The majority of patients in each study drug group had a clinical diagnosis of moderate acne, defined as a baseline EGSS of 3: 84.9% in the IDP-126 gel group, 79.3% in the component A group, 84.9% in the component B group, 86.0% in the component C group, and 85.8% in the vehicle gel group. The remainder of patients in each study drug group had a clinical diagnosis of severe acne, defined as a baseline EGSS of 4: 15.1% in the IDP-126 gel group, 20.7% in the component A group, 15.1% in the component B group, 14.0% in the component C group, and 14.2% in the vehicle gel group.
The mean inflammatory lesion count at baseline was similar between the study drug groups: 39.0 (SD = 11.8) in the IDP-126 gel group, 39.0 (SD = 10.2) in the component A group, 40.0 (SD = 12.8) in the component B group, 38.2 (SD = 7.9) in the component C group, and 38.2 (SD = 9.2) in the vehicle gel group. The inflammatory lesion count range was also similar across all groups, ranging from 30 to 94. The mean noninflammatory lesion count at baseline was similar between the study drug groups: 51.8 (SD = 20.3) in the IDP-126 gel group, 48.0 (SD = 14.7) in the component A group, 49.2 (SD = 17.6) in the component B group, 51.1 (SD = 18.4) in the component C group, and 50.7 (SD = 18.7) in the vehicle gel group. The inflammatory lesion count range was also similar across all groups, ranging from 35 to 150.
Table 29: Summary of Baseline Characteristics of Study 201 (ITT Population)
Characteristic | IDP-126 gel | IDP-126 component A | IDP-126 component B | IDP-126 component C | IDP-126 Vehicle gel |
|---|---|---|---|---|---|
Patient demographic characteristics | |||||
Age (years) | — | — | — | — | — |
Mean (SD) | 19.9 (7.0) | 19.2 (8.0) | 19.6 (6.7) | 19.4 (6.5) | 19.6 (7.1) |
Median (range) | 17 (11 to 46) | 17 (10 to 60) | 17 (10 to 45) | 17 (11 to 50) | 17 (11 to 47) |
Sex, n (%) | — | — | — | — | — |
Male | 52 (35.6) | 64 (42.7) | 55 (37.7) | 57 (38.0) | 59 (39.9) |
Female | 94 (64.4) | 86 (57.3) | 91 (62.3) | 93 (62.0) | 89 (60.1) |
Race, n (%) | — | — | — | — | — |
White | 98 (67.1) | 109 (72.7) | 101 (69.2) | 109 (72.7) | 95 (64.2) |
Black or African American | 24 (16.4) | 26 (17.3) | 30 (20.5) | 20 (13.3) | 26 (17.6) |
Asian | 10 (6.8) | 6 (4.0) | 8 (5.5) | 9 (6.0) | 17 (11.5) |
Native Hawaiian or other Pacific Islander | 2 (1.4) | 4 (2.7) | 0 | 1 (0.7) | 0 |
American Indian or Alaska Native | 1 (0.7) | 1 (0.7) | 1 (0.7) | 1 (0.7) | 2 (1.4) |
Other or multiple | 11 (7.5) | 4 (2.7) | 6 (4.1) | 10 (6.7) | 8 (5.4) |
Patient characteristics relating to acne | |||||
EGSS, n (%) | — | — | — | — | — |
0 (clear) | 0 | 0 | 0 | 0 | 0 |
1 (almost clear) | 0 | 0 | 0 | 0 | 0 |
2 (mild) | 0 | 0 | 0 | 0 | 0 |
3 (moderate) | 124 (84.9) | 119 (79.3) | 124 (84.9) | 129 (86.0) | 127 (85.8) |
4 (severe) | 22 (15.1) | 31 (20.7) | 22 (15.1) | 21 (14.0) | 21 (14.2) |
Inflammatory lesion count | — | — | — | — | — |
Mean (SD) | 39.0 (11.8) | 39.0 (10.2) | 40.0 (12.8) | 38.2 (7.9) | 38.2 (9.2) |
Median (range) | 35.0 (30 to 89) | 36.0 (30 to 79) | 35.5 (30 to 94) | 36.0 (30 to 81) | 35.0 (30 to 74) |
Noninflammatory lesion count | — | — | — | — | — |
Mean (SD) | 51.8 (20.3) | 48.0 (14.7) | 49.2 (17.6) | 51.1 (18.4) | 50.7 (18.7) |
Median (range) | 43.0 (35 to 150) | 43.0 (35 to 132) | 42.0 (35 to 144) | 44.0 (35 to 136) | 42.5 (35 to 126) |
EGSS = Evaluator’s Global Severity Score; IDP-126 component A = benzoyl peroxide plus adapalene; IDP-126 component B = clindamycin phosphate plus benzoyl peroxide; IDP-126 component C = clindamycin phosphate plus adapalene; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; SD = standard deviation.
Sources: Study V01-126A-201 Clinical Study Report (2019).55 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Exposure to Study Treatments
A summary of patient exposure from Study 201 is presented in Table 30. The total amount of study drug used, total number of days of exposure, and total number of applications were similar among the study treatment groups in Study 201. ███████ ████ ███ ██ ████████ ████ ██████████ █████████ ████████ ██ █████ ███ ███ ███ ████ ████ ████ █ ███████████ ████ ██████ ███ ███████ ███ ██ ████ ██ ████████ ████████████ █████ █████████████ ██ ███ ██████ ██████ ███ █████ ████ ███████
Table 30: Summary of Patient Exposure from Study 201 (Safety Population)
Exposure | IDP-126 gel | IDP-126 component A | IDP-126 component B | IDP-126 component C | IDP-126 vehicle gel |
|---|---|---|---|---|---|
Total amount of study drug used (g) | — | — | — | — | — |
|| | ███ | ███ | ███ | ███ | ███ |
████ ████ | ████ ██████ | ████ ██████ | ████ ██████ | ████ ██████ | ████ ██████ |
██████ ███████ | ████ ██ ██ ████ | ████ ██ ██ ████ | ████ ███ ██ ████ | ████ ██ ██ ████ | ████ ██ ██ ████ |
Total number of days of exposure | — | — | — | — | — |
|| | ███ | ███ | ███ | ███ | ███ |
████ ████ | ████ ██████ | ████ ██████ | ███ █████ | ████ ██████ | ████ ██████ |
██████ ███████ | ████ ██ ██ ████ | ████ ██ ██ ████ | ████ ███ ██ ████ | ████ ██ ██ ████ | ████ ██ ██ ████ |
Total number of applications | — | — | — | — | — |
|| | ███ | ███ | ███ | ███ | ███ |
████ ████ | ████ ██████ | ████ ██████ | ████ ██████ | ████ ██████ | ████ ██████ |
██████ ███████ | ████ ██ ██ ████ | ████ ██ ██ ████ | ████ ███ ██ ████ | ████ ██ ██ ████ | ████ ██ ██ ████ |
Complianta | — | — | — | — | — |
|| | ███ | ███ | ███ | ███ | ███ |
████ █ ███ | ███ ██████ | ███ ██████ | ███ ████ | ███ ██████ | ███ ██████ |
IDP-126 component A = benzoyl peroxide plus adapalene; IDP-126 component B = clindamycin phosphate plus benzoyl peroxide; IDP-126 component C = clindamycin phosphate plus adapalene; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; SD = standard deviation.
aA patient was considered compliant with the dosing regimen if the patient did not miss more than 5 consecutive days of dosing and applied 80% to 120% of the expected applications while participating in the study.
Sources: Study V01-126A-201 Clinical Study Report (2019).55 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Concomitant and Prior Therapies
Similar to studies 301 and 302, as presented in Table 6 in the systematic review section, patients in Study 201 were required to undergo specific washout periods for certain topical preparations or physical treatments used on the face, and for certain systemic medications before the study. | █████ ██ █ ████████ ████ ██ ███ ███████ ███ ██████ ██ █████████ █ ██████ ███ █████████ █ █████ ██████ ██ ████ ███ ███████████ ███████ ██ ███████ ██████ ███ ██████████ ████████████ ███ ████ ████ ████████ ████ ███ ██ ███████████
Efficacy
A summary of the key primary efficacy analysis results from Study 201 is presented in Table 31.
Acne Severity
Treatment Success Based on the EGSS
The percentage of patients with at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 was 52.5% in the IDP-126 gel group, 27.8% in the component A group, 30.5% in the component B group, 30.3% in the component C group, and 8.1% in the vehicle gel group. The treatment difference in treatment success based on the EGSS was not reported.
Lesion Count
Inflammatory Lesion Count
The LS mean change from baseline in inflammatory lesion count at week 12 was −29.9 (SD = 11.86) in the IDP-126 gel group, −26.7 (SD = 11.74) in the component A group, −24.8 (SD = 11.71) in the component B group, −26.8 (SD = 11.69) in the component C group, and −19.6 (SD = 12.12) in its vehicle gel group. The treatment difference in the mean absolute change from baseline in inflammatory lesion count at week 12 was not reported.
███ ████ ███████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ████ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ███ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ███ ██ ████ ███████ ██████ ███ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ████ ███ █████████
Noninflammatory Lesion Count
The LS mean change from baseline in noninflammatory lesion count at week 12 was −35.5 (SD = 16.25) in the IDP-126 gel group, −29.9 (SD = 16.40) in the component A group, −27.8 (SD = 15.97) in the component B group, −30.0 (SD = 16.40) in the component C group, and −21.8 (SD = 16.58) in its vehicle gel group. The treatment difference in the mean absolute change from baseline in noninflammatory lesion count at week 12 was not reported.
███ ████ ███████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ████ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ██████ ███ █ ██████ ██ █████████ █ ██████ ███ ██████ ███ █ ██████ ██ ███ ███████ ███ ██████ ███ ██ ████ ███████ ██████ ███ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ████ ███ █████████
Quality of Life
Self-Perception Domain Score in the Acne-Specific Quality of Life Questionnaire
The mean change from baseline in the Acne-QoL self-perception domain score at week 12 was 9.8 (SD = 8.80) in the IDP-126 gel group, 7.3 (SD = 8.34) in the component A group, 7.5 (SD = 7.22) in the component B group, 8.5 (SD = 8.22) in the component C group, and 5.9 (SD = 7.99) in its vehicle gel group. The treatment difference in the absolute change from baseline in the Acne-QoL self-perception domain score at week 12 was not reported.
Symptoms Domain Score in the Acne-Specific Quality of Life Questionnaire
The mean change from baseline in the Acne-QoL symptoms domain score at week 12 was 7.4 (SD = 6.19) in the IDP-126 gel group, 7.3 (SD = 6.52) in the component A group, 6.9 (SD = 5.63) in the component B group, 6.6 (SD = 6.07) in the component C group, and 4.9 (SD = 5.53) in its vehicle gel group. The treatment difference in the absolute change from baseline in the Acne-QoL symptoms domain score at week 12 was not reported.
Table 31: Summary of Key Efficacy Results From Study 201 (ITT Population)
Efficacy outcome | IDP-126 gel | IDP-126 component A | IDP-126 component B | IDP-126 component C | IDP-126 vehicle gel |
|---|---|---|---|---|---|
Acne severity | |||||
Percentage of patients with ≥ 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 | |||||
Treatment success, % | 52.5 | 27.8 | 30.5 | 30.3 | 8.1 |
Treatment failure, % | 47.5 | 72.2 | 69.5 | 69.7 | 91.9 |
Contrast P valuea | — | < 0.001 | 0.001 | 0.001 | < 0.001 |
Lesion count | |||||
Absolute change from baseline in the inflammatory lesion count at week 12 | |||||
Baseline lesion count, mean (SD) | 39.0 (11.84) | 39.0 (10.19) | 40.0 (12.79) | 38.2 (7.91) | 38.2 (9.19) |
Week 12 lesion count, mean (SD) | 9.0 (9.65) | 12.3 (10.23) | 14.4 (16.34) | 11.9 (10.14) | 19.1 (12.94) |
LS change from baseline, mean (SD) | −29.9 (11.86) | −26.7 (11.74) | −24.8 (11.71) | −26.8 (11.69) | −19.6 (12.12) |
P valueb | — | 0.013 | 0.003 | 0.026 | < 0.001 |
Percent change from baseline in the inflammatory lesion count at week 12 | |||||
████ ████ | ████████ | ██████ ███████ | ██████ ███████ | ██████ ███████ | ██████ ███████ |
Absolute change from baseline in the noninflammatory lesion count at week 12 | |||||
Baseline lesion count, mean (SD) | 51.8 (20.26) | 48.0 (14.72) | 49.2 (17.64) | 51.1 (18.36) | 50.7 (18.74) |
Week 12 lesion count, mean (SD) | 15.5 (14.93) | 18.9 (16.15) | 21.6 (22.07) | 20.6 (19.26) | 28.5 (22.77) |
LS change from baseline, mean (SD) | −35.5 (16.25) | −29.9 (16.40) | −27.8 (15.97) | −30.0 (16.40) | −21.8 (16.58) |
P valueb | — | 0.004 | < 0.001 | 0.005 | < 0.001 |
Percent change from baseline in the noninflammatory lesion count at week 12 | |||||
████ ████ | ████████ | ██████ ███████ | ██████ ███████ | ██████ ███████ | ██████ ███████ |
Quality of life | |||||
Absolute change from baseline in the Acne-QoL self-perception domain score at week 12 | |||||
n | 126 | 133 | 134 | 132 | 127 |
█████ ███ ████ ████ | ████ ███ | ████ ██████ | ████ ████ | ████ ██████ | ████ ██████ |
███ ██ █ ████ ████ | ████ ███ | ████ ██████ | ████ ████ | ████ ██████ | ████ ██████ |
Change from baseline, mean (SD) | 9.8 (8.80) | 7.3 (8.34) | 7.5 (7.22) | 8.5 (8.22) | 5.9 (7.99) |
Absolute change from baseline in Acne-QoL acne symptoms domain score at week 12 | |||||
n | 126 | 133 | 134 | 132 | 127 |
███ ████ ████ ████ | ████ ████ | ████ ██████ | ████ ████ | ████ ██████ | ████ ██████ |
███ ██ ███ ████ ████ | ████ ████ | ████ ██████ | ████ ████ | ████ ██████ | ████ ██████ |
Change from baseline, mean (SD) | 7.4 (6.19) | 7.3 (6.52) | 6.9 (5.63) | 6.6 (6.07) | 4.9 (5.53) |
Acne-QoL = Acne-Specific Quality of Life; EGSS = Evaluator’s Global Severity Score; IDP-126 component A = benzoyl peroxide plus adapalene; IDP-126 component B = clindamycin phosphate plus benzoyl peroxide; IDP-126 component C = clindamycin phosphate plus adapalene; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; LS = least squares; NR = not reported; SD = standard deviation.
Note: Multiple imputation (MCMC) was used to impute missing values in the efficacy analyses conducted with the ITT population, missing data on lesion count and EGSS at week 12 (from which treatment success was derived). Contrast P values are shown versus IDP-126 gel; P values are not adjusted for multiple comparisons and, therefore, there is a greater likelihood of type I error.
aContrast P values from a logistic regression with treatment group and analysis centre as factors.
bThis was the primary analysis due to skewness (skewness P value < 0.001), overall P value and contrast P values from a ranked analysis of covariance with treatment group and analysis centre as factors and the respective baseline lesion count as a covariate.
Sources: Study V01-126A-201 Clinical Study Report (2019).55 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Harms
A summary of harms results from Study 201 is presented in Table 32. ███ ███████ ███████████ ██ ████████ ██ ███ ███████ ████ ███████ █████████ ██ ███ ███████ █████████ █ ██████ ███████████ ██████ ██ ███ ██████ █████ █████ ██ ███████ █████████ ███ ██████████████ ████ ██████████ ███████ ██████ ███ ██████ ██████████████ ███ ███████ ███████████ ██ ████████ ██ ███ ███████ █████████ █ ███ ███████ ███████ ███ ██████ ███████████ ██████ ██ ███ ██████ █████ █████ ██ ██████████ ███ ████████████ █████ ███ █████ ██████████████
Adverse Events
The proportion of patients who reported at least 1 TEAE was similar in the IDP-126 gel and IDP-126 component A groups (36.2% and 35.6%, respectively), while the proportion of patients who reported a TEAE in the IDP-126 component B, IDP-126 component C, and IDP-126 vehicle gel groups was 18.1%, 27.0%, and 15.1%, respectively (Table 32). The most common TEAEs reported were application site pain (7.8% of patients in the IDP-126 gel group, 11.0% in the component A group, 0.7% in the component B group, 3.4% in the component C group, and 0.7% in its vehicle gel group), application site dryness (6.4% of patients in the IDP-126 gel group, 5.5% in the component A group, 1.4% in the component B group, 6.1% in the component C group, and 0.7% in its vehicle gel group), and application site exfoliation (3.5% of patients in the IDP-126 gel group, 2.1% in the component A group, 0.0% in the component B group, 1.4% in the component C group, and 0.7% in its vehicle gel group).
Serious Adverse Events
A total of 4 patients were reported with SAEs; 1 patient in the IDP-126 gel group experienced sickle cell anemia with crisis and 3 patients in the component C group experienced an SAE (hyperbilirubinemia, enteritis, and abortion induced; n = 1 each).
Withdrawal Due to Adverse Events
A total of 17 patients (4 in the IDP-126 gel group, 8 in the IDP-126 component A group, 3 in the IDP-126 component C group, and 2 in the IDP-126 vehicle vel group) had their study drug withdrawn due to TEAEs. A total of 16 patients (4 in the IDP-126 gel group, 8 in the IDP-126 component A group, 3 in the IDP-126 component C group, and 1 in the IDP-126 vehicle gel group) discontinued the study due to TEAEs. The most common TEAE reported to have led to the discontinuation of the study drug and/or study was application site pain (reported by 1.4% of patients in the IDP-126 gel group, 3.4% in the component A group, and 1.4% in the component C group), and erythema (0.7% of patients in each of the IDP-126 gel group, component A group, and component C group).
Mortality
There were no reports of patients who died in Study 201.
Notable Harms
| █████ ██ █████ ██ ████████ ██ ███ ███████ ███ ██████ ████ █ ██ █████████ █ ██████ ████ ██ █████████ █ ██████ █████ ██ █████████ █ ██████ ███ ████ ████████ ██ ███ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ███████ ████████ ███ ██████████████ ████ ████████████ █████ ██ ████ ██ ████████ ██ ███ ███████ ███ ██████ ███ █ ██ █████████ █ ██████ ████ ██ █████████ █ ██████ ████ ██ █████████ █ ██████ ███ ████ ██ ███ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ████ ███ ████████████ ██████ █████████
Table 32: Summary of Harms From Study 201 (Safety Population)
Adverse events | IDP-126 gel | IDP-126 component A | IDP-126 component B | IDP-126 component C | IDP-126 vehicle gel |
|---|---|---|---|---|---|
Adverse events, n (%) | |||||
Patients with ≥ 1 TEAE | 51 (36.2) | 52 (35.6) | 26 (18.1) | 40 (27.0) | 22 (15.1) |
Most common TEAEsa | — | — | — | — | — |
General disorders and administration site conditions | 29 (20.6) | 31 (21.2) | 4 (2.8) | 20 (13.5) | 5 (3.4) |
Application site pain | 11 (7.8) | 16 (11.0) | 1 (0.7) | 5 (3.4) | 1 (0.7) |
Application site dryness | 9 (6.4) | 8 (5.5) | 2 (1.4) | 9 (6.1) | 1 (0.7) |
Application site exfoliation | 5 (3.5) | 3 (2.1) | 0 | 2 (1.4) | 1 (0.7) |
Application site dermatitis | 3 (2.1) | 3 (2.1) | 0 | 2 (1.4) | 0 |
Application site irritation | 3 (2.1) | 5 (3.4) | 1 (0.7) | 4 (2.7) | 0 |
Application site pruritus | 3 (2.1) | 2 (1.4) | 1 (0.7) | 1 (0.7) | 0 |
Application site erythema | 2 (1.4) | 2 (1.4) | 1 (0.7) | 5 (3.4) | 0 |
Application site urticaria | 1 (0.7) | 0 | 0 | 2 (1.4) | 0 |
Application site acne | 0 | 1 (0.7) | 0 | 0 | 2 (1.4) |
Infections and infestations | 18 (12.8) | 16 (11.0) | 13 (9.0) | 12 (8.1) | 10 (6.8) |
Viral URTI | 10 (7.1) | 5 (3.4) | 3 (2.1) | 5 (3.4) | 5 (3.4) |
URTI | 5 (3.5) | 5 (3.4) | 5 (3.5) | 2 (1.4) | 0 |
Gastroenteritis viral | 2 (1.4) | 0 | 1 (0.7) | 0 | 0 |
Ear infection | 0 | 2 (1.4) | 0 | 0 | 0 |
Influenza | 0 | 2 (1.4) | 1 (0.7) | 1 (0.7) | 1 (0.7) |
Skin and subcutaneous tissue disorders | 4 (2.8) | 5 (3.4) | 1 (0.7) | 3 (2.0) | 2 (1.4) |
Dermatitis contact | 0 | 2 (1.4) | 0 | 00 | 0 |
Injury, poisoning, and procedural complications | 3 (2.1) | 8 (5.5) | 5 (3.5) | 7 (4.7) | 5 (3.4) |
Sunburn | 2 (1.4) | 5 (3.4) | 1 (0.7) | 2 (1.4) | 1 (0.7) |
Investigations | 1 (0.7) | 2 (1.4) | 1 (0.7) | 0 | 1 (0.7) |
█████ ████████ █████████████ █████████ | | █████ | | █████ | ||| | ||| | | █████ |
SAEs, n (%) | |||||
████████ ████ | | ███ | | █████ | ||| | ||| | | █████ | ||| |
██████ ████ ███████ ████ ██████ | | █████ | ||| | ||| | ||| | ||| |
█████████████████ | ||| | ||| | ||| | | █████ | ||| |
█████████ | ||| | ||| | ||| | | █████ | ||| |
████████ ███████ | ||| | ||| | ||| | | █████ | ||| |
Patients who stopped study drug and/or study due to a TEAE, n (%) | |||||
████████ ███ ███████ ███ ██ | | ████ | | █████ | | █████| | ||| | | █████ | | █████| |
█████████ ████ ████ | ||| | ||| | ||| | ||| | | █████ |
█████ ████ ████████ | ||| | | █████ | ||| | ||| | ||| |
████████ ████ █████ | | █████ | | █████ | ||| | ||| | ||| |
███████ ████ ██████ | | █████ | | █████ | ||| | | █████ | ||| |
█████ ████ ███████ | | █████ | ||| | ||| | ||| | ||| |
███████ ████ ████ | | █████ | | █████ | ||| | | █████ | ||| |
████████ ████ █████ | ||| | ||| | ||| | | █████ | ||| |
██████ ████ ███████ | | █████ | ||| | ||| | ||| | ||| |
█████████ | ||| | ||| | ||| | | █████ | ||| |
███████ | ||| | | █████ | ||| | ||| | ||| |
Deaths, n (%) | |||||
████████ ███ ████ | ||| | ||| | ||| | ||| | ||| |
IDP-126 component A = benzoyl peroxide plus adapalene; IDP-126 component B = clindamycin phosphate plus benzoyl peroxide; IDP-126 component C = clindamycin phosphate plus adapalene; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; SAE = serious adverse event; TEAE = treatment-emergent adverse event; URTI = upper respiratory tract infection.
aTEAEs experienced by more than 1 patient in either study drug group by system organ class and preferred term. At each level of system organ class or preferred term, patients reporting more than 1 TEAE were counted only once.
