Drugs, Health Technologies, Health Systems
Reimbursement Review
Risperidone Long-Acting Injection (Okedi)
Sponsor: Bausch Health, Canada Inc.
Therapeutic area: Schizophrenia, adults
Abbreviations
ANCOVA
analysis of covariance
AE
adverse event
AUC
area under the curve
AUCtau
area under the curve during the dosing interval
BIA
budget impact analysis
BMI
body mass index
CCEIP
Canadian Consortium for Early Intervention in Psychosis
CDA-AMC
Canada’s Drug Agency
CGI-I
Clinical Global Impression – Improvement
CGI-S
Clinical Global Impression – Severity
CHW
Cui, Hung, Wang
CI
confidence interval
C-SSRS
Columbia-Suicide Severity Rating Scale
DB
double blind
DSM-5
Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition)
HEOR
health economics and outcomes research
HRQoL
health-related quality of life
IAM
Institute for Advancements in Mental Health
IM
intramuscular
ISM
in situ microparticle
ITT
intention to treat
IWRS
Interactive Web Response System
LAI
long-acting injection
LH
Lawrence and Hung
LS
least squares
MID
minimally important difference
mITT
modified intention to treat
MMRM
mixed effect model with repeated measurements
mPP
modified per protocol
mSAF
modified safety population
OLE
open-label extension
OLP
open-label population
ORR
overall response rate
PANSS
Positive and Negative Syndrome Scale
PK
pharmacokinetic
PP
per protocol
RAI-MH
Resident Assessment Instrument-Mental Health
SAE
serious adverse event
SAF
safety population
SD
standard deviation
SWN-20
Subjective Well-Being Under Neuroleptics – Short Form
TEAE
treatment-emergent adverse event
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Table 1: Background Information of Application Submitted for Review
Item | Description |
|---|---|
Drug product | Risperidone long-acting injection (Okedi),a 75 mg or 100 mg, powder for extended-release suspension, intramuscular injection |
Sponsor | Bausch Health, Canada Inc. |
Indication | The treatment of schizophrenia in adults |
Reimbursement request | As per the indication |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | March 21, 2024 |
Recommended dose | 75 mg or 100 mg once q.4.w. by IM injection |
IM = intramuscular; ISM = in situ microparticle; NOC = Notice of Compliance; q.4.w. = every 4 weeks.
aThe term risperidone ISM is used for Okedi throughout this review report.
Sources: Sponsor’s Application Overview,1 Okedi Product Monograph.2
Introduction
Schizophrenia is a complex and chronic mental health condition involving a broad range of symptoms and fluctuating symptom severity over time.3,4 Symptoms of schizophrenia are grouped into domains. The positive domain involves symptoms of reality distortion (hallucinations and delusions, including impaired insight into the illness) and disorganized thoughts and behaviours.5 The negative symptoms impact emotional expression and motivation and include reduction in spontaneous speech, diminished functioning, and social withdrawal.3,5 Cognitive symptoms may include impaired attention, memory, and verbal comprehension.5 Affective symptoms, such as depression and anxiety, are also often experienced by people with schizophrenia.5,6 The course of illness in schizophrenia is heterogeneous and influenced by various factors.5 The onset of schizophrenia is often characterized by a prodromal phase with negative symptoms that precedes the initial episode of psychosis.5,7,8 The acute phase, characterized more by positive symptoms, typically follows.8 Positive symptoms decrease over time and are replaced by negative symptoms, which are most prevalent during the residual phase.7,8 Breakthrough psychotic episodes (relapses), which are experienced by most patients during the course of their illness, can be triggered by nonadherence to therapy, substance use, or stress.9 Schizophrenia significantly affects patients’ quality of life as a result of its impacts on physical, mental, and social well-being.10,11 Schizophrenia is characterized by increased mortality, compared to the general population, with the predominant causes comorbid illnesses (e.g., cardiovascular disease) and death due to unnatural causes, including suicide.5,7,12,13 In 2016 to 2017, the all cause-mortality rate in Canada for individuals with schizophrenia was reported to be 2.8 times higher than for those without the disorder.14 In 2022 to 2023, the prevalence of schizophrenia in people in Canada aged 10 years and older was approximately 1% and the incidence rate in the same population was estimated to be 53 cases per 100,000.15
The treatment of schizophrenia is multimodal and includes antipsychotic medications, which are classified as first-generation antipsychotics or second-generation antipsychotics (which are also known as atypical antipsychotics).16 Second-generation antipsychotics are less likely than first-generation antipsychotics to cause extrapyramidal symptoms.17 Clozapine (a second-generation antipsychotic) is more effective than other antipsychotic medications in the treatment of schizophrenia but has significant side effects and health risks that require careful monitoring.17-19 According to the clinical expert consulted by Canada’s Drug Agency (CDA-AMC) for this review, the choice of antipsychotic medication is mainly determined by side-effect profile, patient preference, and availability. The clinical expert noted that an atypical antipsychotic medication is usually prescribed as initial therapy because this class carries lower risk of side effects. Clinical practice guidelines in Canada include the use of long-acting injection (LAI) antipsychotics as a strategy to address nonadherence and note the potential place for LAIs earlier in the course of treatment and not only for patients for whom suboptimal adherence is a concern. The guidelines also state that clozapine should be offered to patients with schizophrenia that meets the criteria for treatment resistance.20 According to the clinical expert, LAIs are considered a first-line option in the treatment of schizophrenia, including during an acute exacerbation or as maintenance therapy. The clinical expert also commented that, for the minority of patients with schizophrenia who have good or partial insight into their illness and are adherent to an oral antipsychotic, either an oral or an injectable treatment may be appropriate. Psychosocial interventions, such as specialty care programs, cognitive behavioural therapy, support for the development of self-management skills, family interventions, psychoeducation, access to safe and affordable housing, and supportive employment services, are also recommended as components of effective treatment for patients with schizophrenia.21-23
The clinical expert stated that, in the treatment of schizophrenia, there is an unmet need for treatments that improve function and quality of life as well as negative and cognitive symptoms and that have minimal (or no) adverse effects.
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects, as well as the costs, of risperidone LAI (Okedi), 75 mg or 100 mg powder for extended-release suspension, intramuscular (IM) injection, in the treatment of schizophrenia in adults.
Risperidone LAI (Okedi, also referred to as risperidone in situ microparticle [ISM]) has not been previously reviewed by CDA-AMC. Risperidone tablets for oral administration have been approved in Canada since 1993,24,25 and risperidone LAI for IM administration every 2 weeks (Risperdal Consta) has been approved in Canada since 2004;26 both are used in clinical practice. Risperidone for extended-release injectable suspension for subcutaneous administration once monthly (Perseris) was reviewed by CADTH for the indication of treatment of schizophrenia in adults and received a recommendation to “reimburse with clinical criteria and/or conditions” (September 24, 2021).27,28 According to the Health Canada Drug Product Database, the current status of Perseris is “cancelled post market.”29 Another risperidone product, a monthly subcutaneous risperidone extended-release formulation (Longavo), reviewed by CDA-AMC for the treatment of schizophrenia in adults received a recommendation to “reimburse with conditions” (June 25, 2025).30
Perspectives of Patients, Clinicians, and Drug Programs
The information in this section is a summary of input provided by the patient and clinician groups who responded to our call for input and from the clinical expert consulted by for the purpose of this review.
Patient Input
No patient input was received for this review. Patient input received for a previous CADTH review (November 2021) on risperidone for extended-release injectable suspension (SR0671 Perseris) for the treatment of schizophrenia in adults is included in this report.31 As such, the available input may not accurately reflect patients’ current unmet needs.
Patient group input was provided by the Institute for Advancements in Mental Health (IAM). The IAM submission was informed by IAM’s 40-year history of serving adults with schizophrenia and also draws on information from a 2018 survey of members of IAM’s client network. The survey respondents self-described as personally diagnosed (12%), caregivers (50%), family or friends of someone with a diagnosis (63%), or working in social services (18%).
The survey respondents indicated that many patients experience symptoms of psychosis, which have a significant impact on day-to-day functioning. Patient experiences vary widely but typically involve some levels of cognitive impairment, delusions, and hallucinations. Many patients also experience a lack of insight into their illness, which often impacts their ability and motivation to access treatment and supports. This symptom can cause significant strain on relationships, including those with caregivers and family members, ultimately leading to social isolation and a lack of supports for the individual with the illness.
Patients indicated that the most common side effects of antipsychotic drugs were drowsiness (reported by 58%), dry mouth (50%), restlessness (50%), dizziness (45%), muscle stiffness (45%), constipation (43%), and anxiety (43%).
Twenty-three percent of patients identified the cost of medications as a significant barrier to access; 63% of respondents indicated that it is difficult to pay for health care bills, including for medication, visits to specialists, counselling, and so forth; 20% of patients indicated that not having their preferred medication covered by public drug programs is a challenge.
Patients expect new, quick, simple, convenient, and affordable access to a wide range of treatments and medications that suit their unique needs, can improve adherence, and allow for the highest level of daily functioning and symptom reduction while managing side effects.
Clinician Input
Input From the Clinical Expert Consulted for This Review
The clinical expert stated that there remains an unmet need for treatments for schizophrenia that improve function and quality of life as well as negative and cognitive symptoms and have minimal (or no) adverse effects.
The clinical expert stated that risperidone ISM (Okedi) would be suitable for patients who are experiencing an acute exacerbation of schizophrenia and those who may benefit from transitioning from a stable oral risperidone dose to an LAI. Given that there are currently available atypical LAI antipsychotics that can be administered less frequently than risperidone ISM (i.e., up to every 3 months), the clinical expert expected that risperidone ISM would be used for patients who are already receiving an LAI and are experiencing an inadequate response but who are not willing to take clozapine. The clinical expert also expected risperidone ISM may be used in the treatment of patients with schizophrenia that is responding well to oral risperidone but for whom an LAI would be preferable. The clinical expert noted that participants in the PRISMA-3 pivotal trials included patients who were known to have previously experienced a clinically significant response to an antipsychotic and that the overall population in the trials was healthier than patients in clinical practice. The clinical expert acknowledged the dosing information from the Okedi product monograph,2 noting that risperidone ISM would not be suitable for patients receiving oral risperidone doses greater than 4 mg/day (or its equivalent).
According to the clinical expert, the outcome measures used in the PRISMA-3 pivotal trials are not typically used by physicians in clinical practice. The clinical expert noted that the Brief Psychiatric Rating Scale (BPRS) and Clinical Global Impression – Improvement (CGI-I) tools may be used in certain clinical settings and many hospitals use the Resident Assessment Instrument-Mental Health (RAI-MH) version 2.032 tool to evaluate progress; however, others may assess progress based on clinical interviews rather than an assessment scale. The clinical expert commented that the definition of a clinically meaningful response to treatment can vary among physicians and patients and that the following would all be considered important responses to treatment: reduction in the frequency or severity of symptoms, improvement in symptoms, stabilization of symptoms, and the ability to perform activities of daily living. According to the clinical expert, in inpatient settings patients receive frequent monitoring for treatment response, whereas in outpatient settings, patients initiating treatment with an LAI antipsychotic would initially be assessed in person every 2 weeks. Once a patient is stable, assessments by a psychiatrist may occur every 1 to 3 months.
The clinical expert commented that there are no specific parameters to define loss of response, absence of clinical benefit, and disease progression in clinical practice; these would be determined on an individual basis, typically based on the patient’s symptoms, ability to function, and other factors.
The clinical expert stated that LAI antipsychotics are initially prescribed and monitored by psychiatrists (and in some cases family physicians with specialized practices). Once a patient with schizophrenia has stable disease, they may continue to see a psychiatrist or may be followed by a family physician and receive injections in the family practice setting.
Clinician Group Input
Two clinicians from the Canadian Consortium for Early Intervention in Psychosis (CCEIP) provided input for this review, and information was gathered from a literature review of risperidone ISM, comparison of risperidone LAI formulations, data on the use of long-acting antipsychotic injections in schizophrenia and first 5 years of illness, clinical experience, and a conference presentation by the author of the CCEIP submission.
CCEIP also noted that atypical LAI antipsychotic medications all require some type of titration or loading protocol to ensure therapeutic levels are achieved as quickly as possible, highlighting that early attainment of therapeutic levels is correlated with more positive outcomes and better persistence with treatment. According to the clinician group, the desired outcomes of early intervention during the critical period are to improve the course of psychosis and lead to a period of stability; return to pre-illness social and occupational levels of functioning; have a better outcome compared with the intervention after the critical period; and decrease the risk of suicide.
CCEIP commented that risperidone ISM allows for achievement of therapeutic levels within 24 hours of administration and that it would be recommended to try early and as monotherapy (rather than as a last option or in polypharmacy) for the majority of patients. In selecting the best drug for an individual patient, considerations include achieving a balance between efficacy and tolerability, ease of use, quality of life, functionality, and patient acceptance. According to CCEIP, patients are likely to demonstrate adherence issues at some point during treatment, and it is recommended that LAIs be considered at the appropriate place in therapy per the Canadian guidelines.
Regarding determining response to treatment, CCEIP commented that outcomes in clinical practice are mainly determined by multidisciplinary clinical observations supplemented by reports from patients and caregivers. CCEIP identified differences in clinical trial outcomes (e.g., Positive and Negative Syndrome Scale [PANSS]) and patients compared to those in clinical practice, noting that symptom control and improvement in quality of life and functionality are more relevant goals in clinical practice, and that patients have significant psychiatric and physical comorbidity and experience greater variability in treatment response. CCEIP noted that measures of successful treatment include gaining stability of illness and preventing recurrences and/or relapses and that symptom and functional response to treatment is a priority in a population with early phase psychosis.
CCEIP identified the factors for discontinuing or switching treatment with risperidone ISM as the same as with other treatments, and would include nonresponse or suboptimal response to treatment or intolerability to side effects.
CCEIP stated that the inpatient (hospital) and outpatient (hospital outpatient and community clinic) settings are appropriate for administration of risperidone ISM, including in acute care settings and early in the course of hospitalization, given the rapid onset of action of risperidone ISM. According to CCEIP, risperidone ISM would typically be administered by nurses (and pharmacists in certain jurisdictions) under the supervision of a physician (specialist or general practitioner). CCEIP noted that most early intervention for psychosis programs are with specialty teams located in community outpatient settings and that, given the ease of use and rapid onset of action of risperidone ISM, it is ideally suited for initiation in the community for any type of program.
Drug Program Input
Input was obtained from the drug programs that participate in the reimbursement review process. The following items were identified as key factors that could potentially impact the implementation of a recommendation for risperidone ISM: relevant comparators; considerations for prescribing, initiation, continuation or renewal, and discontinuation of therapy; care provision issues; and system and economic issues. The details of the drug program input along with advice from the clinical expert consulted for this review are available in Table 3.
Clinical Evidence
Pivotal Studies
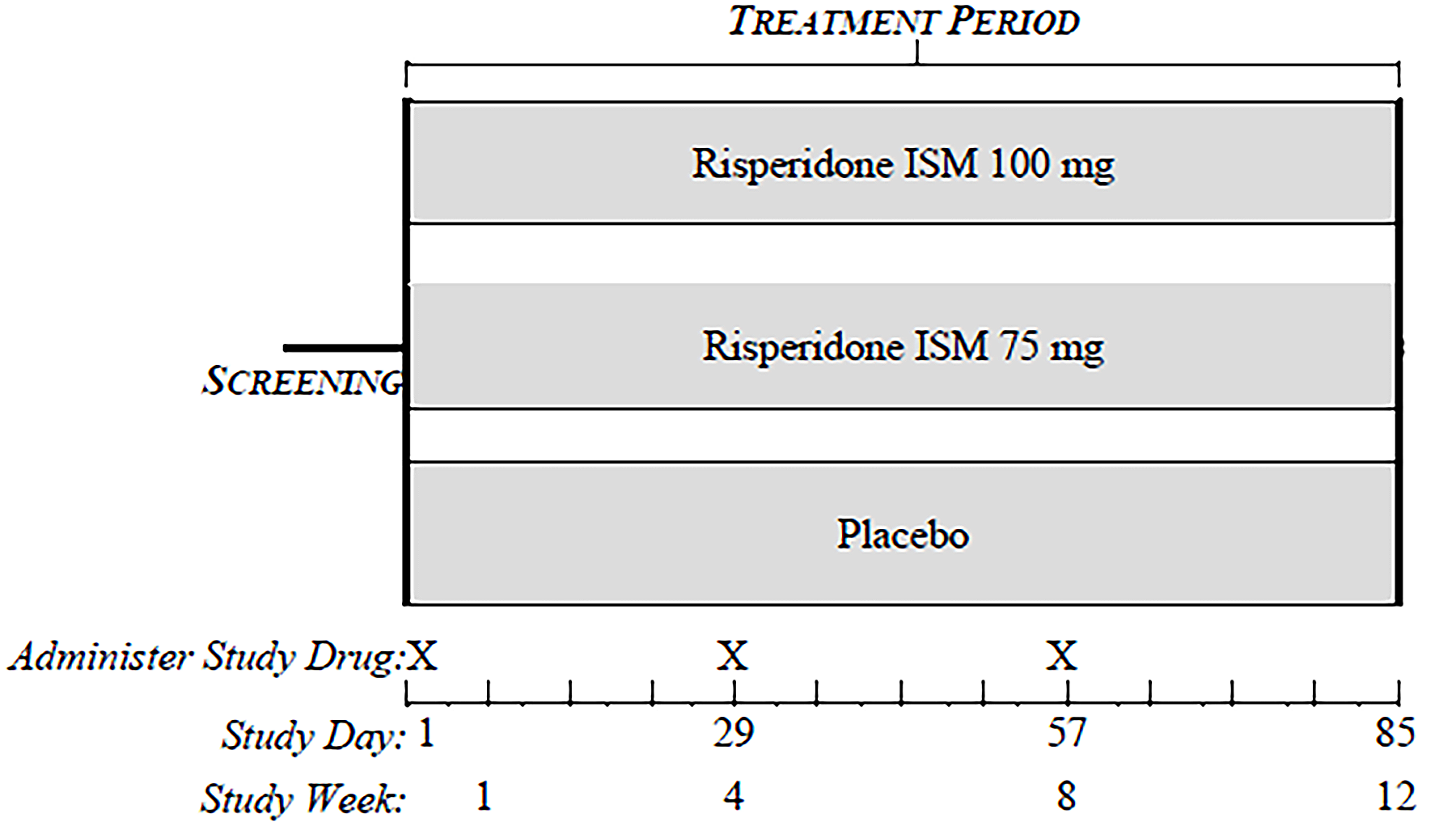
The PRISMA-3 Trial
Description of Study
The PRISMA-3 trial (N = 438) was a multicentre, randomized, double-blind (DB), placebo-controlled study investigating the efficacy and safety of risperidone ISM in the treatment of adult patients with schizophrenia who were experiencing an acute exacerbation. Both the PRISMA-3 trial and its extension study, the PRISMA-3 open-label extension (OLE) trial, were conducted at 26 centres in Ukraine and the US. In the PRISMA-3 trial, patients were randomized 1:1:1 to receive risperidone ISM 75 mg, risperidone ISM 100 mg, or matched placebo by IM injection every 4 weeks for a total of 3 injections during the 12-week treatment phase. The primary end point was the PANSS total score mean change from baseline at day 85, and the key secondary end point was the Clinical Global Impression – Severity (CGI-S) score mean change from baseline at day 85. The PANSS is a 30-item scale with total scores ranging from 30 to 210; higher scores represent greater symptom severity. PANSS subscales include the positive (7 items), negative (7 items), and general psychopathology (16 items) subscales. The CGI-S is a clinician-rated scale of severity of illness with overall scores ranging from 0 (not assessed) to 7 (among the most extremely ill patients). Other secondary end points included overall response rate (ORR), defined as a decrease in PANSS total score of greater than or equal to 30% from baseline or a CGI-I score of 2 (much improved) or 1 (very much improved), at day 85; PANSS positive subscale score mean change from baseline and PANSS negative subscale score mean change from baseline, each at day 85; and health-related quality of life (HRQoL) at the end of the trial, as measured by the Subjective Well-Being Under Neuroleptics – Short Form (SWN-20) total score change from baseline. The CGI-I is a clinician-rated global improvement scale with overall scores ranging from 1 (very much improved) to 7 (very much worse). The SWN-20 is a scale used by patients to rate well-being items that have been identified as related to antipsychotic treatment. Total scores range from 20 to 120, with higher scores indicating better HRQoL. Harms outcomes were also assessed.
Patients eligible for participation in the PRISMA-3 trial were aged 18 years to 65 years, with a current diagnosis of schizophrenia and who were actively experiencing an acute exacerbation or relapse with a PANSS total score between 80 and 120 and a score of greater than or equal to 4 points for 2 or more of the following positive symptom items: delusions, conceptual disorganization, hallucinatory behaviour, and suspiciousness and/or persecution. All patients had a CGI-S score of greater than or equal to 4 (moderately ill or worse). Patients previously had a clinically significant beneficial response after treatment with an antipsychotic other than clozapine, no history of inadequate clinical response to treatment with risperidone or paliperidone, and no history of treatment resistance. The mean age of patients in the PRISMA-3 trial was 42.0 years. Approximately two-thirds of the patients were male (67.0%) and one-third were female (33.0%). Approximately half of the patients were white (48.5%) and half were Black or African American (49.9%). The majority of patients were in the US (61.1%), and the remainder were in Ukraine (38.9%). The mean body mass index (BMI) of patients was 28.33 kg/m2. The mean time since acute exacerbation or relapse was 0.4 weeks, and the mean time since schizophrenia diagnosis was 15.5 years.
Efficacy Results
PANSS total score change from baseline to day 85 (primary end point): The mean change from baseline to day 85 was –11.0 (95% confidence interval [CI], –14.1 to –8.0) for the placebo group, –24.6 (95% CI, –27.5 to –21.6) for the risperidone ISM 75 mg group, and –24.7 (95% CI, –27.7 to –21.6) for the risperidone ISM 100 mg group. The Lawrence and Hung (LH) mean difference from the placebo group was –13.0 (95% CI, –17.3 to –8.8) for the risperidone ISM 75 mg group and –13.3 (95% CI, –17.6 to –8.9) for the risperidone ISM 100 mg group (Hommel-adjusted P value < 0.0001 for both groups). The within-group changes for risperidone ISM exceeded the PANSS within-group minimally important difference (MID) estimate of 15 points; however, in the absence of an estimated between-group MID, there is uncertainty as to whether the advantage of risperidone ISM over placebo is clinically meaningful.
CGI-S score change from baseline to day 85 (key secondary end point): The mean change from baseline at day 85 was –0.6 (95% CI, –0.8 to –0.4) for the placebo group, –1.3 (95% CI, –1.5 to –1.2) for the risperidone ISM 75 mg group, and –1.3 (95% CI, –1.5 to –1.2) for the risperidone ISM 100 mg group. The LH mean difference from the placebo group was –0.7 (95% CI, –1.0 to –0.5) for the risperidone ISM 75 mg group and –0.7 (95% CI, –1.0 to –0.5) for the risperidone ISM 100 mg group (Hommel-adjusted P value < 0.0001 for both groups). The within-group changes exceeded the 1-point improvement on the CGI-S that is considered clinically meaningful; however, in the absence of an estimated between-group MID, there is uncertainty as to whether the advantage of risperidone ISM over placebo is clinically meaningful.
PANSS positive and negative subscale score change from baseline to day 85: The PANSS positive subscale score mean change from baseline at day 85 was –4.1 (95% CI, –5.1 to –3.1) for the placebo group, –8.0 (95% CI, –9.0 to –7.1) for the risperidone ISM 75 mg group, and –8.7 (95% CI, –9.7 to –7.8) for the risperidone ISM 100 mg group. The least squares (LS) mean difference from placebo was –3.9 (95% CI, –5.3 to –2.5; P < 0.0001) for the risperidone ISM 75 mg group and –4.6 (95% CI, –6.0 to –3.2; P < 0.0001) for the risperidone ISM 100 mg group. At day 85, the PANSS negative subscale score mean change from baseline was –1.7 (95% CI, –2.5 to –0.9) for the placebo group, –3.8 (95% CI, –4.5 to –3.0) for the risperidone ISM 75 mg group, and –3.7 (95% CI, –4.4 to –2.9) for the risperidone ISM 100 mg group. The LS mean difference from placebo was –2.1 (95% CI, –3.1 to –1.0; P < 0.001) for the risperidone ISM 75 mg group and –2.0 (95% CI, –3.1 to –0.9; P < 0.001) for the risperidone ISM 100 mg group. These end points were not part of the multiple testing procedure and are considered supportive evidence. In the absence of MID estimates, the clinical meaningfulness of the between-group differences is uncertain.
ORR at day 85: At day 85, the ORR was █████ ████ ███ ████ ██ █████ ██ ███ ███████ ██████ █████ ████ ███ ████ ██ █████ ██ ███ ███████████ ███ ██ ██ ██████ ███ █████ ████ ███ ████ ██ █████ ██ ███ ███████████ ███ ███ ██ ██████ ███ ███████████ ██ ███████████ ████ ███████ ████ █████ ████ ███ ████ ██ █████████ ███ ███████████ ███ ██ ██ ███ █████ ████ ███ ████ ██ ███████ ███ ███████████ ███ ███ ██. This end point was not part of the multiple testing procedure and is considered as supportive evidence. In the absence of MID estimates, the clinical meaningfulness of the between-group differences is uncertain.