██ ████ ██████ █ ██████ ████████ ████████████ █████ ████ ██████ ███ █████ ███ ██ █ ██ ████ ██████ █ █████ ██ █ ████████ ████████████ ████ ███ █████ ████ ███ ███ ██████ █ ███████ ████████████ █████ ████ ███ ███ ███ ██████ ███ █ ███████ ████████████ ███ █████ ███ ███ ███ █████ █████ || ██ ████ ██████ █ ██████ ████████ ████████████ █████ ████ ██████ ███ █████ ███ ██ █ ██ ████ ██████ ████ ████████ █ ███████ ███ ████████████ ████ ███ █████ ████ ███ ███ █████ ███ ██ █ ████ ███ █ ███████ ███ ████████████ █████ ████ ███ ███ ███ █████ ███ ██ █ █████
For notable harms, refer to preceding data on general disorders and administration site conditions and skin and subcutaneous tissues disorders (TEAE categories) and SAEs.
Sources: Study V01-126A-201 Clinical Study Report (2019)55 and Adverse Events Listings.57 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Critical Appraisal
Internal Validity
The randomization and masking procedures in Study 201 were considered appropriate. Since it was a phase II trial aiming to provide preliminary evidence about the efficacy and harms of the study drug, the results cannot be considered confirmatory. Relevant patient demographic characteristics at baseline appeared to be well balanced between the study drug groups. No notable differences in the baseline EGSS and lesion counts between the study drug groups were identified. Similar to the pivotal trial, the washout periods used in the studies were considered adequate (i.e., prior use of therapies for the treatment of acne was not expected to have an impact on the efficacy results) and the list of prohibited treatments for acne (topical preparations, physical treatments, and systemic therapies) was considered comprehensive by the clinical expert. Only 3 patients had protocol deviations regarding the requirement related to the washout period for prohibited medications. No adjustments were made for multiple comparisons for primary and secondary outcomes and, therefore, there is a greater likelihood of type I error. For the outcomes on quality of life (Acne-QoL self-perception and symptom domains), no inferential analyses or multiplicity adjustments were conducted, as per the statistical analysis plan, so these data are considered supportive and no definitive conclusions could be drawn. Study discontinuation rates were similar between the pivotal trials and Study 201 (i.e., not high in the context of this patient population and rates are similar between groups for overall study discontinuation and reasons for study discontinuation). Similar to the pivotal trials, in consultation with the clinical expert, it was concluded that the study discontinuation rates are reasonable in the context of the therapeutic area and, as such, the risks of attrition bias and possible unblinding are unlikely.
Overall, no major concern for bias in the results was identified; however, the results cannot be interpreted as conclusive evidence due to the phase II trial design.
External Validity
The inclusion criteria used in Study 201 include the population of interest identified in the indication for IDP-126 gel: patients aged 9 years and older with a clinical diagnosis of moderate to severe acne vulgaris, defined as a baseline of EGSS of 3 (moderate) or 4 (severe) for facial acne. In consultation with the clinical expert, it was concluded that the inclusion criteria adequately capture (and, consequently, the study population is representative of) the patients seen in practice who would be candidates for IDP-126 gel. Similar to the pivotal trials, the majority of patients from each study (approximately ≥ 79.3% of patients in each study drug group) had moderate acne. The age range of patients was similar across 5 groups, ranging from 10 to 60 years in Study 201. In consultation with the clinical expert, it was concluded that topical fixed-dose combination therapies (i.e., retinoid plus BPO, antibiotic plus BPO, and retinoid plus antibiotic) are relevant comparators for IDP-126 gel in the Canadian practice setting. Similar to the pivotal trials, in consultation with the clinical expert, it was concluded that the outcome measures of acne severity and lesion counts used in Study 201 are applicable to Canadian clinical practice.
Overall, no notable concern on the generalizability of results to the population of interest in the Canadian setting was identified in the appraisal of Study 201.
Description of Study 202
Study 202 is a phase II, multicentre, randomized, double-blind, vehicle-controlled study that assessed the safety and efficacy of IDP-126 gel in comparison with adapalene 0.3% plus BPO 2.5% gel and vehicle in patients aged 12 years and older with a clinical diagnosis of moderate to severe acne (N = 686). Patients were enrolled at a total of 42 study centres in North America, including 6 centres in Canada. The duration of the study was 12 weeks and included the following study visits: screening (day −35 to 0), baseline (day 0), and weeks 2, 4, 8, and 12. Patients who met the inclusion criteria and none of the exclusion criteria were randomized in a 2:2:1:1 ratio, using an interactive web response system, to receive treatment with either IDP-126 gel, adapalene 0.3% plus BPO 2.5% gel, IDP-126 vehicle gel (stored at 2°C to 8°C), or IDP-126 vehicle gel (stored at a controlled room temperature).
For evaluation and reporting purposes, data on the 2 IDP-126 vehicle gel groups were combined and are referred to throughout this report as the combined vehicle gel group.
Populations
The inclusion and exclusion criteria for Study 202 were identical to the criteria in studies 301 and 302, as presented in Table 5, with the exception of age limit, as Study 202 enrolled patients aged 12 years and older.
Interventions
Patients applied their assigned study drug topically to the face (i.e., chin, cheeks, nose, and forehead) and, optionally, to the trunk once daily in the evening for 12 weeks. The study drug was applied in the same manner as in studies 301 and 302, described in the intervention subsection of the systematic review section in this report.
Outcomes
The outcomes assessed in Study 202 were similar to those assessed in studies 301 and 302.
There were 3 coprimary efficacy outcomes:
absolute change in inflammatory lesion count from baseline at week 12
absolute change in noninflammatory lesion count from baseline at week 12
percentage of patients with at least a 2-grade reduction in the EGSS from baseline and an EGSS that equated to clear or almost clear at week 12 (i.e., treatment success).
The secondary outcomes of interest for this review included the following:
absolute changes in inflammatory and noninflammatory lesion counts from baseline at weeks 2, 4, and 8
percentage of patients with treatment success defined using the EGSS at weeks 2, 4, and 8
percentage of patients with at least a 2-grade reduction in the EGSS from baseline at weeks 2, 4, 8, and 12
percent changes in the inflammatory and noninflammatory lesion counts from baseline at weeks 2, 4, 8, and 12
The Acne-QoL questionnaire was included as part of other assessments in this study.
Harms
AEs and SAEs were assessed as part of the safety assessments in this study. Information on the notable harms selected for the purpose of this review is described in the Outcomes subsection of the systematic review section in this report.
Statistical Analysis
Coprimary End Points
The overall type I error rate was controlled by testing the coprimary efficacy end points in a sequential process. The testing process terminated whenever a statistical test for a step was not significant. All subsequent tests for the remaining steps were then considered to be not significant. The order of testing was as follows:
IDP-126 gel versus the combined vehicle gel:
superiority based on treatment success in the dichotomized EGSS
superiority based on absolute changes in inflammatory lesion counts
superiority based on absolute changes in noninflammatory lesion counts
Adapalene 0.3% plus BPO 2.5% gel versus the combined vehicle gel:
superiority based on treatment success in the dichotomized EGSS
superiority based on absolute changes in inflammatory lesion counts
superiority based on absolute changes in noninflammatory lesion counts
IDP-126 gel versus adapalene 0.3% plus BPO 2.5% gel:
noninferiority based on treatment success in the dichotomized EGSS
noninferiority based on absolute changes in inflammatory lesion counts
noninferiority based on absolute changes in noninflammatory lesion counts
superiority based on treatment success in the dichotomized EGSS
superiority based on absolute changes in inflammatory lesion counts
superiority based on absolute changes in noninflammatory lesion counts.
Tests of superiority and noninferiority for the absolute changes from baseline in inflammatory and noninflammatory lesion counts were based on parametric methods. Specifically, the tests of superiority and noninferiority were based on an ANCOVA, with the study drug and analysis centre as factors and the respective baseline lesion count as a covariate. Nonparametric methods were included to confirm the consistency of the results and the nonparametric tests of superiority and noninferiority were based on ranked data submitted to an ANCOVA, with the study drug and analysis centre as factors and the respective baseline lesion count as a covariate. For the analysis of the coprimary efficacy end points, if a treatment by analysis centre interaction effect was significant (based on pooling analysis) at an alpha of less than 0.10, then the effect was included in the model; otherwise, it was removed.
The superiority of IDP-126 gel over the combined vehicle gel, adapalene 0.3% plus BPO 2.5% gel over the combined vehicle gel, and IDP-126 gel over the adapalene 0.3% plus BPO 2.5% gel for the absolute changes from baseline in lesion counts was based on 2-tailed tests of the null hypothesis, resulting in contrast P values of 0.05 or less.
The noninferiority of IDP-126 gel to adapalene 0.3% plus BPO 2.5% gel for the analyses of absolute changes from baseline to week 12 in both inflammatory and noninflammatory lesion counts was assessed with 95% CIs using noninferiority margins of 2.1 for the comparisons of each lesion type. The CIs of the difference in the means (calculated as IDP-126 gel minus adapalene 0.3% plus BPO 2.5% gel) for the absolute changes from baseline in inflammatory and noninflammatory lesion counts were based on an ANCOVA, with the study drug and analysis centre as factors and the respective baseline lesion count as a covariate.
In the analyses of the lesion counts, a skewness test was applied to the residuals resulting from the ANCOVA. A 2-sided P value for the skewness test that was significant at an alpha of 0.01 implied the use of nonparametric methods to check the consistency of the results with the parametric methods. If a nonparametric analysis was indicated, the absolute changes in noninflammatory and inflammatory lesion counts were rank transformed before submitting them to the ANCOVA.
For the superiority analyses of the percentage of patients achieving success in the EGSS, a logistic regression test was used with study drug group and analysis centre as factors. If there was a quasi-complete separation of the data that prevented the model from converging, Firth’s penalized likelihood was applied to allow the model to converge.
The noninferiority of IDP-126 gel to adapalene 0.3% plus BPO 2.5% gel for the percentage of patients with treatment success according to the EGSS was assessed with 95% CIs and a noninferiority margin of 10%. In this analysis, if the study drug treatment by analysis centre interaction effect was significant based on the pooling analysis at an alpha of less than 0.10, then the effect was included in the model; otherwise, it was removed. The steps of the analyses were analogous to those used in the analyses of lesion counts, with logistic regression replacing the ANCOVA.
Secondary End Points
The secondary efficacy end points compared IDP-126 gel with the combined vehicle gel. To control for the overall type I error, failure of any 1 of the coprimary efficacy end points invalidated the statistical significance of all secondary efficacy end points. To control for multiplicity in the secondary efficacy end points, the analyses were conducted in a stepwise manner following the order presented subsequently. The testing process terminated whenever a statistical test for a step was not significant. The secondary efficacy end points were assessed for differences between IDP-126 gel and the combined vehicle gel.
Percent change in inflammatory lesion counts from baseline at week 12.
Percent change in noninflammatory lesion counts from baseline at week 12.
Percentage of patients who achieved at least a 2-grade reduction at week 8 from baseline in the EGSS and with an EGSS at the same visit that equated to clear or almost clear.
Absolute change in inflammatory lesion counts from baseline at week 8.
Absolute change in noninflammatory lesion counts from baseline at week 8.
Percent change in inflammatory lesion counts from baseline at week 8.
Percent change in noninflammatory lesion counts from baseline at week 8.
Percentage of patients who achieved at least a 2-grade reduction at week 4 from baseline in the EGSS and with an EGSS at the same visit that equated to clear or almost clear.
Absolute change in inflammatory lesion counts from baseline at week 4.
Absolute change in noninflammatory lesion counts from baseline at week 4.
Percent change in inflammatory lesion counts from baseline at week 4.
Percent change in noninflammatory lesion counts from baseline at week 4.
Percentage of patients who achieved at least a 2-grade reduction at week 2 from baseline in the EGSS and with an EGSS at the same visit that equated to clear or almost clear.
Absolute change in inflammatory lesion counts from baseline at week 2.
Absolute change in noninflammatory lesion counts from baseline at week 2.
Percent change in inflammatory lesion counts from baseline at week 2.
Percent change in noninflammatory lesion counts from baseline at week 2.
The methods used to analyze the lesion count and EGSS end points were the same as those used to analyze the variables in the coprimary efficacy end point analysis, except that the study drug treatment by analysis centre interaction effect was not tested and, therefore, was not included in the model.
Other End Points
For the other efficacy comparisons, the secondary efficacy end points were repeated to assess differences in the same end points between IDP-126 gel and adapalene 0.3% plus BPO 2.5% gel and between adapalene 0.3% plus BPO 2.5% gel and the combined vehicle gel. The methods used to analyze the lesion count and EGSS end points were the same as those used to analyze the variables in the coprimary efficacy end point analysis, except that the study drug treatment by analysis centre interaction effect was not tested and, therefore, was not included in the model.
Descriptive statistics were used to summarize the data reported for each domain of the Acne-QoL questionnaire; no inferential analyses were conducted. Each domain was the sum of the values from the questions it comprised, provided at least 3 of its questions had nonmissing results. For domains that met this criterion, any missing values were imputed through mean substitution before computing the domain score. Individual Acne-QoL scores and the domain scores were presented within separate by-patient listings.
Analysis Populations
The analysis populations (ITT, PP, and safety) used in Study 202 are the same as those defined in studies 301 and 302.
Results
███████ █████████████ ███████ ██ ███████ ███████████ ████ █████ ███ ██ █████████ ██ █████ ███ █ █████ ██ ███ ████████ ████ ██████████ ███ ███ ███████ █████████ ███ ██████ █████ ███████████████ █████ ████ ███████████ ███████ ██ ███ █████████ ████████ ████ ███ █████ ███████ ████████ ██ ███ ███████ ███ █████ ███████ ███ ████████ ███████ ███ █████ ████████ ███ ████ ██████ ███████ ███ █████ ███████████████ ████ █████ ████ ██ █████████ ██████ █████ ███ ████ ██ ███ ███████ ███ ██████ █████████ ████████ ████ ███ ██████ ███ ████████ ███████ ███ ██████ ██████████████ ███████ ███████ ██████ █████ ███ ████ ██ ███ ███████ ███ ██████ █████████ ████████ ████ ███ ██████ ███ ████████ ███████ ███ ██████ █████████████ ███ ██████████ ██ ███████████████ ██████ █████ ███ ████ ██ ███ ███████ ███ ██████ █████████ ████████ ████ ███ ██████ ███ ████████ ███████ ███ ██████ ██████████████
Table 33: Summary of Patient Disposition From Study 202 (All Randomized Patients)
Patient disposition | IDP-126 gel | Adapalene 0.3% plus BPO 2.5% gel | Combined vehicle gel |
|---|---|---|---|
█████████ | | ███ | ||
███████████ █ ███ | ███ █████ | ███████████ █ ███ | ███ █████ |
████████████ ████ ██████ █ ███ | ██ ██████ | ███████ ████ ██████ █ ███ | ██ ██████ |
██████ ███ ███████████ █ ███ | — | ███████████ | — |
████ ██ █████████ | ██ █████ | ████ ██ █████████ | ██ █████ |
███████ ███████ | | █████ | █████ ██████ | | █████ |
███████ ██████ | | █████ | ██████ █████ | | █████ |
██████████ ██ ███████████████ | | █████ | ███████████ | | █████ |
████████ █████████ | | █████ | ███████ ████ | | █████ |
█████████ | || | █████████ | || |
█████████████████ ██ ██████ | | █████ | █████████ ██████████ ███████ | | █████ |
█████ | | █████ | █████ | | █████ |
████ | | ███ ███████ | ████ | | ███ ███████ |
███ | | ███ ██████ | ███ | | ███ ██████ |
███████ | | ███ ███████ | ███████ | | ███ ███████ |
AE = adverse event; BPO = benzoyl peroxide; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; PP = per-protocol.
Sources: Study V01-126A-202 Clinical Study Report (2022)58 and Patients by Site Study 202 (2022).59 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Baseline Characteristics
A summary of baseline characteristics of patients from Study 202 is presented in Table 34.
██ █████ ████ ███ ████ ███ ██ ████████ ██████████ ██ ████ █████ ████ █████ ███ ███████ █ ████ █████ ███ █ ███ ██████ ██ ███ ███████ ███ ██████ ████ █████ ███ █ ███ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ █████ ███ █ ███ ██████ ██ ███ ████████ ███████ ███ ██████ ███ ███ █████ ██ ████████ ███ ███████ ██████ ███ ███████ ███████ ████ ██ ██ ██ ██████ ███ █████ ██████████ ██ ████ █████ ████ ██████ ████████ ████ ████████ ████ ██████ █ █████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ██ ███ ████████ ███████ ███ ██████ ███ █████████ ██ ████████ ████ ████ █ █████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ██ ███ ████████ ███████ ███ ███████████ ████████ ██████████ ██ ████ █████ ████ █████ ████ █████ █ █████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ██ ███ ████████ ███████ ███ ██████ █████ █████ ████████ ██ ███ █████ ████ █████ ██ ███████ █████████ ██████ ███ ████████ ██████ ██ ██████ ███████ ██ ██████ ████████ ██ █████ ███████ ████████ ████████ ████ ████████ ██ █████ ████████ ████████ ██ ████████ ██ ████ █████ ████ █████ ███ █ ████████ █████████ ██ ████████ █████ ███████ ██ █ ████████ ████ ██ | | █████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ██ ███ ████████ ███████ ███ ██████ ███ █████████ ██ ████████ ██ ████ █████ ████ █████ ███ █ ████████ █████████ ██ ██████ █████ ███████ ██ █ ████████ ████ ██ | | █████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ██ ███ ████████ ███████ ███ ██████████ ████ ████████████ ██████ █████ ██ ████████ ███ ███████ ██████ ███ █████ ████ ██████ █ ████ ███ █ █████ ██ ███ ███████ ███ ██████ ████ ███ █ ████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ███ █ █████ ██ ███ ████████ ███████ ███ ██████ ███ ████████████ ██████ █████ █████ ███ ████ ███████ ██████ █████ ████ ███████ ███████ ████ ██ ██ ███ ███ ████ ███████████████ ██████ █████ ██ ████████ ███ ███████ ███████ █████ ████ ██████ █ ████ ███ █ █████ ██ ███ ███████ ███ ██████ ████ ███ █ █████ ██████ █████████ ████████ ████ ███ ██████ ███ ████ ███ █ █████ ██ ███ ████████ ███████ ███ ██████ ███ ████████████ ██████ █████ █████ ███ ████ ████████ ███████ ████ ██ ██ ████
Table 34: Summary of Baseline Characteristics of Study 202 (ITT Population)
Characteristic | IDP-126 gel | Adapalene 0.3% plus BPO 2.5% gel | Combined vehicle gel |
|---|---|---|---|
Patient demographic characteristics | |||
███ ███████ | — | — | — |
████ ████ | ████ █████ | ████ █████ | ████ █████ |
██████ ███████ | ███ ██ ██ ███ | ███ ██ ██ ███ | ███ ███ ██ ██ |
████ █ ███ | — | — | — |
████ | ██ ██████ | ██ ██████ | ██ ██████ |
██████ | ███ ██████ | ███ ██████ | ███ ██████ |
█████ █ ███ | — | — | — |
█████ | ███ ██████ | ███ ██████ | ███ ██████ |
█████ ██ ████ ████████ | ██ ██████ | ██ ██████ | ██ ██████ |
█████ | ██ █████ | ██ █████ | ██ ██████ |
█████ ██████ ██ ███ ██████ ████████ | || | || | || |
████████ ██████ ██ ██████ ██████ | || | || | | █████ |
███ ██████████████ | | █████ | | █████ | | █████ |
Patient characteristics relating to acne | |||
█████ █ ███ | |||
| | █████ | || | || | || |
| | ██████ █████ | || | || | || |
| | ████ | || | || | || |
| | ████████ | ███ ██████ | ███ ██████ | ███ ██████ |
| | ██████ | ██ ██████ | ██ ██████ | ██ ██████ |
████████ █████ █████ | — | — | — |
████ ████ | ████ ██████ | ████ █████ | ████ ██████ |
██████ ███████ | ████ ███ ██ ███ | ████ ███ ██ ███ | ████ ███ ██ ███ |
███████ ██████ ████ | — | — | — |
████ ████ | ████ ██████ | ████ ██████ | ████ ██████ |
██████ ███████ | ███ ██ ██ ████ | ███ ██ ██ ████ | ███ ██ ██ ████ |
BPO = benzoyl peroxide; EGSS = Evaluator’s Global Severity Score; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; ITT = intention to treat; SD = standard deviation.
Sources: Study V01-126A-202 Clinical Study Report (2022).58 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Exposure to Study Treatments
A summary of patient exposure from Study 201 is presented in Table 35. The total amount of study drug used, total number of days of exposure, and total number of applications were similar between the study drug groups in Study 202. Greater than 88% of patients were considered compliant (did not miss more than 5 consecutive days of dosing and applied 80% to 120% of the expected applications while participating in the study) across study drug groups.
Table 35: Summary of Patient Exposure From Study 202 (Safety Population)
Exposure | IDP-126 gel | Adapalene 0.3% plus BPO 2.5% gel | Combined vehicle gel |
|---|---|---|---|
█████ ██████ ██ █████ ████ ████ ███ | — | — | — |
|| | ███ | ███ | ███ |
████ ████ | ████ █████ | ████ ██████ | ████ ██████ |
██████ ███████ | ████ ██ ██ ████ | ████ ██ ██ ████ | ████ ██ ██ ████ |
█████ ██████ ██ ████ ██ ████████ | — | — | — |
|| | ███ | ███ | ███ |
████ ████ | ████ ████ | ████ ██████ | ████ ██████ |
██████ ███████ | ████ █ ████ | ████ ██ ██ ████ | ████ ███ ██ ████ |
█████ ██████ ██ █████████ | — | — | — |
|| | ███ | ███ | ███ |
████ ████ | ████ █████ | ████ ██████ | ████ ██████ |
██████ ███████ | ████ ██ ██ ████ | ████ ██ ██ ████ | ████ ███ ██ ████ |
█████████| | — | — | — |
|| | ███ | ███ | ███ |
████ █ ███ | ███ ██████ | ███ ██████ | ███ ██████ |
BPO = benzoyl peroxide; IDP-126 gel = clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel; SD = standard deviation.
aA patient was considered compliant with the dosing regimen if the patient did not miss more than 5 consecutive days of dosing and applied 80% to 120% of the expected applications while participating in the study.
Sources: Study V01-126A-202 Clinical Study Report (2022).58 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Concomitant and Prior Therapies
███████ ██ █████ ███ ███ ███ ██ █████████ ██ █████ █ ██ ███ ███████ █████ ████████ ██ █████ ████ ████████ ████ ████████ ██ ███████ ████████ ███████ ███████ ███ ███████ ███████ ████████████ ██ ████████ ██████████ ████ ██ ███ █████ ███ ███ ███████ ████████ ███████████ █████ ██ ███ ██████ █ █████ ██ █ ████████ ██████ ██ █████ ███████ ███ █████████ ███████ ██████ ███ █████ ███████████ ███ ███ ████████ ████ ███ ██ ███████████
Efficacy
███████ ██ ███ ███ ███████ ████████ ████████ ███████ ████ █████ ███ ██ █████████ ██ █████ ███
Acne Severity
Treatment Success Based on the EGSS
███ ██████████ ██ ████████ ████ ██ █████ █ ███████ █████████ ████ ████████ ██ ███ ████ ███ ██ ████ ██ █████ ██ ██████ █████ ██████ █████████ ████████ ██ ████ ██ ███ █████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ██ ████████ ███████ ███ ██████ ███ █████████ ██████████ ██ █████████ ███████ █████ ██ ███ ████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ █████ ███ ██████ ████ ███ ██████ ██ ████████ ███ █████████ ██████████ ██ █████████ ███████ █████ ██ ███ ████ ██ ████ ██ ███████ ███████ ███ ███ █████████ ████████ ████ ███ ███ ██████ ████ ███ █████ ██ ████████
Lesion Count
████████████ ██████ █████████ ██ ████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███ █████ ███ █ ██████ ██ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ █████████ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ █████ ███ ██████ ████ ███ ██████ ██ ███████ ███ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ █████████ ████████ ████ ███ █████ ███ █████ ████ ███ █ ████ ██ ██████████ ██ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███ ██████ ███ █ ███████ ██ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ ███ ███████ ████ ███ ███████ ██ █████████ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ █████████ ████████ ████ ███ █████ ███ ██████ ████ ███ ██████ ██ ██████████████████████ ██████ █████████ ██ ████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███ █████ ███ █ ██████ ██ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ █████████ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ █████ ███ ██████ ████ ███ ██████ ██ ████████ ███ █████████ ██████████ ██ ███ ████ ████████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ █████████ ████████ ████ ███ █████ ███ █████ ████ ███ █ ████ ██ █████ █ █████ █ ███████████ ██ ████ ███████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███ ██████ ███ █ ███████ ██ ███ ███████ ███ ██████ █████ ███ █ ██████ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ ███ █ ██████ ██ ████████ ███████ ███ █████████ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ████████ ███████ ███ ███ ███████ ████ ███ ███████ ██ █████████ ███ █████████ ██████████ ██ ███ ████ ███████ ██████ ████ ████████ ██ ███████████████ ██████ █████ ██ ████ ██ ███████ ███████ ███ ███ ███ █████████ ████████ ████ ███ █████ ███ ██████ ████ ███ ██████ ██ ██████████████ ██ ████████████████████ ██████ █████ ██ █████████████ ███████ ██ ████ █████████████████ ████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ ███ █ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ███ ███ █ █████ ██ ███ ████████ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ███████████████ ██████ █████ ██ ████ ██ ████ ███ ██████████████████ ██████ █████ ██ █████████████ ███████ ██ ████ █████████████████ ████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ████ ███ ███ █ █████ ██ ███ ███████ ███ ██████ ███ ███ █ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ███ ███ █ █████ ██ ███ ████████ ███████ ███ ██████ ███ █████████ ██████████ ██ ███ ████████ ██████ ████ ████████ ██ ███ ████████ ████████ ██████ █████ ██ ████ ██ ████ ███ █████████
Table 36: Summary of Key Efficacy Results From Study 202 (ITT Population)
Efficacy outcome | IDP-126 gel | Adapalene 0.3% plus BPO 2.5% gel | Combined vehicle gel |
|---|---|---|---|
Acne severity | |||
Percentage of patients with ≥ 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear (i.e., treatment success) at week 12 | |||
█████████ ████████ | | ████ | ████ | ████ |
█████████ ████████ | | ████ | ████ | ████ |
███████ █████████ ████ █ ████████ █████ ████ █ ████ ███ | █████ ██████ ██ ██████ | █████ █████ ██ ██████ | || |
| ██████ ██████ ██ ████████ ███████ █ | | █████ | | █████ | || |
█████████ ███████████ ███████ █ █████████ ████████ ████ ████ █ ████ ███| | █████ █████ ██ ██████ | || | || |
| ██████ ███████ ███ ██ █████████ ████████ ████ ███ | | █████ | || | || |
Lesion count | |||
Absolute change from baseline in the inflammatory lesion count at week 12 | |||
████████ ██████ ██████ ████ ████ | ████ ███████ | ████ ██████ | ████ ███████ |
████ ██ ██████ ██████ ████ ████ | ███ ██████ | ████ ███████ | ████ ███████ |
██ ████ ██████ ████ ████████ ████| | █████ ███████ | █████ ███████ | █████ ███████ |
███████ ████████ ██████ █ ████████ █████ ███ ████ ███ | ██████ ███████ ██ ██████ | █████ ███████ ██ ██████ | || |
| ██████ ██████ ██ ████████ ███████ ███ | | █████ | | █████ | || |
███████ █████████ █████ █ ████ ████████ ████ ███ ████ ███ | █████ ██████ ██ █████ | || | || |
| ████ ███████ ███ ██ ███████ ████████ ██ ███ | █████ | || | || |
Percent change from baseline in the inflammatory lesion count at week 12 | |||
██ ████ ██████ ████ ████████ ████ | █████ ███████ | █████ ███████ | █████ ███████ |
███████████ ███████ ███ █ ████████ ███████ ███ ████ ███ | ██████ ███████ ██ ███████ | || | || |
█████ ████ ███ ███████ ██████ ███ | | █████ | || | || |
█████████ ████████ █████ █ ████████ ████████ ████ ███ ████ ███ | █████ ██████ ██ █████ | || | || |
█████ ███ ███ ██████ █████ ████ ███ | █████ | || | || |
Absolute change from baseline in the noninflammatory lesion count at week 12 | |||
████████ ██████ ██████ ████ ████ | ████ ███████ | ████ ███████ | ████ ███████ |
████ ██ ██████ ██████ ████ ████ | ████ ███████ | ████ ███████ | ████ ███████ |
██ ████ ██████ ████ ████████ ████ | █████ ███████ | █████ ███████ | █████ ███████ |
█████████ ██████ ███████ ███ █ ███████████ ███ ████ ███ | ██████ ███████ ██ ███████ | ██████ ███ ██ ██████ | || |
██████ ███████ ███ ██ ████████ ███████ ███| | | █████ | | █████ | || |
██████ █████████ ██████ █ ██████ ████████ ████ ███ ████ ███ | █████ ██████ ██ █████ | || | || |
████ ████ ████████ ███████ ████ ███ | █████ | || | || |
Percent change from baseline in the noninflammatory lesion count at week 12 | |||
██ ████ ██████ ████ ████████ ████ | █████ ███████ | █████ ███████ | █████ ███████ |
███████████ ███████ ███ █ ████████ ███████ ███ ████ ███ | ██████ ███████████ | || | || |
████ ██████ ████████ ███████ ███| | | █████ | || | || |
███████ ████ █████ █ ██████ ██████ ████ ███ ████ ███ | █████ ███████ █████ | || | || |
██████ ███████ ███ ██ █████████ ████████ ████ ███ | █████ | || | || |
Quality of life | |||
Absolute change from baseline in Acne-QoL self-perception domain score at week 12 | |||
|| | ███ | ███ | ███ |
██████ ██████ ████ ████ | ████ ██████ | ████ ██████ | ████ ██████ |
████ ██ █████ ███ ██ | ████ ██████ | ████ ██████ | ████ ██████ |
██████ ████ █████████ ████ ████| | ███ ██████ | ███ ██████ | ███ ██████ |
Absolute change from baseline in Acne-QoL acne symptoms domain score at week 12 | |||
|| | ███ | ███ | ███ |
██████ █████ ████ ████ | ████ ██████ | ████ ██████ | ████ ██████ |
████ ██ █████ ████ ██ | ████ ██████ | ████ ██████ | ████ ██████ |
██████ ████ █████████ ████ ████ | ███ ██████ | ███ ██████ | ███ ██████ |
██ █ ██████████ █████████ ████ █ ███████████ ██████ ████████ ██████ ███ █ ████████████████ ██ █ █████ ████████ ██ █ ████████ ██████████| █████████ ██████████ ████████ ███ █████ █████████ ████████ ████ ████ ███ █████████████ ███ ██ █████ ██ ███ ████████ ██████████ ██████████ ███ ██████████████ ██████ ███ ██████ ████████ █ ██████ ████ █ ████████ ██████████ ██████ ███████ █████████ ███████████ ████ ███████ ██ █████████ █████ ███ ████████ ███████ ████████ ███ ████ ████████ ███ ████████ ████████████ ██ ███████ ███ ███ ███████ ████ █ ██████ ███████ ██ ███ ███ ██ ███ █████████ ████████ ███ ██████ ███████████ ███ ███████████ ████████████ ██ ███ █████████ ████████ ███ █████████ ███ ██ ██████ ████ ████████ █ ███████ ███ █████████ ██████████ ████████ ███ █████ █████████ ████████ ████ ████ ██ ██ █████ ███ ██████████ ███ ██ ████ ██ ████████ ██ ██████████ ████ ███████ ██ █████████ █████ ███ ████████ ██████ ███ ███ ██████████ ████████ ██████ █████ ██ █ ██████████ ██████ ████ ████ ████████ ███ ████████ ███████████ ████████ ██ █████ █████████ █ ████████ ████ █████████ ███ ██████████████ ██████ ███ ██████ ██ ██████████ ██ ███████ ███████ ██████ ████ ████████ ██████████ ██ ████ ██ █████ █████████ █ ████████ ████ ██████ █████████ ██████████ ███████
Sources: Study V01-126A-202 Clinical Study Report (2022).58 Details included in the Table are from the sponsor’s summary of clinical evidence.15
███████ ███████ ██ █████ ███████ ████ █████ ███ ██ █████████ ██ █████ ███ ███ ████████ █████████ ██ ██████ █████████ ██ ███ ██████ █████ ████ ██████ ████ ██ ███ ██████ █████ █████ ██ ███████ █████████ ███ ██████████████ ████ ██████████ ██████ ███ █████ ██ ███ ███████ ███ ███ █████████ ████████ ████ ███ ███████ ██████████████ ███ ████████ █████████ ██ ██████ █████████ ██ ███ ████████ ███████ ███ █████ ███ ██ ███ ██████ █████ █████ ██ ██████████ ███ ████████████ ███████████████ ██████████ ██████████ ██ ████████ ████ ████████ ██ █████ ███ ████ ████ ███████ ██ ███ ███████ ███ ███ █████████ ████████ ████ ███ ██████ ██████ ███ ██████ ██████████████ █████ ███ ██████████ ██ ████████ ████ ████████ █ ████ ██ ███ ████████ ███████ ███ █████ ███ ██████ ███ ████ ██████ █████ ████████ ████ ███████████ ████ ████ ██████ ██ ████████ ██ ███ ███████ ███ ██████ ████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███ ███████ ███ ███████████ ████ ███████ █████ ██ ████████ ██ ███ ███████ ███ ██████ ████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███████ ███ ███████████████ ███████ ██████████ ███████ ███ ████████ ████ ███ ██ ███ ███████ ███ █████ ███ ████ ███████ █████████████████████ ███ ██ ███████ ███████| █████ ██ ██ █████████ █ ██ ███ ███████ ███ █████ ███ █ ██ ███ █████████ ████████ ████ ███ ██████ ███ █████ █████ ████ █████████ ███ ██ ██████ ███ ██ ██ █████ ████████ ██ ██ ███ ███████ ███ █████ ███ █ ██ ███ █████████ ████████ ████ ███ ███████ ███ █████ ████ ███ ██ █████ ████ ██████████ ████ ███ ██ █████ ████████████████████████████████ ████ ██ ███████ ██ ████████ ███ ████ ██ █████ ████████████ ██████| █████ ██ █████ ██ ████████ ██ ███ ███████ ███ ██████ █████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ███████ ████████ ███ ██████████████ ████ ████████████ █████ ██ ████ ██ ████████ ██ ███ ███████ ███ ██████ ████ ██ ███ █████████ ████████ ████ ███ ██████ ███ ████ ██ ███ ████████ ███████ ███ █████ ████ ████████ ████ █ ████ ███████████ ██ █ ████ ███ ████████████ ██████ █████████
Table 37: Summary of Harms from Study 202 (Safety Population)
Adverse events | IDP-126 gel | Adapalene 0.3% plus BPO 2.5% gel | Combined vehicle gel |
|---|---|---|---|
Adverse events, n (%) | |||
████████ ████ | | ████ | ██ ██████ | ██ ██████ | ██ ██████ |
████ ██████ █████ | |||
███████ █████████ ███ ██████████████ ████ ██████████ | ██ ██████ | ██ ██████ | | █████ |
████████ ████ ████ | ██ ██████ | ██ █████ | || |
████████ ████ ███████ | | █████ | ██ █████ | | █████ |
████████ ████ █████████ | | █████ | | █████ | || |
███████ ████ █████████ | | █████ | | █████ | || |
███████ ████ █████████ | | █████ | | █████ | | █████ |
████████ ████ ████████ | | █████ | | █████ | || |
██████████ ████ ███████ | | █████ | | █████ | | █████ |
████████ ████ ████ | | █████ | || | || |
██████████ ████ ███████ | | █████ | | █████ | || |
███████ | || | || | | █████ |
██████████ ███ ████████████ | ██ ██████ | ██ █████ | ██ █████ |
████████ | | █████ | | █████ | ██ █████ |
███████████████ | | █████ | | █████ | | █████ |
████ | | █████ | | █████ | | █████ |
██████████ | | █████ | | █████ | | █████ |
█████████ | | █████ | | █████ | || |
███████ █████ ███████ | | █████ | | █████ | || |
████ ███ █████████ ███████ ████████ | ██ █████ | ██ █████ | | █████ |
████ | | █████ | | █████ | | █████ |
██████████ ███████ | | █████ | | █████ | || |
████████ | | █████ | | █████ | || |
███████ ██████ ███ ██████████ ███████████ | | █████ | | █████ | | █████ |
███████ | | █████ | | █████ | || |
████████ ██████ | | █████ | || | || |
███████ ██████ ███████ | | █████ | | █████ | | █████ |
████████ | | █████ | | █████ | | █████ |
██████████████ | | █████ | | █████ | | █████ |
████ ██████ ██████████ █████████ | | █████ | || | | █████ |
█████████ █████ ███ ██████████ █████████ | | █████ | | █████ | | █████ |
█████ | | █████ | | █████ | | █████ |
█████ ██████████ | | █████ | | █████ | || |
██████████████ ███████ | | █████ | | █████ | | █████ |
███████ | | || | || | | █████ |
███████ ███████ ███████ █ ███ | |||
████████ ████ | | ███ | | █████ | || | || |
████ █████ ██████ | | █████ | || | || |
████████ ███ ███████ █████ ████ ██████ █████ ███ ██ █ █████ █ ███ | |||
████████ ███ ███████ ███ ██ | | ████ | | █████ | | █████ | || |
███████ █████████ ███ ██████████████ ████ ██████████ | | █████ | | █████ | || |
███████████ ████ ██████████ | || | | █████ | || |
██████████ ████ ██████ | || | | █████ | || |
███████████ ████ ██████ | | █████ | | █████ | || |
███████████ ████ ██████████ | — | | █████ | || |
████████ ███ ████ | — | | █████ | || |
█████████ ████ ████████ | | █████ | | █████ | | |
██████████ ████ ████████ | — | | █████ | || |
██████████ ███ ███████████ | | █████ | || | || |
████████ ██████████ | | █████ | || | || |
████████ █████ ███ ████████ ████████ | | █████ | || | || |
█████ | | █████ | || | || |
████ ███ ████████████ ██████ █████████ | || | | █████ | || |
████ █████████ | || | | █████ | || |
█████████ | || | | █████ | || |
███████ █ ███ | |||
████████ ███ ████ | || | || | || |
███████ █ ███ | |||
█████ ██ ████ ██ ███████ ███████ ███ ██████████ ████ ███████ ███ ████ ███ ██████████ ███████ █████████ █████████ ██ █████ ███ ███ ██████ | |||
███ █ ███████ ███████ ██████ ████ █ █████████ ████████ ███████ ██████ ████ █ █████ ███████████ █████ ███████████| █████ ███████████ ██ ████ ████ █ ███████ ██ ██████ █████ ████ █████ ██ ██████ █████ █████ ███ █████████ █████ ██ ████ █████ ██ ██████ █████ █████ ██ █████████ █████ ████████ █████████ ████ ████ █ ████ ████ ███████ ████ ██████| ██ ████ ██████ █ ██████ ████████ ████████████ █████ ████ ██████ ███ █████ ███ ██ █ ██ ████ ██████ █ █████ ██ █ ████████ ████████████ ████ ███ █████ ████ ███ ███ ██████ █ ███████ ████████████ █████ ████ ███ ███ ███ ██████ ███ █ ███████ ████████████ ███ █████ ███ ███ ███ █████ █████ || ██ ████ ██████ █ ██████ ████████ ████████████ █████ ████ ██████ ███ █████ ███ ██ █ ██ ████ ██████ ████ ████████ █ ███████ ███ ████████████ ████ ███ █████ ████ ███ ███ █████ ███ ██ █ ████ ███ █ ███████ ███ ████████████ █████ ████ ███ ███ ███ █████ ███ ██ █ █████
Sources: Study V01-126A-202 Clinical Study Report (2019).58 Details included in the Table are from the sponsor’s summary of clinical evidence.15
Critical Appraisal
Internal Validity
The randomization and masking procedures in Study 202 were considered appropriate. The coprimary and secondary end points were controlled for multiplicity. Type I error was controlled by requiring all the coprimary efficacy end points to be statistically significant, and failure of any 1 of the coprimary efficacy end points invalidated the statistical significance of all secondary efficacy end points. Since it was a phase II trial aiming to provide preliminary evidence about the efficacy and harms of the study drug, the results cannot be considered confirmatory. Relevant patient demographic characteristics at baseline appeared to be balanced among the study drug groups. No notable differences in the baseline EGSS and lesion counts among the study drug groups were identified. Similar to the pivotal trial, the washout periods used in the studies were considered adequate (i.e., prior use of therapies for the treatment of acne was not expected to have an impact on the efficacy results) and the list of prohibited treatments for acne (topical preparations, physical treatments, and systemic therapies) was considered comprehensive by the clinical expert. A total of 7 patients had protocol deviations regarding the specified washout period for prior medications. For the outcomes on quality of life (Acne-QoL self-perception and symptom domains), no inferential analyses were conducted, as per the statistical analysis plan, so no conclusions could be drawn from these data.
Of note, while all data were summarized in listings presented by patient, data collected at early discontinuation and unscheduled visits occurring before study day 8 were not included in the analyses of efficacy and safety, except for baseline values.
External Validity
The inclusion criteria used in Study 202 included the population of interest identified in the indication for IDP-126 gel. In consultation with the clinical expert, it was concluded that the inclusion criteria adequately capture (and, consequently, the study population is representative of) the patients seen in practice who would be candidates for IDP-126 gel.
In consultation with the clinical expert, it was concluded that topical fixed-dose combination therapies, including retinoid plus BPO, are relevant comparators for IDP-126 gel in the Canadian practice setting. Similar to the pivotal trial and Study 201, in consultation with the clinical expert, it was concluded that the outcome measures of acne severity and lesion counts used in Study 202 are applicable to Canadian clinical practice. Similar to the pivotal trial, the majority of patients from each study ██████████████ █ █████ ██ ████████ ██ ████ █████ ████ ██████ ███ ████████ ████.
Overall, no notable concerns on the generalizability of the results to the population of interest in the Canadian setting were identified in the appraisal of Study 202.
Discussion
Summary of Available Evidence
Two phase III, multicentre, double-blind, RCTs (Study 301, N = 183; Study 302, N = 180) assessed whether there is a difference in the proportion of patients with treatment success (defined by ≥ 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear) and change from baseline in inflammatory and noninflammatory lesion counts in patients aged 9 years and older with moderate to severe acne applying IDP-126 topical gel once daily for 12 weeks when compared with its vehicle gel. Other outcomes of interest include change in HRQoL measured by the self-perception and symptom subscales of the Acne-QoL questionnaire. Notable harms include general disorders and administration site conditions, skin and subcutaneous tissue disorders, and SAEs. The mean age of patients randomized to each study drug group was similar, approximately 20 years, and ranged from 10 to 48 years across studies. The majority of patients in each study drug group had moderate acne, defined as a baseline EGSS of 3, ranging from 87.7% to 95.1% of patients across studies. The remainder of the patients in each study drug group had severe acne, defined as a baseline EGSS of 4, ranging from 4.9% to 12.3% of patients across studies.
In the absence of direct comparative evidence of IDP-126 gel versus relevant comparators from the pivotal phase III RCTs in the treatment of moderate to severe acne (a score of 3 or 4 on the EGSS, IGA, or ISGA), the sponsor submitted an ITC in the form of Bayesian NMAs that assessed treatment effect on outcomes of interest, including patients with treatment success (defined by a ≥ 2-grade reduction from baseline and a score of clear or almost clear on the IGA or equivalent scales) and changes in inflammatory and noninflammatory lesion counts. The NMAs in the base-case analysis included 46 to 50 trials and included the following comparators: topical fixed-dose combinations (retinoid plus BPO, antibiotic plus BPO, antibiotic plus retinoid), topical monotherapies (antibiotic, retinoid, BPO, other topical monotherapy), oral antibiotic monotherapy, combinations of oral antibiotic and either physical or topical treatment, physical treatment only, other treatments, and vehicle or placebo. Base-case analyses incorporated random-study, fixed class–effect models estimating relative treatment effects for each outcome of interest. One published NMA of 221 trials conducted by Huang et al.17 was also submitted by the sponsor for this review. The analysis adopted a frequentist approach to assess the effect of different treatments for acne on outcomes of interest, including reductions in total, inflammatory, and noninflammatory lesions; treatment success defined using the IGA; and discontinuation due to any AEs.