SWN-20 total score change from baseline: The mean change in SWN-20 total score from baseline to the end of the PRISMA-3 trial was ███ ████ ███ █ ██ ███ ███ ███ ███████████ ███ █████ ███ ████ ████ ███ █ ██ ██ ███ ███ ███████ █████; for the comparison of risperidone ISM versus placebo, P = ██████. This end point was not part of the multiple testing procedure and is considered as supportive evidence. The clinical meaningfulness of the between-group differences is uncertain because no MID has been established for the SWN-20 scale and the clinical expert could not comment on the clinical meaningfulness of these results.
Harms Results
The proportion of patients who experienced at least 1 treatment-emergent adverse event (TEAE) was █████ ██ ███ ███████████ ███ ██ ██ ██████ █████ ██ ███ ███████████ ███ ███ ██ ██████ ███ █████ ██ ███ ███████ █████. The most common TEAEs were blood prolactin increased (███████████ ███ ██ ███ █████ ███████████ ███ ███ ███ █████ ███ ████████ ██), headache (██████ ████ ███ ████), hyperprolactinemia (█████ ████ ███ ████), akathisia (█████ ████ ███ ████), and weight increased (█████ ████ ██ ████). The proportion of patients who experienced at least 1 serious adverse event (SAE) was 1.4% in the risperidone ISM 75 mg group, 3.4% in the risperidone ISM 100 mg group, and 3.4% in the placebo group and the proportions of patients who discontinued risperidone ISM due to a TEAE were 4.2%, 6.2%, and 6.8% in the risperidone ISM 75 mg, risperidone ISM 100 mg, and placebo groups, respectively. No patient died during the PRISMA-3 trial.
Critical Appraisal
The PRISMA-3 trial was a randomized DB, placebo-controlled study. Methods of randomization and treatment allocation were adequate for limiting risk of bias in the randomization process. The reported baseline characteristics were generally balanced across the groups, and the clinical expert consulted for this review did not identify any imbalances that would be expected to impact the interpretation of the results. Because the individuals who prepared and administered the study drug were not blinded to the contents, there is potential risk that patients and/or outcome assessors could have become aware of the treatment group assignment, which could introduce risk of bias in the measurement of subjective outcomes (including PANSS, CGI-S, and SWN-20 measures and adverse events [AEs]); however, the magnitude and direction of this risk could not be predicted. Multiplicity of testing was accounted for in the primary and key secondary end points and included hierarchical ordering. Analyses of other secondary end points (and SWN-20) were not adjusted for multiplicity and the analyses results were considered supportive evidence. Study completion rates were low with imbalances noted between groups; however, loss to follow-up was generally low and similar across treatment groups. The clinical expert consulted for this review stated that the discontinuation rates and reasons for discontinuation in the PRISMA-3 study were reasonably aligned with rates that would be expected for patients receiving LAI antipsychotics for treatment of schizophrenia in clinical practice. For the SWN-20 end point, the proportion of patients with missing data increased over time, with a greater proportion of patients in the risperidone ISM group than in the placebo group completing the SWN-20; as such, the HRQoL results could be biased as a result of missing outcome data, although the direction of bias is uncertain. Evidence is available to support the psychometric properties of the PANSS and SWN-20 outcome measures (evidence regarding responsiveness of the PANSS was not available); however, there is limited information available on the validity, reliability, and responsiveness of the CGI-S and CGI-I measures in patients with schizophrenia.
Patients in the PRISMA-3 trial had received a diagnosis of schizophrenia and were experiencing an acute exacerbation. Eligibility criteria included a history of clinically significant beneficial response to an antipsychotic other than clozapine, no history of inadequate clinical response to risperidone or paliperidone, and no history of treatment resistance. The clinical expert commented that these criteria do not reflect patients in clinical practice whose history of treatment response may not be known and who often have a history of no or partial response to previously tried antipsychotics. All patients in the PRISMA-3 trial had a history of receiving an antipsychotic, some of whom had experience with risperidone. The clinical expert highlighted that the trial includes a highly selected population of patients with schizophrenia that would be more likely to respond to treatment with risperidone ISM. The clinical expert also noted that the typical patient with schizophrenia would have many comorbidities, often including substance use disorder; however, patients with a current diagnosis or history of substance use disorder were excluded from the PRISMA-3 trial.
According to the clinical expert, the age of patients randomized in the trial reflects that of the indicated population in clinical practice and that the sex of the participants was slightly skewed toward male patients. The clinical expert noted that the race of patients in the trial is not representative of the population in Canada as Asian patients were under-represented. An important limitation of the PRISMA-3 trial is that risperidone ISM was compared to placebo, which does not represent the standard of care for treatment of patients with schizophrenia who are experiencing an acute exacerbation. As identified by the clinical expert, appropriate comparators to risperidone ISM would be risperidone LAI every 2 weeks, aripiprazole LAI every month, and the paliperidone LAIs. Although many of the outcomes assessed in the PRISMA-3 trial align with key outcomes of importance to patients and clinicians, the clinical expert stated that the outcome measures used in the PRISMA-3 trial are not typically used in clinical practice and physician assessment of response to treatment may be based on clinical interviews rather than an assessment scale. The clinical expert also highlighted that the definition of a clinically meaningful response to treatment can vary among physicians and patients.
The PRISMA-3 OLE Trial
Description of Study
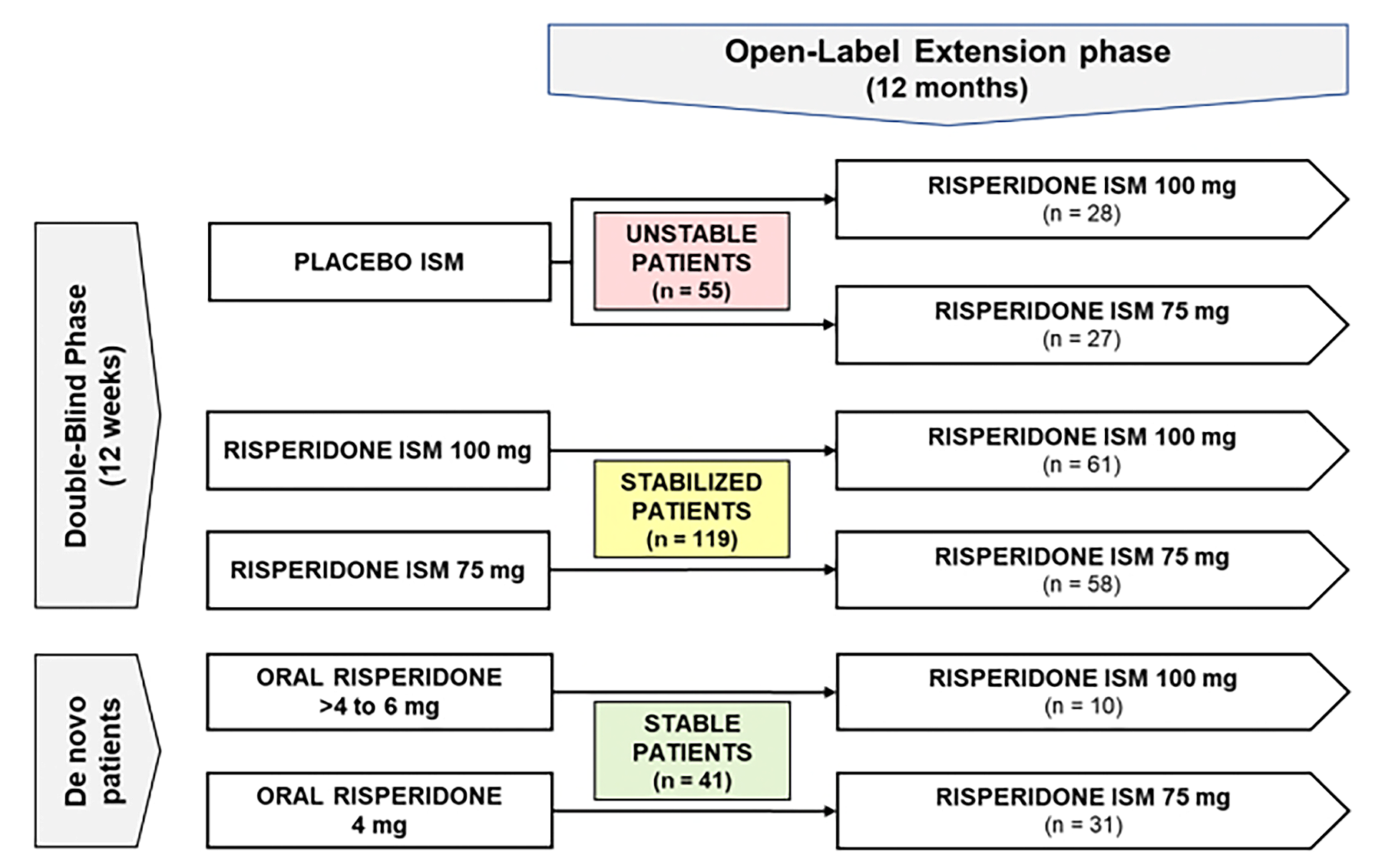
The PRISMA-3 OLE trial (N = 215) was an open-label extension study of the PRISMA-3 trial evaluating the long-term efficacy and safety of risperidone ISM in adult patients with schizophrenia. All patients received either risperidone ISM 75 mg or risperidone ISM 100 mg by IM injection every 4 weeks over 12 months. End points from the PRISMA-3 OLE trial included those for the PRISMA-3 trial (but at the OLE end point [i.e., day 365 or the last postbaseline OLE assessment]) and also relapse (defined as a PANSS total score increase of ≥ 30% from baseline; or rehospitalization for psychotic symptoms or use of adjunctive antipsychotic medication after stabilization) and remitters (defined as the simultaneous attainment of a score of ≤ 3 for 6 months or longer on 8 main items of the PANSS [delusions, conceptual disorganization, hallucinations, blunted affect, passive apathetic social withdrawal, lack of spontaneity and flow of conversations, mannerisms and posturing, unusual thought content]).
Patients eligible for participation in the PRISMA-3 OLE trial included “rollover patients” [from original source] who had completed the PRISMA-3 trial; those who had been receiving placebo (”unstable patients” [from original source]) were randomly assigned to receive either risperidone ISM 75 mg or 100 mg, while those who had been treated with risperidone ISM ( “stabilized patients” [from original source]) continued to receive risperidone ISM at the same dose as during the DB phase. In addition, newly eligible de novo patients [from original source] were aged 18 years to 65 years, with a current diagnosis of schizophrenia that was clinically stable at screening (“stable patients” [from original source]) and with a PANSS total score of less than 70 and CGI-S score of less than or equal to 3 (mild). De novo patients previously had a clinically significant beneficial response after treatment with an antipsychotic other than clozapine, no history of inadequate clinical response to treatment with risperidone or paliperidone, and no history of treatment resistance. De novo patients were also receiving a stable dose of 4 mg/day to 6 mg/day of oral risperidone; patients taking 4 mg/day received risperidone ISM 75 mg and those taking doses greater than 4 mg/day and up to 6 mg/day received risperidone ISM 100 mg. The mean age of patients in the PRISMA-3 OLE trial was 39.3 years. Male patients comprised 60.9% and female patients 39.1% of participants; the majority (84.7%) of patients were white and 14.9% were Black or African American. The majority of patients were in Ukraine (81.4%) and the remainder were in the US (18.6%); all de novo patients were in Ukraine. The mean BMI was 26.88 kg/m2, and the mean time since schizophrenia diagnosis was 10.9 years.
Efficacy Results
At the PRISMA-3 OLE trial end point, the mean PANSS total score change from baseline in the open-label population (OLP) was −11.2 (standard deviation [SD] = 14.67) and the positive and negative subscale score mean changes from baseline in the OLP were −3.4 (SD = 5.11) and −2.2 (SD = 4.00), respectively. Also at the PRISMA-3 OLE end point, the mean CGI-S score change from baseline in the OLP was –0.5 (SD = 0.91). The ORR was 45.1% in the OLP (42.2% [95% CI, 33.1% to 51.8%] in the risperidone ISM 75 mg group and 48.5% [95% CI, 38.3% to 58.7%] in the risperidone ISM 100 mg group).
Relapse during the OLE phase occurred in 11.2% (95% CI, 6.1% to 18.4%) of patients in the risperidone ISM 75 mg group and 10.1% (95% CI, 5.0% to 17.8%) of patients in the risperidone ISM 100 mg group. The relapse rate in the OLP was 10.7% (95% CI, 6.9% to 15.6%).
Kaplan-Meier estimated remittance rates after 365 days or less of treatment were █████ ████ ███ ████ ██ █████ ██ ███ ███████████ ███ ██ ██ ██████ █████ ████ ███ ███ █████████ █████ ██ ███ ███████████ ███ ███ ██ ██████ ███ █████ ████ ███ ████ ██ █████ ██ ███ ████.
During the OLE phase, the mean SWN-20 total score change from baseline to the end of the OLE phase was ████ ████ ███ ██ ██ ████████ ███ ███ ██ ████ ██████ ████ ████ ███ █ ███████ ███ ███ ███████████████ ██████ ███████████ ███ ██████ ███ ████ ████ ███ █ ██ ██████████ ███ ███ ███████████ ██████.
Harms Results
The sponsor-provided summary of harms results presented combined data for patients who received risperidone ISM in the PRISMA-3 and PRISMA-3 OLE trials. In both phases, 68.4% experienced at least 1 TEAE. The most common TEAEs were headache (█████), hyperprolactinemia (█████), increased blood prolactin (████), nasopharyngitis (████), increased weight (████), insomnia (████), and akathisia (████). In both phases, ████ ██ ████████ ███████████ ██ █████ █ ████ ████ ██ ████████ ████████████ ███████████ ███ ███ ██ █ █████ ███ █ ███████ ██████ ███████████ █ ████ ████ ████████ ██ ██████.
Critical Appraisal
In the PRISMA-3 OLE trial, the open-label extension of the PRISMA-3 trial, all patients received either risperidone ISM 75 mg or risperidone ISM 100 mg. Results for subjective end points, including the PANSS, CGI-S, SWN-20, and AEs may have been biased as a result patients and investigators knowing which treatment was being received. No adjustments for multiplicity were made for the end points in the PRISMA-3 OLE trial, and the efficacy analyses were descriptive. Results for within-group comparisons (i.e., comparing baseline to the end of the OLE phase within a treatment group) are at increased risk of type 1 errors (false-positive results). The lack of control arm precludes the ability to draw conclusions regarding the magnitude of effect attributable to risperidone ISM. According to the clinical expert, the discontinuation rates and reasons for discontinuation in the PRISMA-3 OLE trial were reasonably aligned with rates that would be expected for the indicated population in clinical practice. Missing data for the SWN-20 end point over time could have introduced bias.
In the PRISMA-3 OLE trial, the patient population comprised rollover (i.e., “stabilized” and “unstable”) patients from PRISMA-3 and de novo (i.e., “stable”) patients, which represents a broader population of patients with schizophrenia than those in the DB phase. Given that the other eligibility criteria were aligned for patients in the DB and OLE phases, the generalizability considerations noted for the PRISMA-3 trial (e.g., a generally healthier and selected population that would be expected to have better response to risperidone) would be expected to be relevant to the PRISMA-3 OLE trial. In addition, rollover patients represent a selected population who tolerated or continued to receive the study drug long enough to complete the DB phase. Regarding de novo patients, the clinical expert observed that the inclusion criteria reflect patients with schizophrenia that would be considered stable. The clinical expert noted that, as in the DB phase, a higher proportion of patients in the PRISMA-3 OLE trial were male and that Asian patients were under-represented. De novo patients on a stable dose of oral risperidone of 4 mg/day received risperidone ISM 75 mg, and those taking a stable oral risperidone dose greater than 4 mg/day and up to 6 mg/day received risperidone ISM 100 mg. However, the Okedi product monograph states that risperidone ISM 75 mg is similar to oral risperidone 3 mg/day and that risperidone ISM 100 mg is similar to oral risperidone 4 mg/day (to maintain similar plasma concentrations), and that “patients who are stable on oral risperidone doses outside this range may not be candidates for risperidone ISM.”2 The clinical expert stated that, in practice, a significant portion of patients with schizophrenia that is stabilized when taking oral risperidone would be receiving doses greater than 4 mg/day. As for the PRISMA-3 trial, many of the outcomes assessed in the extension phase align with key outcomes of importance to patients and clinicians, including relapse.
Cost Information
The sponsor submitted a cost comparison evaluating the annual drug and administration costs associated with risperidone ISM compared to other LAI antipsychotics approved for the treatment of adults with schizophrenia, as well as to oral risperidone. At the submitted prices of $468.32 and $624.40 for the 75 mg and 100 mg vials, the annual drug cost of risperidone ISM is $6,088 or $8,117 per patient per year, with an estimated annual administration cost of $88. Based on publicly available list prices, the annual drug costs for the other antipsychotic LAIs (i.e., risperidone, aripiprazole, paliperidone) ranged from $3,815 to $10,834 per patient, with annual administration costs ranging from $81 to $175. The incremental cost of risperidone ISM every 4 weeks compared to the recommended doses of the LAI comparators ranged from a savings of $4,746 annually to an additional cost of $4,302 annually, per patient, depending on the doses being compared. The annual drug acquisition cost of risperidone ISM is $5,644 to $7,821 more expensive than that of risperidone tablets ($222 to $445 per patient).
The sponsor’s cost comparison was associated with limitations including uncertainty in the assumption of clinical similarity, uncertainty in the equivalency of doses across comparators, and the potential confidential prices of the comparators. All incremental costs or savings are based on publicly available list prices and may not reflect actual prices paid by public drug plans in Canada.
Conclusions
Evidence from the PRISMA-3 trial demonstrated that, in patients with schizophrenia who are experiencing an acute exacerbation, treatment with risperidone ISM resulted in greater improvements in symptoms and disease severity compared to placebo, as measured by the change in PANSS and CGI-S total scores after 12 weeks of treatment. In both risperidone ISM groups (75 mg and 100 mg), improvements in PANSS and CGI-S total scores exceeded the within-group MIDs estimated in the literature. However, in the absence of estimated between-group MIDs, there is uncertainty regarding the clinical meaningfulness of the advantage of risperidone ISM over placebo. In the PRISMA-3 OLE trial, improvements in PANSS and CGI-S scores were observed after 12 months in a population of patients with both acute and stable disease; however, the ability to draw conclusions is limited by the open-label study design and lack of comparator arm. No conclusions can be made regarding the effects of risperidone ISM on HRQoL in patients with schizophrenia based on the evidence available from the trials due to missing data and the study design limitations. According to clinical expert input, the types of AEs that occurred in the PRISMA-3 trial are as expected for risperidone and considered manageable; however, higher rates of AEs may be expected in clinical practice. Improved adherence was identified as an outcome of importance to patients and clinicians; however, no evidence assessing this outcome for risperidone ISM was available from the PRISMA-3 pivotal trials and no evidence evaluating adherence to risperidone ISM compared to other LAI antipsychotics was submitted. In the absence of any submitted direct or indirect evidence, the comparative efficacy and harms of risperidone ISM relative to relevant comparators, including oral risperidone and other LAI antipsychotics, remain unknown.
At the submitted price, the annual cost of risperidone ISM is $6,088 or $8,117 per patient for the 75 mg and 100 mg doses, respectively, which is more expensive than that of risperidone tablets ($222 to $445 per patient), but within the range of other LAI atypical antipsychotics ($3,815 to $10,834 per patient). The sponsor’s cost comparison assumes clinical similarity between risperidone ISM and comparators; however, no direct or indirect evidence was submitted to support this assumption. Incremental savings are based on publicly available list prices and may not reflect actual prices paid by public drug plans in Canada. The sponsor’s cost comparison was associated with limitations including uncertainty in the assumption of clinical similarity and uncertainty in the equivalency of doses across comparators. Overall, given the lack of any submitted direct or indirect comparative efficacy data for risperidone ISM versus other LAI antipsychotics, there is insufficient evidence for risperidone ISM to have a price premium over other LAIs reimbursed for the treatment of schizophrenia.
Introduction
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects as well as the costs of risperidone LAI, 75 mg or 100 mg, powder for extended-release suspension, IM injection in the treatment of schizophrenia in adults.
Disease Background
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following have been summarized and validated by the review team.
Schizophrenia is a complex and chronic mental health condition with a broad range of symptoms and fluctuating symptom severity over time.3,4 Schizophrenia is a polygenic disorder (with heritable factors estimated to account for 80% of risk) and the etiology of schizophrenia is believed to be influenced by interactions between genetic and environmental factors (particularly during early brain development), although the mechanisms of these interactions are unclear.3,33,34
Symptoms of schizophrenia are grouped into domains. The positive domain involves symptoms of reality distortion (hallucinations and delusions, including impaired insight into the illness) and disorganized thoughts and behaviours.5 The negative symptoms impact emotional expression and motivation and include reduction in spontaneous speech, diminished functioning, and social withdrawal.3,5 Cognitive symptoms may include impaired attention, memory, and verbal comprehension.5 Affective symptoms, such as depression and anxiety, are also often experienced by people with schizophrenia, and depressive symptoms can increase the risk of suicide.5,6
The course of illness in schizophrenia is heterogeneous and influenced by various factors.5 The onset of schizophrenia is often characterized by a prodromal phase with negative symptoms, although transient and/or attenuated psychotic symptoms, memory and concentration problems, and unusual behaviour may also be present.5,8 The prodromal phase precedes the initial episode of psychosis and can last from a few days to approximately 18 months.7,8 The acute phase, characterized more by positive symptoms (e.g., hallucinations, delusions, behavioural disturbances, agitation), typically follows.8 Positive symptoms decrease over time (due to treatment or the natural course of the disease) and are replaced by negative symptoms that are most prevalent during the residual phase.7,8 Breakthrough psychotic episodes (relapses), which are experienced by most patients during the course of their illness, can be triggered by nonadherence to therapy, substance use, or stress.9
The diagnosis of schizophrenia is clinical, with diagnostic criteria defined in the Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition, Text Revision) (DSM-5-TR).35,36 Diagnostic criteria include persistence of the disorder for at least 6 months, including at least 1 month of active-phase symptoms. The evaluation may involve additional tests such as brain imaging and genetic testing.35 The clinical expert also noted that drug testing is common to rule out substance-induced psychosis.
Schizophrenia significantly affects patients’ quality of life as a result of its impacts on their physical, mental, and social well-being.10,11 The factors contributing to lower quality of life may include psychotic symptoms; comorbidities (e.g., depression, metabolic syndrome); negative symptoms; social isolation; employment challenges; difficulties with personal and social relationships; and stigma (which may be related to health care disparities, incarceration, homelessness, and violence).7,10,37,38 Schizophrenia is characterized by increased mortality, compared to the general population, with the predominant causes comorbid illnesses (e.g., cardiovascular disease) and death due to unnatural causes, including suicide.5,7,12,13 In 2016 to 2017, the all cause-mortality rate in Canada for individuals with schizophrenia was reported to be 2.8 times higher than for those without the disorder.14
In 2022 to 2023, the prevalence of schizophrenia in people in Canada aged 10 years and older was approximately 1% and the incidence rate in the same population was estimated to be 53 cases per 100,000.15 In Canada, 56% of individuals with schizophrenia are male and 44% are female. The onset of schizophrenia usually occurs in early adulthood, with approximately 30% of new diagnoses among people aged between 20 and 34 years (males generally experience earlier onset of schizophrenia than females and, in this age group, were twice as likely to be diagnosed).8,14
Standards of Therapy
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following have been summarized and validated by the review team.
According to the clinical expert consulted by CDA-AMC for this review, the main goals in the treatment of schizophrenia are maintenance of function in daily life, prevention of readmissions to hospital (i.e., relapses), supported community living and integration, and medication adherence for symptom control.