Two phase II, multicentre, double-blind, RCTs (Study 201, N = 741, and Study 202, N = 686) were submitted by the sponsor to further address the evidence gap and provide direct comparative evidence of IDP-126 gel versus active comparators. The comparators in Study 201 were IDP-126 component A (BPO 3.1% plus adapalene 0.15% gel), IDP-126 component B (clindamycin phosphate 1.2% plus BPO 3.1% gel), IDP-126 component C (clindamycin phosphate 1.2% plus adapalene 0.15% gel), and IDP-126 vehicle gel. The comparator in Study 202 was adapalene 0.3% plus BPO 2.5%. Studies 201 and 202 assessed whether there is a difference in the proportion of patients with treatment success (same definition that was used in studies 301 and 302) and change from baseline in inflammatory and noninflammatory lesion counts in patients aged 9 years and older (Study 201) or 12 years and older (Study 202) with moderate to severe acne applying IDP-126 topical gel once daily for 12 weeks. Other efficacy and safety outcomes assessed in studies 201 and 202 are similar to those assessed in studies 301 and 302. The mean age of patients randomized to each study drug group was similar — approximately 20 years across studies — and ranged from 10 to 60 years in Study 201 and 12 to 56 years in Study 202. The majority of patients in each study drug group had moderate acne, ranging from 79.3% to 86.0% in Study 201 and █████ ██ █████ ██ █████ ███. The remainder of patients had severe acne, ranging from 14.0% to 20.7% in Study 201 and █████ ██ █████ ██ █████ ████.
Interpretation of Results
Efficacy
Studies 301 and 302 demonstrated that 12 weeks of treatment with IDP-126 gel applied once daily results in a clinically meaningful improvement in acne, as measured by the proportion of patients with treatment success and change in inflammatory and noninflammatory lesion counts when compared with its vehicle gel. While the absolute change from baseline in inflammatory lesion counts was statistically significant in the trials, the GRADE assessment for this end point concluded there is little to no clinically meaningful difference in inflammatory lesion count, since the point estimates for the absolute difference did not exceed the suggested threshold of importance (i.e., 10 lesions) (Table 2). In contrast, the trials showed IDP-126 gel results in a clinically meaningful reduction in inflammatory lesion count based on the percent change from baseline when compared with its vehicle gel. Additionally, uncertainty in the evidence for a clinically meaningful reduction in noninflammatory lesion count is primarily due to the CIs, including the potential that the absolute difference compared with vehicle is small and unimportant. Overall, no serious concern regarding risk of bias and no serious concern about the generalizability of the results to the population of interest were identified in the appraisal of the trials. Of note, the clinical expert indicated that placebo or vehicle response rates in acne trials generally range from 20% to 25%, as moisturizers alone have a clinically meaningful effect on acne.
The subgroup analysis results for the coprimary efficacy outcomes (patients with treatment success and absolute change in lesion counts) at week 12 from studies 301 and 302 provide supportive evidence of the favourable treatment effect of IDP-126 gel versus vehicle in patients with moderate acne. The small sample size of the subset of patients with severe acne limits the ability to interpret the results for this subgroup.
The patient groups identified an unmet need for early, effective treatment to prevent acne scarring and hyperpigmentation. The other analysis results for the HRQoL outcomes (change in Acne-QoL self-perception and symptom domain scores) at week 12 from studies 301 and 302 are suggestive of little to no clinically meaningful difference in the HRQoL of patients with acne applying IDP-126 gel versus its vehicle gel. Of note, the domain structure of acne symptoms in the Acne-QoL questionnaire comprises bumps on the face, bumps full of pus on the face, scabbing from facial acne, scarring from facial acne, and oily facial skin. Therefore, the change from baseline in the acne symptoms domain score reflects treatment effects beyond acne scarring. The effect of IDP-126 gel versus any comparator on the rate of treatment failure, change in skin pigmentation, and on mental health (other important outcomes according to patient and clinical expert input) in patients with acne is unknown, as they were not formally assessed in the evidence submitted by the sponsor.
The sponsor-conducted NMA results showed a favourable treatment effect with IDP-126 gel versus vehicle or placebo, oral antibiotic, and topical monotherapies on treatment success and lesion counts, based on a median treatment period of 12 weeks across the included trials. While the effect estimates are suggestive of a possible favourable treatment effect with IDP-126 gel versus topical fixed-dose combination dual therapies, there is some uncertainty in the evidence, as the 95% CrIs were close to the null for certain dual-combination therapies and outcomes, and a definitive conclusion could not be drawn for those results with 95% CrIs that included the null for certain dual-combination therapies and outcomes. Of note, the literature review informing the NMA analyses applied somewhat restrictive exclusion criteria that ruled out the inclusion of single and small sample trials. The NMA appears to have included study populations that vary greatly in terms of their disease severity and distribution by sex, which raises concerns for heterogeneity across trials. A literature review and meta-regression were performed to identify and assess the influence of effect-modifying variables (i.e., duration of treatment, severity of acne, diverse treatments); however, their impact on the overall estimates could not be properly addressed due to limited reporting in the included trials. Further, the treatment group nodes included monotherapies and combination therapies that are unavailable in Canada, thereby limiting the generalizability of the results. In consideration of these notable limitations that suggest some concerns for bias in the estimates, the magnitude of effect associated with IDP-126 gel is uncertain. In consultation with the clinical expert, additional relevant comparators were found to be missing in the network, including oral isotretinoin, azelaic acid, and topical dapsone; as such, the relative effect of IDP-126 gel versus these other comparators of interest is unknown.
In the published NMA conducted by Huang et al.,17 the estimates comparing triple therapy (topical antibiotic plus retinoid and BPO) with placebo were generally consistent with the sponsor-conducted ITC. The limitations of the published NMA included the lack of prespecification of study methods through a review protocol and notable heterogeneity in the effect-modifying factors across the studies included in the network. The exploration of between-study differences and potential biases was further limited by missing information on patient and study characteristics. Notable generalizability issues, including NMA estimates coming from comparisons with placebo only and the presence of treatments without market approval in Canada in the network node of interest, further limit the applicability of these analyses to the Canadian clinical context. The reported results comparing triple therapy with placebo from Huang et al. appeared to be similar in magnitude to the results comparing IDP-126 with placebo in the sponsor-conducted NMA, which suggests some level of consistency between the 2 NMAs. However, the comparisons of the NMAs as they relate to IDP-126 are limited, primarily due to Huang et al. combining IDP-126 with other triple therapies but reporting only results comparing IDP-126 gel with placebo.
While definitive conclusions cannot be drawn from phase II trials, as they provide preliminary evidence on a drug under review to inform a phase III trial design, results from studies 201 and 202 provide supportive evidence for the favourable treatment effect of IDP-126 gel versus vehicle gel that was demonstrated in studies 301 and 302 and is consistent with the sponsor-conducted NMA results for IDP-126 gel versus vehicle or placebo. In consultation with the clinical expert, it was concluded that topical fixed-dose dual-combination therapies (i.e., retinoid plus BPO, antibiotic plus BPO, and retinoid plus antibiotic) are relevant comparators for IDP-126 gel in the Canadian practice setting, which were the comparators in studies 201 and 202. Overall, results from the phase II studies appear to be directionally aligned with the sponsor-conducted NMA results that are suggestive of a possible favourable treatment effect with IDP-126 gel versus topical fixed-dose dual-combination therapies (antibiotic plus retinoid, retinoid plus BPO, and antibiotic plus BPO) on treatment success and lesion counts, although associated with some uncertainty. Consistent with the NMA results for the comparison between IDP-126 gel and a topical retinoid plus BPO fixed-dose combination, a definitive conclusion cannot be drawn for those results with 95% CIs that included the null for the comparison between IDP-126 gel and adapalene 0.3% plus BPO 2.5% gel on lesion counts in Study 202. Results from studies 201 and 202 are aligned with studies 301 and 302 on HRQoL exploratory outcomes (i.e., suggestive of possibly little to no difference in the HRQoL of patients with acne applying IDP-126 gel versus the comparators assessed in the phase II studies, based on the effect estimates and size of variance around the difference). Of note, results for all end points in Study 201 (not controlled for multiple comparisons) and ███ ███ ██████ ██████ █████ ███ ████████ ██ ███████ ███ ██████ █████████ ████████ ████ ███ ██ ████████ ██████ ██ ██████ ██████ ██ ████ ██ ██ █████ ███ ██████ ██ ██████ ██ ████ ███████████ ████████████ ██ ███ ███████████ ██████████ ██████ ██ ███████████ ███████ ███████████ ███ █████████ ████ ██ ████ █ ██████
The clinical expert identified the following challenges in the treatment of patients with acne: not all patients’ acne responds to current topical therapies and adherence to acne therapies is generally low, often due to associated side effects, treatment modality, and complicated treatment regimens (i.e., requiring ≥ 2 different products with the potential for active ingredient cross-reactivity and reduced efficacy). Based on the available evidence and input from the clinical expert, once-daily topical application of IDP-126 gel likely addresses these concerns, as it provides an additional treatment option among the current first-line therapies for moderate to severe acne in patients aged 12 years and older.
Harms
The patient groups concluded that patients weigh the side effects associated with treatment against effectiveness when deciding to start, stop, or continue their therapy. Based on the evidence from studies 301 and 302, once-daily topical application of IDP-126 gel likely results in an increase in general disorders and administration site conditions as well as skin and subcutaneous tissue disorders and likely results in little to no difference in SAEs when compared with its vehicle gel. Uncertainty in the evidence is primarily due to imprecision; the total sample size and number of events did not reach the optimal information size.11
Results from the published NMA conducted by Huang et al.17 are suggestive of an increase in the frequency of drug discontinuation with topical antibiotic plus retinoid and BPO therapy compared with placebo; however, these results are of limited generalizability to the Canadian clinical context, as described earlier. Based on the evidence from studies 301 and 302 as well as studies 201 and 202, the impact of IDP-126 gel on the frequency of drug discontinuation due to any TEAE is difficult to interpret, as the numbers of these events during the trials are small. The safety of IDP-126 gel was not assessed in the sponsor-conducted NMA.
The safety results from studies 201 and 202 are consistent with the safety results from studies 301 and 302; application site pain was the most frequently reported TEAE in patients applying IDP-126 gel once daily for 12 weeks. In consultation with the clinical expert, it was concluded that there was no clinically meaningful difference in the frequency of AEs between the study drug groups that would be expected to impact clinical decisions (i.e., no concern with the safety profile based on the safety results from studies 301 and 302 as well as studies 201 and 202 was identified).
Conclusion
Studies 301 and 302 demonstrated that 12 weeks of treatment with IDP-126 gel applied once daily results in a clinically meaningful improvement in acne, when compared with its vehicle gel, in patients aged 10 years and older with moderate to severe acne. Treatment effect was measured by the proportion of patients with treatment success, defined by at least a 2-grade reduction from baseline in the EGSS and an EGSS of clear or almost clear, and a reduction in inflammatory and noninflammatory lesion counts. While a statistically significant reduction in the absolute change in inflammatory lesion count was demonstrated in the trials, the GRADE assessment for this end point concluded there is little to no clinically meaningful difference, as the effect estimates for the absolute difference did not exceed the suggested threshold of importance. In contrast, the trials showed IDP-126 gel results in a clinically meaningful reduction in inflammatory lesion count based on the percent change from baseline when compared with its vehicle gel. The sponsor-conducted NMA results showed a favourable treatment effect with IDP-126 gel versus vehicle plus placebo, oral antibiotic, and topical monotherapies on treatment success and change in lesion counts. While the effect estimates are suggestive of a possible favourable treatment effect with IDP-126 gel versus topical fixed-dose dual-combination therapies, there is some uncertainty, as the 95% CrIs included the null or were close to the null for certain dual-combination therapies. Due to the possible exclusion of relevant studies, heterogeneity across trials in the networks, and omission of relevant comparators in the Canadian setting that suggest concerns for bias in the NMA estimates, the magnitude of effect associated with IDP-126 gel is uncertain. Findings from the NMA by Huang et al. aligned with the sponsor-conducted NMA results; however, these findings are of limited applicability to the Canadian context, as IDP-126 gel was combined with other triple therapies in the analyses and only comparisons with placebo were reported. Results from the phase II trials (studies 201 and 202) are supportive evidence suggesting a possible favourable treatment effect with IDP-126 gel versus vehicle gel and topical dual-combination therapies (fixed-dose combination of retinoid plus BPO, antibiotic plus BPO, and retinoid plus antibiotic), based on treatment success and change in lesion counts at week 12. No concerns with the safety profile of IDP-126 gel were identified based on the safety results from all submitted trials.
References
1.Arora MK, Yadav A, Saini V. Role of hormones in acne vulgaris. Clin Biochem. 2011;44(13):1035-1040. PubMed
2.DermNet. Inflammatory lesions in acne. 2014; https://dermnetnz.org/topics/inflammatory-lesions-in-acne. Accessed 2024 Jun 4.
3.National Institute of Arthritis and Musculoskeletal and Skin Diseases. Acne. 2020; https://www.niams.nih.gov/health-topics/acne. Accessed 2024 Jun 4.
4.Canadian Dermatology Association. Acne. https://dermatology.ca/public-patients/skin/acne/. Accessed 2024 Jun 4.
5.Asai Y, Baibergenova A, Dutil M, et al. Management of acne: Canadian clinical practice guideline. CMAJ. 2016;188(2):118-126. PubMed
6.Acne vulgaris: establishing effectiveness of drugs intended for treatment guidance for industry. Silver Spring (MD): U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER); 2018: https://www.fda.gov/files/drugs/published/Acne-Vulgaris—Establishing-Effectiveness-of-Drugs-Intended-for-Treatment.pdf. Accessed 2024 May 27.
7.Fehnel SE, McLeod LD, Brandman J, et al. Responsiveness of the Acne-Specific Quality of Life Questionnaire (Acne-QoL) to treatment for acne vulgaris in placebo-controlled clinical trials. Qual Life Res. 2002;11(8):809-816. PubMed
8.Martin AR, Lookingbill DP, Botek A, Light J, Thiboutot D, Girman CJ. Health-related quality of life among patients with facial acne -- assessment of a new acne-specific questionnaire. Clin Exp Dermatol. 2001;26(5):380-385. PubMed
9.Balshem H, Helfand M, Schunemann HJ, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011;64(4):401-406. PubMed
10.Santesso N, Glenton C, Dahm P, et al. GRADE guidelines 26: informative statements to communicate the findings of systematic reviews of interventions. J Clin Epidemiol. 2020;119:126-135. PubMed
11.Guyatt GH, Oxman AD, Kunz R, et al. GRADE guidelines 6. Rating the quality of evidence--imprecision. J Clin Epidemiol. 2011;64(12):1283-1293. PubMed
12.Clinical Study Report: V01-126A-301. A phase 3, multicenter, randomized, double-blind, vehicle-controlled, parallel-group, clinical study comparing the efficacy and safety of IDP-126 gel in the treatment of acne vulgaris [internal sponsor's report]. Petaluma (CA): Bausch Health Americas, Inc.; 2021 Dec 15.
13.Clinical Study Report: V01-126A-302. A phase 3, multicenter, randomized, double-blind, vehicle-controlled, parallel-group, clinical study comparing the efficacy and safety of IDP-126 gel in the treatment of acne vulgaris [internal sponsor's report]. Petaluma (CA): Bausch Health Americas, Inc.; 2021 Dec 15.
14.Common Technical Document section 2.7.4: Summary of clinical safety [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: IDP-126 (TBC) (clindamycin phosphate, benzoyl peroxide, adapalene) gel, 1.2%/3.1%/0.15%. Laval (QC): Bausch Health, Canada Inc.
15.Summary of clinical evidence clindamycin phosphate, adapalene and benzoyl peroxide (IDP-126). In: Drug Reimbursement Review sponsor submission: IDP-126 gel. Laval (QC): Bausch Health, Canada Inc.; 2024 Mar 8.
16.Bausch Health response to April 8, 2024 CADTH request for additional information regarding IDP-126 Gel CADTH review [internal additional sponsor's information]. Laval (QC): Bausch Health; 2024 Apr 22.
17.Huang C-Y, Chang IJ, Bolick N, et al. Comparative Efficacy of Pharmacological Treatments for Acne Vulgaris: A Network Meta-Analysis of 221 Randomized Controlled Trials. Ann Fam Med. 2023;21(4):358-369. PubMed
18.Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74(5):945-973 e933. PubMed
19.Dréno B, Bettoli V, Araviiskaia E, Sanchez Viera M, Bouloc A. The influence of exposome on acne. J Eur Acad Dermatol Venereol. 2018;32(5):812-819. PubMed
20.Lai JJ, Chang P, Lai KP, Chen L, Chang C. The role of androgen and androgen receptor in skin-related disorders. Arch Dermatol Res. 2012;304(7):499-510. PubMed
21.Moradi Tuchayi S, Makrantonaki E, Ganceviciene R, Dessinioti C, Feldman SR, Zouboulis CC. Acne vulgaris. Nat Rev Dis Primers. 2015;1:15029. PubMed
22.Clinical Study Report: V01-126A-301. A Phase 3, Multicenter, Randomized, Double-Blind, Vehicle-Controlled, Parallel-Group, Clinical Study Comparing the Efficacy and Safety of IDP-126 Gel in the Treatment of Acne Vulgaris [Confidential Information] [internal sponsor's report]. Petaluma (CA): Bausch Health Americas, Inc.; 2021 Dec 15.
23.Mancini AJ, Baldwin HE, Eichenfield LF, Friedlander SF, Yan AC. Acne life cycle: the spectrum of pediatric disease. Semin Cutan Med Surg. 2011;30(3 Suppl):S2-5. PubMed
24.Picardo M, Eichenfield LF, Tan J. Acne and Rosacea. Dermatol Ther (Heidelb). 2017;7(Suppl 1):43-52. PubMed
25.Fried RG. Acne vulgaris: The psychosocial & psychological burden of illness [accessed by sponsor]. 2013; https://www.the-dermatologist.com/content/acne-vulgaristhe-psychosocial-and-psychological-burden-illness.
26.Layton AM, Thiboutot D, Tan J. Reviewing the global burden of acne: how could we improve care to reduce the burden? Br J Dermatol. 2021;184(2):219-225. PubMed
27.Cabtreo (Clindamycin Phosphate, Adapalene and Benzoyl Peroxide 1.2% w/w, 0.15% w/w and 3.1% w/w) Topical Gel [draft product monograph]. Laval (QC): Bausch Health Canada, Inc.; 2023 Aug 28.
28.TBC (clindamycin phosphate, adapalene and benzoyl peroxide 1.2% w/w, 0.15% w/w and 3.1% w/w): topical gel [product monograph]. Laval (QC): Bausch Health, Canada Inc.; 2024 Mar 8.
29.Arazlo™ (tazarotene) lotion 0.045% w/w topical [product monograph]. Laval (QC): Bausch Health Canada, Inc; 2021 Jul 7.
30.Biacna® (Clindamycin Phosphate and Tretinoin) Topical gel 1.2% w/w, 0.025% w/w [product monograph]. Laval (QC): Bausch Health Canada, Inc; 2020 Oct 2.
31.BENZACLIN®(Clindamycin, as phosphat/and benzoyl peroxide) Topical gel 1%/5% [product monograph]. Laval (QC): Bausch Health Canada, Inc; 2021 Mar 29.
32.Retin-A® (Tretinoin) Topical Cream 0.1% w/w, 0.05% w/w, 0.025% w/w and 0.01% w/w and Gel 0.025% w/w and 0.01% w/w [product monograph]. Laval (QC): Bausch Health Canada, Inc.; 2022 Jan 24.
33.Retin-A Micro® (Tretinoin) Gel (microsphere) 0.1% w/w and 0.04% w/w [product monograph]. Laval (QC): Bausch Health Canada, Inc.; 2023 Aug 23.
34.TactuPump® and TactuPump® Forte (adapalene and benzoyl peroxide) topical gel 0.1%/2.5% w/w, 0.3%/2.5% w/w [product monograph]. Thornhill (ON): Galderma Canada, Inc; 2019 May 17.
35.CLINDOXYL ADV and CLINDOXYL (clindamycin and benzoyl peroxide) Topical Gel 1%/3% ww, 1%/5% ww [product monograph]. Mississauga (ON): GlaxoSmithKline, Inc; 2018 Nov 2.
36.Differin® 0.1% w/w topical cream, gel, and lotion/DifferinXP® 0.3% w/w topical gel [product monograph]. Thornhill (ON): Galderma Canada, Inc; 2021 Sep 28.
37.Taro-Clindamycin (clindamycin phosphate) Topical Solution 1% w/v [product monograph]. Brampton (ON): Taro Pharmaceuticals, Inc; 2020 Oct 19.
38.Stein Gold L, Lain E, Del Rosso JQ, et al. Clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel for moderate-to-severe acne: Efficacy and safety results from two randomized phase 3 trials. J Am Acad Dermatol. 2023;89(5):927-935. PubMed
39.Girman CJ, Hartmaier S, Thiboutot D, et al. Evaluating health-related quality of life in patients with facial acne: development of a self-administered questionnaire for clinical trials. Qual Life Res. 1996;5(5):481-490. PubMed
40.McLeod LD, Fehnel SE, Brandman J, Symonds T. Evaluating minimal clinically important differences for the acne-specific quality of life questionnaire. Pharmacoeconomics. 2003;21(15):1069-1079. PubMed
41.Higgins JPT, Green S. Cochrane handbook for systematic reviews of interventions version 5.1.0. Updated Mar, 2011 [accessed by sponsor]. London (GB): The Cochrane Collaboration; 2011: www.handbook.cochrane.org.
42.Harper J.C., Baldwin H., J. T, et al. Clinical effectiveness and cost-effectiveness of treatments for the management of moderate to severe acne vulgaris: A systematic review and network meta-analysis (CRD42023430668) [accessed by sponsor]. PROSPERO: International prospective register of systematic reviews. York (GB): University of York Centre for Reviews and Dissemination; 2023: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42023430668.
43.U.S. Food & Drug Administration. Development & Approval Process | Drugs [accessed by sponsor]. 2022; https://www.fda.gov/drugs/development-approval-process-drugs.
44.Cope S, Zhang J, Saletan S, Smiechowski B, Jansen JP, Schmid P. A process for assessing the feasibility of a network meta-analysis: a case study of everolimus in combination with hormonal therapy versus chemotherapy for advanced breast cancer. BMC Med. 2014;12(1):93. PubMed
45.Jansen JP, Trikalinos T, Cappelleri JC, et al. Indirect treatment comparison/network meta-analysis study questionnaire to assess relevance and credibility to inform health care decision making: an ISPOR-AMCP-NPC Good Practice Task Force report. Value Health. 2014;17(2):157-173. PubMed
46.Phillippo DM. Multinma: An R package for Bayesian network meta-analysis of individual and aggregate data [accessed by sponsor]. Paper presented at: Evidence Synthesis and Meta-Analysis in R Conference. 2021.
47.Phillippo DM, Dias S, Welton NJ, Caldwell DM, Taske N, Ades A. Threshold analysis as an alternative to GRADE for assessing confidence in guideline recommendations based on network meta-analyses. Annals of internal medicine. 2019;170(8):538-546. PubMed
48.Network meta-analysis: comparative effectiveness of IDP-126 gel for patients with moderate-to-severe acne vulgaris [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: IDP-126 gel. Laval (QC): Bausch Health; 2023 Nov 30.