Antipsychotic medications are classified as first-generation antipsychotics or second-generation antipsychotics; second-generation antipsychotics are also known as atypical antipsychotics.16 Second-generation antipsychotics are less likely than first-generation antipsychotics to cause extrapyramidal symptoms such as akathisia, dystonia, and parkinsonism.17 Clozapine (a second-generation antipsychotic) is more effective than other antipsychotic medications in the treatment of schizophrenia but has significant side effects and health risks that require careful monitoring.17-19
Clinical practice guidelines published in 2017 provide recommendations for the pharmacotherapy of schizophrenia in Canada.20 These guidelines recommend the use of antipsychotic medication for first-episode psychosis, with the choice of antipsychotic being made by the patient and physician together, with the views of the caregiver (where appropriate) taken into consideration. The guidelines note that the selection of antipsychotic medication in this setting is often guided by the side-effect profile because the clinical superiority of a specific antipsychotic agent or antipsychotic class (i.e., first- or second-generation antipsychotic) has not been established. Assessment of response should take into account factors such as antipsychotic nonadherence. Strategies to address nonadherence are noted in the guidelines; these include psychoeducation, simplified dosing regimens, blister packs, and caregiver support as well as consideration of different formulations, such as LAIs. The guidelines in Canada also note the potential place for LAIs earlier in the course of treatment, and not only for patients for whom suboptimal adherence is a concern. The guidelines recommend maintenance treatment with antipsychotics for at least 18 months once the positive symptoms of the first episode of schizophrenia have resolved, noting that relapse rates are high with antipsychotic discontinuation and that response can be compromised upon relapse.20
In the event of an acute exacerbation of schizophrenia, the dose of the antipsychotic may be adjusted or treatment may be switched to another antipsychotic. In instances where the acute episode is triggered by nonadherence, the guidelines suggest a switch in formulation to a depot or LAI antipsychotic as a preferred strategy. Maintenance treatment with an antipsychotic at a daily low or moderate dose of 4 mg to 6 mg of risperidone, or other equivalents, is recommended following an acute episode. Upon resolution of the episode, maintenance treatment is recommended for 2 years and possibly up to 5 years or longer. In this setting, the guidelines recommend that the option of oral or LAI antipsychotic should be offered to all patients (that is, LAI formulations should not be restricted to those for whom nonadherence to oral formulations is a concern). According to the guidelines, clozapine should be offered to patients with schizophrenia that meets the criteria for treatment resistance and should be considered for patients with schizophrenia that has not responded to treatment with 2 antipsychotics.20
The Health Quality Ontario standard for the care of adults with schizophrenia in the community (updated in 2023) states that LAI antipsychotic medications can improve treatment adherence and prevent relapse. Quality standards within this document include offering the option of an LAI antipsychotic medication to patients, including early in the course of antipsychotic treatment. This quality standard also states that patients whose symptoms have improved with antipsychotic medication are advised to continue receiving this treatment in the long term.39
Psychosocial interventions, such as specialty care programs, cognitive behavioural therapy, support for the development of self-management skills, family interventions and education, psychoeducation, access to safe and affordable housing, and supportive employment services, are also recommended as components of effective treatment for patients with schizophrenia.21-23
The clinical expert consulted by CDA-AMC for this review stated that the choice of antipsychotic medication is mainly determined by side-effect profile, patient preference, and availability. The clinical expert noted that an atypical antipsychotic medication is usually prescribed as initial therapy because this class carries less risk of side effects such as extrapyramidal symptoms. The clinical expert highlighted that adherence to antipsychotic medication is important in preventing relapse but is a concern for oral medications because patients with schizophrenia may lack insight into their illness. The clinical expert noted that in current clinical practice, LAIs are not reserved for patients with nonadherence or previous oral medication treatment failures, and that early initiation of an LAI may be preferable to maintain medication adherence. According to the clinical expert, LAIs would be considered first-line options in the treatment of schizophrenia, including during an acute exacerbation or as maintenance therapy. The clinical expert commented that, for the minority of patients with schizophrenia who have good or partial insight into their illness and experience adherence to an oral antipsychotic, the choice of either an oral or injectable treatment may be appropriate. For patients who do not experience an adequate response to 2 different antipsychotic treatment options, the clinical expert stated that clozapine is an effective therapy but is associated with adherence challenges and requires additional health care resources to ensure adherence and safety. The clinical expert conveyed that the current approach to treating schizophrenia is multimodal; medications are 1 component, but psychosocial therapy, including support systems to help patients remain functional, is a significant aspect of treatment.
Drug Under Review
In this report, Okedi is referred to as “risperidone long-acting injection” (per the sponsor application1), “risperidone for extended-release injectable suspension” (per the Okedi product monograph2), and “risperidone ISM” (per the PRISMA-3 pivotal trials40).
Key characteristics of risperidone (including risperidone for extended-release injectable suspension) and LAI antipsychotic medications available for the treatment of schizophrenia are summarized in Table 2.
Risperidone for extended-release injectable suspension (Okedi) has been approved by Health Canada for the treatment of schizophrenia in adults (Notice of Compliance date: March 21, 2024).1,2 The reimbursement request is per the Health Canada–approved indication. Risperidone is an atypical antipsychotic medication.41 Although the mechanism of action of risperidone in the treatment of schizophrenia is unclear, its therapeutic activity may be mediated through antagonism of dopamine type 2 and serotonin type 2 receptors.2
Risperidone for extended-release injectable suspension (Okedi) is available as a prefilled syringe of powder for extended-release suspension for IM deltoid or gluteal injection and is available in 75 mg and 100 mg strengths. The product monograph recommends that, for patients who have never taken risperidone, tolerability with oral risperidone be established before initiating risperidone for extended-release injectable suspension (Okedi). The recommended dose of risperidone for extended-release injectable suspension (Okedi) is 75 mg or 100 mg administered once every 4 weeks. The 75 mg dosing regimen is similar to an oral risperidone dose of 3 mg/day and the 100 mg dosing regimen is similar to an oral risperidone dose of 4 mg/day (to maintain similar plasma concentrations); the product monograph notes that “patients who are stable on oral risperidone doses of less than 3 mg/day or greater than 4 mg/day may not be candidates for risperidone extended-release injectable suspension (Okedi).” The product monograph states that a loading dose should not be administered, and that the injection should not be supplemented with oral risperidone. Each injection should be administered by a health care professional only.2
Risperidone for extended-release injectable suspension (Okedi) has not been previously reviewed by CDA-AMC. Risperidone tablets for oral administration have been approved in Canada since 1993,24,25 and risperidone LAI for IM administration every 2 weeks (Risperdal Consta) has been approved in Canada since 2004;26 both are used in clinical practice. Risperidone for extended-release injectable suspension for subcutaneous administration once monthly (Perseris) was reviewed by CADTH for the indication of treatment of schizophrenia in adults and received a recommendation to “reimburse with clinical criteria and/or conditions” (September 24, 2021).27,28 According to the Health Canada Drug Product Database, the current status of Perseris is “cancelled post market.”29 Another risperidone product, a monthly subcutaneous risperidone extended-release formulation (Longavo), reviewed by CDA-AMC for the treatment of schizophrenia in adults, received a recommendation to “reimburse with conditions” (June 25, 2025).30
Table 2: Key Characteristics of Risperidone and LAI Antipsychotic Medications
Characteristic | Risperidone ISM (Okedi) | Risperidone LAI q.2.w. (Risperdal Consta) | Risperidone for oral administration | Aripiprazole LAI q.m. (Abilify Maintenaa) | Paliperidone palmitate LAI q.m. (Invega Sustenna) | Paliperidone palmitate LAI q.3.m. (Invega Trinza) |
|---|---|---|---|---|---|---|
Mechanism of action | The mechanism of action of risperidone in the treatment of schizophrenia is unclear; its therapeutic activity could be mediated through antagonism of D2 and 5-HT2 receptors. Antagonism at other receptors may explain some of the other effects of risperidone. | Risperidone binds with high affinity to 5-HT2, D2, and alpha1-adrenergic receptors and with a lower affinity to alpha2-adrenergic and histamine H1 receptors. | The mechanism of action of aripiprazole in the treatment of schizophrenia is unknown; it has been proposed that it is mediated through partial agonist activity at D2 and 5-HT1A receptors and antagonist activity at 5-HT2A receptors. | The mechanism of action of paliperidoneb in the treatment of schizophrenia is unknown but has been proposed to be mediated through antagonism of D2 and 5-HT2A receptors. Antagonism at other receptors may explain some of the other effects of paliperidone. | ||
Indicationc | Treatment of schizophrenia in adults. | Management of the manifestations of schizophrenia and related psychotic disorders. | Acute treatment and maintenance treatment of schizophrenia and related psychotic disorders. | Treatment of schizophrenia in adult patients. | Treatment of schizophrenia. | Treatment of schizophrenia in adult patients. |
Route of administration | IM (deltoid or gluteal) | IM (deltoid or gluteal) | p.o. | IM (deltoid or gluteal) | IM (deltoid or gluteal) | IM (deltoid or gluteal) |
Recommended dose | 75 mg or 100 mg q.4.w.d For patients who have never taken risperidone, tolerability with p.o. risperidone should be established before initiating treatment. A loading dose should not be administered. Okedi should not be supplemented with p.o. risperidone. | The recommended dose is 25 mg q.2.w. “Some patients not responding to 25 mg may benefit from a higher dose of 37.5 mg or 50 mg” (maximum dose: 50 mg q.2.w.). A lower initial dose of 12.5 mg or a dose reduction to as low as 12.5 mg may be appropriate when clinical factors warrant dose adjustment (e.g., hepatic/renal impairment, drug interactions). For patients who have never taken p.o. risperidone, tolerability with p.o. risperidone should be established before initiating treatment. Risperidone p.o. (or another antipsychotic medication) should be given with the first injection and continued for 3 weeks. | Can be administered on a once daily or b.i.d. schedule, generally beginning with 1 mg/day to 2 mg/day. The dose should be adjusted gradually over several days based on clinical response to a target dose of 4 mg/day to 6 mg/day. Some patients may benefit from lower initial doses and/or a slower adjustment schedule. | Treatment initiation:
Maintenance dose: 400 mg once monthly. Dosage reductions may be recommended (e.g., for adverse reactions, drug interactions). For patients who have never taken aripiprazole, tolerability with p.o. aripiprazole should be established before initiating treatment. | Initiation regimen: 150 mg on treatment day 1 and 100 mg on day 8 (1 week later), both administered in the deltoid muscle. Subsequent monthly maintenance dose: 75 mg; this dose can be higher or lower within the recommended range of 25 mge to 150 mg based on individual patient tolerability and/or efficacy. Starting 1 month after the second initiation regimen dose, monthly maintenance doses can be administered in either the deltoid or gluteal muscle. For patients who have never taken p.o. paliperidone or p.o. or injectable risperidone, tolerability with p.o. paliperidone or p.o. risperidone should be established before initiating treatment. | Initial dose: The equivalent 3.5-fold multiplier of the previous Invega Sustenna monthly dose, administered at the scheduled time of the Invega Sustenna dose. Following the initial dose, Invega Trinza should be administered every 3 months. If needed, dose adjustment can be made every 3 months in increments within the range of 175 mg to 525 mg based on individual patient tolerability and/or efficacy. Invega Trinza is to be used only after Invega Sustenna has been established as adequate treatment for at least 4 months. |
Serious adverse effects or safety issues |
| |||||
Other | To be administered by a health care professional. | — | To be administered by a health care professional. | |||
5-HT1 = serotonin 1; 5-HT2 = serotonin 2; b.i.d. = twice a day; CDA-AMC = Canada's Drug Agency; D2 = dopamine type 2; IM = intramuscular; ISM = in situ microparticle; LAI = long-acting injection; p.o. = orally; q.m. = every month; q.3.m. = every 3 months; q.2.w. = every 2 weeks; q.4.w. = every 4 weeks.
aAnother aripiprazole LAI (Abilify Asimtufii) is marketed in Canada (as of March 20, 2025) and was noted in the clinician group input; however, this drug has not been reviewed by CDA-AMC and it is not currently publicly funded by jurisdictions in Canada.42-44
bFollowing injection, paliperidone palmitate is hydrolyzed to paliperidone.45,46
cHealth Canada–approved indication.
dAn Okedi injection of 75 mg once q.4.w. is similar to an oral risperidone dose of 3 mg/day. An Okedi injection of 100 mg q.4.w. is similar to an oral risperidone dose of 4 mg/day. Patients who are stable on oral risperidone doses of less than 3 mg/day or greater than 4 mg/day may not be candidates for Okedi.2
eNot currently available in Canada.46
Sources: Product monographs for Okedi,2 Risperdal Consta,47 Risperidone,48 Abilify Maintena,44 Invega Sustenna,46 and Invega Trinza.45
Perspectives of Patients, Clinicians, and Drug Programs
The full patient and clinician group submissions received are available in the consolidated patient and clinician group input document for this review on the project website.
Patient Group Input
This section was prepared by the review team based on the input provided by patient groups.
No patient input was received for this review. Patient input received for a previous CADTH review (November 2021) of risperidone for extended-release injectable suspension (SR0671 Perseris) for the treatment of schizophrenia in adults is included in this report.31 As such, the available input may not accurately reflect patients’ current unmet needs.
One patient group, IAM (formerly the Schizophrenia Society of Ontario), responded to the call for patient input for the reimbursement review for Perseris. IAM is a connector, collaborator, and solutions-driven organization supporting, innovating, and driving change for better mental health. IAM innovates in mental health with a focus on returning solutions back to communities through partnership and collaboration. Historically serving those impacted by psychotic illnesses such as schizophrenia and psychosis, IAM serves anyone affected by serious mental illness and their families, caregivers, supports, and communities. IAM’s direct services include one-on-one and group counselling using cognitive behavioural therapy–based interventions, information and system navigation, training for front-line workers, community education, and more.
This submission was informed by IAM’s 40-year history of serving adults with schizophrenia and other psychotic illnesses as well as their families, service providers, and communities. Much of this knowledge came directly from IAM’s work with clients and the expertise of IAM front-line staff. IAM’s clients are primarily adults from the southern and central regions of Ontario and those from northern regions of Ontario, who are served virtually, and health service and social service providers who work with individuals with psychotic illnesses, including schizophrenia. IAM also drew on information from a survey of members of its client network conducted by Ipsos in 2018. In the Ipsos survey, respondents self-described as personally diagnosed (12%), caregivers (50%), family members or friends of someone with a diagnosis (63%), or working in social services (18%). About 77% of survey respondents were born in Canada, and 83% self-described as white. A total of 55% of respondents came from the Greater Toronto Area, while 20% came from southwestern Ontario, 14% from Eastern Ontario, 5% from Central Ontario, and 4% from Northern Ontario. The remaining 2% came from outside of Ontario.
The funding information for the patient group is included in the patient group input submission published on the CDA-AMC project website.
Disease Experience
The IAM patient input submission included individuals with schizophrenia and other related illnesses with psychosis as a main feature. The patient input indicated that many of patients experience symptoms of psychosis without having a formal diagnosis. Often, their symptoms have a significant impact on day-to-day functioning. The patients’ experiences vary widely but typically involve some levels of cognitive impairment, delusions, and hallucinations. The cognitive impairment can range from mild to severe but is typically strongest in the cognitive domains of working and episodic memory, attention, processing speed, problem-solving, and social cognition. Many patients also experience anosognosia, a lack of insight into their illness, which often impacts their ability and motivation to access treatment and supports. This symptom can cause significant strain on relationships, including those with caregivers and family members, ultimately leading to social isolation and a lack of supports for the individual with the illness.
Experience With Treatment
Of the respondents to the IAM client network survey, 55% were taking long-acting injectable medications or had previously taken them, while 98% were taking or had taken oral medications to manage their schizophrenia or psychosis symptoms. The most commonly reported side effects were drowsiness (58%), dry mouth (50%), restlessness (50%), dizziness (45%), muscle stiffness (45%), constipation (43%), and anxiety (43%).
Among those taking long-acting injectable medications, the most common stated benefits were convenience and not needing to remember to take a medication every day. The difficulties that were reported most frequently were pain at the injection site and frequent travel to clinics.
While 63% of the IAM survey respondents said that they paid for their medications with government insurance, 23% paid out of pocket for their medications. A total of 63% of respondents agreed it is difficult to pay for health care bills, including medications, visits to specialists, counselling, and so forth.
Twenty-three percent of respondents identified the cost of medications as a significant barrier to access. Other identified barriers included their preferred medication not being covered by public drug programs (20%) and the wait times for approvals of certain medications under the Ontario Public Drug Program Exceptional Access Program and Health Canada Special Access Program (13%).
Patients expect new, quick, simple, convenient, and affordable access to a wide range of treatments and medications that suit their unique needs, can improve adherence, and allow for the highest level of daily functioning and symptom reduction while managing side effects.
Improved Outcomes
The patient group indicated that the right medication allows for the highest level of daily functioning while managing side effects. This is often achieved through a trial-and-error process to find which medications and dosages work best. This process can make treatment adherence difficult for patients. Difficulties with adherence can be exacerbated by challenges with accessing psychiatrists, obtaining prescriptions, and understanding medication options; the cost of medications; and the wait times to access medications through public drug programs. When the right combination of therapy and medication is determined, individuals may still experience relapse and may require extensive supports to adhere to the treatment plan. This trial-and-error process of finding the right medication for each individual would be improved by having quick, simple, and affordable access to a wide range of treatments and medications to suit individuals’ unique needs.
Additional Information
The patient group indicated that the greater the variety and affordability of medications on the market, the better the treatment adherence is likely to be among patients with schizophrenia and psychosis and, by extension, the greater the levels of recovery. IAM advocates for a wide selection of reimbursable medications in the marketplace in Canada.
Clinician Input
Input From the Clinical Expert Consulted for This Review
All CDA-AMC review teams include at least 1 clinical specialist with expertise in the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 1 clinical specialist with expertise in the diagnosis and management of schizophrenia.
Unmet Needs
The clinical expert stated that, for schizophrenia, there remains an unmet need for treatments with minimal (or no) adverse effects, that improve function and quality of life, and that improve negative and cognitive symptoms.
Place in Therapy
According to the clinical expert, most patients with schizophrenia, whether or not they have insight into their illness, would be expected to benefit from an LAI antipsychotic because it allows for treatment adherence; therefore, LAIs would be considered first-line options for many patients with a diagnosis of schizophrenia, including during an acute exacerbation or as maintenance therapy. The clinical expert stated that risperidone ISM would be suitable for patients who are experiencing an acute exacerbation of schizophrenia and for patients who may benefit from transitioning from a stable oral risperidone dose to an LAI antipsychotic. The clinical expert also noted that patients are required to trial the oral formulation of an antipsychotic medication to ensure tolerability before switching to the LAI formulation.
Patient Population
There are atypical LAI antipsychotics currently available that can be administered less frequently than risperidone ISM (i.e., up to every 3 months). Based on this, the clinical expert expected that risperidone ISM would be prescribed for patients who are already receiving an LAI but experiencing an inadequate response and who are not willing to take clozapine. The clinical expert also expected that risperidone ISM may be prescribed for patients with schizophrenia that was responding well to oral risperidone treatment but for whom an LAI would be preferred; in this situation, dosing with risperidone ISM every 4 weeks may be advantageous compared to dosing with risperidone LAI every 2 weeks (Risperdal Consta), provided that the patient’s oral risperidone dose is within the recommended range to switch to risperidone ISM. The clinical expert noted that the PRISMA-3 pivotal trials included patients with schizophrenia that had previously shown a clinically significant response to an antipsychotic; however, in clinical practice previous treatment response may not be known and the patient population receiving risperidone ISM would include patients with schizophrenia that had not responded to an initial antipsychotic. Furthermore, the clinical expert also noted that the overall population in the PRISMA-3 and PRISMA-3 OLE trials was healthier than patients in clinical practice as patients with schizophrenia often have many comorbidities, which may include substance and polysubstance use disorders. The clinical expert acknowledged the dosing information from the Okedi product monograph,2 noting that based on the instructions, risperidone ISM treatment would not be suitable for patients receiving oral risperidone doses greater than 4 mg/day (or its equivalent).
Assessing the Response Treatment
According to the clinical expert, the outcome measures used in the PRISMA-3 pivotal trials (PANSS, CGI-S, CGI-I, and SWN-20 measures) are not typically used by physicians in clinical practice. For example, the PANSS is a research tool that lacks feasibility in clinical practice as it takes a considerable length of time to complete. The clinical expert noted that the BPRS and the CGI-I may sometimes be used in certain clinical settings while use of the RAI-MH version 2.032 is mandatory during psychiatric admissions in Ontario to capture data. The clinical expert also noted that some hospitals may use the RAI-MH tool to evaluate progress; however, others may assess progress based on clinical interviews rather than an assessment scale.
The clinical expert noted that the definition of a clinically meaningful response to treatment can vary among physicians and patients. For example, for a patient who is very ill and at risk of harm to self or others, attenuation of symptoms without resolution would be a significant difference. The clinical expert identified the following as important responses to treatment: reduction in the frequency or severity of symptoms (e.g., experiencing fewer hallucinations or a decrease in the intensity of symptoms); improvement in symptoms; stabilization (no deterioration) of symptoms; and the ability to perform activities of daily living.
According to the clinical expert, the frequency of treatment response assessment in clinical practice depends on the treatment setting. In inpatient settings, patients receive frequent monitoring for treatment response. In outpatient settings, patients initiating treatment with an LAI antipsychotic would initially be assessed in person every 2 weeks, with phone consultations in between as needed; once a patient with schizophrenia has stable disease, assessments by a psychiatrist may occur every 1 to 3 months, although patients may see the nurse in the injection clinic more frequently.
Discontinuing Treatment
According to the clinical expert, there are no specific parameters to define loss of response, absence of clinical benefit, and disease progression in clinical practice. These would be determined on an individual basis and would typically be based on the patient’s symptoms (e.g., delusions, hallucinations), ability to function, and other factors (e.g., threatening behaviour, eviction, hospitalization).
Prescribing Considerations
The clinical expert stated that LAI antipsychotics are initially prescribed and monitored by psychiatrists (and in some cases family physicians with specialized practices). Once a patient with schizophrenia has stable disease, they may continue to see a psychiatrist or may be followed by a family physician and receive injections in the family practice setting.
Clinician Group Input
This section was prepared by the review team based on the input provided by clinician groups.
Two clinicians from CCEIP provided input for this review. CCEIP is a national not-for-profit organization of clinicians and researchers who are associated with early psychosis programs. CCEIP represents 22 early phase psychosis programs and is a national leader in early phase psychosis service delivery, research, and education. For this clinician group input submission, information was gathered from a literature review of risperidone ISM, comparison of risperidone LAI formulations, data on the use of long-acting antipsychotic injections in schizophrenia and first 5 years of illness, clinical experience, and a conference presentation by the author of the CCEIP submission.
CCEIP highlighted that schizophrenia is a complex and heterogeneous psychiatric disorder that impairs multiple domains of cognitive, perceptual, emotional, and behavioural functioning. CCEIP noted that life-long treatment is typically required; however, rates of nonadherence to oral antipsychotics range from approximately 60% to 70% (particularly when partial adherence is taken into account). CCEIP identified “lack of insight” as the most common symptom of acute schizophrenia and arguably the main factor driving partial adherence and nonadherence.
CCEIP commented that long-acting antipsychotic injection formulations of first-generation antipsychotics were developed in the 1960s to address these adherence issues; however, these oil-based formulations were associated with injection site complications. CCEIP identified newer long-acting formulations of atypical antipsychotics (Risperdal Consta, Invega Sustenna, Invega Trinza, Abilify Maintena, Abilify Asimtufii) as offering advantages (atypicality, aqueous-based vehicles) over LAI formulations of first-generation antipsychotics. CCEIP also noted that these newer medications all require some type of titration or loading protocol (e.g., simultaneous or spaced double injections, oral supplementation) to ensure therapeutic levels are achieved as quickly as possible, highlighting that early attainment of therapeutic levels is correlated with more positive outcomes and better persistence with treatment. CCEIP also commented that other risperidone LAI formulations may be associated with more discomfort due to the required needle size (Risperdal Consta) and subcutaneous injections (Perseris), which is a consideration for patient persistence with treatment.
The desired outcomes of early intervention during the critical period, as identified by CCEIP, are to improve the course of psychosis and lead to a period of stability; return to pre-illness social and occupational levels of functioning; have a better outcome compared with the intervention after the critical period; and decrease the risk of suicide.
The obstacles to improving outcomes identified by CCEIP include delay in obtaining adequate treatment (duration of untreated psychosis); delay in treatment response and remission not being achieved or sustained (inadequate dosing, titration); partial adherence and nonadherence to treatment (use of oral medications); inadequate persistence with treatment (discontinuing before an adequate trial); and poor treatment of comorbidities, including substance use disorder. CCEIP expressed that patients with early psychosis have the highest rates of discontinuation and relapse and that, given the variability and evolution of schizophrenia, it is necessary that patients have access to and reimbursement of the full range of antipsychotic medications.
CCEIP commented that risperidone ISM allows for achievement of therapeutic levels within 24 hours of administration and that it would be recommended to for patients early in the course of treatment and as monotherapy (rather than as a last option or in polypharmacy) for the majority of patients. In selecting the best drug for an individual patient, considerations include achieving a balance between efficacy and tolerability, ease of use, quality of life, functionality, and patient acceptance.
According to CCEIP, patients are likely to demonstrate adherence issues at some point during treatment and it is recommended that LAIs be considered at the appropriate place in therapy per the guidelines in Canada (e.g., QAAPAPLE [Stip E., et al. Can J Psychiatry. 2019]). CCEIP also noted that the use of a long-acting antipsychotic injection, particularly compared to an oral medication, can be helpful in achieving a degree of stability and providing protection against future decompensation in patients with comorbid substance use disorders.
Regarding determining response to treatment, CCEIP commented that outcomes in clinical practice are mainly determined by multidisciplinary clinical observations supplemented by reports from patients and caregivers. CCEIP identified differences in clinical trial outcomes (e.g., PANSS) and patients compared to those in clinical practice, noting that symptom control, improvement in quality of life, and functionality are the more relevant goals in clinical practice, and that patients have significant psychiatric and physical comorbidity and experience greater variability in treatment response. CCEIP noted that measures of successful treatment include gaining stability of illness and preventing recurrences and/or relapses, and that symptom and functional response to treatment is a priority in a population with early phase psychosis.