49.Bausch Health response to May 13, 2024 CADTH request for additional information regarding IDP-126 Gel CADTH review [internal additional sponsor's information]. Laval (QC): Bausch Health, Canada Inc.; 2024 May 15.
50.Stein Gold L, Baldwin H, Kircik LH, et al. Efficacy and Safety of a Fixed-Dose Clindamycin Phosphate 1.2%, Benzoyl Peroxide 3.1%, and Adapalene 0.15% Gel for Moderate-to-Severe Acne: A Randomized Phase II Study of the First Triple-Combination Drug. Am J Clin Dermatol. 2022;23(1):93-104. PubMed
51.Bowman S, Gold M, Nasir A, Vamvakias G. Comparison of clindamycin/benzoyl peroxide, tretinoin plus clindamycin, and the combination of clindamycin/benzoyl peroxide and tretinoin plus clindamycin in the treatment of acne vulgaris: a randomized, blinded study. J Drugs Dermatol. 2005;4(5):611-618. PubMed
52.Draelos ZD, Shalita AR, Thiboutot D, Oresajo C, Yatskayer M, Raab S. A multicenter, double-blind study to evaluate the efficacy and safety of 2 treatments in participants with mild to moderate acne vulgaris. Cutis. 2012;89(6):287-293. PubMed
53.Del Rosso JQ. Study results of benzoyl peroxide 5%/clindamycin 1% topical gel, adapalene 0.1% gel, and use in combination for acne vulgaris. J Drugs Dermatol. 2007;6(6):616-622. PubMed
54.Tanghetti EA, Werschler WP, Lain T, Guenin E, Martin G, Pillai R. Tazarotene 0.045% Lotion for Once-Daily Treatment of Moderate-to-Severe Acne Vulgaris: Results from Two Phase 3 Trials. J Drugs Dermatol. 2020;19(1):70-77. PubMed
55.Clinical Study Report: V01-126A-201. A PHASE 2, MULTICENTER, RANDOMIZED, DOUBLE-BLIND, VEHICLE CONTROLLED, PARALLEL-GROUP, CLINICAL STUDY COMPARING THE EFFICACY AND SAFETY OF IDP-126 GEL IN THE TREATMENT OF ACNE VULGARIS [Confidential Information] [internal sponsor's report]. Petaluma (CA): Bausch Health Americas, Inc.; 2019 Oct 24.
56.Patient by Site. Study V01-126A-201. [Confidential Information] [sponsor supplied reference]. Petaluma (CA): Bausch Health Americas, Inc. Accessed 2022 Sep 09.
57.Adverse Events Listings [Confidential Information] [sponsor supplied reference]. Petaluma (CA): Bausch Health Americas, Inc. Accessed 2019 May.
58.Clinical Study Report: V01-126A-202. A Phase 2, Multicenter, Randomized, Double-Blind, Vehicle-Controlled Clinical Study to Compare the Safety and Efficacy of IDP-126 Gel to Epiduo Forte Gel (0.3% adapalene/2.5% BPO) in the Treatment of Acne Vulgaris [Confidential Information] [internal sponsor's report]. Petaluma (CA): Bausch Health Americas, Inc.; 2022 Nov 14.
59.Patient by Site. Study V01-126A-202. [Confidential Information] [sponsor supplied reference]. Petaluma (CA): Bausch Health Americas, Inc. Accessed 2022 Dec 08.
60.Dreno B, Kaufmann R, Talarico S, et al. Combination therapy with adapalene-benzoyl peroxide and oral lymecycline in the treatment of moderate to severe acne vulgaris: a multicentre, randomized, double-blind controlled study. Br J Dermatol. 2011;165(2):383-390. PubMed
61.Del Rosso J, Levy-Hacham O, Mizrahi O. Efficacy and safety of microencapsulated benzoyl peroxide 3% and microencapsulated tretinoin 0.1%(E-Bpo/E-Atra) in acne vulgaris: results from two randomized controlled clinical trials. SKIN The Journal of Cutaneous Medicine. 2021;5(1):s24-s24.
62.George M. Selected Poster Abstracts from MauiDerm 2021 for Dermatologists. J Clin Aesthet Dermatol. 2021;14(5 Suppl 1):S8-S30. PubMed
63.Raoof TJ, Hooper D, Moore A, et al. Efficacy and safety of a novel topical minocycline foam for the treatment of moderate to severe acne vulgaris: A phase 3 study. J Am Acad Dermatol. 2020;82(4):832-837. PubMed
64.Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate to severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. Journal of drugs in dermatology: JDD. 2018;17(9):987-996. PubMed
65.Alexis A, Del Rosso JQ, Desai SR, et al. BPX-01 minocycline topical gel shows promise for the treatment of moderate-to-severe inflammatory acne vulgaris. J Clin Aesthet Dermatol. 2018;11(11):25. PubMed
66.Bissonnette R, Poulin Y, Drew J, Hofland H, Tan J. Olumacostat glasaretil, a novel topical sebum inhibitor, in the treatment of acne vulgaris: A phase IIa, multicenter, randomized, vehicle-controlled study. J Am Acad Dermatol. 2017;76(1):33-39. PubMed
67.Eichenfield LF, Gold LS, Nahm WK, Cook-Bolden FE, Pariser DM. Results of a phase 2, randomized, vehicle-controlled study evaluating the efficacy, tolerability, and safety of daily or twice daily sb204 for the treatment of acne vulgaris. J Drugs Dermatol. 2016;15(12):1496-15027. PubMed
68.Thiboutot D, Zaenglein A, Weiss J, Webster G, Calvarese B, Chen D. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 2.5% for the once-daily treatment of moderate to severe acne vulgaris: assessment of efficacy and safety in 2813 patients. J Am Acad Dermatol. 2008;59(5):792-800. PubMed
69.Dogra S, Sumathy TK, Nayak C, et al. Efficacy and safety comparison of combination of 0.04% tretinoin microspheres plus 1% clindamycin versus their monotherapy in patients with acne vulgaris: a phase 3, randomized, double-blind study. J Dermatolog Treat. 2021;32(8):925-933. PubMed
70.Stewart DM, Torok HM, Weiss JS, Plott RT, Solodyn Phase 2 Study G. Dose-ranging efficacy of new once-daily extended-release minocycline for acne vulgaris. Cutis. 2006;78(4 Suppl):11-20. PubMed
71.Leyden JJ, Sniukiene V, Berk DR, Kaoukhov A. Efficacy and Safety of Sarecycline, a Novel, Once-Daily, Narrow Spectrum Antibiotic for the Treatment of Moderate to Severe Facial Acne Vulgaris: Results of a Phase 2, Dose-Ranging Study. J Drugs Dermatol. 2018;17(3):333-338. PubMed
72.Tanghetti EA, Kircik LH, Green LJ, et al. A Phase 2, Multicenter, Double-Blind, Randomized, Vehicle-Controlled Clinical Study to Compare the Safety and Efficacy of a Novel Tazarotene 0.045% Lotion and Tazarotene 0.1% Cream in the Treatment of Moderate-to-Severe Acne Vulgaris. J Drugs Dermatol. 2019;18(6):542. PubMed
73.Gold LS, Cruz A, Eichenfield L, et al. Effective and safe combination therapy for severe acne vulgaris: a randomized, vehicle-controlled, double-blind study of adapalene 0.1%-benzoyl peroxide 2.5% fixed-dose combination gel with doxycycline hyclate 100 mg. Cutis. 2010;85(2):94-104. PubMed
74.Tanghetti E, Dhawan S, Green L, et al. Randomized comparison of the safety and efficacy of tazarotene 0.1% cream and adapalene 0.3% gel in the treatment of patients with at least moderate facial acne vulgaris. J Drugs Dermatol. 2010;9(5):549-558. PubMed
75.Leyden JJ, Bruce S, Lee CS, et al. A randomized, phase 2, dose-ranging study in the treatment of moderate to severe inflammatory facial acne vulgaris with doxycycline calcium. Journal of Drugs in Dermatology: JDD. 2013;12(6):658-663. PubMed
76.Picardo M, Cardinali C, La Placa M, et al. Efficacy and safety of N-acetyl-GED-0507-34-LEVO gel in patients with moderate-to severe facial acne vulgaris: a phase IIb randomized double-blind, vehicle-controlled trial. Br J Dermatol. 2022;187(4):507-514. PubMed
77.Stein Gold L, Weiss J, Rueda MJ, Liu H, Tanghetti E. Moderate and Severe Inflammatory Acne Vulgaris Effectively Treated with Single-Agent Therapy by a New Fixed-Dose Combination Adapalene 0.3%/Benzoyl Peroxide 2.5% Gel: A Randomized, Double-Blind, Parallel-Group, Controlled Study. Am J Clin Dermatol. 2016;17(3):293-303. PubMed
78.Hebert A, Thiboutot D, Stein Gold L, et al. Efficacy and Safety of Topical Clascoterone Cream, 1%, for Treatment in Patients With Facial Acne: Two Phase 3 Randomized Clinical Trials. JAMA Dermatol. 2020;156(6):621-630. PubMed
79.Webster GF, Sugarman J, Levy-Hacham O, Toledano O. Microencapsulated Benzoyl Peroxide and Tretinoin for the Treatment of Acne Vulgaris: Results from a Phase 2 Multicenter, Double-Blind, Randomized, Vehicle-Controlled Study. Skinmed. 2020;18(6):343-351. PubMed
80.Feldman SR, Werner CP, Ab AS. The efficacy and tolerability of tazarotene foam, 0.1%, in the treatment of acne vulgaris in 2 multicenter, randomized, vehicle-controlled, double-blind studies. Journal of drugs in dermatology: JDD. 2013;12(4):438-446. PubMed
81.Gold LS, Dhawan S, Weiss J, Draelos ZD, Ellman H, Stuart IA. A novel topical minocycline foam for the treatment of moderate-to-severe acne vulgaris: Results of 2 randomized, double-blind, phase 3 studies. J Am Acad Dermatol. 2019;80(1):168-177. PubMed
82.Moore A, Ling M, Bucko A, Manna V, Rueda MJ. Efficacy and Safety of Subantimicrobial Dose, Modified-Release Doxycycline 40 mg Versus Doxycycline 100 mg Versus Placebo for the treatment of Inflammatory Lesions in Moderate and Severe Acne: A Randomized, Double-Blinded, Controlled Study. J Drugs Dermatol. 2015;14(6):581-586. PubMed
83.Pariser DM, Eichenfield LF, Bukhalo M, Waterman G, Jarratt M, Group PDTS. Photodynamic therapy with methyl aminolaevulinate 80 mg g(−1) for severe facial acne vulgaris: a randomized vehicle-controlled study. Br J Dermatol. 2016;174(4):770-777. PubMed
84.Pariser DM, Rich P, Cook-Bolden FE, Korotzer A. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 3.75% for the once-daily treatment of moderate to severe acne vulgaris. J Drugs Dermatol. 2014;13(9):1083-1089. PubMed
85.Tyring SK, Kircik LH, Pariser DM, Guenin E, Bhatt V, Pillai R. Novel Tretinoin 0.05% Lotion for the Once-Daily Treatment of Moderate-to-Severe Acne Vulgaris: Assessment of Efficacy and Safety in Patients Aged 9 Years and Older. J Drugs Dermatol. 2018;17(10):1084-1091. PubMed
86.Schmidt N, Gans EH. Clindamycin 1.2% Tretinoin 0.025% Gel versus Clindamycin Gel Treatment in Acne Patients: A Focus on Fitzpatrick Skin Types. J Clin Aesthet Dermatol. 2011;4(6):31-40. PubMed
87.Botanix Pharmaceuticals. NCT03573518: Evaluation of BTX 1503 in Patients With Moderate to Severe Acne Vulgaris [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2018: https://classic.clinicaltrials.gov/show/NCT03573518. Accessed Jun 26.
88.Photocure. NCT00933543: Efficacy and Safety Study With Visonac Photodynamic Therapy (PDT) [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2009: https://classic.clinicaltrials.gov/show/NCT00933543. Accessed Aug.
89.Bausch Health Americas, Inc. NCT02965456: Efficacy and Safety of IDP-121 and IDP-121 Vehicle Lotion in the Treatment of Acne Vulgaris [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2015: https://classic.clinicaltrials.gov/show/NCT02965456. Accessed Nov 4.
90.Bausch Health Americas, Inc. NCT02932306: Efficacy and Safety of IDP-121 and IDP-121 Vehicle Lotion in the Treatment of Acne Vulgaris [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2017: https://classic.clinicaltrials.gov/ct2/show/NCT02932306.
91.Galderma R&D. NCT01616654: Dose Range Study of CD5789 in Acne Vulgaris. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine: https://classic.clinicaltrials.gov/ct2/show/NCT01616654. Accessed 2021 Sep 20.
92.Galderma R&D. NCT00598832: Clinical Study to Test the Efficacy and Safety of Adapalene, 0.1%. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine: https://classic.clinicaltrials.gov/ct2/show/NCT00598832. Accessed 2021 Feb 18.
93.Dermira, Inc. NCT03073486: A Study of Olumacostat Glasaretil Gel in Subjects With Acne Vulgaris [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2017: https://classic.clinicaltrials.gov/show/NCT03073486. Accessed Feb 6.
94.Dermira, Inc. NCT02431052: A Dose-ranging Study of DRM01 in Subjects With Acne Vulgaris [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2015: https://classic.clinicaltrials.gov/show/NCT02431052. Accessed Apr.
95.Tan J, Thiboutot D, Popp G, et al. Randomized phase 3 evaluation of trifarotene 50 mug/g cream treatment of moderate facial and truncal acne. J Am Acad Dermatol. 2019;80(6):1691-1699. PubMed
96.Eichenfield LF, Draelos Z, Lucky AW, et al. Preadolescent moderate acne vulgaris: a randomized trial of the efficacy and safety of topical adapalene-benzoyl peroxides. J Drugs Dermatol. 2013;12(6):611-618. PubMed
97.Gold LS, Tan J, Cruz-Santana A, et al. A North American study of adapalene-benzoyl peroxide combination gel in the treatment of acne. Cutis. 2009;84(2):110-116. PubMed
98.Koltun W, Lucky AW, Thiboutot D, et al. Efficacy and safety of 3 mg drospirenone/20 mcg ethinylestradiol oral contraceptive administered in 24/4 regimen in the treatment of acne vulgaris: a randomized, double-blind, placebo-controlled trial. Contraception. 2008;77(4):249-256. PubMed
99.Eichenfield LF, Hebert AA, Schachner L, Paller AS, Rossi AB, Lucky AW. Tretinoin microsphere gel 0.04% pump for treating acne vulgaris in preadolescents: a randomized, controlled study. Pediatr Dermatol. 2012;29(5):598-604. PubMed
100.Gollnick HP, Draelos Z, Glenn MJ, et al. Adapalene-benzoyl peroxide, a unique fixed-dose combination topical gel for the treatment of acne vulgaris: a transatlantic, randomized, double-blind, controlled study in 1670 patients. Br J Dermatol. 2009;161(5):1180-1189. PubMed
101.Maleszka R, Turek-Urasinska K, Oremus M, Vukovic J, Barsic B. Pulsed azithromycin treatment is as effective and safe as 2-week-longer daily doxycycline treatment of acne vulgaris: a randomized, double-blind, noninferiority study. Skinmed. 2011;9(2):86-94. PubMed
102.Maloney JM, Dietze P, Jr., Watson D, et al. Treatment of acne using a 3-milligram drospirenone/20-microgram ethinyl estradiol oral contraceptive administered in a 24/4 regimen: a randomized controlled trial. Obstet Gynecol. 2008;112(4):773-781. PubMed
103.AndroScience Corp. NCT01289574: Topical ASC-J9 Cream for Acne [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2011: https://classic.clinicaltrials.gov/show/NCT01289574. Accessed Feb.
104.Actavis Inc. NCT02651220: Clinical End Point Study of Generic Adapalene and Benzoyl Peroxide Gel Versus Epiduo® Forte Gel in Treatment of Acne Vulgaris [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2015: https://classic.clinicaltrials.gov/show/NCT02651220. Accessed Nov.
105.Bayer. NCT00818519: GA YAZ ACNE in China Phase III [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2008: https://classic.clinicaltrials.gov/show/NCT00818519. Accessed Dec.
106.Padagis LLC. NCT01138514: Clinical Study Between Two Clindamycin 1%/Benzoyl Peroxide 5% Topical Gels [accessed by sponsor]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2009: https://classic.clinicaltrials.gov/show/NCT01138514. Accessed Oct.
Appendix 1: Detailed Outcome Data
Please note that this appendix has not been copy-edited.
Table 38: Description of Included Studies (Sponsor-Submitted NMA)
Study ID and country | Interventions | Patients at baseline | Outcomes |
|---|---|---|---|
Dréno et al. (2011)60 France, Italy, Belgium, Sweden, Germany, Poland, Mexico, Brazil, and Australia | Adapalene 0.1% plus benzoyl peroxide 2.5% and lymecycline 300 mg | 191 |
|
Lymecycline 300 mg with vehicle | 187 | ||
Rosso et al. (2021)61 (SGT-65-04), US | E-BPO plus E-ATRA (Benzoyl peroxide 3% plus topical tretinoin 0.1%) | 281 |
|
Vehicle | 143 | ||
Rosso et al. (2021)61 (SGT-65-05), US | E-BPO plus E-ATRA (benzoyl peroxide 3% plus topical tretinoin 0.1%) | 290 |
|
Vehicle | 144 | ||
Rosso et al. (2021),62 US | FCD105 (minocycline 3% + adapalene 0.3%) | 140 |
|
Vehicle | 83 | ||
Raoof et al. (2020),63 US | FMX101 (minocycline 0.4%) | 738 |
|
Vehicle | 750 | ||
Moore et al. (2018)64 (NCT02322866), US | Sarecycline 1.5 mg/kg | 519 |
|
Placebo | 515 | ||
Moore et al. (2018)64 (NCT02320149), US | Sarecycline 1.5 mg/kg | 483 |
|
Placebo | 485 | ||
Alexis et al. (2018) (NCT02815332),65 US | BPX-01 1% (minocycline hydrochloride [HCl] gel) | 73 |
|
BPX-01 2% (minocycline hydrochloride [HCl] gel) | 72 | ||
Vehicle control | 74 | ||
Bissonnette et al. (2016),66 Canada | Olumacostat glasaretil 7.5% | 53 |
|
Vehicle | 55 | ||
Eichenfield et al. (2016),67 US | SB204 gel 2% twice daily | 53 |
|
SB204 gel 4% twice daily | 51 | ||
SB204 gel 4% once daily | 52 | ||
Vehicle | 56 | ||
Thiboutot et al. (2008),68 US | Clindamycin 1.2% plus benzoyl peroxide 2.5% | 797 |
|
Clindamycin phosphate 1.2% | 812 | ||
Benzoyl peroxide 2.5% | 809 | ||
Vehicle | 395 | ||
Dogra et al. (2021),69 India | Tretinoin microsphere 0.025% plus clindamycin 1% | 300 |
|
Tretinoin 0.025% | 300 | ||
Clindamycin 1% | 150 | ||
Stewart Daniel M et al. (2006),70 US | Extended-release minocycline hydrochloride formulation 1 mg/kg | 59 |
|
Extended-release minocycline hydrochloride formulation 2 mg/kg | 59 | ||
Extended-release minocycline hydrochloride formulation 3 mg/kg | 60 | ||
Placebo | 55 | ||
Leyden et al. (2018),71 US | Sarecycline 0.75 mg/kg | 76 |
|
Sarecycline 1.5 mg/kg | 70 | ||
Sarecycline 3 mg/kg | 66 | ||
Placebo | 72 | ||
Tanghetti et al. (2019),72 US | Tazarotene 0.045% lotion | 69 |
|
Tazarotene 0.1% cream | 72 | ||
Vehicle | 69 | ||
Gold (2010),73 US and Canada | Adapalene 0.1% plus benzoyl peroxide 2.5%+ doxycycline hyclate 100 mg | 232 |
|
Vehicle + doxycycline hyclate 100 mg | 227 | ||
Tanghetti et al. (2010),74 US and Canada | Tazarotene 0.1% | 90 |
|
Adapalene 0.3% | 90 | ||
Leyden et al. (2013),75 US | Doxycycline 0.75 mg/kg | 64 |
|
Doxycycline 1.5 mg/kg | 65 | ||
Doxycycline 3 mg/kg | 61 | ||
Placebo | 67 | ||
Picardo et al. (2022),76 Germany, Italy, Poland | NAC-GED gel 2% | 150 |
|
NAC-GED gel 5% | 150 | ||
Vehicle | 150 | ||
Gold (2016),77 US and Canada | Adapalene 0.3% plus benzoyl peroxide 2.5% | 217 |
|
Vehicle | 69 | ||
Hebert et al. (2020) (NCT02608450),78 US, Bulgaria, Romania, Poland, Serbia, Georgia | Clascoterone 1% | 353 |
|
Vehicle | 355 | ||
Hebert et al. (2020) (NCT02608476),78 US, Ukraine, Georgia | Clascoterone 1% | 369 |
|
Vehicle | 363 | ||
Webster et al. (2020),79 US | E-BP (microencapsulated benzoyl peroxide 3%) | 118 |
|
E-ATRA low (Microencapsulated tretinoin 0.05%) | 118 | ||
E-ATRA high (microencapsulated tretinoin 0.1%) | 118 | ||
TWIN low (E- ATRA 0.5% + E-B 3%) | 121 | ||
TWIN high (E- ATRA 0.1% + E-B 3%) | 120 | ||
Vehicle | 115 | ||
Feldman et al. (2013)80 Study 301, US and Canada | Tazarotene 0.1% | 371 |
|
Vehicle | 372 | ||
Feldman et al. (2013)80 Study 302, US and Canada | Tazarotene 0.1% | 373 |
|
Vehicle | 369 | ||
Gold et al. (2018)81 (NCT02815267) (S-04), US | Topical minocycline foam 4% | 307 |
|
Vehicle | 159 | ||
Gold et al. (2018)81 (NCT02815280) (S-05), US | Topical minocycline foam 4% | 333 |
|
Vehicle | 162 | ||
Moore et al. (2015),82 US | Modified release doxycycline 400 mg | 216 |
|
Doxycycline 100 mg | 224 | ||
Placebo | 222 | ||
Pariser et al. (2016),83 US | (Methyl aminolevulinate, low dose) MAL + PDT (photodynamic therapy) | 100 |
|
Vehicle | 53 | ||
Pariser et al. (2014),84 US | Clindamycin 1.2% plus benzoyl peroxide 3.75% | 253 |
|
Vehicle | 245 | ||
Tyring et al. (2018),85 US | Tretinoin 0.05% | 819 |
|
Vehicle | 821 | ||
Schmidt et al. (2011),86 US | Clindamycin 1.2% plus tretinoin 0.025% | 1,008 |
|
Clindamycin 1.2% | 1,002 | ||
Vehicle | 245 | ||
Pharmaceuticals, Botanix (2018),87 US | BTX 1503 5% once daily | 92 |
|
BTX 1503 5% twice daily | 92 | ||
BTX 1503 2.5% once daily | 92 | ||
Vehicle | 92 | ||
Photocure (2009),88 US and Canada | Visonac cream with PDT | 54 |
|
Vehicle | 53 | ||
V01 to 126A-301 (2021),38 North America | IDP-126 gel (clindamycin phosphate 1.2% plus benzoyl peroxide 2.5% and adapalene 0.3%) | 122 |
|
Vehicle | 61 | ||
V01 to 126A-302 (2021),38 North America | IDP-126 gel (clindamycin phosphate 1.2% plus benzoyl peroxide 2.5% and adapalene 0.3%) | 120 |
|
Vehicle | 60 | ||
Bausch Health Americas, Inc. (2019),89 US | IDP-121 lotion (tretinoin 0.5%) | 413 |
|
Vehicle | 407 | ||
Bausch Health Americas, Inc. (2020),90 US | IDP-121 lotion (tretinoin 0.5%) | 406 |
|
Vehicle | 414 | ||
Galderma R&D (2021) (NCT01616654),91 US | CD5789 cream 25 mcg/g | 61 |
|
CD5789 cream 50 mcg/g | 61 | ||
CD5789 cream 100 mcg/g | 60 | ||
Tazarotene gel 25 mcg/g | 61 | ||
Vehicle | 61 | ||
Galderma R&D (2008) (NCT00599521 and NCT00598832),92 US and Canada | Adapalene 0.1% | 535 |
|
Vehicle | 531 | ||
Adapalene 0.1% | 533 | ||
Vehicle | 542 | ||
Dermia, Inc. (NCT03073486 and NCT03028363),93 US, Canada, Australia | Olumacostat glasaretil gel 5% | 493 |
|
Vehicle | 251 | ||
Olumacostat glasaretil gel 5% | 498 | ||
Vehicle | 261 | ||
Dermira, Inc. (2016),94 US, Canada | Olumacostat glasaretil gel 4% | 106 |
|
Olumacostat glasaretil 7.5% once daily | 110 | ||
Olumacostat glasaretil gel 7.5% twice daily | 101 | ||
Vehicle once daily | 52 | ||
Vehicle twice daily | 50 | ||
Tanghetti et al. (2020)54 (Study 1), US and Canada | Tazarotene 0.045% | 402 |
|
Vehicle | 411 | ||
Tanghetti et al. (2020)54 (Study 2), US and Canada | Tazarotene 0.045% | 397 |
|
Vehicle | 404 | ||
Tan et al. (2019)95 (PERFECT 1), US, Canada, Europe, and Russia | Trifarotene 50 mcg/g | 612 |
|
Vehicle | 596 | ||
Tan et al. (2019)95 (PERFECT 2), US, Canada, Europe, and Russia | Trifarotene 50 mcg/g | 602 |
|
Vehicle | 610 | ||
Gold et al. (2021),50 US and Canada | IDP-126 gel [clindamycin phosphate 1.2% plus benzoyl peroxide 2.5% and adapalene 0.3%] | 146 |
|
Topical benzoyl peroxide 3.1% plus adapalene 0.15% | 150 | ||
Topical clindamycin phosphate 1.2% plus benzoyl peroxide 3.1% | 146 | ||
Topical clindamycin phosphate 1.2% plus adapalene 0.15% | 150 | ||
Vehicle | 148 | ||
Eichenfield et al. (2013),96 US and Canada | Topical adapalene 0.1% plus benzoyl peroxide gel 2.5% | 142 |
|
Vehicle | 143 | ||
Stein Gold et al. (2009),97 US, Puerto Rico, and Canada | Topical adapalene 0.1% plus benzoyl peroxide 2.5% | 415 |
|
Topical adapalene gel 0.1% | 420 | ||
Topical benzoyl peroxide gel 2.5% | 415 | ||
Vehicle | 418 | ||
Koltun et al. (2008),98 US | Drospirenone 3 mg plus ethinyl estradiol 20 mcg | 266 |
|
Placebo | 268 | ||
Eichenfield et al. (2012),99 US | Tretinoin microsphere gel 0.04% | 55 |
|
Vehicle | 55 | ||
Gollnick et al. (2009),100 US Canada, Europe | Adapalene 0.1% plus benzoyl peroxide 2.5% | 419 |
|
Adapalene gel 0.1% | 418 | ||
Benzoyl peroxide 2.5% | 415 | ||
Vehicle | 418 | ||
Maleszka et al. (2011),101 Poland | Azithromycin 500 mg | 109 |
|
Doxycycline 100 mg | 115 | ||
Maloney et al. (2008),102 US | Drospirenone 3 mg plus ethinyl estradiol 20 mcg | 270 |
|
Placebo | 268 | ||
AndroScience Corp (2014),103 US, Taiwan | ASC-J9 cream 0.025% | 60 |
|
ASC-J9 cream 0.1% | 63 | ||
Vehicle | 58 | ||
Actavis Inc. (2020),104 US | Adapalene 0.3% plus benzoyl peroxide 2.5% | 337 |
|
Adapalene 0.3% plus benzoyl peroxide 2.5% | 335 | ||
Vehicle | 329 | ||
Bayer (2010),105 China | Drospirenone 3 mg plus ethinyl estradiol 20 mcg | 89 |
|
Placebo | 90 | ||
Padagis LLC (2021),106 NR | Clindamycin 1% plus benzoyl peroxide 5% (Perrigo) | 430 |
|
Clindamycin 1% plus benzoyl peroxide 5% (Benzaclin) | 419 | ||
Vehicle | 427 |
IL = inflammatory lesion; NIL = noninflammatory lesion; NR = not reported.