CCEIP identified the factors for discontinuing or switching treatment with risperidone ISM as the same as with other treatments, and would include nonresponse or suboptimal response to treatment or intolerability to side effects. CCEIP also noted that switching medications can be a complex process that requires a collaborative approach with the care team, the patient, and caregivers.
CCEIP stated that the inpatient (hospital) and outpatient (hospital outpatient and community clinic) settings are appropriate for administration of risperidone ISM, including in acute care settings and early in the course of hospitalization, given the rapid onset of action of risperidone ISM. According to CCEIP, risperidone ISM would typically be administered by nurses (and pharmacists in certain jurisdictions) under the supervision of a physician (specialist or general practitioner). CCEIP also commented that most patients will receive most of their treatment in community outpatient settings during the course of their illness, and that to be effective, LAI antipsychotics should ideally be started before discharge from inpatient units. CCEIP noted that most early intervention for psychosis programs are specialty teams located in community outpatient settings and that, given the ease of use and rapid onset of action of risperidone ISM, it is ideally suited for initiation in the community for any type of program.
Drug Program Input
The drug programs provide input on each drug being reviewed through the reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted for this review are summarized in Table 3.
Table 3: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Relevant comparators | |
Issues with the choice of comparator in the submitted trial(s) The PRISMA-3 trial: 12-week placebo-controlled trial in patients with schizophrenia experiencing an acute exacerbation. Eligible patients received risperidone ISM 75 mg, risperidone ISM 100 mg, or placebo injected into the gluteal or deltoid muscle every 4 weeks on days 1, 29, and 57. Clinical expert: What is an appropriate comparator for patients with schizophrenia, given that there are other long-acting antipsychotics on the market in Canada? | The clinical expert stated that the most appropriate comparators to risperidone ISM would be risperidone LAI q.2.w. (Risperdal Consta), aripiprazole LAI q.m. (Abilify Maintena), and the paliperidone LAIs. |
Other implementation issues regarding relevant comparators (e.g., access and/or funding, covered population) Abilify Maintena received a positive reimbursement recommendation from CDA-AMC (2014) - Concluded with an LOI. Invega Sustenna received a negative reimbursement recommendation from CDA-AMC (2011). Invega Trinza concluded with an LOI. Risperdal Consta has not been reviewed by CDA-AMC. CDEC: There is a variability of listing status across the federal, provincial, and territorial jurisdictions and, as such, a lack of consistency. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for initiation of therapy | |
Other patient characteristics for eligibility (e.g., age restrictions, comorbidities) The inclusion criteria for the PRISMA-3 trial include:
The exclusion criteria for the PRISMA-3 trial include:
The inclusion criteria for de novo patients in the PRISMA-3 OLE trial include:
Clinical expert: Should the CDEC reimbursement criteria align with PRISMA-3 trial inclusion and exclusion criteria? Please elaborate specifically given the trial participants’ age (can patients aged < 18 years and > 65 years access this medication?) and BMI thresholds (can patients with BMI < 18.5 kg/m2 and > 40.0 kg/m2 access this medication?). The Health Canada indication states “treatment of schizophrenia.” In the PRISMA-3 trial, the mean time since acute exacerbation or relapse was approximately 3 days. Clinical practice suggests offering long-acting injectables as an option in all phases of illness, including the first episode, after first establishing tolerability with oral formulations. Clinical expert: Would you be able to elaborate on patient selection and the place in therapy of Okedi? Is Okedi to be used only in the setting of an acute exacerbation in patients with schizophrenia? Can Okedi be used for patients with schizophrenia who may benefit from transitioning from a stable oral risperidone dose to an antipsychotic LAI? | The clinical expert could not comment on the use of Okedi for patients aged younger than 18 years. The clinical expert commented that many patients with schizophrenia would be older than 65 years and that reimbursement should extend to these patients (noting that increased risk of side effects would be a clinical consideration in this age group). According to the clinical expert, reimbursement should not necessarily be limited to patients with BMI within the range of the inclusion criteria of patients in the PRISMA-3 pivotal trials. The clinical expert stated that LAIs would be considered a first-line option for most patients with a diagnosis of schizophrenia, including during an acute exacerbation or as maintenance therapy. The clinical expert stated that Okedi would be suitable for patients who are experiencing an acute exacerbation of schizophrenia as well as for patients who may benefit from transitioning from a stable oral risperidone dose to an antipsychotic LAI. |
Prior therapies required for eligibility Patients who had never taken risperidone had a brief trial of oral risperidone 2 mg/day for 3 days during the screening period to ensure lack of any hypersensitivity reactions before the first dose of the study drug. Clinical expert: Should a trial of oral antipsychotic (any) be initiated before the administration of Okedi be a part of the CDEC reimbursement criteria? Or can the patient receive Okedi for the acute episode without this trial? | According to the clinical expert, it would be reasonable for the reimbursement criteria to require a safety trial of oral risperidone to ensure lack of any hypersensitivity reaction or severe side effects before administration of risperidone ISM, including in patients being treated for an acute episode. |
Consistency with initiation criteria associated with other drugs reviewed by CDA-AMC in the same therapeutic space Abilify Maintena (2014): Positive reimbursement recommendation from CADTH “recommends that aripiprazole long-acting injection (LAI) be listed for the maintenance treatment of schizophrenia in adult patients who are stabilized on oral aripiprazole, if the following condition is met: List in a manner similar to other long-acting atypical antipsychotic drugs.” Clinical expert: Should there be a requirement in the criteria for a patient’s disease to be stabilized on oral risperidone at a specific dose range before accessing Okedi? Noted as per the product monograph:
Patients receiving higher or lower p.o. risperidone doses may not be candidates for treatment with Okedi. Clinical expert: If the dose of p.o. risperidone is not in the range indicated in the product monograph as suitable to transition to Okedi, what other options do patients have? | The clinical expert stated that there should not necessarily be a requirement in the criteria for patients with schizophrenia to be stabilized when receiving oral risperidone at a specific dose range before accessing risperidone ISM; however, it might be reasonable to be consistent with other medications. According to the clinical expert, for patients receiving a dose of oral risperidone that is not in the recommended range to transition to risperidone ISM, per the product monograph, the options would be other atypical LAI antipsychotics (risperidone, paliperidone, or aripiprazole) or, for patients who have received these but without adequate response, first-generation antipsychotic injections. |
Considerations for continuation or renewal of therapy | |
Challenges related to assessment and monitoring of therapeutic response Efficacy in the PRISMA-3 trial was assessed using PANSS, CGI-S, and CGI-I measures. Clinical expert: Is the assessment and monitoring of therapeutic response to LAI performed through PANSS, CGI-S, and CGI-I measures in clinical practice? Please include comments on feasibility and other tools used for consideration in this population context. Clinical expert: How often in clinical practice do you assess and/or monitor therapeutic response for LAI? This may help inform the approval period for the initial period and renewal period. | The clinical expert stated that the outcome measures used in the PRISMA-3 pivotal trials (PANSS, CGI-S, and CGI-I measures) are not typically used by physicians in clinical practice. For example, the PANSS is a research tool that lacks feasibility in clinical practice as it takes a considerable length of time to complete. The clinical expert noted that the BPRS and CGI-I may sometimes be used in certain clinical settings. The clinical expert also commented that the RAI-MH version 2.032 is mandatory for use during psychiatric admissions in Ontario to capture data. The clinical expert noted that some hospitals may use the RAI-MH tool to evaluate progress, although others may assess progress based on clinical interviews rather than an assessment scale. According to the clinical expert, the frequency of assessment for treatment response in clinical practice depends on the treatment setting. People receiving inpatient care receive frequent monitoring for treatment response. In an outpatient setting, patients initiating treatment with an LAI antipsychotic would initially be assessed in person every 2 weeks (with phone consultations in between, as needed); once a patient with schizophrenia has stable disease, assessments by a psychiatrist may occur every 1 to 3 months (although patients may see the nurse in the injection clinic more frequently). |
Considerations for discontinuation of therapy | |
Definition of loss of response, absence of clinical benefit, or disease progression Clinical expert: What definition would you use for loss of response, absence of clinical benefit, and disease progression in clinical practice? Based on what parameters? Note: If a dose is delayed by a week, the median trough concentration decreases by approximately 50% during that week. Clinical expert: If the patient is nonadherent, is this a consideration for treatment discontinuation? How is the disease managed in patients with treatment nonadherence? | According to the clinical expert, there are no specific parameters to define loss of response, absence of clinical benefit, and disease progression in clinical practice. These would be determined based on the individual patient and would typically be based on the patient’s symptoms (e.g., delusions, hallucinations), ability to function, and other factors (e.g., threatening behaviour, eviction, hospitalization). The clinical expert stated that management of nonadherence depends on the cause. Nonadherence due to adverse effects would be managed by changing the medication. Nonadherence due to lack of insight into the illness may result in a detailed assessment of the individual's ability to consent to treatment and, as needed, a community treatment order. |
Considerations for prescribing of therapy | |
Dosing, schedule/frequency, dose intensity Clinical expert: Is there any evidence to increase or reduce the frequency of medication administration for this indication (i.e., from every 4 weeks to every 3 weeks)? | The clinical expert noted that this occurs in clinical practice for other antipsychotics; however, no comment could be made regarding risperidone ISM. |
Drug administration Note: There is a cost of administration as Okedi is to be administered by a health care professional. It is not self-administered. | Comment from the drug programs to inform CDEC deliberations. |
Concerns related to accessing clinical specialists and/or special settings Clinical expert: Please describe which specialists will be seeing these patients and in which setting Okedi would be administered? | LAI antipsychotics are initially prescribed and monitored by psychiatrists (and in some cases, by family physicians with specialized practices). Once a patient with schizophrenia has stable disease, they may continue to see a psychiatrist or they may be followed by a family physician and receive injections in the family practice setting. When prescribed by a psychiatrist, an LAI would typically be administered by a nurse working with the psychiatrist in the clinic setting. LAIs may also be administered in the home in the case of mental health outreach teams. |
Care provision issues | |
Drug preparation, storage, administration or dispensing Clinical expert: Where would Okedi be initiated for administration (e.g., specialist) and where would it be maintained for administration (e.g., primary care office)? | LAI antipsychotics are initially prescribed and monitored by psychiatrists (and in some cases, by family physicians with specialized practices). Once a patient with schizophrenia has stable disease, they may continue to see a psychiatrist or they may be followed by a family physician and receive injections in the family practice setting. When prescribed by a psychiatrist, an LAI would typically be administered by a nurse working with the psychiatrist in a clinic setting, but an LAI may also be administered in the home in the case of mental health outreach teams. |
System and economic issues | |
Presence of confidential negotiated prices for comparators Abilify Maintena and Invega Trinza have successfully gone through price negotiations for the same indication. | Comment from the drug programs to inform CDEC deliberations. |
BMI = body mass index; BPRS = Brief Psychiatric Rating Scale; CDA-AMC = Canada's Drug Agency; C-SSRS = Columbia-Suicide Severity Rating Scale; CDEC = Canadian Drug Expert Committee; CGI-I = Clinical Global Impression – Improvement; CGI-S = Clinical Global Impression-Severity; DSM-5 = Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition); ISM = in situ microparticle; LAI = long-acting injection; LOI = letter of intent; OLE = open-label extension; PANSS = Positive and Negative Syndrome Scale; p.o. = orally; q.m. = every month; q.2.w. = every 2 weeks; RAI-MH = Resident Assessment Instrument — Mental Health.
Sponsor’s Summary of Clinical Evidence
Pivotal Studies
Table 4: Details of Included Studies
Characteristics | PRISMA-3 | PRISMA-3 OLE | |
|---|---|---|---|
Study design | Multicentre, randomized, DB, placebo-controlled study | Multicentre, open-label study extension | |
Locations | 26 centres in the US and Ukraine | ||
Patient enrolment dates | June 2, 2017 | August 25, 2017 | |
Assigned/randomized (N) | 438 | 215 | |
Key inclusion criteria |
| Rollover Patients:
De Novo Patients:
| |
Key exclusion criteria |
| Rollover Patients:
De Novo Patients:
| |
Intervention | Okedi 75 mg IM every four weeks Okedi 100 mg IM every four weeks | Okedi 75 mg IM every four weeks Okedi 100 mg IM every four weeks | |
Comparator(s) | Placebo | NA | |
Duration | |||
Phase | Screening | 1 to 8 days | NA |
Double-blind | 12 weeks | NA | |
Follow-up | 2 weeksa | NA | |
Open-label extension | NA | 12 months | |
Outcomes | |||
Primary end point | PANSS total score mean change from baseline to end pointb | NA | |
Secondary and additional end points | Key secondary efficacy end point:
Other secondary efficacy end points:
|
| |
Notes | |||
Publications | NCT03160521 Correll CU, Litman RE, Filts Y, et al. Efficacy and safety of once-monthly Risperidone ISM® in schizophrenic patients with an acute exacerbation. NPJ Schizophr. 2020;6(1):37.50 | NCT03870880 Filts Y, Litman RE, Martinez J, Anta L, Naber D, Correll CU. Long-term efficacy and safety of once-monthly Risperidone ISM® in the treatment of schizophrenia: Results from a 12-month open-label extension study. Schizophr Res. 2022;239:83-91.51 | |
BMI = body mass index; C-SSRS = Columbia Suicide Severity Rating Scale; CGI-I = Clinical Global Impression-Improvement; CGI-S = Clinical Global Impression-Severity; DB = double-blind; ECG = electrocardiogram; IM = intramuscular; NA = not applicable; OLE = open-label extension; PANSS = Positive and Negative Syndrome Scale; US = United States.
a Not applicable for patients who enter into the long-term extension.
b For the DB phase, end point was defined as study day 85 or the last postbaseline DB assessment.
Source: PRISMA-3 Clinical Study Report40
Description of Studies
The efficacy and safety of Okedi in treating patients with schizophrenia were assessed in a multicentre, randomized, double-blind (DB), placebo-controlled Phase III study (PRISMA-3). PRISMA-3 also included an open-label extension (OLE) phase to evaluate the long-term safety and tolerability of Okedi. Both the PRISMA-3 and OLE phases were conducted across 26 centres in the United States (US) and Ukraine.
PRISMA-3
PRISMA-3 was a multicentre, randomized, DB, placebo-controlled trial conducted to assess the efficacy and safety of intramuscular (IM) injections of Okedi (75 mg or 100 mg) compared to placebo in patients with acute exacerbation of schizophrenia. The study consisted of a screening period, a 12-week treatment phase, and a 2-week follow-up period.40
Eligible patients were 18 to 65 years old, with a current diagnosis of schizophrenia, according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria and a body mass index (BMI) between 18.5 and 40.0 kg/m2. Patients were actively experiencing an acute exacerbation or relapse with a total score between 80 and 120 on the Positive and Negative Syndrome Scale (PANSS) and a score of ≥ 4 points for ≥ 2 of the following positive symptom items: delusions, conceptual disorganization, hallucinatory behaviour, and suspiciousness/persecution. All patients had a score of ≥ 4 (moderately ill or worse) on the Clinical Global Impression-Severity (CGI-S) and had previously had a clinically significant beneficial response after treatment with an antipsychotic other than clozapine.
Eligible patients were randomized in a 1:1:1 ratio to receive Okedi 75 mg, Okedi 100 mg, or placebo under DB conditions. Randomization was managed via an Interactive Web Response System (IWRS), which assigned unique randomization numbers immediately upon confirming patient eligibility. Randomization was stratified by country where enrolled and PANSS total score (≥ 95 versus < 95) at baseline.40 The IM study drug (Okedi or placebo) was administered into the deltoid or gluteal muscle once every four weeks for a total of three injections during the 12-week treatment phase. To ensure a lack of any clinically significant hypersensitivity, patients with no prior exposure to risperidone were required to complete a 3-day trial of oral risperidone (2 mg/day) during the screening period before receiving the study drug. A trial of oral risperidone was not required for patients who had previously taken any formulation of risperidone. Patients who did not enter the OLE had a final safety follow-up phone contact approximately 2 weeks after the end-of-treatment visit. The overall study design is depicted in Figure 1.
PRISMA-3 OLE
The PRISMA-3 OLE is a multicentre, long-term, OLE of the DB PRISMA-3 study conducted in 26 centres in the US and Ukraine between August 2017 and January 2020. The study aimed to evaluate long-term efficacy, safety and tolerability of Okedi in patients with schizophrenia.
Eligible patients were 18 to 65 years old, with a current diagnosis of schizophrenia, according to the DSM-5 criteria. Those who completed the DB phase of the PRISMA-3 study and had been receiving placebo (“unstable patients”) were randomly assigned to Okedi at a dose of either 75 mg or 100 mg. Patients treated with Okedi in the DB phase continued to receive Okedi every four weeks in the OLE phase at the same dose (75 mg or 100 mg) as during the DB phase (“stabilized patients”). In addition, patients who had not participated in the DB phase (de novo patients), were newly enrolled into the OLE if they met the age and diagnostic criteria and were clinically stable at screening (“stable patients”). Like the rollover patients, the de novo patients were allocated to receive either 75 mg or 100 mg Okedi every four weeks for approximately 12 months.
To preserve the blinding condition of the PRISMA-3 DB phase, if a participating patient entered the OLE prior to locking the database of the DB phase of the study, the allocation to either 75 mg or 100 mg of this patient was still to be blinded at least up to locking the database. Rollover patients from the DB phase entered the OLE upon completion of the end-of-treatment visit assessments and procedures. The overall study design is depicted in Figure 2.
Filts Y, Litman RE, Martínez J, Anta L, Naber D, Correll CU. © 2021 Laboratorios Farmacéuticos Rovi, S.A. Published by Elsevier B.V. Used under Creative Commons Licensing CC BY 4.0 https://creativecommons.org/licenses/by/4.0/ Please find material here: https://www.sciencedirect.com/science/article/pii/S0920996421004515?via%3Dihub
Populations
PRISMA-3
Inclusion Criteria
Eligible patients were 18 to 65 years old, with a current diagnosis of schizophrenia, according to the DSM-5 criteria and a BMI between 18.5 and 40.0 kg/m2. Patients were currently experiencing an acute exacerbation or relapse with a total score between 80 and 120 on the PANSS and a score of ≥ 4 points for ≥ 2 of the following positive symptom items: delusions, conceptual disorganization, hallucinatory behaviour, and suspiciousness/persecution. All patients had a score of ≥ 4 (moderately ill or worse) on the CGI-S and had previously had a clinically significant beneficial response after treatment with an antipsychotic other than clozapine.
Exclusion Criteria
Patients were excluded if improvement in PANSS total score was ≥ 20% between the screening visit and baseline. Additionally, patients with active suicidality were excluded, as indicated by having answered “yes” on item 4 or 5 of the Columbia-Suicide Severity Rating Scale (C-SSRS) with the most recent episode occurring within the past 2 months or having answered “yes” to any of the five items (suicidal behaviour) with an episode occurring within the last year. Patients were also excluded for the presence of clinically significant comorbid neuropsychiatric disorder, lifetime history of schizoaffective or bipolar disorders, or a history of any unstable medical condition or laboratory abnormality that could interfere with the conduct of the study or compromise the wellbeing of the patient. Women who were pregnant or breastfeeding were also excluded.
PRISMA-3 OLE
Inclusion Criteria
Eligible patients were 18 to 65 years old, with a current diagnosis of schizophrenia, according to the DSM-5 criteria. Those who completed the DB phase of the PRISMA-3 study according to the protocol and had been receiving placebo in the DB phase (“unstable patients”) were randomly assigned to receive either 75 mg or 100 mg of Okedi. Patients treated with Okedi in the DB phase continued to receive Okedi every four weeks in the OLE phase at the same dose (75 mg or 100 mg) as during the DB phase (“stabilized patients”).
In addition, patients who had not participated in the DB phase (de novo patients), were newly enrolled into the OLE if they met the age and diagnostic criteria and were clinically stable at screening (“stable patients”).
Exclusion Criteria
Rollover patients were excluded if they missed more than one scheduled study visit during PRISMA-3 or had clinically relevant abnormal findings in laboratory values, vital signs, or electrocardiograms (ECGs) that were deemed related to the study drug and could compromise their well-being in the extension phase. Rollover patients were also excluded if they had clinically significant or unstable medical conditions during PRISMA-3 that, in the investigator's opinion, could jeopardize patient safety in the OLE. Additionally, patients requiring or anticipated to require prohibited concomitant medications were excluded.
For de novo patients, exclusion criteria included a history of proven inadequate clinical response to therapeutic doses of risperidone or paliperidone with good compliance. Patients with a history of treatment resistance, defined as failure to respond to two separate adequate trials of different antipsychotic medications, or a history of clozapine use (unless not due to treatment resistance or refractory psychotic symptoms), were also excluded. Other exclusion criteria for de novo patients included a current diagnosis or history of substance use disorder and recent suicidal ideation or behavior as assessed by the C-SSRS. This included a “yes” response on item 4 or 5 of the ideation scale within the past two months, or any “yes” response on the behavior scale within the past year.
Baseline Characteristics
PRISMA-3
PRISMA-3 included 437 patients. The mean age of patients in PRISMA-3 was 42.0 (standard deviation [SD] = 11.02) years. Two-thirds of patients were male (67.0%) and one-third female (33.0%). Approximately half of patients were white (48.5%) and half were Black or African American (49.9%), with small percentages Asian (0.9%) or Other (0.7%). A small number of patients were Hispanic or Latino (5.0%), with the remainder not Hispanic or Latino. The study was conducted in 2 countries, with the majority of patients in the United States (61.1%) and the remainder in Ukraine (38.9%). The mean BMI of patients was 28.33 (SD = 5.247) kg/m2.40 The mean time since acute exacerbation or relapse was 0.4 (SD = 0.33) weeks. The mean years since schizophrenia diagnosis was 15.5 (SD = 10.15) years for patients overall. The demographic and other baseline characteristics were similar in each treatment group (Table 5).
Table 5: Summary of PRISMA-3 Baseline Characteristics (SAF)
Baseline Characteristics | Placebo N = 147 | Okedi 75 mg N = 144 | Okedi 100 mg N = 146 | Okedi N = 290 | Overall N = 437 | |
|---|---|---|---|---|---|---|
Age (years), mean (SD) | 40.5 (11.18) | 42.5 (10.86) | 42.9 (10.93) | 42.7 (10.88) | 42.0 (11.02) | |
Sex, n (%) | Male | 98 (66.7) | 98 (68.1) | 97 (66.4) | 195 (67.2) | 293 (67.0) |
Female | 49 (33.3) | 46 (31.9) | 49 (33.6) | 95 (32.8) | 144 (33.0) | |
Race, n (%) | White | 72 (49.0) | 73 (50.7) | 67 (45.9) | 140 (48.3) | 212 (48.5) |
Black or African American | 72 (49.0) | 68 (47.2) | 78 (53.4) | 146 (50.3) | 218 (49.9) | |
Asian | 1 (0.7) | 2 (1.4) | 1 (0.7) | 3 (1.0) | 4 (0.9) | |
Other | 2 (1.4) | 1 (0.7) | 0 | 1 (0.3) | 3 (0.7) | |
Ethnicity, n (%) | Hispanic or Latino | 11 (7.5) | 8 (5.6) | 3 (2.1) | 11 (3.8) | 22 (5.0) |
Not Hispanic or Latino | 136 (92.5) | 136 (94.4) | 143 (97.9) | 279 (96.2) | 415 (95.0) | |
Country, n (%) | Ukraine | 56 (38.1) | 57 (39.6) | 57 (39.0) | 114 (39.3) | 170 (38.9) |
United States | 91 (61.9) | 87 (60.4) | 89 (61.0) | 176 (60.7) | 267 (61.1) | |
BMI (kg/m2)a, mean (SD) | 28.36 (4.909) | 28.04 (5.418) | 28.58 (5.423) | 28.31 (5.418) | 28.33 (5.247) | |
Weight (kg), mean (SD) | 83.30 (15.018) | 83.88 (18.112) | 85.48 (18.636) | 84.68 (18.363) | 84.22 (17.305) | |
Years since schizophrenia diagnosis, mean (SD) | 14.6 (9.49) | 16.1 (10.58) | 15.9 (10.35) | 16.0 (10.44) | 15.5 (10.15) | |
Time since acute exacerbation or relapse (weeks), mean (SD) | 0.4 (0.21) | 0.4 (0.25) | 0.4 (0.46) | 0.4 (0.37) | 0.4 (0.33) | |
Antipsychotic medication taken within 30 days prior to first study drug dose in the trialb, n (%) | 128 (87.1) | 121 (84.0) | 137 (93.8) | 258 (89.0) | 386 (88.3) | |
Risperidone | 80 (54.4) | 92 (63.9) | 87 (59.6) | 179 (61.7) | 259 (59.3) | |
BMI = body mass index; SAF = safety population; SD = standard deviation.
a BMI (kg/m2) is calculated as BMI = 1002 × weight (kg)/ [height (cm)2].
b Per the inclusion criteria, all patients had received treatment with an antipsychotic medication in the past; these data apply to medications taken in the 30 days prior to screening.