Source: Sponsor summary of clinical evidence.15
Figure 5: NMA League Table for Proportion of Patients Experiencing Treatment Success Outcome

BPO = benzoyl peroxide; IDP-126 gel = clindamycin 1.2% plus BPO 3.1% and adapalene 0.15% topical gel; OA1 = oral antibiotic monotherapy; ORx = other treatment; PH1 = physical treatment only; TFDCAB2 = topical antibiotic and BPO fixed-dose combination; TFDCABR3 = topical antibiotic plus BPO and retinoid fixed-dose combination; TFDCAR2 = topical antibiotic plus retinoid fixed-dose combination; TFDCRB2 = topical retinoid and BPO fixed-dose combination; TMA1 = topical antibiotic monotherapy; TMB1 = topical BPO monotherapy; TMO1 = other topical (prescription) monotherapies; TMR1 = topical retinoid monotherapy; TOA3 = combinations of dual-drug fixed-dose topical treatments with an oral antibiotic; V/P = vehicle or placebo.
Note: Results should be interpreted as row versus column. Values in cells represent estimated log-odds ratios with their 95%credible intervals. All bolded values are statistically meaningful at the 0.05 significant level.
Source: IDP-126 ITC technical report.48
Figure 6: NMA League Table for Inflammatory Lesion Count Reduction Outcome
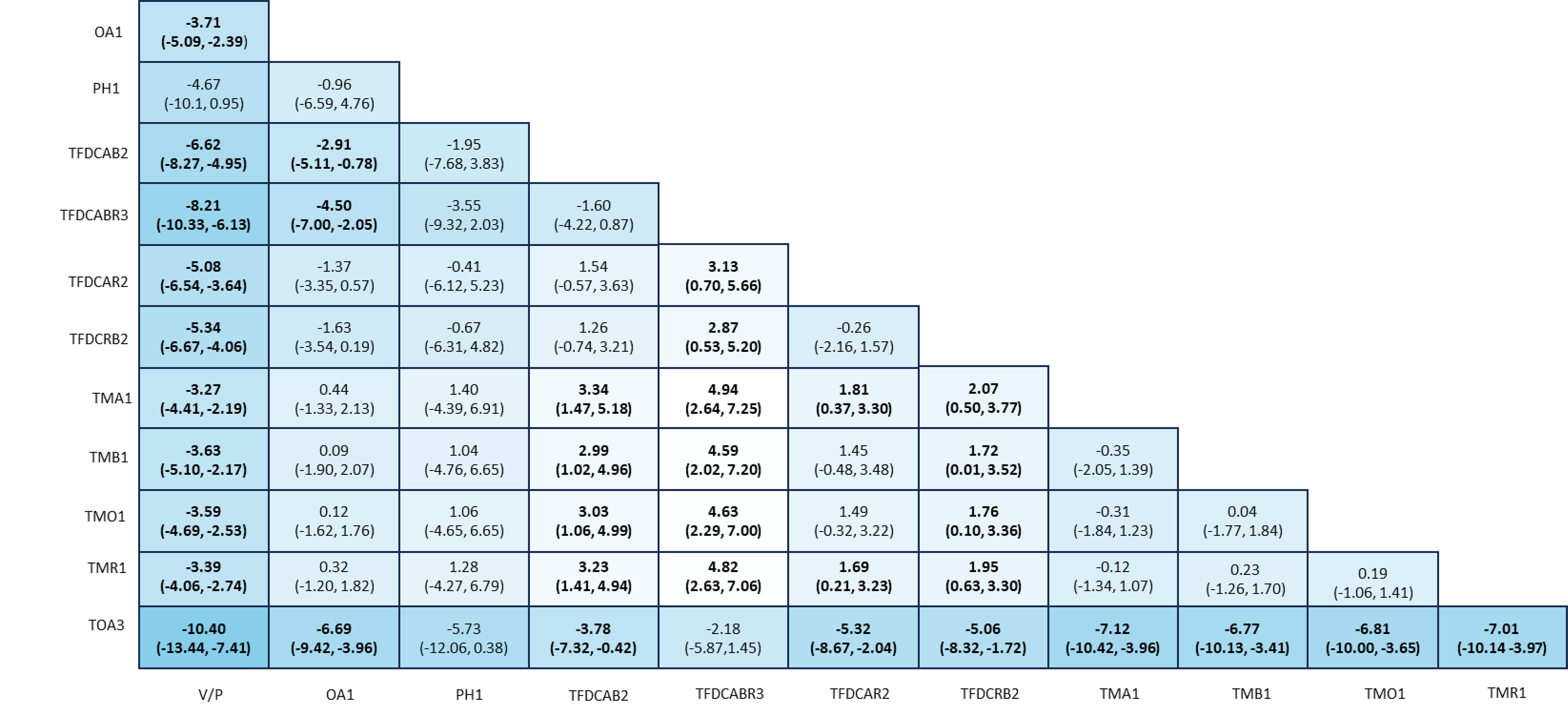
BPO = benzoyl peroxide; IDP-126 gel = clindamycin 1.2% plus BPO 3.1% and adapalene 0.15% topical gel; OA1 = oral antibiotic monotherapy; ORx = other treatment; PH1 = physical treatment only; TFDCAB2 = topical antibiotic and BPO fixed-dose combination; TFDCABR3 = topical antibiotic plus BPO and retinoid fixed-dose combination; TFDCAR2 = topical antibiotic plus retinoid fixed-dose combination; TFDCRB2 = topical retinoid and BPO fixed-dose combination; TMA1 = topical antibiotic monotherapy; TMB1 = topical BPO monotherapy; TMO1 = other topical (prescription) monotherapies; TMR1 = topical retinoid monotherapy; TOA3 = combinations of dual-drug fixed-dose topical treatments with an oral antibiotic; V/P = vehicle or placebo.
Note: Results should be interpreted as row versus column manner. Values in cells represent estimated mean difference with their 95% credible intervals. All bolded values are statistically meaningful at the 0.05 significant level.
Source: IDP-126 ITC technical report.48
Figure 7: NMA League Table for Noninflammatory Lesion Count Reduction Outcome

BPO = benzoyl peroxide; IDP-126 gel = clindamycin 1.2% plus BPO 3.1% and adapalene 0.15% topical gel; OA1 = oral antibiotic monotherapy; ORx = other treatment; PH1 = physical treatment only; TFDCAB2 = topical antibiotic and BPO fixed-dose combination; TFDCABR3 = topical antibiotic plus BPO and retinoid fixed-dose combination; TFDCAR2 = topical antibiotic plus retinoid fixed-dose combination; TFDCRB2 = topical retinoid and BPO fixed-dose combination; TMA1 = topical antibiotic monotherapy; TMB1 = topical BPO monotherapy; TMO1 = other topical (prescription) monotherapies; TMR1 = topical retinoid monotherapy; TOA3 = combinations of dual-drug fixed-dose topical treatments with an oral antibiotic; V/P = vehicle or placebo.
Note: Results should be interpreted as row versus column manner. Values in cells represent estimated mean difference with their 95% credible intervals. All bolded values are statistically meaningful at the 0.05 significant level.
Source: IDP-126 ITC technical report.48
PharmacoeconomicReview
Abbreviations
BIA
budget impact analysis
BPO
benzoyl peroxide
BSC
best supportive care
CDA-AMC
Canada’s Drug Agency
DLQI
Dermatology Life Quality Index
ICER
incremental cost-effectiveness ratio
IDP-126
clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% topical gel
NMA
network meta-analysis
QALY
quality-adjusted life-year
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% (Cabtreo) topical gel |
Indication | For the topical treatment of acne vulgaris in patients 12 years of age and older |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | August 15, 2024 |
Reimbursement request | As per indication |
Sponsor | Bausch Health, Canada Inc. |
Submission history | Previously reviewed: No |
NOC = Notice of Compliance.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation |
|
Target population | Patients aged 12 years and older with acne vulgaris |
Treatment | Clindamycin 1.2% plus BPO 3.1% and adapalene 0.15% (IDP-126) topical gel |
Dose regimen | A pea-sized amount of gel (1.5 g) once daily |
Submitted price | IDP-126 gel: $147.42 per 50 g pump |
Submitted treatment cost | $1,614 per year |
Comparators |
|
Perspective | Canadian publicly funded health care payer |
Outcomes | QALYs, LYs |
Time horizon | 60 weeks (1.15 years) |
Key data source |
|
Submitted results | Among the optimal treatments (on the efficiency frontier): IDP-126 gel was the most costly and most effective, with an ICER of $62,967 per QALY gained (incremental costs = $1,133; incremental QALYs = 0.02) compared with topical antibiotic plus BPO fixed-dose combinations. |
Key limitations |
|
CDA-AMC reanalysis results |
|
BPO = benzoyl peroxide; CDA-AMC = Canada’s Drug Agency; HRQoL = health-related quality of life; ICER = incremental cost-effectiveness ratio; IDP-126 = clindamycin plus benzoyl peroxide and adapalene; LY = life-year; NMA = network meta-analysis; QALY = quality-adjusted life-year.
Conclusions
Based on the Clinical Review by Canada’s Drug Agency (CDA-AMC), clindamycin 1.2% plus benzoyl peroxide (BPO) 3.1% and adapalene 0.15% (IDP-126) gel results in a clinically meaningful improvement in acne, when compared with its vehicle gel, in patients aged 10 years and older with moderate to severe acne based on data from Study 301 and Study 302. The sponsor-conducted network meta-analysis (NMA) results showed a favourable treatment effect of IDP-126 gel versus vehicle or placebo, oral antibiotic, and topical monotherapies on treatment success and change in lesion counts. While the effect estimates are suggestive of a possible favourable treatment effect with IDP-126 gel versus topical fixed-dose dual-combination therapies, there is some uncertainty, as the 95% credible intervals included the null or were close to the null for certain dual-combination therapies. However, several notable limitations in the NMA, including the possible exclusion of relevant studies, heterogeneity across trials in the network, and the omission of relevant comparators for the Canadian setting suggest the resulting estimates may be biased. As a result, the Clinical Review concluded that while IDP-126 gel was favoured over other active therapies on treatment success and change in lesion count, the magnitude of benefit associated with IDP-126 gel is uncertain.
CDA-AMC identified several limitations with the submitted economic evaluation that could not be addressed through reanalysis. As a result, the cost-effectiveness of IDP-126 gel relative to active therapies is highly uncertain. According to the sponsor’s base case, IDP-126 gel was associated with the highest total costs and highest total quality-adjusted life-years (QALYs) versus all comparators. As noted in the CDA-AMC Clinical Review Report, while the sponsor’s NMA favoured IDP-126 gel over several alternative active therapies for acne vulgaris, the magnitude of benefit remains uncertain. That, combined with limitations in the sponsor’s model structure and approach to health state utility values, means that the magnitude of the QALY gain associated with IDP-126 gel is highly uncertain and could be overestimated. The sponsor’s analysis predicted that, compared with topical antibiotic plus BPO fixed-dose combinations, IDP-126 would lead to a 0.02 QALY gain, which is equivalent to approximately 7 days spent in perfect health over the 1.15-year time horizon and which could be overestimated. Additionally, in the sponsor’s base case, all of the incremental costs were attributable to treatment acquisition costs, meaning that the sponsor is not predicting that IDP-126 gel will result in any cost offsets or reductions in health care resource use. As such, based on public list prices for comparators and the sponsor’s submitted price for IDP-126, there is high certainty that IDP-126 is more costly than comparators and there is significant unresolved uncertainty regarding the quantity of QALY benefit it is expected to yield. If a decision-maker is willing to accept the assumptions from the sponsor’s submitted base case, including the magnitude of benefit associated with IDP-126 versus comparators, then a 13% price reduction would be required for IDP-126 gel to be considered cost-effective. However, given the limitations identified by CDA-AMC that could not be addressed, a higher price reduction is likely required for IDP-126 to be considered cost-effective. Finally, public drug plan coverage of many of the comparator treatments included in the analysis is variable. Based on current list prices of comparators and the sponsor’s submitted price for IDP-126, IDP-126 is more expensive than all currently listed treatments, apart from adapalene 0.3%, which is reimbursed by only 1 drug plan that participates in the CDA-AMC review process.
Input Relevant to the Economic Review
This section is a summary of the feedback received from patient and clinician groups and the drug plans that participate in the CDA-AMC review process.
Patient input was provided as a joint submission from the Acne and Rosacea Society of Canada and the Canadian Skin Patient Alliance. Information was obtained from patients with acne using 2 electronic surveys. The first targeted any individual living with acne in Canada and collected information from 154 respondents. The second survey, which targeted patients living in Canada who are participating in the clinical trial for IDP-126 gel, collected information from 3 respondents. In terms of experience with currently available treatments, more than half of respondents in the first survey reported experience with isotretinoin pills to treat their acne and it was generally viewed to be an effective option. Hormone pills (birth control and spironolactone) were also reported as effective treatment options. A total of 89% of respondents reported they had used some form of prescription gel or cream with varying degrees of success. In terms of hopes for improved outcomes, respondents noted that top priorities were the ability to enjoy personal relationships and improvements in scarring and skin pigmentation, along with having clearer skin and improved self-confidence and mental health. Three respondents to the second survey all had experience with IDP-126 gel as participants in clinical trials in Canada. All 3 respondents indicated that IDP-126 gel was an effective way to treat their acne. Each respondent expressed a willingness to accept some form of adverse event (skin irritation, dry skin, redness) if the treatment reduced the extent and severity of their acne. There was a consensus that IDP-126 gel may be an effective treatment option that offers relief in a short amount of time.
No clinician input was received for this review.
The drug plan input raised concerns regarding the implementation of IDP-126 gel, including its place in therapy as a first or subsequent line of treatment.
Several of these concerns were addressed in the sponsor’s model:
The model tracks the amount of lesion reduction achieved on each treatment.
CDA-AMC was unable to address the following concerns raised in the input:
There is concern as to the place in therapy for IDP-126 gel. The submitted model structure does not consider the possibility that patients will discontinue 1 active therapy and switch to another.
Economic Review
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The sponsor submitted an economic evaluation comparing IDP-126 gel with topical acne treatments, grouped by categories of comparator treatments. The model population comprised individuals aged 12 years and older with acne vulgaris.1 This was aligned with the Health Canada indication and the reimbursement request.
IDP-126 is available as a triple-combination fixed-dose topical gel consisting of an antibiotic (clindamycin 1.2%), a retinoid (adapalene 0.15%), and BPO (3.1%). The submitted price was $147.42 per 50 g pump, corresponding to $2.95 per gram. For the indicated population, the recommended dosage is the application of a pea-sized amount of gel (assumed to be 1.5 g in the sponsor’s submission) to the affected area once daily.1,2 In the model, the sponsor calculated that IDP-126 gel would cost $1,614 per year ($4.42 per day).1 Drug wastage was not considered in the sponsor’s economic evaluation.
For the submitted economic evaluation, comparators were defined in terms of their treatment category, rather than individual treatments, based on the sponsor’s assumption that treatments within the same category would share similar treatment mechanisms and were unlikely to differ in terms of their effectiveness.1 The recommended dose for each comparator was the application of a pea-sized amount of gel, lotion, or cream (assumed to be 1.5 g) to the affected area once or twice daily (depending on the product). A total of 5 treatment categories were considered for the present submission. The weighted cost and included treatments are summarized as follows:1
Topical retinoid monotherapy. Weighted cost: $884 per year ($2.42 per day).
Included treatments: tazarotene, adapalene, tretinoin.
Topical antibiotic monotherapy. Weighted cost: $285 per year ($0.78 per day).
Included treatments: clindamycin.
Topical antibiotic plus retinoid fixed-dose combinations. Weighted cost: $672 per year ($1.84 per day).
Included treatments: clindamycin and tretinoin.
Topical retinoid and BPO fixed-dose combinations. Weighted cost: $800 per year ($2.19 per day).
Formulations of adapalene and BPO.
Topical antibiotic plus BPO fixed-dose combinations. Weighted cost: $599 per year ($1.64 per day).
Included treatments: erythromycin and BPO, formulations of clindamycin and BPO.
Modelled outcomes included life-years and QALYs. Costs were estimated from the perspective of the public health care payer in Canada. Model outputs were generated over a time horizon of 60 weeks (1.15 years), with a cycle length of 12 weeks. Costs and outcomes were discounted at 1.5% per year.1
Model Structure
The sponsor submitted a Markov model that tracked a hypothetical cohort of patients with acne vulgaris eligible for topical therapy (Figure 1). The Markov structure consisted of 2 mutually exclusive health states: active treatment and best supportive care (BSC), which was defined as the absence of treatment with an active ingredient. Upon model entry, patients initiated 1 of the eligible active therapies (IDP-126 gel or 1 of the comparators). During the first cycle, patients faced a risk of discontinuation because of adverse events, lack of efficacy or nonadherence. Following discontinuation, patients transitioned to the BSC state where they remained for the remainder of the model time horizon.1 Patients who did not discontinue in the first cycle remained in the active treatment health state for the remainder of the model time horizon.
In addition to the treatments, the model also tracked the total lesion counts due to acne. This was calculated as the difference between the baseline lesion count and the treatment-specific estimates of lesion reduction in the first cycle. Afterward, it was assumed patients on active therapy would experience no change in lesion count. Meanwhile, patients who discontinued active therapy were assumed to experience no change in lesion count in the first cycle of the model. In the second cycle, patients on BSC experienced an increase in lesion count that remained constant for the remainder of the model time horizon.1
Model Inputs
Costs and effects were estimated using a homogeneous baseline population. All data summarizing baseline characteristics of the cohort were obtained from studies 301 and 302.1,3 Both studies were randomized, phase III trials that involved the direct comparison of IDP-126 gel with vehicle gel (a gel without an active ingredient).3 Baseline characteristics of interest included the noninflammatory and inflammatory lesion counts (49 and 37, respectively), which were used to calculate the combined baseline lesion count (86).1,3
Transitions from the active treatment state to the BSC state were informed by treatment discontinuation risk, which was assumed to be attributable to adverse events, a lack of efficacy, or nonadherence.1 The discontinuation parameter was calculated as the average (2.9%) between the discontinuation rates due to adverse events in studies 301 (2.5%) and 302 (3.3%), respectively.1,3
Comparative treatment efficacy in the model was based on the number of lesions that patients who remained on active treatment experienced at week 12 and beyond. Estimates of relative efficacy for the economic evaluation were obtained from the sponsor-submitted systematic review and NMA.1,4 Consistent with the economic evaluation, comparators for the indirect treatment comparisons were defined in terms of existing categories of comparator treatments. The outcome of interest was the relative reduction in inflammatory and noninflammatory lesion counts from baseline at 12 weeks.4 Estimates of relative lesion reduction obtained from the NMA were converted to treatment-specific estimates of absolute lesion change from baseline. This was calculated as the sum of the relative lesion reduction change from baseline and the average reduction in lesion count (at 12 weeks) for patients in the vehicle arm of the included trials.1,4
Health-related quality of life was incorporated into the model as a function of the predicted lesion count. The sponsor adopted an approach that adjusted general population utility values based on the predicted number of lesions. This approach meant that a lower number of lesions would reflect a higher utility and, by extension, superior quality of life. Three series of inputs were necessary to implement this relationship. First, general population utility values were obtained from a study of estimates based in Canada measured using the Health Utilities Index (Mark III).5 Given the short time horizon, the sponsor assumed the cohort would be represented by the median value (0.927) for all individuals aged 12 to 19 years.5 Second, the sponsor fitted a linear regression data to model the relationship between the Dermatology Life Quality Index (DLQI) and EQ-5D informed by data from patients with acne in specialist care.1,6 Third, the sponsor fitted an additional linear regression to model the relationship between the DLQI and lesion count (to link the lesion count to the EQ-5D) across 5 studies among acne patients.1,7-11
The submission considered costs associated with treatment acquisition and monitoring, expressed in 2023 Canadian dollars. Treatment acquisition costs were calculated from the price per unit consumed, following the recommended dosage for each alternative considered in the model. It was assumed that each application was equivalent to 1.5 g of a gel, cream, or lotion for all identified comparators. In the event more than 1 commercial option was available for a specific treatment category, the unit cost was estimated as a weighted average where the weights were informed by public claims data. Unit prices reflected the sponsor’s submitted price and values from the Ontario Drug Benefit Formulary and the IQVIA Pharmastat database.1,12,13 Monitoring costs included family medicine and specialist dermatologist visits, with costs obtained from the Ontario Schedule of Benefits: Physician Services.14 Visit frequency was dependent on a patient’s acne severity and informed by input from clinical experts in Canada.
Summary of Sponsor’s Economic Evaluation Results
All analyses were run probabilistically (1,000 iterations for the base-case and scenario analyses). The deterministic and probabilistic results were similar. The probabilistic findings are presented subsequently.
Base-Case Results
Results from the sponsor’s base case of the submitted economic evaluation are presented in Table 3. Three alternatives were identified on the efficiency frontier: topical antibiotic monotherapy, topical antibiotic plus BPO fixed-dose combinations, and IDP-126 gel. Among these comparators, IDP-126 gel was the costliest and most effective, and the incremental cost-effectiveness ratio (ICER) relative to topical antibiotic plus BPO fixed-dose combinations was $62,927 per QALY gained (incremental costs = $1,133; incremental QALYs = 0.02). At a willingness-to-pay threshold of $50,000 per QALY gained, IDP-126 gel had a 29.5% probability of being cost-effective.
Additional results from the sponsor’s submitted economic evaluation base case are presented in Appendix 3. As monitoring costs were similar across all treatments (Table 8), all differences in total costs are attributable to differences in treatment acquisition costs among comparators.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total | Total QALYs | Sequential ICER ($/QALY) |
|---|---|---|---|
Topical antibiotic monotherapy | 1,221 | 0.923 | Reference |
Topical antibiotic plus BPO fixed-dose combination | 1,567 | 0.943 | 17,432 |
IDP-126 gel | 2,700 | 0.961 | 62,967 |
Dominated treatments | |||
Topical antibiotic plus retinoid fixed-dose combinations | 1,651 | 0.939 | Dominated by topical antibiotic plus BPO fixed-dose combinations |
Topical retinoid plus BPO fixed-dose combinations | 1,795 | 0.943 | Extendedly dominated by IDP-126 gel |
Topical retinoid monotherapy | 1,887 | 0.931 | Dominated by topical antibiotic plus BPO fixed-dose combinations, topical antibiotic plus retinoid fixed-dose combinations, topical retinoid plus BPO fixed-dose combinations |
BPO = benzoyl peroxide; ICER = incremental cost-effectiveness ratio; IDP-126 = clindamycin plus benzoyl peroxide and adapalene; QALY = quality-adjusted life-year.
Note: The submitted analysis was based on publicly available prices of the comparator treatments.
Source: Sponsor’s pharmacoeconomic submission.1
Sensitivity and Scenario Analysis Results
In addition to the submitted base case, the sponsor considered 4 distinct scenario analyses. Three of these scenarios explored discount rates of 0% and 3% and a time horizon of 240 weeks. While each scenario had a slight impact on the expected costs and benefits, none had a meaningful effect on the conclusion for the cost-effectiveness of IDP-126 gel.
In addition, the sponsor conducted a scenario analysis from a societal perspective. This scenario incorporated additional indirect costs associated with lost wages due to absenteeism from lesions. In this scenario, relative to topical antibiotic plus BPO fixed-dose combinations, the ICER for IDP-126 gel was $62,726 per QALY gained. This was similar to the sponsor’s base-case analysis using a health care payer perspective.
CDA-AMC Appraisal of the Sponsor’s Economic Evaluation
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
The comparative efficacy of IDP-126 gel is uncertain: The submitted economic evaluation compared IDP-126 gel with alternative categories of treatments for acne vulgaris. These alternatives included: topical retinoid monotherapy, topical antibiotic monotherapy, topical antibiotic plus retinoid fixed-dose combinations, topical retinoid plus BPO fixed-dose combinations, and topical antibiotic plus BPO fixed-dose combinations. Comparisons between multiple topical treatments (for example, between a topical antibiotic and BPO fixed-dose combination alongside a topical retinoid monotherapy) were not included in the submitted NMA or model. Relative treatment effects in the economic evaluation were informed by relative lesion count reductions obtained from the sponsor-submitted NMA.
According to the CDA-AMC Clinical Review, the sponsor-conducted NMA results showed a favourable treatment effect of IDP-126 gel versus topical monotherapies on change in lesion counts. While the effect estimates are suggestive of a possible favourable treatment effect with IDP-126 gel versus topical fixed-dose dual-combination therapies, there is some uncertainty, as the 95% credible intervals included the null or were close to the null for certain dual-combination therapies.
The Clinical Review noted that these estimates were subject to considerable uncertainty. This was attributable to heterogeneous trial populations, the exclusion of potentially relevant alternatives from the NMA, and imprecision in the estimates of relative treatment effect. As a result, the Clinical Review concluded that while IDP-126 gel was favoured over other active therapies, the magnitude of benefit associated with IDP-126 gel was uncertain.
The Clinical Review also noted that, due to the sponsor’s approach of pooling different treatments together within an individual node, the efficacy of specific formulations and specific dosing schedules could not be assessed. Additionally, certain nodes in the network included comparators that are not relevant to the clinical context in Canada.
Due to the limitations with the sponsor’s NMA, there is uncertainty in the relative magnitude of effect with IDP-126 used in the pharmacoeconomic model. This clinical uncertainty could not be addressed in reanalyses.
The model does not reasonably reflect the disease area and current management of acne: The sponsor submitted a Markov model with health states based on patients being on or off active treatment. The model also tracked the number of lesions that patients experienced within each health state. In the first cycle, all patients experienced the baseline number of lesions (86). In subsequent cycles, those who remained on treatment experienced the number of lesions associated with the lesion reduction for their given treatment, which was the sum of the reduction in inflammatory lesions (8.21 for IDP-126 gel) and noninflammatory lesions (13.41 for IDP-126 gel), relative to vehicle, plus the sum of the reductions in inflammatory and noninflammatory lesions with the vehicle gel (28), as reported in the sponsor’s submitted NMA. As such, patients who remained on IDP-126 gel would experience a lesion reduction of 49, leaving them with 37 lesions for the remainder of the model time horizon. Those who transitioned to the BSC health state in cycle 2 all experienced a lesion increase of 19 (total of 105 lesions) regardless of which treatment they initially received. This modelling approach is associated with uncertainty for several reasons.
One, it assumes that the experience of acne and the impact of its treatments are valued solely based on the number of lesions that patients experience. This contradicts the patient input received for this review, which highlighted improvements in scarring and skin pigmentation as important outcomes, and the clinical expert feedback noted that the goals of acne treatment are to reduce symptom severity and prevent scar formation. Furthermore, because health state utility values were based on lesion counts, the modelling approach assumes that a single reduction in lesion counts results in an improvement in patient quality of life. The uncertainty of this assumption is highlighted by clinical expert feedback noting that the number of lesions is estimated by clinicians and not specifically counted. Two, it assumes that reductions in inflammatory and noninflammatory lesion counts are valued equally by patients, which contradicts clinical expert feedback noting that inflammatory lesions cause more discomfort than noninflammatory lesions. Three, the approach meant that cost-effectiveness is highly dependent on the number of baseline lesions. For example, if someone was classified as having mild acne (30 lesions), the model predicts equal QALYs across all treatments, meaning that IDP-126 gel is dominated by all other comparators (associated with equal benefit but highest costs). This is a function of the lesion reduction being capped in the model by the baseline number of lesions (i.e., if the lesion reduction is greater than the baseline lesion count, then the lesion reduction is based on the baseline lesion count, and everyone has 0 lesions after cycle 1); otherwise, the sponsor’s model would predict a negative number of acne lesions. As such, analyses based on baseline acne severity may be relevant.
These issues, combined with a number of simplifying assumptions made by the sponsor (refer to Table 4) meant that IDP-126 gel was certain to lead to an incremental benefit versus all comparators in probabilistic analyses (refer to Figure 2), regardless of the testing of alternative assumptions, such as the time horizon.
CDA-AMC was unable to address this limitation, as revisions to the model structure are beyond the scope of the review. As such, the relative cost-effectiveness of IDP-126 gel for the treatment of acne vulgaris is highly uncertain.
Missing comparators: Alternatives to IDP-126 gel considered in the sponsor’s base case were restricted to existing categories of topical treatments (gels, solutions, creams, or lotions). CDA-AMC guidelines state that the identification of comparators should not be limited to a specific class of interventions. Instead, all interventions that may be used for treatment or displaced by a new technology should be considered in an economic evaluation.15 According to clinical expert feedback received for this review, several hormone therapies (oral contraceptives) are often used in the treatment of acne vulgaris and would represent relevant comparators for the decision model. Additionally, oral antibiotic monotherapy and combinations of dual-drug fixed-dose topical treatments with oral antibiotics were identified as relevant comparators. While both alternatives were included in the submitted NMA (meaning that there were data available to incorporate these treatments into the sponsor’s pharmacoeconomic model), a rationale supporting their exclusion from the economic evaluation could not be identified.1,4 The low acquisition cost of these treatments may have a meaningful impact on the results of the economic evaluation, as they could make the results less favourable to IDP-126. While oral antibiotics were favoured over IDP-126 gel, the credible intervals for combinations of dual-drug fixed-dose topical treatments with oral antibiotics included the null.
Finally, clinical expert feedback indicated that combinations of different topical treatments were also relevant comparators, though this can be associated with limitations such as compliance concerns and potential incompatibility of formulations. The use of multiple topical therapies was not considered in the sponsor’s submitted NMA, so their relative efficacy versus IDP-126 gel is not known. Based on the cost comparison table, sums of combinations of the components of dual therapies and monotherapies used in IDP-126 gel (i.e., clindamycin, BPO, and adapalene) are less costly than the IDP-126 gel fixed-dose combination (ranging from $1.53 per gram for adapalene and BPO [generic] fixed-dose combination plus clindamycin to $2.95 per gram for adapalene [Differin] plus the fixed-dose combination of clindamycin phosphate and BPO), which is lower than or similar to the submitted price for IDP-126 gel ($2.95 per gram). Note that these combinations are not formulated with the exact strengths of the active ingredients used in IDP-126 gel.
CDA-AMC was unable to address this limitation, as these comparators were not included in the NMA or as options in the sponsor’s economic model.
Impact of IDP-126 gel on health-related quality of life is highly uncertain: The sponsor’s base-case analysis predicted an incremental gain of 0.02 QALYs with IDP-126 gel compared with topical antibiotic plus BPO fixed-dose combination over the time horizon of 1.15 years (equivalent to approximately 7 days spent in perfect health over the course of 1.15 years, compared with topical antibiotic plus BPO fixed-dose combinations). As the sponsor’s model valued acne treatments based on lesion reductions, the sponsor was required to map the DLQI to the EQ-5D to derive utility values associated with lesion counts. The resulting health state utility values quantified the expected utility associated with a decrease in 1 lesion (i.e., a reduction in lesion count by 1 will improve utility by 0.0029). This approach is uncertain for several reasons. One, it is unclear whether there is a perfectly linear relationship between number of lesions and patient health-related quality of life. Two, the sponsor’s approach led to implausible utility values at higher lesion counts that required capping. Specifically, for all lesion counts of 71 or greater, utilities were capped at 0.72, which was the utility for patients with moderate to severe acne from the published literature.1 If uncapped, the sponsor’s approach would have led to a utility of 0.67 at the baseline lesion count of 86 and a utility of 0.62 for all patients who switch to BSC after cycle 1, which are utility values similar to having chronic obstructive pulmonary disease and the effects of a stroke, respectively.16 Finally, according to CADTH guidelines for economic evaluations, it is not recommended to use mapping to derive health state utilities.17 Further, the sponsor did not explore results by stratifying by acne severity, which could have different implications on patient quality of life.
CDA-AMC was unable to address this limitation, given the sponsor’s approach to modelling acne relied on lesion counts.
Additionally, the following key assumptions were made by the sponsor and have been appraised by CDA-AMC (Table 4).
Table 4: Key Assumptions of the Submitted Economic Evaluation (Not Noted as Limitations to the Submission)
Sponsor’s key assumption | CDA-AMC comment |
|---|---|
The number of lesions at baseline was assumed to be 86. | Uncertain. Because of the sponsor’s modelling approach, the model is highly sensitive to baseline lesion count. The cost-effectiveness of IDP-126 gel is therefore dependent on lesion count, which is highly uncertain. |
All interventions were assumed to use 1.5 g of medication per application. | Uncertain. The relative amount of product used per application was not evaluated in the clinical trials. |
It was assumed patients will try only a single treatment. If a patient discontinues their initial therapy, they are not permitted to switch to 1 of the other possible alternative active treatments. | Inappropriate. Clinical expert feedback obtained by CDA-AMC indicated that while there is some uncertainty regarding the extent of treatment switching, it is unlikely that patients will be prescribed only a single therapy over the modelled time horizon. |
It was assumed the risk of discontinuation was restricted to the first 12 weeks of treatment. | Inappropriate. Clinical expert feedback obtainedby CDA-AMC indicated that patients would remain at risk for adverse events, a loss of efficacy, or nonadherence for as long as they remain on treatment. It was also noted that these discontinuation risks may also depend on the amount of time spent on treatment, which was not modelled. |
When patients who discontinue treatment in the first cycle complete their first cycle on BSC, it was assumed that their acne will always get worse (a 19-unit increase in lesion count from their baseline lesion count). | Inappropriate. Clinical expert feedback obtainedby CDA-AMC suggested that no change, an increase, or a decrease in lesion count are all possible outcomes following discontinuation of active treatment. The sponsor’s model was not sufficiently flexible to consider multiple potential outcomes among those who discontinue treatment and receive only BSC (i.e., the sponsor’s model only allowed those who discontinue and receive BSC to either all increase, decrease, or have no change in lesion count). |
It was assumed there is no long-term benefit from any active treatment and patients experience no further change in lesions, even if they used treatment for an indefinite period. | Inappropriate. Clinical expert feedback indicated that, all things being equal, further reductions in lesion count would be expected, as patients continued treatment beyond 12 weeks. There is a lack of indirect comparative data for IDP-126 versus topical treatments beyond 12 weeks. If IDP-126 had a greater reduction in lesion counts beyond 12 weeks versus comparators, this assumption would be conservative. |
Beyond the 19 -lesion increase in lesion count in the following model cycle upon discontinuation, the model assumed no further changes to the lesion count following discontinuation. | Inappropriate. Clinical expert feedback obtained by CDA-AMC indicated that, all things being equal, it is unlikely for these lesion counts to remain unchanged for the duration of the simulated time horizon. |
Time horizon was 1.15 years. | Uncertain, but CDA-AMC determined the time horizon was not influential in cost-effectiveness conclusions. |
Adverse events were not included in the model. | Uncertain. The sponsor’s submitted NMA did not consider safety. As such, the comparative safety of IDP-126 gel versus other active comparators is uncertain. |
All treatments were assumed to have an equal discontinuation rate of 2.9%. | Uncertain. The sponsor’s submitted NMA did not consider discontinuation. As such, the comparative discontinuation rate for IDP-126 gel versus other active comparators is uncertain. |
BSC = best supportive care; CDA-AMC = Canada’s Drug Agency; IDP-126 = clindamycin plus benzoyl peroxide and adapalene; NMA = network meta-analysis.
CDA-AMC Reanalyses of the Economic Evaluation
Base-Case Results
As described earlier, CDA-AMC was unable to address the limitations identified in the sponsor’s submitted analysis, which prevented CDA-AMC from specifying a reanalysis. As such, the cost-effectiveness of IDP-126 is highly uncertain.
Scenario Analysis Results
CDA-AMC conducted a series of scenario analyses to explore the price reductions required to obtain an ICER for IDP-126 gel below a threshold of $50,000 per QALY gained, using the sponsor’s submitted model structure and base-case assumptions. As summarized in Table 9, a 13% price reduction is required for IDP-126 gel to be considered cost-effective in the sponsor’s base case. However, as detailed elsewhere in the report, these results are based on assumptions that are unlikely to hold in actual practice and that overestimate the benefits associated with IDP-126 gel.
Issues for Consideration
Coverage of topical acne treatments varies across drug plans and not all drug plans may cover all topical treatments considered in the sponsor’s analysis.
Overall Conclusions
Based on the CDA-AMC Clinical Review, IDP-126 gel results in a clinically meaningful improvement in acne, when compared with its vehicle gel, in patients aged 10 years and older with moderate to severe acne based on data from Study 301 and Study 302. The sponsor-conducted NMA results showed a favourable treatment effect of IDP-126 gel versus vehicle or placebo, oral antibiotic, and topical monotherapies on treatment success and change in lesion counts. While the effect estimates are suggestive of a possible favourable treatment effect with IDP-126 gel versus topical fixed-dose dual-combination therapies, there is some uncertainty, as the 95% credible intervals included the null or were close to the null for certain dual-combination therapies. However, several notable limitations in the NMA, including the possible exclusion of relevant studies, heterogeneity across trials in the network, and the omission of comparators relevant to the Canadian setting suggest the resulting estimates may be biased. As a result, the Clinical Review concluded that while IDP-126 gel is favoured over other active therapies on treatment success and change in lesion count, the magnitude of benefit associated with IDP-126 gel is uncertain.
CDA-AMC identified several limitations with the submitted economic evaluation that could not be addressed through reanalysis. As a result, the cost-effectiveness of IDP-126 gel relative to active therapies is highly uncertain. According to the sponsor’s base case, IDP-126 gel was associated with the highest total costs and highest total QALYs versus all comparators. As noted in the CDA-AMC Clinical Review Report, while the sponsor’s NMA favoured IDP-126 gel over several alternative active therapies for acne vulgaris, the magnitude of benefit remains uncertain. That, combined with limitations in the sponsor’s model structure and approach to health state utility values, means that the magnitude of the QALY gain associated with IDP-126 gel is highly uncertain and could be overestimated. The sponsor’s analysis predicted that, compared with topical antibiotic plus BPO fixed-dose combination, IDP-126 would lead to a 0.02 QALY gain, which is equivalent to approximately 7 days spent in perfect health over a time horizon of 1.15 years and which could be overestimated. Additionally, in the sponsor’s base case, all of the incremental costs were attributable to treatment acquisition costs, meaning the sponsor is not predicting that IDP-126 gel will result in any cost offsets or reductions in health care resource use. As such, based on public list prices for comparators and the sponsor’s submitted price for IDP-126, there is high certainty that IDP-126 is more costly than comparators, and there is significant unresolved uncertainty regarding the quantity of QALY benefit it is expected to yield. If a decision-maker is willing to accept the assumptions from the sponsor’s submitted base case, including the magnitude of benefit associated with IDP-126 versus comparators, then a 13% price reduction would be required for IDP-126 gel to be considered cost-effective. However, given the limitations identified by CDA-AMC that could not be addressed, a higher price reduction is likely required for IDP-126 to be considered cost-effective. Finally, public drug plan coverage of many of the comparator treatments included in the analysis is variable. Based on current list prices of comparators and the sponsor’s submitted price for IDP-126, IDP-126 is more expensive than all currently listed treatments, apart from adapalene 0.3%, which is only reimbursed by 1 drug plan that participates in the CDA-AMC review process.
References
1.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Cabtreo (IDP-126: clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15%), topical gel in 50-gram pumps. Laval (PC): Bausch Health, Canada Inc; 2024 Mar 8.
2.Cabtreo (Clindamycin Phosphate, Adapalene and Benzoyl Peroxide 1.2% w/w, 0.15% w/w and 3.1% w/w) Topical Gel [draft product monograph]. Laval (QC): Bausch Health Canada, Inc; 2023 Aug 28.
3.Stein Gold L, Lain E, Del Rosso JQ, et al. Clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel for moderate-to-severe acne: Efficacy and safety results from two randomized phase 3 trials. J Am Acad Dermatol. 2023;89(5):927-935. PubMed
4.Network meta-analysis: Comparative effectiveness of IDP-126 gel for patients with moderate-to-severe acne vulgaris [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Cabtreo (IDP-126: clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15%), topical gel in 50 gram pumps. Laval (QC): Bausch Health, Canada Inc; 2024 Mar 8.