Source: Okedi Global Value Dossier52, PRISMA-3 Clinical Study Report40, sponsor’s response to CDA request for additional information53
PRISMA-3 OLE
The PRISMA-3 OLE included 215 patients (Table 6). Patients had a mean age of 39.3 (SD = 10.84) years old, mean BMI of 26.88 (SD = 4.69) kg/m2, and the majority of participants were male (60.9%). Most patients were white (84.7%) and nearly all patients were not Hispanic or Latino (96.3%). The mean years since schizophrenia diagnosis was 10.9 (SD = 8.09) years for patients overall. Most demographic and baseline characteristics were similar in each treatment group, with exceptions subsequently noted. The proportion of male patients ranged from 39.3% to 74.1% across groups. All de novo patients in the OLE were white and not Hispanic or Latino. Among rollover patients, the proportion of Black or African American patients ranged from 12.1% to 23.0% and the proportion of Hispanic or Latino patients ranged from 0% to 8.6%. Across all groups, the proportion of patients from Ukraine ranged from 73.8% to 100%. The mean time since schizophrenia diagnosis among all groups ranged from 6.4 years to 12.1 years. Differences across groups are expected since randomization did not occur across all OLE groups.
Table 6: Summary of PRISMA-3 OLE Baseline Characteristics (OLP)a
Baseline Characteristics | Rollover | De Novo | Total N = 215 | |||||
|---|---|---|---|---|---|---|---|---|
Placebo/ Okedi 75 mg N = 27b | Okedi 75 mg/ Okedi 75 mg N = 58c | Okedi 100 mg/ Okedi 100 mg N = 61c | Placebo/ Okedi 100 mg N = 28b | Okedi 75 mg N = 31 | Okedi 100 mg N = 10 | |||
Age (years), mean (SD) | 38.7 (9.29) | 40.9 (11.98) | 40.3 (11.04) | 38.4 (10.42) | 37.0 (10.91) | 35.5 (6.40) | 39.3 (10.84) | |
Sex, n (%) | Male | 20 (74.1) | 38 (65.5) | 38 (62.3) | 11 (39.3) | 20 (64.5) | 4 (40.0) | 131 (60.9) |
Female | 7 (25.9) | 20 (34.5) | 23 (37.7) | 17 (60.7) | 11 (35.5) | 6 (60.0) | 84 (39.1) | |
Race, n (%) | White | 22 (81.5) | 50 (86.2) | 47 (77.0) | 22 (78.6) | 31 (100) | 10 (100.0) | 182 (84.7) |
Black or African American | 5 (18.5) | 7 (12.1) | 14 (23.0) | 6 (21.4) | 0 | 0 | 32 (14.9) | |
Asian | 0 | 1 (1.7) | 0 | 0 | 0 | 0 | 1 (0.5) | |
Ethnicity, n (%) | Hispanic or Latino | 1 (3.7) | 5 (8.6) | 0 | 2 (7.1) | 0 | 0 | 8 (3.7) |
Not Hispanic or Latino | 26 (96.3) | 53 (91.4) | 61 (100) | 26 (92.9) | 31 (100) | 10 (100) | 207 (96.3) | |
Country, n (%) | Ukraine | 20 (74.1) | 47 (81.0) | 45 (73.8) | 22 (78.6) | 31 (100) | 10 (100) | 175 (81.4) |
United States | 7 (25.9) | 11 (19.0) | 16 (26.2) | 6 (21.4) | 0 | 0 | 40 (18.6) | |
BMI (kg/m2), mean (SD) | 27.53 (4.49) | 26.52 (4.74) | 27.53 (5.06) | 27.65 (4.95) | 25.28 (3.7) | 26.06 (3.94) | 26.88 (4.69) | |
Weight (kg), mean (SD) | 82.23 (14.231) | 78.32 (14.299) | 80.50 (16.890) | 77.96 (11.956) | 75.58 (14.142) | 74.58 (12.707) | 78.81 (14.721) | |
Time since schizophrenia diagnosis (years), mean (SD) | 11.8 (6.81) | 12.1 (9.17) | 11.5 (7.98) | 9.3 (7.45) | 9.6 (8.12) | 6.4 (5.34) | 10.9 (8.09) | |
Time since acute exacerbation or relapse (weeks), mean (SD) | 0.4 (0.18) | 0.3 (0.21) | 0.4 (0.24) | 0.4 (0.20) | 0 | 0 | 0.4 (0.21) | |
BMI = body mass index; OLE = open-label extension; OLP = open-label population; SD = standard deviation.
a For rollover patients, OLE baseline was the same day as day 85 of the DB phase. DB day 85 assessments served as the OLE baseline values.
b Unstable patients
c Stablized patients
Source: PRISMA-3 Clinical Study Report40
Interventions
Okedi is a long-acting formulation of risperidone that uses in situ microparticle (ISM®) technology. Okedi required a reconstitution procedure to be performed at the study site for each dose to be administered. Okedi 75 mg and 100 mg and matching placebo were administered IM in the deltoid or gluteal muscle every four weeks. Results from previously conducted phase I and phase II studies as well as pharmacokinetic (PK) modeling were considered to support the doses selected for this study. Each of the IM injections of study drug was to be administered at approximately the same time of day, if possible. Generally, at a given study visit, the IM injection of study drug was to be administered after efficacy evaluations had been performed and after any required blood samples (e.g., PK sample) had been collected.
Prior and Concomitant Therapy
All concomitant medications (including those used during ophthalmological examination, if any) and other therapies received during a patient’s participation in this study (i.e., from the date of the informed consent form was signed through to the date of the final study visit) were to be recorded. The investigator could discontinue or adjust the dose of a concomitant medication to ensure patient safety; however, unnecessary changes in concomitant medications for reasons other than safety were generally discouraged to avoid confounding study evaluations.
Patients who had never taken risperidone had to have a brief trial of oral risperidone 2 mg/day for 3 days during the screening period to ensure a lack of any hypersensitivity reactions before the first dose of IM study drug was administered. A trial of oral risperidone was not required or indicated for patients who had previously taken any formulation of risperidone. Risperidone tablets were provided by study sponsor to all participating sites for risperidone-naïve patients testing.
Prohibited Medications
Use of the following medications was prohibited by patients for the duration of the DB and open-label extension (OLE) phases: cytochrome P450 3A4 inducers and inhibitors, 2D6 inhibitors, antipsychotic medications other than the study drugs administered, other psychotropic medications, amphetamines, barbiturates, cocaine, methadone, opioids, and phencyclidine.
Psychotherapy was not to be started or changed during a patient’s participation in the study. It was acceptable for a patient already receiving psychotherapy to participate in the study.
Rescue Medication
Permitted rescue medications for this study included benzodiazepines, anticholinergics, and propranolol (Table 7).
Medications | Indication(s) | Maximum Dosage |
|---|---|---|
Benzodiazepines | agitation, anxiety, insomnia, and/or restlessness | 6 mg/day lorazepam or equivalent for other benzodiazepines |
Anticholinergics | extrapyramidal symptoms | 4 mg/day benztropine or equivalent for other anticholinergics |
Propranolol | akathisia or tremor | 20 mg 3 times daily (60 mg/day total) |
Source: PRISMA-3 Clinical Study Report40
Outcomes
Efficacy variables in the DB and OLE phases included the PANSS, PANSS subscale scores, Clinical Global Impression-Improvement (CGI-I), CGI-S, Overall response, and PANSS response (Table 4). The PANSS, CGI-S, and CGI-I were performed at screening, DB phase study days 1 (pre-dose; except for CGI-I), 4, 8, 15, 29, 43 (CGI-I only), 57, 71 (CGI-I only), and 85 and OLE phase study days 1 (except for CGI-I), 29, 57, 85, 113, 141, 169, 197, 225, 253, 281, 309, 337, and 365.40
The primary efficacy outcome in the PRISMA-3 trial was PANSS total score mean change from baseline to end point (defined as study day 85 or the last postbaseline DB assessment). The key secondary efficacy outcome is mean change in CGI-S from baseline to end point during the DB phase.40
PANSS
The PANSS is a well-known and validated rating scale used in numerous drug evaluation trials. This 30-item scale provides a total score (sum of the scores of all 30 items) and scores for 3 subscales. The subscales include the positive subscale (7 items), negative subscale (7 items), and general psychopathology subscale (16 items). The severity of each of the 30 items of the PANSS is rated on a scale of 1 (absent) to 7 (extreme). Higher scores represent greater symptom severity. The maximum total score equals 210 and the total minimum score equals 30, when all items are completed.
CGI-S
The CGI-S severity of illness scale requires the clinician to rate the severity of illness on a 7-point ordinal scale (0 = Not assessed, 1 = Normal, not at all ill; 2 = Borderline mentally ill; 3 = Mildly ill; 4 = Moderately ill; 5 = Markedly ill; 6 = Severely ill; 7 = Among the most extremely ill patients).
CGI-I
The CGI-I global improvement scale requires the clinician to assess how much the patient's illness has improved or worsened relative to a baseline state at the beginning of the intervention. The clinician rates the degree of change observed on a 7-point ordinal scale (1 = Very much improved; 2 = Much improved; 3 = Minimally improved; 4 = No change; 5 = Minimally worse; 6 = Much worse; or 7 = Very much worse). Both CGI scales provide a summary measure that takes into consideration all available information, including a knowledge of the patient’s history, psychosocial circumstances, behaviour, symptoms, and the effect of symptoms on the patient’s functional ability.54
Statistical Analysis
Primary Outcome of PRISMA-3
Power Calculation
The modified intent-to-treat (mITT) population included all patients in the intent-to-treat (ITT) population for whom blinding was not compromised. It was used for analyses of efficacy end points. A sample size of 124 patients in the mITT population for each treatment group would provide 90% power to detect a difference in means of 9 (standard deviation = 20, effect size = 0.45) at a 2.5% two-sided significance level for comparisons between Okedi groups and placebo. The power to demonstrate superiority of both Okedi doses over placebo was estimated to be at least 81%.55
A relatively low post-randomization dropout rate was anticipated, as most patients in the inpatient setting were expected to remain in the study through day 4 (patients who withdrew before day 4 were excluded from the mITT population). Based on this assumption, a 5% dropout rate was considered reasonable. With this assumption, a randomized sample size of 131 patients per treatment group (393 patients total across all three treatment groups) was planned for analysis, assuming no compromise of blinding. One planned unblinded interim analysis re-estimated the sample size required for the final analysis in the mITT population. This analysis was conducted when 196 (approximately 50%) randomized patients, for whom blinding was not compromised, had reached study day 85 or had withdrawn from the study. At the time of the interim analysis, the independent data monitoring committee decided to continue the study without modifying the sample size.40
On September 28, 2017, due to a noted error in the IWRS system, potential accidental unblinding of treatment allocation to patients for blinded personnel was reported. Including the 43 patients for whom blinding was potentially compromised, a total of 436 randomized patients were required for the study.
Statistical Test or Model
The primary efficacy analysis was designed to show superiority of active treatment versus placebo in the primary efficacy variable. The two hypotheses tested were:
H0,A: μOkedi 75 mg - μplacebo = 0 vs. H1,A: μOkedi 75 mg - μplacebo ≠ 0
H0,B: μOkedi 100 mg - μplacebo = 0 vs. H0,B: μOkedi 100 mg - μplacebo ≠ 0
with μ = PANSS total score mean change from baseline for identified treatment group.
A mixed effect model with repeated measurements (MMRM) approach was fitted for patients in the mITT population with country where enrolled, visit, treatment, and treatment-by-visit interaction as fixed effects and baseline PANSS total score as a covariate. An unstructured covariance structure shared across treatment groups was used to model the within-patient errors with Kenward-Rogers correction to degrees of freedom. This model was applied separately for patients in stage 1 (patients included in the interim analysis) and stage 2 (all subsequent patients) of the study, as well as for overall patients. The treatment differences from the model at study day 85 were evaluated to test the hypotheses, maintaining a 2-sided type 1 error rate of 5%.
To maintain the type 1 error rate for the primary analysis, each hypothesis was tested using a weighted test statistic in accordance with Cui, Hung, Wang (CHW) methodology which comprises of combining the z-statistics from the respective study day 85 treatment comparison at each stage. The weighting was equal for each stage and predefined as square root (0.5).
The nominal 2-sided p-value for each hypothesis was calculated for the respective weighted CHW statistic using the cumulative normal distribution.
To account for multiplicity of testing and to keep the type 1 error rate at 5%, the Hommel’s closed-testing correction procedure was used to provide adjusted p-values to assess for superiority of either dose.
Confirmatory findings from the Hommel adjusted CHW analyses only remained so for a particular dose if p < 0.05 in both the mITT and ITT analyses.
All analyses for the mITT were repeated for the ITT population using CHW stage 1 weighting of square root (0.55) and stage 2 weighting of square root(0.45).40,55 Analyses in the ITT and mITT populations were repeated for the per-protocol (PP) and modified per-protocol (mPP) populations, respectively. Additional sensitivity analyses to assess missing data assumptions were performed.
The MMRM (without CHW or Hommel adjustments) was repeated in the mITT and ITT populations incorporating all main effects and interactions between treatment, visit, and the subgroup (i.e., country or baseline PANSS total score [< 95 vs. ≥ 95]) to explore the treatment differences within subgroups.
Data Imputation Methods
If a patient had a PANSS assessment recorded, but any of the 30 items were missing, the last non-missing score for the respective item from previous assessments were to be carried forward. If the total or subscale score had > 30% of the items missing at a particular visit, the respective total or subscale score at the visit would not be calculated and would be treated as missing data in the analysis. With regard to efficacy variables that refer to end point, to account for postbaseline end points occurring before study day 85 a mixed model for repeated measurements was used as the primary method and the estimates from the model at day 85 were used for the primary efficacy analysis. It was assumed that a majority of the missing values were “missing completely at random” or “missing at random.” For overall and PANSS response, the response rate at each visit was presented using 2 differing approaches for a patient that has withdrawn early from treatment: 1) non-responder imputation, and 2) excluded from the analysis following early withdrawal. For health economics and outcomes research (HEOR) outcomes, there was no imputation for the Subjective Well-Being Under Neuroleptics-Short Form (SWN-20) outcome; changes from baseline and distribution of patients across levels of the SWN-20 were calculated based on available data only.55
Subgroup Analyses
The primary and key secondary efficacy analysis were repeated to assess the treatment differences within country and baseline PANSS total score (≥ 95 versus < 95).
Sensitivity Analyses
Sensitivity analyses were conducted to assess the robustness of treatment effects under different assumptions for handling missing data. These analyses utilized a pattern mixture model (PMM) and a multiple imputation (MI) approach to address potential biases. The same CHW and Hommel adjustments were applied across all sensitivity analyses, which were repeated for the ITT, PP, and mPP populations. Weighting for the CHW and Lawrence and Hung (LH) calculations for the modified populations was consistent with the mITT population, while for non-modified populations, it was consistent with the ITT population.
For the mITT population, a sensitivity analysis of the mean change in PANSS total score from baseline to day 85 was performed using observed cases (OC) only, based on the last post-baseline DB assessment. This analysis employed an Analysis of Covariance (ANCOVA) model with country of enrollment and treatment as fixed effects and baseline PANSS total score as a covariate. Similarly, for the modified randomized population, an ANCOVA model was used to analyze the PANSS total score mean change from baseline to day 85, with missing data imputed using an MI approach. Imputed PANSS scores were rounded to the nearest integer within the valid range (30 to 210) and generated following a general three-step PMM framework, as outlined below:
Non-Monotone Missing Data: Missing intermediate visits were imputed first using the Monte Carlo Markov Chain (MCMC) method under the Missing at Random (MAR) assumption across all treatment arms. A multivariate normal model was used for the PANSS total score at baseline and all post-baseline visits within each treatment group, alongside stratification variables (country of enrollment and baseline PANSS score ≥ 95 or < 95). One hundred imputations were generated using the MCMC statement in PROC MI.
Monotone Missing Data: Remaining monotone missing data were imputed under the Missing Not at Random (MNAR) assumption. For withdrawn patients in the Okedi groups (75 mg and 100 mg), correlations with future (post-withdrawal) visits were assumed to align with patterns observed in the placebo group, adjusted for baseline covariates and prior outcomes. Missing data from placebo patients were imputed under the MAR assumption, based on patterns of placebo completers. A regression imputation model for PANSS scores at each time point included randomization stratification factors and prior PANSS scores. Imputations were performed using a sequence of regression-based methods (MONOTONE REG in PROC MI) for each post-baseline time point.
ANCOVA Analysis of Imputed Data: Imputed datasets were analyzed using an ANCOVA model, with country of enrollment and treatment as fixed effects and baseline PANSS total score as a covariate. Results from all imputed datasets were combined using Rubin’s combination rule (PROC MIANALYZE) to generate the final analysis.
Secondary Outcomes of the Studies
PRISMA-3 Secondary Efficacy Outcomes
As per the primary efficacy analysis, the CGI-S was analyzed taking the interim assessment into account by using CHW methodology and the multiplicity of testing into account using Hommel’s closed-testing correction procedure. The interim analysis was conducted when 196 patients for whom the blinding was not compromised reached day 85 or withdrew from the study. Mean change in CGI-S from baseline to day 85, the key secondary variable, was analyzed at day 85 for the mITT and ITT populations in a hierarchical order. Confirmatory testing was only performed sequentially on the mITT and ITT population if the primary efficacy analysis showed superiority of both doses of Okedi versus the placebo treatment at day 85.
Confirmatory sequential testing of the key secondary efficacy outcome was performed on the mITT and ITT populations only if the primary efficacy analysis showed the superiority of the Okedi treatment versus the IM placebo treatment for both doses. Otherwise, all analyses for the key secondary efficacy variable were to be deemed supportive or exploratory evidence. The CGI-S mean change was analyzed using identical methods as described for the primary, sensitivity, and subgroup analyses of the primary efficacy variable. The only exception to this was that the baseline CGI-S was used as a covariate in the MMRM/ANCOVA models instead of the baseline PANSS score.
Secondary efficacy variables were analyzed in the mITT, ITT, and PP populations. Nominal p-values, unadjusted for interim assessments or multiple comparisons, were used, and these results are considered exploratory.
CGI-I Score Analysis
CGI-I scores were summarized at each postbaseline assessment and day 85 using descriptive statistics (n, mean, median, SD, minimum, and maximum), along with distribution across categories by treatment group and visit.55 Analyses employed the MMRM model, with fixed effects for country of enrollment, visit, treatment, and treatment-by-visit interaction, and baseline CGI-S score as a covariate.
Overall and PANSS Response
PANSS response was defined as a ≥ 30% reduction in total score from baseline, while overall response included either PANSS response or a CGI-I score of 2 (much improved) or 1 (very much improved). Response rates were reported by treatment group for each visit and day 85 using frequencies, percentages, and exact 95% confidence intervals (CIs; Clopper-Pearson method). Pairwise comparisons of Okedi doses versus placebo were conducted using a Mantel-Haenszel test, stratified by country and baseline PANSS score (≥ 95 or < 95).
Time to Response
Time to achieve overall or PANSS response was summarized using cumulative frequencies and percentages, with medians and 95% CIs estimated via the Kaplan-Meier method. Non-responders were censored at 85 days. Kaplan-Meier plots were presented by treatment group, and group comparisons were performed using a log-rank test adjusted for randomization stratification factors.
PANSS Subscale Changes
Descriptive statistics (n, mean, median, SD, minimum, and maximum) for PANSS subscale scores and changes from baseline were reported for each postbaseline time point and end point. Analyses followed the MMRM model, with fixed effects for country, visit, treatment, treatment-by-visit interaction, and the corresponding baseline subscale score as a covariate.
PRISMA-3 OLE Efficacy Outcomes
Efficacy assessments, including PANSS and CGI-S, were collected at baseline, while all efficacy scales (PANSS, CGI-S, and CGI-I) were evaluated at all postbaseline time points during the OLE phase. All efficacy analyses in the OLE phase utilized the open-label population (OLP). For rollover patients, the baseline for the OLE phase corresponded to day 85 of the DB phase.
Efficacy variables, including PANSS, PANSS subscale scores, CGI-I, CGI-S, overall response, and PANSS response, were summarized and listed during the OLE phase in a manner consistent with the DB phase. However, no formal statistical testing between treatment groups was conducted during the OLE phase.
HEOR Outcomes
All health economics and outcomes research (HEOR) data were analyzed using the ITT population, ensuring that no patients or measurements were excluded from the analysis. The analysis included all available data from the start to the end of the study. HEOR outcomes included healthcare resource utilization (HCRU), Personal and Social Performance (PSP), and Subjective Well-Being Under Neuroleptics-Short Form (SWN-20).55
The Subjective Well-Being Under Neuroleptics Treatment Scale (SWN) is a 38-item instrument developed to measure subjective effects of neuroleptic medications in patients with schizophrenia and consists of 20 positive statements and 18 negative statements.56 The short form of the SWN, the 20-item SWN-20, was developed in order to allow for quick assessment of subjective side effects in a clinical setting. Like in the original SWN, with the SWN-20 the patient is asked to rate well-being items that have been identified as related to antipsychotic treatment on a 6-point scale ranging from “Not at all” to “Very much.” The SWN-20 is scored on a scale ranging from 20 to 120, with higher scores indicating better health-related quality of life (HRQoL). The SWN-20 contains five 4-item subscales: mental functioning, self-control, emotional regulation, physical functioning, and social integration. Each subscale score ranges from 4 to 24, with higher scores indicating better HRQoL.
The PSP is a 100-point single-item rating scale that is based on 4 areas: family and social functioning, self-care, work and socially useful activities, and disturbing and aggressive behaviours. Higher scores indicate better social functioning.
As the analyses were exploratory and not hypothesis-driven, no adjustments were made to account for multiple testing thresholds of significance. Missing data for select outcomes were documented as a proportion of all responses collected. The analysis method accounted for incomplete data (e.g., partially completed instruments).55
Analysis Populations
SAF
The safety population (SAF) includes all patients who received at least 1 dose of study drug. Analyses performed on the SAF were as treated. The SAF was used for analyses of all safety end points and all patient listings unless specified otherwise.
mSAF
The modified safety population (mSAF) consists of all patients in the SAF for whom blinding was not compromised. Analyses performed on the mSAF were as treated. This population was used for selected safety analyses.
Randomized Population
The randomized population consists of all patients who were randomized in the study. Analyses performed on the randomized population were as randomized. This population was used for sensitivity analysis of the primary variable and for summaries/listings of patient disposition post-screening.
Modified Randomized Population
The modified randomized population consists of all patients in the randomized population for whom blinding was not compromised. This population was used for sensitivity analysis of the primary variable.
ITT Population
The ITT population consists of all randomized patients who received at least 1 dose of study drug with a baseline measurement and ≥ 1 postbaseline evaluation of the PANSS. Analyses performed on the ITT population were as randomized. The ITT population was used for analyses of efficacy end points, including HEOR end points.
mITT Population
The mITT population consists of all patients in the ITT population for whom blinding was not compromised. Analyses performed on the mITT population were as randomized. The mITT population was used for analyses of efficacy end points.
PP Population
The PP population consists of patients from the ITT population who received ≥ 2 injections of study drug (active or placebo) during the DB phase and had no important protocol deviations that may have impacted the primary efficacy variable.
mPP Population
The mPP population consists of all patients in the PP population for whom blinding was not compromised. Analyses performed on the mPP population were as treated. This population was used for selected efficacy analyses.
OLP
The open-label population (OLP) consists of all patients who received at least 1 dose of study drug in the OLE phase of the study and was used for presentation of OLE analyses and listings. Analyses performed on the OLP were as treated, and by DB phase treatment arm/de novo entry at the time of the OLE phase.
Sponsor’s Summary of the Results
Patient Disposition
PRISMA-3
A total of 565 patients were screened for the DB phase, of which 127 (22.5%) were screen failures, and 438 (77.5%) passed screening and were randomized in the DB phase. The most common reasons for failing screening were not meeting an eligibility criterion (81 patients, 14.3%), and withdrawal of consent (37 patients, 6.5%) (Table 8).
Patients were evenly distributed across the treatment groups, with 147 patients in the placebo group, 145 patients in the Okedi 75 mg group, and 146 patients in the Okedi 100 mg group. One patient was assigned treatment (assigned to the Okedi 75 mg group) but did not receive study treatment because consent was withdrawn before receiving a dose of study drug.
Study completion was highest in the Okedi 75 mg group (73.8%), and lowest in the placebo group (59.9%). Study completion in the Okedi 100 mg group was 65.1%. Of the 147 patients who received placebo, 59 patients (40.1%) did not complete the study. The most common reasons for patients in the placebo group not completing the study were withdrawal of consent (20 patients in placebo group, 13.6%) and insufficient clinical response (15 patients, 10.2%). Of the 144 patients who received Okedi 75 mg, 37 patients (25.5%) did not complete the study. The most common reasons were withdrawal of consent (14 patients, 9.7%), insufficient clinical response (6 patients, 4.1%), lost to follow-up (6 patients, 4.1%), and other (6 patients, 4.1%). Of the 146 patients who received Okedi 100 mg, 51 did not complete the study. The most common reasons were withdrawal of consent (23 patients, 15.8%), and insufficient clinical response (9 patients, 6.2%).
Table 8: Patient Disposition – PRISMA-3 (Randomized Population)
Characteristics | Placebo N = 147 | Okedi 75 mg N = 145 | Okedi 100 mg N = 146 | |
|---|---|---|---|---|
Completed DB phase, n (%) | Yes | 88 (59.9) | 107 (73.8) | 95 (65.1) |
No | 59 (40.1) | 37 (25.5) | 51 (34.9) | |
Reason(s) for discontinuation, n (%) | ||||
Adverse event | 3 (2.0) | 5 (3.4) | 5 (3.4) | |
Positive pregnancy or urine drug screen test result | 3 (2.0) | 0 | 2 (1.4) | |
Withdrawal of consent | 20 (13.6) | 14 (9.7) | 23 (15.8) | |
Lost to follow-up | 6 (4.1) | 6 (4.1) | 5 (3.4) | |
Physician decision | 1 (0.7) | 0 | 0 | |
Non-compliance | 1 (0.7) | 0 | 1 (0.7) | |
Hospitalization due to worsening of schizophrenia | 3 (2.0) | 0 | 2 (1.4) | |
Insufficient clinical response | 15 (10.2) | 6 (4.1) | 9 (6.2) | |
Other | 7 (4.8) | 6 (4.1) | 4 (2.7) | |
Analysis Populations, n | Placebo | OKEDI 75 mg | OKEDI 100 mg | |
SAF | 147 | 144 | 146 | |
mSAF | 134 | 130 | 130 | |
ITT | 145 | 143 | 145 | |
mITT | 132 | 129 | 129 | |
PP | 119 | 126 | 121 | |
mPP | 109 | 114 | 112 | |
Screened, N | 565 | |||
Randomized, N | 438 | |||
DB = double-blind; ITT = intent-to-treat; mITT = modified intent-to-treat; mPP = modified per protocol; mSAF = modified safety; PP = per protocol; SAF = safety.
Source: PRISMA-3 Clinical Study Report40
PRISMA-3 OLE
A total of 174 patients signed the informed consent form for the OLE phase, of which 44 patients were de novo patients (did not take part in the DB phase). Three of these patients failed screening, and the rest continued into the OLE (Table 9).
A total of 215 patients received at least 1 dose in the OLE phase (OLP). Of these 215 patients, 161 patients (74.9%) completed the OLE phase and 54 did not complete the OLE phase. The most common reasons for the 54 patients (25.1%) not completing the OLE phase were withdrawal of consent (26 patients, 12.1%), adverse events (AEs) (7 patients, 3.3%), hospitalization for worsening, relapse, or exacerbation of schizophrenia symptoms (6 patients, 2.8%) and lost to follow-up (5 patients, 2.3%). Other reasons for not completing were insufficient clinical response (4 patients, 1.9%), other (3 patients, 1.4%), and death, positive pregnancy or urine drug screen test result, or noncompliance (each 1 patient, 0.5%). Median duration in the OLE phase was 365 days for all groups.
Table 9: Patient Disposition – PRISMA-3 OLE (OLP)
Characteristics | Rollover | De Novo | Total N = 215 | ||||||
|---|---|---|---|---|---|---|---|---|---|
Placebo/ Okedi 75 mg N = 27 | Okedi 75mg/ Okedi 75 mg N = 58 | Okedi 100 mg/ Okedi 100 mg N = 61 | Placebo/ Okedi 100 mg N = 28 | Okedi 75 mg N = 31 | Okedi 100 mg N = 10 | ||||
Completed OLE phase, n (%) | Yes | 21 (77.8) | 43 (74.1) | 44 (72.1) | 20 (71.4) | 26 (83.9) | 7 (70.0) | 161 (74.9) | |
No | 6 (22.2) | 15 (25.9) | 17 (27.9) | 8 (28.6) | 5 (16.1) | 3 (30.0) | 54 (25.1) | ||
Reason(s) for discontinuation, n (%) | |||||||||
Adverse event | 1 (3.7) | 3 (5.2) | 2 (3.3) | 1 (3.6) | 0 | 0 | 7 (3.3) | ||
Positive pregnancy or urine drug screen test result | 0 | 0 | 1 (1.6) | 0 | 0 | 0 | 1 (0.5) | ||
Withdrawal of consent | 2 (7.4) | 6 (10.3) | 8 (13.1) | 6 (21.4) | 3 (9.7) | 1 (10.0) | 26 (12.1) | ||
Lost to follow-up | 0 | 3 (5.2) | 2 (3.3) | 0 | 0 | 0 | 5 (2.3) | ||
Death | 0 | 1 (1.7) | 0 | 0 | 0 | 0 | 1 (0.5) | ||
Administrative reasons | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
Protocol deviation | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
Physician decision | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
Pregnancy | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
Non-compliance | 1 (3.7) | 0 | 0 | 0 | 0 | 0 | 1 (0.5) | ||
Development of a medical condition that requires treatment with a prohibited medication | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
Hospitalization for worsening, relapse, or exacerbation of schizophrenia symptoms | 1 (3.7) | 1 (1.7) | 2 (3.3) | 0 | 1 (3.2) | 1 (10.0) | 6 (2.8) | ||
Insufficient clinical response | 1 (3.7) | 1 (1.7) | 1 (1.6) | 0 | 1 (3.2) | 0 | 4 (1.9) | ||
Termination of the study | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
Other | 0 | 0 | 1 (1.6) | 1 (3.6) | 0 | 1 (10.0) | 3 (1.4) | ||
Analysis Population | Okedi 75 mg | Okedi 100 mg | Overall | ||||||
OLP, N | 116 | 99 | 215 | ||||||
Screened, N | 218 | ||||||||
Assigned/ Randomized, N | 215 | ||||||||
OLE = open-label extension; OLP = open-label population.
Source: PRISMA-3 Clinical Study Report40
Exposure to Study Treatments
Study Treatments
PRISMA-3
The majority of patients in all 3 treatment groups received 3 doses of study medication (63.3% of patients in the placebo group, 77.8% in the Okedi 75 mg group, 67.8% in the Okedi 100 mg group) over the 12-week study period. In all treatment groups, more patients received one dose than 2 doses (Table 10).
Mean exposure to Okedi was 194.3 mg (SD = 59.11) in the Okedi 75 mg group and 245.9 mg (SD = 82.35) in the Okedi 100 mg group over 12 weeks. Respective median values were 225.0 mg (range, 75 to 225) and 300.0 mg (range, 100 to 300).40
Table 10: Treatment Exposure – PRISMA-3 (SAF)
Treatment exposure | Placebo N = 147 | Okedi 75 mg N = 144 | Okedi 100 mg N = 146 | |
|---|---|---|---|---|
Number of IM doses, n (%) | 1 | 38 (25.9) | 27 (18.8) | 31 (21.2) |
2 | 16 (10.9) | 5 (3.5) | 16 (11.0) | |
3 | 93 (63.3) | 112 (77.8) | 99 (67.8) | |
Total exposure to OKEDI, n (%) | Mean (SD) | NA | 194.3 (59.11) | 245.9 (82.35) |
Median (min, max) | NA | 225.0 (75, 225) | 300.0 (100, 300) | |
IM = intramuscular NA = not applicable; SAF = safety population; SD = standard deviation.
Source: PRISMA-3 Clinical Study Report40
PRISMA-3 OLE
During the OLE phase, 215 patients have been exposed to treatment with Okedi, either 75 mg or 100 mg. In the OLE phase, patients received between 1 and 13 doses of Okedi 75 mg and 100 mg over 12 months (Table 11). The majority of 164 patients (76.3%) in the OLP received 13 doses of Okedi, of which Okedi 75 mg was administered to 92 patients (79.3%) and Okedi 100 mg to 72 patients (72.7%). The total number of Okedi doses was 2358 (Okedi 75 mg: 1303 doses, Okedi 100 mg: 1055 doses).
Median exposure to Okedi 75 mg was 975.0 mg (range, 75 mg to 975 mg) and 1300.0 mg (range, 100 mg to 1300 mg) for Okedi 100 mg during the OLE phase. The respective mean values were 842.5 mg (SD = 278.24) in the Okedi 75 mg group and 1065.7 mg (SD = 425.02) in the Okedi 100 mg group.40
Table 11: Treatment Exposure – PRISMA-3 OLE (OLP)
Treatment exposure | Okedi 75 mg N = 116 | Okedi 100 mg N = 99 |
|---|---|---|
Number of IM doses, N | 1303 | 1055 |
Number of IM doses during OLE phase, n (%) | ||
1 | 5 (4.3) | 5 (5.1) |
2 | 3 (2.6) | 6 (6.1) |
3 | 2 (1.7) | 3 (3.0) |
4 | 1 (0.9) | 5 (5.1) |
5 | 7 (6.0) | 0 |
6 | 1 (0.9) | 2 (2.0) |
7 | 0 | 1 (0) |
8 | 2 (1.7) | 0 |
9 | 1 (0.9) | 0 |
10 | 2 (1.7) | 3 (3.0) |
11 | 0 | 0 |
12 | 0 | 2 (2.0) |
13 | 92 (79.3) | 72 (72.7) |
Total exposure to Okedi, n (%) | ||
Mean (SD) | 842.5 (278.24) | 1065.7 (425.02) |
Median (min, max) | 975.0 (75, 975) | 1300.0 (100, 1300) |
IM = intramuscular (injection); OLE = open-label extension; OLP = open-label population; SD = standard deviation.
Source: PRISMA-3 Clinical Study Report40
Concomitant Medications
PRISMA-3
Concomitant medications were taken by 56.5% of overall patients during the PRISMA-3 study. The most common Anatomical Therapeutic Chemical (ATC) Class 3 groups were anxiolytics (37.8% of overall patients), anti-inflammatory and antirheumatic products, non-steroids (14.9%), and other analgesics and antipyretics (10.3%). The most common preferred terms (PTs) were lorazepam (34.6% of overall patients), ibuprofen (13.7%), paracetamol (9.2%), and lisinopril (8.0%).
PRISMA-3 OLE
Concomitant medications were taken by 61.9% of the OLP. The most common ATC Class 3 groups were local anesthetics (51 patients, 23.7%), mydriatics and cycloplegics (27 patients, 12.6%) other analgesics and antipyretics (27 patients, 12.6%), anti-infectives (24 patients, 11.2%), anxiolytics (17 patients, 7.9%), angiotensin-converting enzyme inhibitors, plain (16 patients, 7.4%), anti-inflammatory and antirheumatic products, non-steroids (15 patients, 7.0%), and blood glucose lowering drugs, excluding insulins (14 patients, 6.5%). The most common PTs were proxymetacaine (39 patients, 18.1%), tropicamide (25 patients, 11.6%), silver protein (22 patients, 10.2%), paracetamol (21 patients, 9.8%), metformin (12 patients, 5.6%), and oxybuprocaine (12 patients, 5.6%).
Efficacy
PRISMA-3
Primary Efficacy Outcomes
The primary efficacy variable was PANSS total score mean change from baseline to end point (defined as study day 85 or the last postbaseline DB assessment; Table 12).40 The mean PANSS score at baseline was 96.4 (SD = 7.21) for the placebo group, 96.3 (SD = 8.47) for the Okedi 75 mg group, and 96.1 (SD = 8.42) for the Okedi 100 mg group. The mean change from baseline to day 85 was −11.0 (95% CI, −14.1 to −8.0) for the placebo group, −24.6 (95% CI, −27.5 to −21.6) for the Okedi 75 mg group, and −24.7 (95% CI, −27.7 to −21.6) for the Okedi 100 mg group. The LH mean difference from the placebo group was −13.0 (95% CI, −17.3 to −8.8) for the OKEDI 75 mg group, and −13.3 (95% CI, −17.6 to −8.9) for the Okedi 100 mg group (Hommel adjusted p-value < 0.0001 for both groups). Statistical superiority of both doses of Okedi compared to placebo was demonstrated for the primary end point.40 Analyses of other populations (Table 12), sensitivity analyses (Table A 1, Table A 2), and subgroup analyses (Table A 3) were consistent with and support the primary analysis.
Table 12: PANSS Total Score Mean Change From Baseline to Day 85 (mITT, ITT, mPP, and PP Populations)
Outcome | Placebo | Okedi 75 mg | Okedi 100 mg | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
mITTa N = 132 | ITT N = 145 | mPP N = 109 | PP N = 119 | mITTa N = 129 | ITT N = 143 | mPP N = 114 | PP N = 126 | mITTa N = 129 | ITT N = 145 | mPP N = 112 | PP N = 121 | ||
LSM (SE; 95% CI) | -11.0 (1.56; −14.1 to −8.0) | -11.8 (1.48; −14.7 to −8.9) | -11.0 (1.64; −14.3 to −7.8) | -11.8 (1.57; −14.9 to −8.7) | -24.6 (1.51; −27.5 to −21.6) | -23.9 (1.44; −26.7 to −21.0) | -24.5 (1.56; −27.5 to −21.4 | -23.7 (1.50; −26.6 to −20.7) | -24.7 (1.54; −27.7 to −21.6) | -24.5 (1.46; −27.4 to −21.6) | -25.1 (1.59; −28.2 to −22.0) | -24.7 (1.52; −27.7 to −21.7) | |
OKEDI vs Placebo | |||||||||||||
LSMD (SE; 95% CI) | NA | NA | NA | NA | -13.6 (2.17; −17.8 to −9.3) | -12.0 (2.06; −16.1 to −8.0) | -13.4 (2.27; −17.9 to −9.0) | -11.8 (2.17; −16.1 to −7.6) | -13.6 (2.19; −17.9 to −9.3) | -12.7 (2.08; −16.8 to −8.6) | -14.1 (2.29; −18.6 to −9.5) | -12.9 (2.19; −17.2 to −8.6) | |
P-value | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | |
LH MD (SE; 95% CI)b | NA | NA | NA | NA | -13.0 (2.19; −17.3 to −8.8) | -11.6 (2.07; −15.7 to −7.5) | -13.1 (2.30; −17.6 to −8.6) | -11.5 (2.18; −15.8 to −7.2) | -13.3 (2.21; −17.6 to −8.9) | -12.3 (2.09; −16.4 to −8.2) | -13.6 (2.32; −18.2 to −9.1) | -12.4 (2.20; −16.7 to −8.1) | |
CHW-adjusted P-valueb | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | |
Hommel-adjusted P-valuec | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | |
CHW = Cui, Hung, Wang; CI = confidence interval; ITT = intent-to-treat; LH = Lawrence and Hung; MD = mean difference; mITT = modified intent-to-treat; mPP = modified per protocol; PP = per protocol; LSM = least squares mean; LSMD = least squares mean difference; PANSS = Positive and Negative Syndrome Scale; SE = standard error.
aPrimary analysis
bCHW and LH methods combine results from stage 1 and stage 2
cHommel adjustment of CHW p-values
Source: PRISMA-3 Clinical Study Report40
Key Secondary Efficacy Outcome
The mean CGI-S score at baseline was 4.9 (SD = 0.54) for the placebo group, 4.9 (SD = 0.63) for the Okedi 75 mg group, and 4.8 (SD = 0.53) for the Okedi 100 mg group. The mean change from baseline at day 85 was −0.6 (95% CI, −0.8 to −0.4) for the placebo group, −1.3 (95% CI, −1.5 to −1.2) for the Okedi 75 mg group, and −1.3 (95% CI, −1.5 to −1.2) for the Okedi 100 mg group. The LH mean difference from the placebo group was −0.7 (95% CI, −1.0 to −0.5) for the Okedi 75 mg group, and −0.7 (95% CI, −1.0 to −0.5) for the Okedi 100 mg group (Hommel adjusted p-value < 0.0001 for both groups).
As statistical superiority of both doses of Okedi versus placebo had been demonstrated for the primary efficacy analysis, confirmatory testing was continued for the key secondary analysis. Statistical superiority of both doses of Okedi treatment versus placebo was also demonstrated for the key secondary end point.40 Analyses conducted in all study populations (Table 13) were consistent.
Table 13: CGI-S Change From Baseline to Day 85 (mITT, ITT, mPP, and PP Populations)
MRMM | Placebo | Okedi 75 mg | Okedi 100 mg | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
mITT N = 132 | ITT N = 145 | mPP N = 109 | PP N = 119 | mITT N = 129 | ITT N = 143 | mPP N = 114 | PP N = 126 | mITT N = 129 | ITT N = 145 | mPP N = 112 | PP N = 121 | |
LSM (SE; 95% CI) | -11.0 (1.56; −14.1 to −8.0) | -11.8 (1.48; −14.7 to −8.9) | -11.0 (1.64; −14.3 to −7.8) | -11.8 (1.57; −14.9 to −8.7) | -24.6 (1.51; −27.5 to −21.6) | -23.9 (1.44; −26.7 to −21.0) | -24.5 (1.56; −27.5 to −21.4 | -23.7 (1.50; −26.6 to −20.7) | -24.7 (1.54; −27.7 to −21.6) | -24.5 (1.46; −27.4 to −21.6) | -25.1 (1.59; −28.2 to −22.0) | -24.7 (1.52; −27.7 to −21.7) |
OKEDI vs Placebo | ||||||||||||
LSMD (SE; 95% CI) | NA | NA | NA | NA | -13.6 (2.17; −17.8 to −9.3) | -12.0 (2.06; −16.1 to −8.0) | -13.4 (2.27; −17.9 to −9.0) | -11.8 (2.17; −16.1 to −7.6) | -13.6 (2.19; −17.9 to −9.3) | -12.7 (2.08; −16.8 to −8.6) | -14.1 (2.29; −18.6 to −9.5) | -12.9 (2.19; −17.2 to −8.6) |
P-value | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
LH MD (SE; 95% CI)a | NA | NA | NA | NA | -13.0 (2.19; −17.3 to −8.8) | -11.6 (2.07; −15.7 to −7.5) | -13.1 (2.30; −17.6 to −8.6) | -11.5 (2.18; −15.8 to −7.2) | -13.3 (2.21; −17.6 to −8.9) | -12.3 (2.09; −16.4 to −8.2) | -13.6 (2.32; −18.2 to −9.1) | -12.4 (2.20; −16.7 to −8.1) |
CHW-adjusted P-valuea | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
Hommel-adjusted P-valueb | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
CGI-S = Clinical Global Impression-Severity; CHW = Cui, Hung, Wang; CI = confidence interval; ITT = intent-to-treat; LH = Lawrence and Hung; mITT = modified intent-to-treat; mPP = modified per protocol; PP = per protocol; LSM = least squares mean; LSMD = least squares mean difference; MD = mean difference; MMRM = mixed model with repeated measures; SE = standard error.
aCHW and LH methods combine results from stage 1 and stage 2
bHommel adjustment of CHW p-values
Source: PRISMA-3 Clinical Study Report40
Other Secondary Efficacy End Points
Data for PRISMA-3’s other secondary efficacy outcomes are presented in the Appendix (Figure 3, Figure 4, Table 24, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9). Statistically significant improvements in PANSS total score were observed as early as day 8 for Okedi 100 mg (LS means difference = −3.9; 95% CI, −6.4 to −1.5; p = 0.001) and by day 15 for Okedi 75 mg (LS means difference = −6.0; 95% CI, −9.0 to −2.9; p < 0.0001), as presented in Figure 1. These differences remained significant through day 85 (refer to the primary end point results). Similarly, placebo-adjusted CGI-S score improvements were statistically significant from day 8 for Okedi 75 mg (LS means difference = █████ ███ ███ ████ ██ █████ █████) and Okedi 100 mg (LS means difference = █████ ███ ███ ████ ██ █████ █████) and onward through day 85 (refer to the key secondary end point results). The PANSS positive subscale score mean change from baseline at day 85 was −4.1 (95% CI, −5.1 to −3.1) for the placebo group, −8.0 (95% CI, −9.0 to −7.1) for the Okedi 75 mg group, and −8.7 (95% CI, −9.7 to −7.8) for the Okedi 100 mg group. The LS means difference from placebo was −3.9 (95% CI, −5.3 to −2.5; p < 0.0001) for the Okedi 75 mg group and −4.6 (95% CI, −6.0 to −3.2; p < 0.0001) for the Okedi 100 mg group. At day 85, the PANSS negative subscale score mean change from baseline was −1.7 (95% CI, −2.5 to −0.9) for the placebo group, −3.8 (95% CI, −4.5 to −3.0) for the Okedi 75 mg group, and −3.7 (95% CI, −4.4 to −2.9) for the Okedi 100 mg group. The LS means difference from placebo was −2.1 (95% CI, −3.1 to −1.0; p < 0.001) for the Okedi 75 mg group and −2.0 (95% CI, −3.1 to −0.9; p < 0.001) for the Okedi 100 mg group. The PANSS general psychopathology subscale score mean change from baseline at day 85 was −5.6 (95% CI, −7.2 to −4.0) for the placebo group, −12.8 (95% CI, −14.4 to −11.3) for the Okedi 75 mg group, and −12.4 (95% CI, −14.0 to −10.8) for the Okedi 100 mg group. The LS means difference from placebo was −7.3 (95% CI, −9.5 to −5.0; p < 0.0001) for the Okedi 75 mg group and −6.8 (95% CI, −9.1 to −4.6; p < 0.0001) for the Okedi 100 mg group.40
CGI-I scores demonstrated statistically significant improvements starting on day 8, with LS mean differences at day 85 of −0.8 (95% CI, −1.0 to −0.5; p < 0.0001) for Okedi75 mg and −0.7 (95% CI, −1.0 to −0.4; p < 0.0001) for Okedi 100 mg compared to placebo. At day 85, overall response rate was 18.2% (95% CI, 12.0 to 25.8) in the placebo group, 53.5% (95% CI, 44.5 to 62.3) in the Okedi 75 mg group, and 46.5% (95% CI, 37.7 to 55.5) in the Okedi 100 mg group. The differences in proportions from placebo for overall response rate were 35.3% (95% CI, 23.7 to 45.2; p < 0.0001) for Okedi 75 mg and 28.3% (95% CI, 16.7 to 38.3; p < 0.0001) for Okedi 100 mg. The first statistically significant difference in overall response rate occurred on day 8 for Okedi 100 mg (difference in proportion = 11.8%; 95% CI, 3.3 to 20.5; p = 0.005) and day 15 for Okedi 75 mg (difference in proportion = 22.0%; 95% CI, 12.1 to 31.5; p < 0.0001).40
PRISMA-3 OLE
PANSS Total Score Change From Baseline
During the OLE phase, the overall OLP had a mean baseline PANSS total score of ████ ███ █ ███████ ██ ███ █████ ██████ ███ ███ ██ ███ ████ ████████████ ███ ████████████ ███ ████ █████ █████ █████ ██████ ███ █████ ███ █ ██████ ███ ███ ████ █████ ███ █ ██████ ███ ███ ██ ██ ██████ ███ █████ ███ █ ██████ ███ ███ ███ ██ ██████. The largest improvements were noted in rollover patients treated with placebo, while de novo patients exhibited the smallest improvements. No formal statistical testing was done for the OLE.40
PANSS Subscale Score Change From Baseline
PANSS subscale scores (positive, negative, general psychopathology) decreased consistently across dose groups, indicating improvement. At end point, the mean changes for positive subscale scores were ████ ███ █ █████ ███ ███ ████ ████ ███ █ █████ ███ ███ ██ ██ ██████ ███ ████ ███ █ █████ ███ ███ ███ ██ ██████ ████████ ████████ ████ ███████ ████ ████ ███ █ █████ ███ ███ ████ ████ ███ █ █████ ███ ███ ██ ██ ██████ ███ ████ ███ █ █████ ███ ███ ███ ██ ██████ ███████ ███████████████ ████████ ███████ ████ ████ ███ █ █████ ███ ███ ████ ████ ███ █ █████ ███ ███ ██ ██ ██████ ███ ████ ███ █ █████ ███ ███ ███ ██ ██████40
CGI-S Change From Baseline
CGI-S scores improved across all groups, with baseline mean scores of ███ ███ █ █████ ███ ███ ████ ███ ███ █ █████ ███ ███ ██ ██ ██████ ███ ███ ███ █ █████ ███ ███ ███ ██ ██████ ██ ███ ██████ ███ ████ ███████ ████ ████ ███ █ █████ ███ ███ ████ ████ ███ █ █████ ███ ███ ██ ██ ██████ ███ ████ ███ █ █████ ███ ███ ███ ██ ██████40
CGI-I Score at Each Time Point
The mean CGI-I score for the OLP at day 29 of the OLE phase was ███ ███ █ █████ ███ ███ ████ ███ ███ █ █████ ███ ███ ██ ██ ██████ ███ ███ ███ █ █████ ███ ███ ███ ██ ██████ ██ ███ ██████ ███ ████ █████ █████ ███ ███ ███ █ █████ ███ ███ ████ ███ ███ █ █████ ███ ███ ██ ██ █████ ███ ███ ███ █ █████ ███ ███ ███ ██ ██████. As observed for other efficacy variables described before, the greatest improvement (lowest CGI-I scores at end point) was found in rollover patients treated with placebo, and the smallest improvement in de novo patients.40
Overall Response at Each Time Point
The overall response rate (ORR) at end point in the OLP was ██████ ████ ██ ████████ █████████ ███ ████████ ██ █████ █████ █████ ██ █ █████ █████ ██ █ ██ ██████ ████████ ██ ██████████ ██ ███ ██ ██ █████ ████ █ ██████ ███ ███ ████ ██ █████ ███ ██ ██████████ ██ ███ ███ ██ █████ ████ █ ██████ ███ ███ ████ ██ ██████ ORR was highest among rollover patients who had previously received placebo and lowest among de novo patients.40
PANSS Response at Each Time Point
At end point in the OLE phase, a PANSS response defined as ≥ 30% decrease from baseline in the PANSS total score was achieved by ██ ████████ ██ ███ ████ ████ ████████ ██ ████████ ██ ███ █████ ██ ██ █████ ███ ██ ████████ ██ ███ █████ ███ ██ ██████ ███ █████ ████████ ████ ███ █████ ████ ███ ████ ██ █████ ███ ███ ████ █████ ████ ███ ████ ██ █████ ███ ███ █████ ██ ██ █████ ███ █████ ████ ███ ███ ██ █████ ███ ███ █████ ███ ██ ██████. Similar to ORR, PANSS response rates were highest in the rollover patients treated with placebo, and lowest in de novo patients.40
Relapse in OLE Phase
Relapse during the OLE phase, defined as a ≥ 30% increase in PANSS total score, rehospitalization for psychotic symptoms, or use of adjunctive antipsychotics, occurred in ██ █████████ ██ ██ ███ ██ ███ █████ ██ ██ █████ ███████ ███ ███ ███ ██ █████ ███ ██ ██ ██ ██ ███ ███ ██ █████ ███████ ███ ███ ███ ██ ██████ The overall relapse rate in the OLP (N = 215) was 10.7% (95% CI, 6.9 to 15.6).40
Remittance in OLE Phase
Remittance, defined as a PANSS score of ≤ 3 on 8 key items for at least 6 months, was observed after 365 days or less of treatment. Kaplan-Meier estimated remittance rates were █████ ████ ███ ████ ██ █████ ██ ███ ███████ ████ █████ ████ ███ ████ ██ █████ ██ ███ █████ ██ ██ ██████ ███ █████ ████ ███ ███ ██ ███ █████ ███ ██ █████ (Figure 10).40
Time to Discontinuation in OLE Phase
Kaplan-Meier analysis showed early treatment discontinuation rates after 365 days or less of treatment were █████ ████ ███ ███ ██ ███ ███████ ████ █████ ██ ███ █████ ██ ██ █████ ████ ███ ████ ███ █████ ██ ███ █████ ███ ██ █████ ████ ███ ███ (Figure 11). ███ ██████ ████ ██ ███████████████ ███ ███ ████ ████ ███ ███ ██ ███ ███ ███ ███ ███ ███ █████ ██ ██ ██████ █████ ███ ██████ ███ ██ ███ ███ █████ ███ ██ ██████40
HEOR Outcomes
The highest incidence of HCRU was reported for outpatient hospital services, with rates of █████ ███ █████ ██████ ███ █ ██████ ██ ███ ██ █████ ███ █████ ███ █████ ██████ ███ █ ██████ ██ ███ ███ █████ ███ ████████ ██ ███████ ███ ███ ███ ████████████.