5.Guertin JR, Feeny D, Tarride JE. Age- and sex-specific Canadian utility norms, based on the 2013-2014 Canadian Community Health Survey. CMAJ. 2018;190(6):E155-E161. PubMed
6.Klassen AF, Newton JN, Mallon E. Measuring quality of life in people referred for specialist care of acne: comparing generic and disease-specific measures. J Am Acad Dermatol. 2000;43(2 Pt 1):229-233. PubMed
7.Wang AS, Wu J, Tuong W, Schupp C, Armstrong AW. Effectiveness of a novel interactive health care education tool on clinical outcomes and quality of life in acne patients: A randomized controlled pilot study. J Dermatolog Treat. 2015;26(5):435-439. PubMed
8.Yang M, Moclair B, Hatcher V, et al. A randomized, double-blind, placebo-controlled study of a novel pantothenic Acid-based dietary supplement in subjects with mild to moderate facial acne. Dermatol Ther (Heidelb). 2014;4(1):93-101. PubMed
9.Oprica C, Emtestam L, Hagstromer L, Nord CE. Clinical and microbiological comparisons of isotretinoin vs. tetracycline in acne vulgaris. Acta Derm Venereol. 2007;87(3):246-254. PubMed
10.Xu JH, Lu QJ, Huang JH, et al. A multicentre, randomized, single-blind comparison of topical clindamycin 1%/benzoyl peroxide 5% once-daily gel versus clindamycin 1% twice-daily gel in the treatment of mild to moderate acne vulgaris in Chinese patients. J Eur Acad Dermatol Venereol. 2016;30(7):1176-1182. PubMed
11.Xu X, Zheng Y, Zhao Z, Zhang X, Liu P, Li C. Efficacy of photodynamic therapy combined with minocycline for treatment of moderate to severe facial acne vulgaris and influence on quality of life. Medicine (Baltimore). 2017;96(51):e9366. PubMed
12.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2024; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2024 Apr 4.
13.PharmaStat. Ottawa (ON): IQVIA; 2023: https://www.iqvia.com/. Accessed 2024 Apr 4.
14.Schedule of benefits for physician services under the Health Insurance Act: (June 29, 2023 (effective July 24, 2023)). Toronto (ON): Ontario Ministry of Health; 2023: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master.pdf. Accessed 2023 Jun 29.
15.Guidelines for the economic evaluation of health technologies: Canada. 4th ed. Ottawa (ON): CADTH; 2017: https://www.cadth.ca/guidelines-economic-evaluation-health-technologies-canada-4th-edition. Accessed 2024 May 09.
16.Statistics Canada. Utility score norms for each of the chronic conditions examined in the 2013 and 2014 Canadian Community Health Survey, household population aged 12 and older, Canada, 2013 and 2014. 2018; https://www150.statcan.gc.ca/n1/pub/82-003-x/2018011/article/00001/tbl/tbl03-eng.htm. Accessed 2024 Jun 19.
17.Guidelines for the Economic Evaluation of Health Technologies: Canada — 4th Edition [accessed by sponsor]. Ottawa (ON): CADTH; 2017: https://www.cadth.ca/guidelines-economic-evaluation-health-technologies-canada-4th-edition.
18.Saskatchewan Drug Plan: search formulary. 2024; https://formulary.drugplan.ehealthsask.ca/SearchFormulary. Accessed 2024 Apr 4.
19.DeltaPA. Ottawa (ON): IQVIA; 2023: https://www.iqvia.com/. Accessed 2024 May 29.
20.BC Government. BC PharmaCare formulary search. 2024; https://pharmacareformularysearch.gov.bc.ca. Accessed 2024 Jun 1.
21.Beleznay K. Acne. Ottawa (ON): Canadian Pharmacists Association; 2022: https://cps.pharmacists.ca. Accessed 2024 Jun 19.
22.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Cabtreo (IDP-126: clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15%), topical gel in 50 gram pumps. Laval (QC): Bausch Health, Canada Inc; 2024 Mar 8.
Appendix 1: Cost Comparison Table
Please note that this appendix has not been copy-edited.
The comparators presented in Table 5 have been deemed to be appropriate based on feedback from clinical expert(s). Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in Table 5, and as such, the table may not represent the actual costs to public drug plans.
Table 5: CDA-AMC Cost Comparison Table for the Topical Treatment of Acne Vulgaris
Treatment | Strength/ | Dosage form | Package form | Price | Recommended dosage | Daily cost ($) | Annual cost ($) |
|---|---|---|---|---|---|---|---|
Clindamycin phosphate + benzoyl peroxide and adapalene (Cabtreo) | 1.2% w/w clindamycin; | Gel | 50 g | 2.9483a | Apply a thin layer of to the affected area once daily | 4.42 | 1,616 |
Topical retinoid monotherapy | |||||||
Tazarotene (Arazlo) | 0.045% w/w | Lotion | 45 g | 1.4051 | Apply a thin layer once daily | 2.11 | 770 |
Adapalene (Differin) | 0.1% w/w | Cream, Lotion, Gel | 60 g | 2.2643b | Apply once daily | 3.40 | 1,240 |
Adapalene (Differin XP) | 0.3% w/w | Gel | 60 g | 3.2473c | Apply once daily | 4.87 | 1,778 |
Tretinoin (Generic) | 0.01% w/w | Gel | 50 g | 0.3788b | Apply daily | 0.57 | 207 |
Tretinoin | 0.025% w/w 0.05% | Cream, gel | 30 g | 0.5090b | Apply daily | 0.76 | 279 |
Tretinoin | 0.04% w/w 0.1% w/w | 50 g gel | 50 g | 1.1495c 1.6139d | Apply once daily | 2.24 | 819 |
Topical antibiotic monotherapy | |||||||
Clindamycin (Generic) | 0.1% w/v | Topical solution | 30 mL 60 mL | 0.2536b | Apply a thin film twice daily | 0.38 | 139 |
Topical antibiotic plus retinoid fixed-dose combinations | |||||||
Clindamycin Phosphate + Tretinoin | 1.2% w/w Clindamycin Phosphate; 0.025% w/w tretinoin | Gel | 60 g | 1.2768c | Apply once daily | 1.92 | 700 |
Topical retinoid plus benzoyl peroxide fixed-dose combinations | |||||||
Adapalene and benzoyl peroxide | 0.1% w/w adapalene plus 2.5% benzoyl peroxide | Gel | 70 g | 1.6669b | Apply once daily | 2.50 | 913 |
Adapalene and benzoyl peroxide (Tactupump Forte) | 0.3% adapalene plus 2.5% benzoyl peroxide | Gel | 70 g | 2.7179b | Apply once daily | 4.08 | 1,488 |
Adapalene and benzoyl peroxide (Generic) | 0.1% w/w adapalene | Gel | 70 g | 1.2718 | Apply once daily | 1.91 | 696 |
Adapalene and peroxide (generic) | 0.3% w/w adapalene | Gel | 70 g | 2.1577 | Apply once daily | 3.24 | 1,181 |
Topical antibiotic plus benzoyl peroxide fixed-dose combinations | |||||||
Erythromycin and benzoyl peroxide (Benzamycin) | 3% w/w erythromycin | Gel | 46.6 g | 1.4129b | Apply twice daily | 4.24 | 1,547 |
Clindamycin phosphate and benzoyl peroxide (Benzaclin, generics) | 1% w/w Clindamycin | Gel | 50 g | 0.7422 | Apply twice daily | 2.23 | 813 |
Clindamycin phosphate and benzoyl peroxide (Clindoxyl, generics) | 1% w/w Clindamycin | Gel | 45 g | 0.6857 | Apply once daily | 1.03 | 376 |
Clindamycin phosphate and benzoyl peroxide (Clindoxyl adv) | 1% w/w Clindamycin | Gel | 45 g | 0.8085 | Apply once daily | 1.21 | 443 |
w/w = grams in 100 g of solution.
Note: All prices are from the Ontario Drug Benefit Formulary (accessed in April 2024),12 unless otherwise indicated, and do not include dispensing fees. Costs in this table assume a year has 365 days and 100% adherence, and all topical comparator costs assume the use of 1.5 g per application.
aSponsor-submitted price.1
bPrice obtained from the Saskatchewan Drug Benefit Formulary (accessed in June 2024).18
cIQVIA DeltaPA wholesale price (accessed in May 2024).19
dPrice obtained from the BC Pharmacare Formulary (accessed in June 2024).20
Table 6: CDA-AMC Cost Comparison Table for the Oral Treatment of Acne Vulgaris
Treatment | Strength | Form | Price ($) | Recommended dosage | Daily cost ($) | Annual cost |
|---|---|---|---|---|---|---|
Hormone therapies (oral contraceptives) | ||||||
Cyproterone acetate plus ethinyl estradiol (Diane-35, various) | 2.0 mg plus 0.035 mg | 21 tablets | 43.8501c | One active tablet daily for 21 days, then 7 days off | 1.57 | 572 |
Desogestrel plus ethinyl estradiol (Marvelon, various) | 0.15 mg plus 0.03 mg | 21 tablets | 7.7700 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.28 | 101 |
Desogestrel plus ethinyl estradiol | Triphasic | 21 tablets | 19.1196 19.1247 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.68 | 249 |
Drospirenone plus ethinyl estradiol (Yasmin, various) | 3.0 mg plus 0.03 mg | 21 tablets | 6.2181 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.22 | 81 |
Drospirenone plus ethinyl estradiol | 3.0 mg plus 0.02 mg | 28 tablets | 8.2600 | One tablet daily for 24 days, then 4 days of inert tablets | 0.30 | 108 |
Levonorgestrel plus ethinyl estradiol | 0.1 mg / 0.02 mg | 21 tablets | 3.9425 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.14 | 51 |
Levonorgestrel plus ethinyl estradiol | 0.15 mg / 0.03 mg | 21 tablets | 7.2800 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.26 | 95 |
Levonorgestrel plus ethinyl estradiol | Triphasic | 21 tablets | 15.7500 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.56 | 205 |
Norgestimate / ethinyl estradiol (Tri-Cira, various) | Triphasic | 21 tablets | 14.3900 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.51 | 188 |
Norgestimate plus ethinyl estradiol (Tricira Lo, various) | Triphasic | 21 tablets | 13.3900 | One tablet daily for 21 days, then 7 days off or 7 days of inert tablets | 0.48 | 175 |
Hormone therapy (spironolactone) | ||||||
Spironolactone (generics) | 25 mg | Tablet | 0.0405 0.0955 | 25 mg to 200 mg daily | 0.04 to 0.19 | 15 to 70 |
Oral antibiotics | ||||||
Minocycline | 50 mg 100 mg | Capsule | 0.5616 1.0836 | Initial: 100 mg daily Maintenance: 50 mg to 200 mg dailye | 0.56 to 2.17 | 205 to 791 |
Doxycycline hyclate | 100 mg | Tablet | 0.4560 | Initial: 100 mg daily Maintenance: 100 mg to 200 mg dailye | 0.46 to 0.91 | 166 to 333 |
Doxycycline monohydrate | 40 mg | Capsule | 2.4277 | 40 mg dailye | 2.43 | 886 |
w/w = grams in 100 g of solution.
Note: All prices are from the Ontario Drug Benefit Formulary (accessed April 2024),12 unless otherwise indicated, and do not include dispensing fees. Costs in this table assume a year has 365 days and 100% adherence, and all topical comparator costs assume the use of 1.5 g per application.
aSponsor-submitted price.1
bPrice obtained from the Saskatchewan Drug Benefit Formulary (accessed in June 2024).18
cIQVIA DeltaPA wholesale price (accessed in May 2024).19
dPrice obtained from the BC Pharmacare Formulary (accessed in June 2024).20
eDosing from e-Therapeutics.21
Appendix 2: Submission Quality
Please note that this appendix has not been copy-edited.
Description | Yes or no | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing. | Yes | No comment. |
Model has been adequately programmed and has sufficient face validity. | No | The model does not reasonably reflect the disease area and current management of acne. Refer to limitation for additional details. |
Model structure is adequate for decision problem. | No | The model does not reasonably reflect the disease area and current management of acne vulgaris. Refer to limitation for additional details. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis). | Yes | No comment. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem. | No | The model does not reasonably reflect the disease area and current management of acne vulgaris. As a result, the characterization of uncertainty in the parameters or structural assumptions cannot provide meaningful information. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details). | Yes | No comment. |
Appendix 3: Additional Information on the Submitted Economic Evaluation
Please note that this appendix has not been copy-edited.
Detailed Results of the Sponsor’s Base Case
Table 8: Disaggregated Summary of the Sponsor’s Economic Evaluation Results
Parameter | IDP-126 | Topical retinoid monotherapy | Topical antibiotic monotherapy | Topical antibiotic / retinoid fixed-dose combinations | Topical retinoid / BPO fixed-dose combinations | Topical antibiotic / BPO fixed-dose combinations |
|---|---|---|---|---|---|---|
Discounted QALYs | ||||||
Total | 0.961 | 0.931 | 0.923 | 0.939 | 0.943 | 0.943 |
By health state | ||||||
Active Treatment | 0.936 | 0.907 | 0.898 | 0.914 | 0.919 | 0.918 |
BSC | 0.025 | 0.025 | 0.025 | 0.025 | 0.025 | 0.025 |
Discounted costs ($) | ||||||
Total | 2,700 | 1,887 | 1,221 | 1,651 | 1,795 | 1,567 |
Acquisition | 1,798 | 984 | 319 | 749 | 892 | 665 |
Monitoring | 902 | 902 | 902 | 902 | 902 | 902 |
BPO = benzoyl peroxide; BSC = best supportive care; IDP-126 = clindamycin plus benzoyl peroxide and adapalene; QALY = quality-adjusted life-year.
Source: Sponsor’s pharmacoeconomic submission.1
Figure 2: Scatterplot for IDP-126 Versus Topical Antibiotic Plus BPO Fixed-Dose Combinations
BPO = benzoyl peroxide; IDP-126 = clindamycin plus benzoyl peroxide and adapalene; QALY = quality-adjusted life-year.
Source: Sponsor’s pharmacoeconomic submission.1
Table 9: CDA-AMC Price Reduction Analyses
Analysis | Unit drug cost ($) | ICERs for IDP-126 gel ($/QALY) |
|---|---|---|
No price reduction | 147 | 62,967a |
10% | 133 | 52,974a |
13% | 128 | 49,976a |
20% | 118 | 42,981a |
30% | 103 | 32,988a |
40% | 88 | 22,994a |
50% | 74 | 15,326b |
60% | 59 | 10,575b |
70% | 44 | 5,824b |
80% | 29 | 1,073b |
90% | 15 | IDP-126 gel dominant |
BPO = benzoyl peroxide; CDA-AMC = Canada’s Drug Agency; IDP-126 = clindamycin plus benzoyl peroxide and adapalene; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
aVersus topical antibiotic plus BPO fixed-dose combinations.
bVersus topical antibiotic monotherapy.
Appendix 4: Additional Details on the CDA-AMC Reanalyses and Sensitivity Analyses of the Economic Evaluation
Please note that this appendix has not been copy-edited.
Given the limitations of the submitted economic model, CDA-AMC was unable to conduct any additional analyses to assess the relative cost-effectiveness of IDP-126 for the treatment of acne vulgaris.
Appendix 5: Submitted Budget Impact Analysis and CDA-AMC Appraisal
Please note that this appendix has not been copy-edited.
Table 10: Summary of Key Take-Aways
Key take-aways of the budget impact analysis |
|---|
|
Summary of Sponsor’s Budget Impact Analysis
The submitted budget impact analysis (BIA) assessed the expected budget impact of reimbursing IDP-126 gel for the topical treatment of acne vulgaris in patients aged 12 years and older. The BIA was undertaken from the perspective of public drug plans (excluding Quebec) in Canada over a 3-year time horizon. A claims-based approach was used to estimate the eligible number of patients in each year of the analysis. Key inputs to the BIA are documented in Table 11.
In the reference scenario, it was assumed that all patients eligible for treatment would receive 1 of the existing topical treatments. In the new drug scenario, it was assumed that IDP-126 gel would displace market share from the existing alternatives.
Key assumptions:
The annual expenditure for each drug product was calculated as a product of the unit price and number of units predicted to be dispensed in each year of the BIA time horizon. These predictions were made using a linear regression model fitted to historical claims data. The sponsor acquired the claims data from January 2019 to September 2023 from the IQVIA Pharmastat database. Separate regression models were fitted for each treatment and province, to explore the relationship between claims and quarter of the calendar year.
Historical claims data were represented in grams, and, by extension, the predicted future consumption of each treatment was also measured in grams. It was therefore necessary to convert the predicted values to “treatments.” This was defined as the amount of topical therapy needed to treat 1 patient over 30 days. The sponsor assumed 1.5 g per application, while the number of applications per day followed the directions specified in the respective product monographs. To calculate the number of units to be dispensed, the sponsor divided the total predicted grams per treatment by the estimated grams per unit.
In the new drug scenario, it was assumed that IDP-126 gel would have a market share of 5% in year 1, 8% in year 2, and 12% in year 3. These estimates were obtained from the sponsor’s internal market research analysis. These adjustments were applied to the total number of units to be dispensed across all comparators.
Table 11: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as year 1 / year 2 / year 3, if appropriate) |
|---|---|
Target population | |
Total predicted claims | 102,874 / 104,759 / 107,156 |
Market uptake (3 years) | |
Uptake (reference scenario) | Refer to Table 12 |
Uptake (new drug scenario) | |
IDP-126 gel | 5% / 8% / 12% |
Comparators | IDP-126 gel assumed to capture market share from all comparators proportionately |
Cost of treatment (per patient, per 30 days) | |
IDP-126 gel | $132.6735 |
Comparators | Refer to Table 12 |
IDP-126 = clindamycin plus benzoyl peroxide and adapalene.
Table 12: Reference Scenario Market Shares
Comparator | Strength | Year 1 | Year 2 | Year 3 | Cost of treatment (per patient, per 30 days) |
|---|---|---|---|---|---|
Retin-a | 0.05% | 1% | 2% | 2% | $23.2785 |
Retin-a | 0.025% | 1% | 1% | 1% | $23.2785 |
Differin | 0.1% | 3% | 3% | 4% | $124.5240 |
Benzamycin | 3/5% | 0% | 0% | 0% | $129.2670 |
Differin | 0.1% | 3% | 3% | 2% | $124.5240 |
Clindoxyl | 5/1% | 10% | 11% | 12% | $30.8565 |
Retin-a micro | 0.1% | 0% | 0% | 0% | $64.6650 |
Benzaclin | 5%/1% | 16% | 17% | 18% | $66.7980 |
Taro-clindamycin | 1% | 7% | 8% | 8% | $23.5170 |
Differin xp | 0.3% | 0% | 0% | 0% | $139.1715 |
Biacna | 1.2%/0.025% | 1% | 1% | 1% | $55.2465 |
TactuPump | 2.5%/0.1% | 0% | 0% | 0% | $81.0045 |
Clindoxyl adv | 3/1% | 1% | 1% | 1% | $36.3780 |
Taro-clinda/benzoyl per | 5%/1% | 27% | 21% | 15% | $30.8565 |
Tactupump forte | 2.5%/0.3% | 0% | 0% | 0% | $132.0930 |
Taro-adapalene/benzoyl | 2.5%/0.1% | 5% | 5% | 6% | $57.2355 |
Taro benzoyl/clinda kit | 5%/1% | 2% | 0% | 0% | $66.7980 |
Sandoz adapalene/benzoyl | 2.5%/0.1% | 6% | 7% | 7% | $57.2310 |
Taro-adapalene/benzyl ft | 2.5%/0.3% | 1% | 2% | 2% | $97.0965 |
Sandoz adapalene/benz ft | 2.5%/0.3% | 1% | 1% | 1% | $97.0929 |
Arazlo | 0.045% | 14% | 17% | 20% | $63.2300 |
BIA = budget impact analysis.
Note: Represents national market share estimates. Calculated the using sponsor’s submitted BIA model by dividing individual product units by total units. While some additional comparators were included in the sponsor’s claims-based approach, all comparators with 0% market shares across all 3 years were not included in the table.
Source: Sponsor’s BIA submission.22
Summary of the Sponsor’s BIA Results
The net budget impact of IDP-126 gel was $444,986 in year 1, $712,533 in year 2, and $1,072,908 in year 3. The 3-year net budget impact of IDP-126 gel was estimated to be $2,230,428.
CDA-AMC Appraisal of the Sponsor’s BIA
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Use of a claims-based approach to estimate market size introduces uncertainty with the anticipated budget impact of IDP-126 gel: The sponsor estimated market size based on public claims data for the relevant comparators. The sponsor assumed that all public claims data for the comparator drugs are for the indication of interest. Given that the claims database does not specify the indication and the proportion of claims pertaining to use for other indications is unknown, using a claims-based approach to estimate market size introduces significant uncertainty in the estimated market size. Furthermore, the sponsor did not convert the claims data into the number of users. Instead, the sponsor sought to predict the total number of dispensations that would be covered by public drug plans. This is unlikely to have a significant impact on the results. However, for transparency and completeness, claims-based BIAs should provide an estimate of the number of active beneficiaries converted from the number of claims.
CDA-AMC was unable to address this limitation.
Uncertainty regarding market shares of IDP-126 gel: The sponsor’s base case assumed that 5%, 8%, and 12% of eligible patients would receive IDP-126 gel in year 1, year 2, and year 3, respectively, based on the sponsor’s market research. According to clinical exert feedback obtained for this review, if a triple therapy were reimbursed, it would be expected to become widely adopted. The main reasons cited by the clinical expert as to why patients may not uptake IDP-126 gel should it be covered were concerns with adherence or previous use of topicals. As such, the market penetrance of IDP-126 gel is uncertain.
CDA-AMC was unable to address this limitation.
CDA-AMC Reanalyses of the BIA
In the absence of more reliable estimates to inform the parameters of the BIA, the sponsor’s base case was maintained. CDA-AMC expects that the budget impact of IDP-126 gel will be sensitive to more reliable inputs, which may affect the market size calculation.
Table 13: Detailed Breakdown of the CDA-AMC Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 (current situation) ($) | Year 1 ($) | Year 2 ($) | Year 3 ($) | Three-year total ($) |
|---|---|---|---|---|---|---|
Submitted base case | Reference | 6,205,518 | 7,155,322 | 7,442,603 | 7,782,393 | 28,585,836 |
New drug | 6,205,518 | 7,600,308 | 8,155,136 | 8,855,301 | 30,816,263 | |
Budget impact | 0 | 444,986 | 712,533 | 1,072,908 | 2,230,428 |
BIA = budget impact analysis; CDA-AMC = Canada’s Drug Agency.
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.
 convergence diagnostic was used to compare between-chain and within-chain estimates for model parameters and other univariate quantities of interest.
convergence diagnostic was used to compare between-chain and within-chain estimates for model parameters and other univariate quantities of interest.