The mean total healthcare costs per patient per 3 months with hospitalization were ███████ ██ ███ ██████ ██████ ███ ██████ █████████ ███████ █████ ██ ████████ ███████ ████████████████ ███ ████ █████ ████ ██████ ███ ███ ████ █████████████
In the ITT population, the mean change in PSP total score from baseline to the end of the DB phase (visit 14) was █████ ████ ███ █ ██ ███ ███ ███ █████ █████ ███ ████ ████ ███ █ ██ ██ ███ ███ ███████ ██████ ███ ███ ██████████ ██ █████ ██████ ████████ ███████ ██ ███ ███ ██████ ███ ████ ███ █████ █████ ██████ ████ ████████ ██ ███ ███ ██ ███ ███ █████ ██████ ███ ███ ████ ████ ███ ██ ██ ██ ███████ ███ ███ ██ ████ ██████ ████ ████ ███ █ ██ ██ ███████ ███ ███ █████████ ██████ ███ ████ ████ ███ █ ██ ███ ███████ ███ ███ ███████████ ██████ █ ██████ █████████ ███ ███ ███ █████ ███ ███ ████████████ ███████████ █████████ ██ ███ ██ ███ ███ ███████40
The mean change in SWN-20 total score from baseline to the end of the DB phase (visit 14) was ███ ████ ███ █ ██ ███ ███ ███ █████ █████ ███ ████ ████ ███ █ ██ ██ ███ ███ ███████ ██████ ███ ███ ██████████ ██ █████ ██████ ████████ ███████ ██████ ███ ███ ██████ ███ ████ ██████ █████ █████ ██████ ████ ████████ ██ ███ ███ ██ ███ ███ █████ ██████ ███ ███ ████ ████ ███ ██ ██ ██ ███████ ███ ███ ██ ████ ██████ ████ ████ ███ █ ██ ██ ███████ ███ ███ █████████ ██████ ███ ████ ████ ███ █ ██ ███ ███████ ███ ███ ███████████ ██████ █ ██████ █████████ ███ ███ ███ █████ ███ ███ ████████████ ███████████ █████████ ██ ███ ██ ███ ███ ███████40
Harms
Safety Evaluation Plan
Safety and tolerability were assessed based on the incidence of treatment-emergent adverse events (TEAEs), adverse events leading to discontinuation, vital sign measurements, physical examination findings, weight changes, abnormal laboratory test results, ECG findings, and abnormal movement scale results (Abnormal Involuntary Movement [AIMS], Barnes Akathisia Rating Scale [BARS], and Simpson-Angus Scale [SAS]). Additional assessments included C-SSRS results, use of concomitant medications, and injection site reactions. Time to early termination was also summarized descriptively.
Overview of Safety
During the 12-week DB and 12-month OLE phases, at least 1 TEAE was reported for ███ ████████ ███████ ██ ███ ███ Table 14 ██ ███ ████████ ███████ ████████ ██ █████ ████ ████ ███ ██ ████████ ██████ ████████ █ ██████ █████ ██ █████ █ █████████ ████ ███ ████████ ███ ███ ████████ ████████ ██ █████ █ ███████ ███████ █████ █████ ███ ████████ ███ ██ ████████ ███████ █ ████ ███ ██ █████ ████ ███████████████ ███ ██ ████████ ██████ ███ ████████ ██ █████ ███ █ ███████ ██████ ██ ███ ███ ███████████40
Table 14: Overall Summary of TEAEs – PRISMA-3 and PRISMA-3 OLE (SAF)
Characteristics | Okedi 75 mg N = 202 | Okedi 100 mg N = 184 | All Okedi N = 386 | ||||
|---|---|---|---|---|---|---|---|
n (%) | AEs, n | n (%) | AEs, n | n (%) | AEs, n | ||
Patients with ≥ 1 TEAE | ███ ██████ | ███ | █████ | ███ | ███ ██████ | ███ | |
Severity | Mild | ██ ██████ | █████ | █████ | ███ | ███ ██████ | ███ |
Moderate | ██ ██████ | █████ | █████ | ██ | ██ ██████ | ███ | |
Severe | █████ | █████ | █████ | █ | █████ | ██ | |
Patients with ≥ 1 serious TEAE | ████ | ██ | █████ | ██ | █████ | ██ | |
Patients with at ≥ 1TEAE leading to treatment discontinuation | ██████ | ██ | █████ | ██ | █████ | ██ | |
Patients with at ≥ 1 TEAE leading to death | █████ | █ | █ | █ | ████ | █ | |
AE = adverse event; SAF = safety population; TEAE = treatment-emergent adverse event.
Source: PRISMA-3 Clinical Study Report40
Adverse Events
At least 1 TEAE was reported for ███ ████████ ███████ in the SAF during the DB and OLE phases (Table 15). The most commonly reported TEAEs were headache (█████ of patients), hyperprolactinemia (█████), blood prolactin increased (████), nasopharyngitis and weight increased (████ ████), insomnia (████), and akathisia (████).
Table 15: Summary of TEAEs – PRISMA-3 and PRISMA-3 OLE (SAF)
TEAE | Okedi 75 mg N = 202 | Okedi 100 mg N = 184 | All Okedi N = 386 | |||
|---|---|---|---|---|---|---|
n (%) | AEs, n | n (%) | AEs, n | n (%) | AEs, n | |
Patients with at least one TEAE | ███ ██████ | ███ | ███ ██████ | ███ | ███ ██████ | ███ |
Endocrine disorders | ██ █████ | ██ | ██ ██████ | ██ | ██ ██████ | ██ |
Hyperprolactinemia | ██ █████ | ██ | ██ ██████ | ██ | ██ ██████ | ██ |
Gastrointestinal disorders | ██ ██████ | ██ | ██ █████ | ██ | ██ ██████ | ██ |
Toothache | █ █████ | ██ | █ █████ | █ | ██ █████ | ██ |
General disorders and administration site conditions | ██ ██████ | ██ | ██ █████ | ██ | ██ █████ | ██ |
Asthenia | █ █████ | ██ | █ █████ | █ | ██ █████ | ██ |
Injection site pain | █ █████ | ██ | █ █████ | █ | ██ █████ | ██ |
Infections and infestations | ██ ██████ | ██ | ██ ██████ | ██ | ██ ██████ | ██ |
Nasopharyngitis | ██ █████ | ██ | ██ █████ | ██ | ██ █████ | ██ |
Investigations | ██ ██████ | ██ | ██ ██████ | ██ | ██ ██████ | ███ |
Alanine aminotransferase increased | █ █████ | █ | ██ █████ | ██ | ██ █████ | ██ |
Aspartate aminotransferase increased | █ █████ | █ | █ █████ | █ | █ █████ | █ |
Blood creatine phosphokinase increased | █ █████ | █ | █ █████ | █ | █ █████ | ██ |
Blood prolactin increased | ██ █████ | ██ | ██ ██████ | ██ | ██ █████ | ██ |
Weight increased | ██ █████ | ██ | ██ █████ | ██ | ██ █████ | ██ |
Nervous system disorders | ██ ██████ | ██ | ██ ██████ | ██ | ███ ██████ | ███ |
Akathisia | █ █████ | ██ | ██ █████ | ██ | ██ █████ | ██ |
Dizziness | █ █████ | █ | █ █████ | █ | ██ █████ | ██ |
Headache | ██ ██████ | ██ | ██ ██████ | ██ | ██ ██████ | ███ |
Somnolence | █ █████ | █ | █ █████ | █ | ██ █████ | ██ |
Psychiatric disorders | ██ ██████ | ██ | ██ ██████ | ██ | ██ ██████ | ██ |
Anxiety | █ █████ | █ | █ █████ | █ | ██ █████ | ██ |
Insomnia | ███████ | ██ | █████ | ██ | ██ █████ | ██ |
Schizophrenia | █████ | ██ | █████ | ██ | ██ █████ | ██ |
AE = adverse event; OLE = open-label extension; SAF = safety population; TEAE = treatment-emergent adverse event.
Source: PRISMA-3 Clinical Study Report40
Serious Adverse Events
During the entire study (DB and OLE study phases), ██ ████████ ██ ███ ███ ██████ ████████ ██ █████ █ ███████ ████ ██████████ ███ █ █████ ██ ██ ██████ Table 16 ██ █████████████ ███ █████████ █████ ███ ███ ████ ████████ ████████ ████ ██████████ ███ ██ ███████ █████ ████ ████ ████████ ███ █ ███████ ████ ██████ ██ ███ ████
Table 16: Summary of Serious TEAEs – PRISMA-3 and PRISMA-3 OLE (SAF)
TEAE | Okedi 75 mg N = 202 | Okedi 100 mg N = 184 | All Okedi N = 386 | |||
|---|---|---|---|---|---|---|
n (%) | AEs, n | n (%) | AEs, n | n (%) | AEs, n | |
Patients with at least one serious TEAE | █ ████ | ██ | ██ █████ | ██ | ██ █████ | ██ |
Infections and infestations | █ ████ | █ | █ ████ | █ | █ ████ | █ |
Appendicitis | █ ████ | █ | █ | █ | █ ████ | █ |
Skin infection | █ | █ | █ ████ | █ | █ ████ | █ |
Injury, poisoning and procedural complications | █ ████ | █ | █ ████ | █ | █ ████ | █ |
Fall | █ | █ | █ ████ | █ | █ ████ | █ |
Humerus fracture | █ | █ | █ ████ | █ | █ ████ | █ |
Intentional overdose | █ ████ | █ | █ | █ | █ ████ | █ |
Psychiatric disorders | █ ████ | █ | █ ████ | █ | ██ █████ | ██ |
Agitation | █ | █ | █ ████ | █ | █ ████ | █ |
Completed suicide | █ ████ | █ | █ | █ | █ ████ | █ |
Insomnia | █ ████ | █ | █ | █ | █ ████ | █ |
Schizophrenia | █ ████ | █ | █ ████ | █ | ██ █████ | ██ |
Suicidal ideation | █ ████ | █ | █ | █ | █ ████ | █ |
Respiratory, thoracic and mediastinal disorders | █ | █ | █ ████ | █ | █ ████ | █ |
Chronic obstructive pulmonary disease | █ | █ | █ ████ | █ | █ ████ | █ |
AE = adverse event; OLE = open-label extension; SAF = safety population; TEAE = Treatment-emergent adverse event
Source: PRISMA-3 Clinical Study Report40
Withdrawals Due to Adverse Events
During the entire study (DB and OLE study phases), a TEAE resulted in permanent study drug discontinuation for ██ ████████ ██████ ██ ███ ███ Table 17 ██ █████████████ ███ █████████ █████ ███ ███ ████ ████████ ████████ ████ ████ ████████ ██ █████ ████ ████████████████ ███ █████ █████ ████ ████████ ██ █████ ████ ███████████████ ████ ████████ ███ █ ███████ ████ ██████ ██ ███ ████.
Table 17: Summary of TEAEs Leading to Study Drug Discontinuation – PRISMA-3 and PRISMA-3 OLE (SAF)
TEAE | Okedi 75 mg N = 202 | Okedi 100 mg N = 184 | All Okedi N = 386 | |||
|---|---|---|---|---|---|---|
n (%) | AEs, n | n (%) | AEs, n | n (%) | AEs, n | |
Patients with at least one TEAE | █ █████ | ██ | █ █████ | ██ | █ █████ | ██ |
Hepatobiliary disorders | █ | █ | █ █████ | █ | █ █████ | █ |
Hepatic steatosis | █ | █ | █ █████ | █ | █ █████ | █ |
Hepatocellular injury | █ | █ | █ █████ | █ | █ █████ | █ |
Infections and infestations | █ | █ | █ █████ | █ | █ █████ | █ |
Abscess limb | █ | █ | █ █████ | █ | █ █████ | █ |
Skin infection | █ | █ | █ █████ | █ | █ █████ | █ |
Injury, poisoning and procedural complications | █ | █ | █ █████ | █ | █ █████ | █ |
Fall | █ | █ | █ █████ | █ | █ █████ | █ |
Humerus fracture | █ | █ | █ █████ | █ | █ █████ | █ |
Investigation | █ █████ | █ | █ █████ | █ | █ █████ | █ |
Liver function test increased | █ █████ | █ | █ | █ | █ █████ | █ |
Neutrophil count decreased | █ | █ | █ █████ | █ | █ █████ | █ |
Weight increased | █ █████ | █ | █ | █ | █ █████ | █ |
Metabolism and nutrition disorders | █ █████ | █ | █ | █ | █ █████ | █ |
Diabetes mellitus | █ █████ | █ | █ | █ | █ █████ | █ |
AE = adverse event; OLE = open-label extension; SAF = safety population; TEAE = treatment-emergent adverse event.
Source: PRISMA-3 Clinical Study Report40
Bioequivalence
The comparative bioavailability of Okedi and oral risperidone was evaluated in the Phase I BORIS study (ROV-RISP-2016-02). This was an open-label, multicentre, one-sequence study in patients with schizophrenia stabilized on oral risperidone.
Description of the BORIS Trial
Study Objectives
The primary objective of this study was to evaluate the steady-state comparative bioavailability of 100 mg Okedi every four weeks compared to once daily 4 mg oral risperidone in patients with schizophrenia stabilized on oral risperidone treatment. The secondary objective was to evaluate the safety and tolerability of 100 mg Okedi compared to once daily 4 mg oral risperidone in patients with schizophrenia stabilized on oral risperidone treatment.
Study Population
The main study inclusion criteria were male and female patients 18 to 65 years old with a BMI of 17 kg/m2 to 35 kg/m2, who had a current diagnosis of schizophrenia per DSM-5 criteria and a CGI-S of ≤ 4 (moderately ill). Participants were required to be outpatients and should not have been hospitalized for worsening of schizophrenia within the last 3 months; medically stable over the last month and psychiatrically stable without significant symptom exacerbation over the last 3 months based on the investigator's judgement; and on oral risperidone 4 mg daily as maintenance therapy for at least the last 4 weeks prior to screening and on 4 mg oral risperidone once daily for at least one week prior baseline. The main criteria for exclusion were uncontrolled, unstable, clinically significant medical condition that could interfere with the interpretation of safety and PK evaluations, or any clinically significant vital sign or physical examination finding that could potentially interfere with the ability to evaluate safety and tolerability of the trial medication or could impair the patient's ability to complete the trial.57
Methods
Patients who were receiving existing oral risperidone (4 mg daily as maintenance therapy for at least the last 4 weeks prior to screening and on 4 mg oral risperidone once daily for at least 1 week prior to baseline [day 1, visit 2]) continued receiving single oral doses of 4 mg risperidone once daily for 7 days. After the 1-week oral risperidone regimen following day 1, visit 2, patients were given a single dose of 100 mg Okedi administered deeply into the gluteal muscle. A total of 4 Okedi doses were given, each separated by 4 weeks. Safety assessments and PK sampling were performed once each dosing day and each outpatient visit. A final follow-up visit was conducted to assess each subject for safety and to obtain PK samples.
Plasma PK parameters were determined for risperidone active moiety at steady-state (i.e., after the seventh dose of oral risperidone [day 7] and after the fourth dose of OKEDI [day 92]).
Outcomes and Statistical Analysis
The following plasma PK parameters were determined for risperidone active moiety at steady-state (i.e., after the seventh dose of oral risperidone [day 7] and after the fourth dose of Okedi [day 92]):
AUCtau: Area under the curve during the dosing interval
The most representative PK parameter for comparison of different administration routes and dosing regimen at steady-state.
Cmax, ss: Maximum plasma concentration at steady-state
Cmin, ss: Minimum plasma concentration (trough level) at steady-state
Cave: Average plasma concentration
tmax, ss: Time to maximum plasma concentration at steady-state
Fluc: Percentage peak to trough fluctuation over a dosing interval
The primary end point was the steady-state area under the curve (AUCtau) for risperidone active moiety. The steady-state AUCtau for oral risperidone was calculated for the 24-hour dosing interval following dosing on day 7 and prior to the first administration of Okedi on day 8. The steady-state AUCtau for Okedi was calculated for the 28-day period following administration of the fourth dose of Okedi on day 92 up to the final blood sample obtained on day 120. The AUCtau was derived by using the trapezoid method based on all quantifiable plasma concentrations over the relevant time frames and actual times that samples were obtained. To enable comparisons between Okedi and oral risperidone, the AUCtau obtained for oral risperidone was multiplied by 28 (Adjusted AUCtau), thus assuming equivalence to Okedi.
To determine the comparative bioavailability of steady-state plasma exposure between oral 4 mg risperidone and 100 mg Okedi, natural log transformation of the adjusted AUCtau, Cmax ss, Cmin ss, Cave, and Fluc for risperidone active moiety was performed and values were compared between treatments by using an analysis of variance (ANOVA) with treatment as a fixed effect and patient as a random effect. The corresponding means, mean differences, and 90% CI for the mean difference (based on the standard error and t-distribution) were back transformed (exponentiated) to derive the geometric means, geometric mean ratio, and 90% CI for the geometric mean ratio for each parameter. Acceptance criteria for the 90% CI for the geometric mean ratio of (0.80, 1.25) was applied.
Results
Patient Disposition and Demographic and Baseline Characteristics
████ █ █████ ██ ███ ████████ ████████ ███ ████████████ ██ ████████ ██ █████ █ ████ ██ █████ █████ ██ ██████ ██ █████████ ███ █████ ███ ██ █████████████ ███████ ███ ███████████████ ████ ██████████ ██ ███ ███████ ██ █ ██ █████████ ███████ ██████ ██ ████████ ████ ██ █████████ ██ ████████ ███ ███████ ████████ ███ ██████████████ ███████ ██ ████████ ███ ██████ ██████████ ██ █ ███ ████████ ████████ ███ ████████ ██ █████ █ ████ ██ █████ █████ ███ ███ ██ ██████████ ██ █ ███ ████████ ████████ ████ ███ ██████ ██████████ ███ █████████ ███ █████ ███ ███ ██ █████ ████████ ██████████ ████ ████████ ████ ████ ███ █████████ Patient demographic and baseline characteristics are summarized in Table 18.58
██████ ████ ███████████ ██████████ ██ ████████ █████ ██ ████████ ███ █ █████ ██ ███ █████ █████ ██████ █████ ██████████ ██ ████████ ███████ ████████ ███ █ █████ ██ ███ █████ █████
Table 18: Demographic and Baseline Characteristics (Safety Population)
Baseline Characteristics | Value N = 81 | |
|---|---|---|
Age (years), mean (SD) | ████ ███████ | |
Sex, n (%) | Male | ██ ██████ |
Female | ██ ██████ | |
Race, n (%) | White | ██ ██████ |
Black or African American | ██ ██████ | |
Asian | █ ██████ | |
Other | █ ██████ | |
Ethnicity, n (%) | Hispanic or Latino | █ ██████ |
Not Hispanic or Latino | ██ ██████ | |
Height (cm), mean (SD) | ██████ █████ | |
Weight (kg), mean (SD) | ████ ██████ | |
BMI (kg/m2), mean (SD) | █████ █████ | |
BMI = body mass index; SD = standard deviation
Source: BORIS Trial Clinical Study Report58, Walling et al. (2021)59
Comparative Bioavailability at Steady-State
████ ██ ██████ ████████ ██ ███████████ ██████ ██████ ███ ████ ██ ███████████ ██████ ██████ ████ ███████ █████████ ██████ ███ ██ █████ █████ ████ █████ ████████ ██ ████ █████ █ ██ ████ ████████████ ██████ ███ ████ ██ ██████ █████████ ██ ███████████ ██████ ██████ ████ ████ ████████ ██████ █████████ ██████ ███ ██ █████ ████████ ██ ████ █████ █ ██ ████ ████████████ ███████████ ██████ ██████ ████ ██ ██████ ███ ███ ██ █████ ████ █████████████ ███████ ████ █████ ███ ████ █████ █ ██ ████ ███████████ (Table 19).
Table 19: Statistical Analysis of Comparative Bioavailability for Risperidone Active Moiety PK Parameters at Steady-State (PK Population)
PK Parameter | Geometric LS Means (N = 48) | Geometric LS Means (90% CI) (OKEDI/oral risperidone) | |
|---|---|---|---|
Adjusted AUCtau (day*ng/mL) | Oral risperidone | █████ | ██████ ███████ ██ ███████ |
OKEDI | ████ | ||
Cmax ss (ng/mL) | Oral risperidone | █████ | ██████ ███████ ██ ███████ |
OKEDI | █████ | ||
Cmin ss (ng/mL) | Oral risperidone | █████ | ██████ ███████ ██ ███████ |
OKEDI | █████ | ||
Cave (ng/mL) | Oral risperidone | █████ | ██████ ███████ ██ ███████ |
OKEDI | █████ | ||
Fluc (%) | Oral risperidone | ███████ | ██████ ███████ ██ ███████ |
OKEDI | ███████ | ||
Median of Difference (90% CI; p-value) | |||
tmax ss (h) | Oral risperidone | █████ | ███████ ████████ ██ █████████ █ ███████ |
OKEDI | ███████ | ||
ANOVA = analysis of variance; AUCtau = area under the curve during the dosing interval; Cave = average plasma concentration; CI = confidence interval; Cmax,ss = maximum plasma concentration at steady-state; Cmin ss = minimum plasma concentration at steady-state; Fluc = percentage peak to trough fluctuation over a dosing interval; LS = least squares; PK = pharmacokinetic; tmax,ss = time to maximum plasma concentration at steady-state.
An ANOVA with treatment as a fixed effect was fitted to each log-transformed PK parameter. Results were back transformed to obtain the geometric LS mean, geometric LS mean ratio, and the 90% CI.
Oral 4 mg dose of risperidone was administered once daily from days 1 to 7. Single intramuscular dose of 100 mg OKEDI was administered every four weeks from days 8 to 92.
PK parameters for oral risperidone were estimated after the seventh oral dose of risperidone. PK parameters for OKEDI were estimated after the fourth dose of risperidone ISM.
Data for 10 patients were excluded because steady-state was not achieved prior to day 7.
Source: BORIS Trial Clinical Study Report58, Health Canada CTD 2.7.260
Critical Appraisal of the Clinical Evidence by CDA-AMC
Internal Validity
The PRISMA-3 Trial
The PRISMA-3 trial was a randomized DB, placebo-controlled study. Methods of randomization and treatment allocation (by interactive voice or web response system) were adequate for limiting risk of bias in the randomization process. The reported baseline characteristics were generally balanced across the groups, and the clinical expert consulted for this review did not identify any imbalances that would be expected to impact the interpretation of the efficacy or safety results. Patients, investigators, and study site staff were blinded to treatment allocation; however, unblinded individuals at each study site prepared and administered the study drug to each patient. As such, there is some risk that patients and/or outcome assessors could have become aware of the treatment group assignment, which could introduce risk of bias in the measurement of subjective outcomes (including PANSS, CGI-S, and SWN-20 measures and AEs); however, the magnitude and direction of this risk cannot be predicted (but would likely favour risperidone ISM for efficacy end points).
The prespecified sample size was achieved and the number of screening failures was lower than anticipated. Multiplicity of testing was accounted for in the primary and key secondary end points and included hierarchical ordering; strategies to adjust for multiplicity were adhered to. All other secondary end points (and SWN-20) were assessed without adjustment for multiple comparisons; the results of these analyses were considered supportive evidence.
Some between-group differences were noted in patient disposition. Study completion was highest in the risperidone ISM 75 mg group (73.8%) and lowest in the placebo group (59.9%), and a higher percentage of patients discontinued the study due to insufficient clinical response in the placebo group (10.2%) than in the risperidone ISM 75 mg and 100 mg groups (4.1% and 6.2%, respectively). Loss to follow-up was generally low and similar across treatment groups. The clinical expert consulted for this review stated that the discontinuation rates and reasons for discontinuation in the PRISMA-3 study were reasonably aligned with rates that would be expected for patients receiving LAI antipsychotics for treatment of schizophrenia in clinical practice. More than half of the patients in all 3 treatment groups received 3 doses of study medication, with the percentage of patients lower in the placebo group (63.3%) than in the risperidone ISM 75 mg and 100 mg groups (77.8% and 67.8%, respectively).
During the PRISMA-3 trial, a range of medications were prohibited, including antipsychotic medications other than risperidone ISM and other psychotropic medications. Rescue medications were permitted and included benzodiazepines, anticholinergics, and propranolol. The clinical expert stated that no specific interpretation could be made regarding the impact of lorazepam and benztropine use within the trial on the efficacy and safety results.
Relatively few patients were reported with important protocol deviations (3.5% to 4.8% of patients across groups in the ITT population), and the deviations were relatively balanced across groups. As such, it is unlikely that these had any important impact on the study results. During the PRISMA-3 trial, blinding was potentially compromised for 43 patients due to an error in the interactive voice or web response system. At Protocol Amendment 3, the total sample size for the trial was increased to account for these patients and the mSAF, mITT, and mPP analysis populations were added, excluding patients for whom blinding was potentially compromised. Confirmatory analyses of primary and key secondary end points had to be substantiated in both the mITT and ITT populations. As the results across these analysis populations were consistent, the CDA-AMC review team judged that bias was not introduced as a result of the error.
Several sensitivity analyses were performed for the primary and key secondary end points and the results supported the primary analyses. Sensitivity analyses were not performed for other end points. The approach to the handling of missing data for the primary and key secondary end points appears to be appropriate, and the results of sensitivity analyses were aligned with the primary analyses, suggesting low risk of bias due to missing outcome data. For the other secondary end points, uncertainty exists as to whether there is a risk of bias due to missing data because the amount of missing data was unknown; in some cases, complete case analysis was used, which may introduce bias because the underlying assumptions are unlikely to be reasonable. Notably, for the SWN-20 end point, the proportion of patients with missing data increased over time, with a greater proportion of patients in the risperidone ISM group than in the placebo group completing the SWN-20. At visit 14 or early termination, 17.9% of patients in the placebo group and 13.5% of patients in the risperidone ISM group (15.0% of patients overall) had not completed the SWN-20 assessment.40 Results of multivariate logistic regression models suggested that, at the end of the DB phase, patients with greater disease severity (as measured using the PANSS) were more likely to have missing data for SWN-20. As such, the HRQoL results could be biased as a result of missing outcome data; the direction of bias is uncertain.
Estimated effects of the prespecified subgroup analyses (by country and baseline PANSS score) for the primary end point were mostly aligned with the primary analysis, with some exceptions. Among those who received risperidone ISM 100 mg, patients in Ukraine had a greater change from baseline and between-group difference compared to placebo than patients in the US. Patients in all groups (including placebo) with a baseline PANSS score greater than or equal to 95 had a greater change from baseline in PANSS total score than patients with a lower baseline PANSS; among patients who received risperidone ISM 100 mg, the between-group difference compared to placebo was also greater for patients with a higher baseline PANSS score. The PRISMA-3 trial was not designed or powered to detect subgroup differences, and the 95% CIs were wide and overlapping across categories within each subgroup. Further, there were no statistical tests for treatment by subgroup interactions and no adjustments for multiple testing, limiting credible conclusions about effect modification.
For the PANSS outcome measure, evidence is available to support the validity and reliability of this scale in patients with schizophrenia61,62 (evidence regarding responsiveness was not provided by the sponsor). According to the literature, the MID for the PANSS is unclear and varies by baseline severity; a 15-point reduction or a 20% reduction in PANSS total score have each been considered to indicate within-group clinical improvement (and to be similar to a 1-point reduction on the CGI-S).63,64 A responder threshold of 30% reduction in the PANSS total score from baseline has been considered clinically relevant in short-term clinical trials with patients with acute or exacerbated schizophrenia symptoms.65 There is limited information available on the validity, reliability, and responsiveness of CGI-S and CGI-I measures in patients with schizophrenia. A 1-point improvement on the CGI-S has often been considered in clinical studies as a meaningful clinical improvement.63,64 For the CGI-I, a rating of “minimally improved” has been considered to align with a 15-point reduction in the PANSS total score.63 Evidence is available to support the validity, reliability, and responsiveness of the SWN-20 scale among patients with schizophrenia;66 however, there is no established point change that constitutes a clinically meaningful difference or MID.67 As such, for all outcome measures there is uncertainty in the clinical meaningfulness of the estimated between-group differences.
The PRISMA-3 OLE Trial
In the PRISMA-3 OLE trial, the open-label extension of the PRISMA-3 trial, all the patients received either risperidone ISM 75 mg or risperidone ISM 100 mg. Results for subjective end points, including the PANSS, CGI-S, and SWN-20 measures and AEs, may have been biased because patients and investigators knew the treatment being received. The lack of a control arm precludes drawing conclusions on the magnitude of effect attributable to risperidone ISM.
No sample size determination was performed for the PRISMA-3 OLE trial. No adjustments for multiplicity were made for the end points in the PRISMA-3 OLE trial, and no formal testing between treatment groups was conducted for the PANSS, CGI-S, CGI-I, and overall response end points. Results for within-group comparisons (i.e., comparing baseline to the end of the OLE phase within a treatment group) are at increased risk of type 1 errors (false-positive results).
The clinical expert consulted for this review stated that the discontinuation rates and reasons for discontinuation in the PRISMA-3 OLE trial reasonably aligned with rates that would be expected for patients receiving LAI antipsychotics for treatment of schizophrenia in clinical practice. As occurred in the DB phase of the trial, the proportion of patients with missing data for SWN-20 increased over time, which could introduce bias. At visit 14 or early termination, 10.7% of patients in the OLE did not have SWN-20 data.40
External Validity
The PRISMA-3 Trial
Patients eligible for inclusion in the PRISMA-3 trial had received a diagnosis of schizophrenia and were experiencing an acute exacerbation, as determined based on PANSS and CGI-S scores (Table 4). According to the clinical expert consulted by CDA-AMC for this review, these inclusion criteria reflect patients with an acute exacerbation and are selective for patients with a greater degree of positive symptoms than negative symptoms. Eligibility criteria included a history of clinically significant beneficial response to an antipsychotic other than clozapine, no history of inadequate clinical response to risperidone or paliperidone, and no history of treatment resistance. The clinical expert commented that these criteria do not reflect patients in clinical practice whose history of treatment response may not be known and who often have a history of no or partial response to previously tried antipsychotics. Within the 30 days before screening, most patients (88.3%) in the PRISMA-3 trial had received treatment with an antipsychotic, including 59.3% of the overall patient population who had received risperidone.40 The clinical expert highlighted that the trial includes a highly selected population of patients with schizophrenia that would be more likely to respond to treatment with risperidone ISM. The clinical expert also noted that the typical patient with schizophrenia would have many comorbidities, often including substance use disorder; however, patients with a current diagnosis or history of substance use disorder were excluded from the trial.
The clinical expert commented that the age of patients reflects that of those who would be expected to receive risperidone ISM in clinical practice and that the sex of the participants was slightly skewed toward males. The clinical expert noted that the race of patients in the trial is not representative of the population in Canada as Asian patients were under-represented.
The clinical expert consulted by CDA-AMC did not have specific concerns regarding the use of concomitant medications (including rescue medications) in the PRISMA-3 trial.
An important limitation of the PRISMA-3 trial is that risperidone ISM was compared to placebo, which does not represent the standard of care for treatment of patients with schizophrenia who are experiencing an acute exacerbation. As identified by the clinical expert, appropriate comparators to risperidone ISM would be risperidone LAI every 2 weeks (Risperdal Consta), aripiprazole LAI every month (Abilify Maintena), and the paliperidone LAIs.
According to the clinical expert, the main goals of therapy for patients with schizophrenia are maintenance of function in daily life, prevention of readmissions to hospital, supported community living and integration, and medication adherence for symptom control. Outcomes of importance identified by the patient group (input from a submission for a previous review) and the clinician group included functionality, symptom reduction or control (including preventing relapses), improved adherence, improved quality of life, and management of side effects. As such, many of the outcomes assessed in the PRISMA-3 trial align with key outcomes of importance to patients and clinicians (relapse was assessed in the PRISMA-3 OLE trial, but neither the DB nor OLE phase included measures of medication adherence).
The clinical expert stated that the outcome measures used in the PRISMA-3 trial (PANSS, CGI-S, CGI-I, and SWN-20 measures) are not typically used by physicians in clinical practice. Some hospitals may use the RAI-MH to evaluate progress; however, others may assess progress based on clinical interviews rather than an assessment scale. The clinical expert also highlighted that the definition of a clinically meaningful response to treatment can vary among physicians and patients and that important responses to treatment include reduction in the frequency or severity of symptoms, improvement in symptoms, stabilization of symptoms, and the ability to perform activities of daily living. According to the clinical expert, 12 weeks is a reasonable amount of time to study the outcomes of interest in the acute phase in a clinical trial setting.
The PRISMA-3 OLE Trial
The PRISMA-3 OLE trial patient population comprised rollover (i.e., “stabilized” and “unstable”) patients from PRISMA-3 and de novo (i.e., “stable”) patients, which represents a broader population of patients with schizophrenia than the population in the DB phase. Results for the OLE are in aggregate, and the study included patients who had been receiving risperidone ISM for various amounts of time. Given that the other eligibility criteria were aligned for patients between the DB and OLE phases, the generalizability considerations noted for the PRISMA-3 trial (e.g., a generally healthier and selected population that would be expected to have better response to risperidone) would be expected to be relevant to the PRISMA-3 OLE trial. In addition, rollover patients represent a selected population who tolerated or continued receiving the study drug for long enough to complete the DB phase. The clinical expert noted that the inclusion criteria for de novo patients reflect patients with schizophrenia that would be considered stable. The clinical expert also noted that, as in the DB phase, a higher proportion of patients in the PRISMA-3 OLE trial were male and that Asian patients were under-represented.
In the OLE phase, de novo patients who were taking a stable dose of oral risperidone of 4 mg/day received risperidone ISM 75 mg, and those taking a stable oral risperidone dose greater than 4 mg/day and up to 6 mg/day received risperidone ISM 100 mg. According to the Okedi product monograph, risperidone ISM 75 mg is similar to oral risperidone 3 mg/day and risperidone ISM 100 mg is similar to oral risperidone 4 mg/day (to maintain similar plasma concentrations), and “patients who are stable on oral risperidone doses outside this range may not be candidates for treatment with risperidone ISM.”2 The clinical expert consulted for this review commented that, based on this information, they would not initiate risperidone ISM treatment for patients who are receiving oral risperidone at a dose greater than 4 mg/day (or its equivalent). The clinical expert also stated that in practice a significant portion of patients with schizophrenia stabilized when receiving oral risperidone would be receiving doses greater than 4 mg/day. The clinical expert noted that in the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) trial,68 the mean dose of risperidone was 3.9 mg/day, but that 122 of 305 (40%) patients randomized to treatment with risperidone received the maximum dose of 6 mg/day.
As described for the PRISMA-3 trial, many of the outcomes assessed in the extension phase align with key outcomes of importance to patients and clinicians, including relapse(an end point assessed in PRISMA-3 OLE). According to the clinical expert, 12 months is a reasonable amount of time to study the long-term effects.
The PRISMA-3 and PRISMA-3 OLE trials were conducted in Ukraine and the US; all de novo patients in the extension phase were from Ukraine.40 The clinical expert was not able to comment on the generalizability of the study findings to the population in Canada based on the study locations.
Sponsor-Submitted Cost Comparison
The sponsor submitted a cost comparison for risperidone ISM (Okedi) compared to other LAI antipsychotics approved for the treatment of adults with schizophrenia, including risperidone every 2 weeks (Risperdal Consta), aripiprazole monthly (Abilify Maintena), paliperidone monthly (Invega Sustenna), paliperidone every 3 months (Invega Trinza), and oral risperidone (generic). The unit prices of risperidone ISM were submitted by the sponsor, while those of its comparators were from the Ontario Drug Benefit Formulary.69 Administration costs were derived from the Ontario Schedule of Benefits for Physicians Services.70
At the submitted price of $468.32 and $624.40 for the 75 mg and 100 mg vials, the annual drug cost of risperidone ISM is $6,088 or $8,117 per patient for the 75 mg and 100 mg doses every 4 weeks, respectively. When compared with doses of alternative LAIs deemed comparable by the sponsor, risperidone ISM 75 mg every 4 weeks was more costly than aripiprazole LAI 300 mg monthly, paliperidone LAI 75 mg every month, paliperidone 263 mg every 3 months, and risperidone 25 mg every 2 weeks (incremental cost range of $194 to $671 per patient per year), while risperidone ISM 100 mg every 4 weeks was more costly than aripiprazole 400 mg monthly, paliperidone 100 mg monthly, and paliperidone LAI 350 mg and 525 mg every 3 months, but less costly than paliperidone 150 mg monthly and risperidone 37.5 mg and 50 mg every 2 weeks (incremental savings of $2,716 to incremental costs of $2,395 per patient per year) (refer to Table 25). Risperidone ISM 75 mg every 4 weeks was $5,866 more costly per patient per year than oral risperidone 3 mg daily, while risperidone ISM 100 mg was $7,821 more costly per patient per year than oral risperidone 4 mg daily. The estimated annual administration cost for risperidone ISM was $88 per patient, while annual administration costs for the comparators ranged from $0 for oral risperidone to $175 per patient per year for risperidone LAI every 2 weeks (Table 26).
Critical Appraisal of Cost Information
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the cost comparison:
Assumption of clinical similarity is uncertain: The sponsor’s cost comparison assumes that there is clinical similarity between risperidone ISM and its comparators. The clinical trial (PRISMA-3) compared risperidone ISM 75 mg and 100 mg to placebo, and no direct or indirect evidence was submitted comparing risperidone ISM with other LAI antipsychotics or oral risperidone. According to the CDA-AMC Clinical Review, in the absence of any submitted direct or indirect evidence, the comparative efficacy and harms of risperidone ISM relative to relevant comparators, including oral risperidone and other LAI antipsychotics used in clinical practice remain, unknown. Given the lack of evidence regarding clinical similarity, the appropriateness of conducting a cost comparison is unknown.
CDA-AMC was unable to address this limitation.
Assumption of equivalent doses across comparators: The sponsor compared the cost of risperidone ISM 75 mg and 100 mg with comparators at doses the sponsor considered most closely aligned with each dose, citing clinical expert opinion. For example, the sponsor compared the annual cost of risperidone ISM 75 mg every 4 weeks to that of aripiprazole 300 mg monthly, paliperidone 75 mg monthly, and paliperidone 263 mg every 3 months, but not to aripiprazole 400 mg monthly, paliperidone 50 mg monthly, or paliperidone 175 mg every 3 months. In doing so, the sponsor implicitly assumes these doses are equally effective. According to clinical expert opinion obtained by CDA-AMC, dose equivalencies between the various LAI antipsychotics are uncertain. For example, a 2018 study reported that the minimum effective dose for preventing relapse was aripiprazole LAI 400 mg every 4 weeks, paliperidone LAI 50 mg every 4 weeks, and paliperidone LAI 175 mg every 12 weeks, which are different than the doses that are all considered by the sponsor to be equivalent to risperidone ISM 75 mg every 4 weeks.71 As such, it is uncertain what recommended doses of risperidone ISM may be clinically similar to the available doses of the included comparators. Of note, in the submitted budget impact analysis (BIA), the sponsor assumed that either dose of risperidone ISM could displace any dose of the comparator LAIs.
CDA-AMC compared the cost of both risperidone ISM 75 mg and 100 mg to all recommended doses of the included comparators.
Confidential pricing agreements: While the annual cost of risperidone ISM at the submitted price is within the range of the annual costs of the other LAI antipsychotics at publicly available list prices, these list prices are likely higher than the price paid by jurisdictional drug plans due to confidential pricing agreements that likely exist for at least some of the comparators.72,73 Therefore, the submitted price of risperidone ISM may require additional price reductions to avoid incurring additional costs relative to its comparators.
CDA-AMC was unable to address this limitation in reanalyses, as the negotiated prices of the comparators are unknown.
CDA-AMC Reanalyses
The CDA-AMC reanalysis was derived in consultation with clinical experts and compares both doses of risperidone ISM 75 mg to all recommended doses of the comparators (Table 20). At the submitted price, the annual drug acquisition cost of risperidone ISM ($6,088 to $8,117 per patient) is more expensive than risperidone oral tablets ($222 to $445 per patient). However, it is within the range of other LAI antipsychotics ($3,815 to $10,834 per patient, annually) at publicly available list prices, although less expensive doses of some LAI comparators exist (i.e., risperidone 25 mg every 2 weeks, aripiprazole monthly, paliperidone 50 mg monthly, and paliperidone 175 mg, 263 mg, and 350 mg every 3 months).
In terms of administration costs, when compared with risperidone LAI every 2 weeks, the use of risperidone ISM every 4 weeks is associated with a savings of approximately 13 administrations per year. The amount of savings associated with less frequent injections will depend on the jurisdictional cost per injection and whether the injection is administered as part of a consultation appointment or as the sole reason for the visit.
Table 20: CDA-AMC Cost Comparison Table
Generic name (brand name) | Strength | Dosage form | Price ($) | Recommended dosage regimen | Annual drug cost ($) | Difference in annual drug cost ($, %) vs. risperidone ISM 75 mg | Difference in annual drug cost ($, %) vs. risperidone ISM 100 mg |
|---|---|---|---|---|---|---|---|
Risperidone ISM (Okedi) | 137 mg/mL | 75 mg 100 mg Powder for injection | 468.3200a 624.4000a | 75 mg or 100 mg every 4 weeks | 6,088 8,117 | Reference | Reference |
Long-acting injectable comparators | |||||||
Risperidone (Risperdal Consta) | 6.25 mg/mL 12.5 mg/mL 18.75 mg/mL 25 mg/mL | 12.5 mg 25 mg 37.5 mg 50 mg Powder for injection | 100.3400b 208.3500 312.5100 416.6800 | 25 mg to 50 mg every 2 weeks | NA 5,417 8,125 10,834 | NC –671 (–11%) 2,037 (33%) 4,746 (78%) | NC –2,700 (–33%) 8 (0%) 2,716 (33%) |
Aripiprazole (Abilify Maintena) | 200 mg/mL | 300 mg 400 mg Powder for injection | 487.2002 487.2002 | 300 mg to 400 mg monthly | 5,846 5,846 | –242 (–4%) –242 (–4%) | –2,271 (–28%) –2,271 (–28%) |
Paliperidone (Invega Sustenna) | 100 mg/mL | 50 mg 75 mg 100 mg 150 mg Prefilled syringes | 327.4500 491.1800 544.4400 725.9100 | 50 mg to 150 mg monthly | 3,929 5,894 6,533 8,711 | –2,159 (–35%) –194 (–3%) 445 (7%) 2,623 (43%) | –4,188 (–52%) 2,223 (–27%) –1,584 (–20%) 594 (7%) |
Paliperidone (Invega Trinza) | 200 mg/mL | 175 mg 263 mg 350 mg 525 mg prefilled syringes | 953.7300 1,430.6100 1,430.6100 1,907.4900 | 175 mg to 525 mg every 3 months | 3,815 5,722 5,722 7,630 | –2,273 (–37%) –366 (–6%) –366 (–6%) 1,542 (25%) | –4,302 (–53%) –2,395 (–30%) –2,395 (–30%) –487 (–6%) |
Oral comparator | |||||||
Risperidone (generics) | 0.25 mg 0.5 mg 1 mg 2 mg 3 mg 4 mg | Tablets | 0.0878 0.1470 0.2031 0.4062 0.6083 0.8111 | 3 mg/day to 6 mg/day in 1 to 2 dosesc | 222 to 445 | –5,644 (–93%) to –5,866 (–96%)d | –7,673 (–95%) to –7,821 (–96%)d |
CDA-AMC = Canada’s Drug Agency; ISM = in situ microparticle; NA = not applicable; NC = not comparable; vs. = versus.
Note: All prices are from the Ontario Drug Benefit Formulary, accessed May 2025, unless otherwise specified.69 Annual drug cost calculations assume 12 monthly injections, 13 injections if given every 4 weeks, 26 injections if given every 2 weeks, and 4 injections if given every 3 months. Negative annual differences and percentages in cost indicate that the comparator is less expensive than risperidone ISM (Okedi).
aSponsor’s submitted price.74
bSaskatchewan Formulary, accessed May 2025.75
cThe product monograph for oral risperidone recommends a target dose range after titration of 4 mg/day to 6 mg/day, administered in 1 to 2 doses, with a maximum of 16 mg/day.48 However, as risperidone ISM 75 mg is intended to replace doses of 3 mg/day of oral risperidone,2 the recommended dose range has been extended to include this dose.
dThe monograph for risperidone ISM states that 75 mg every 4 weeks is similar to 3 mg/day of oral risperidone while 100 mg every 4 weeks is similar to 4 mg/day of oral risperidone. The monograph also states that “patients who are stable on higher or lower doses of oral risperidone may not be candidates for risperidone ISM”.2 The annual incremental cost of risperidone ISM 75 mg every 4 weeks is $5,866 more per patient than the incremental cost of 3 mg daily of oral risperidone, while the annual incremental cost of risperidone ISM 100 mg every 4 weeks is $7,821 per patient more than the incremental cost of 4 mg daily of oral risperidone.
Source: Sponsor’s submission.74
Price Reduction Analyses
Given the sponsor’s claim of clinical similarity between risperidone ISM and other LAIs and given the lack of any submitted direct or indirect comparative efficacy data for risperidone ISM versus other LAIs, there is insufficient evidence for risperidone ISM to have a price premium over other LAIs currently reimbursed for the treatment of schizophrenia.
Issues for Consideration
Lack of loading dose or cross-tapering requirement: As with other LAIs, patients initiating risperidone ISM require an established tolerance to the oral form of their medication. However, risperidone ISM does not require concomitant use with an oral antipsychotic therapy.2 In contrast, concomitant use of an oral antipsychotic drug is recommended during the initial 2 to 3 weeks of therapy with risperidone every 2 weeks and aripiprazole monthly.76,77 Risperidone ISM also does not require an additional injection as a loading dose, while a loading dose is recommended when initiating paliperidone monthly injections or as an alternate to concomitant oral therapy when initiating aripiprazole.76,78 This benefit eliminates the costs associated with cross-tapering and loading doses and may be an advantage in reducing the monitoring intensity required for outpatients or the length of stay required for inpatients when initiating LAI antipsychotics.
Generic versions of aripiprazole LAI may soon be available: A generic version of aripiprazole LAI was recently approved by Health Canada.79 According to the pan-Canadian Pharmaceutical Alliance (pCPA) Tiered Pricing Framework,80 after 3 months of funding, the price of a single source generic product automatically reduces to 55% of the brand reference price. Should a generic product become available, the cost of aripiprazole LAI would be reduced to approximately $268 per 300 mg or 400 mg vial, or $3,216 per patient per year, and the incremental cost of risperidone ISM would be increased. Should additional generic brands of aripiprazole LAI become available, the cost would be further reduced.
Alternate risperidone LAI is currently under review by CDA-AMC: A subcutaneous risperidone LAI is currently under review for the treatment of schizophrenia in adults (Longavo).81,82 Should this product become funded, it may impact the potential market uptake of risperidone ISM (Okedi).
Alternate form of aripiprazole LAI has been approved by Health Canada: An alternate form of aripiprazole LAI (Abilify Asimtufii) has been approved by Health Canada and is reported as being marketed for the treatment of schizophrenia in adults.44 The recommended maintenance dose of this product is 960 mg intramuscularly every 2 months, with the option to reduce to 720 mg every 2 months if necessary due to adverse reactions. Abilify Asimtufii had not been submitted to CDA-AMC at the time of this review.
Discussion
Summary of Available Evidence
The PRISMA-3 trial (N = 438) was a multicentre, randomized, DB, placebo-controlled study investigating the efficacy and safety of risperidone ISM in treating adult patients with schizophrenia who were experiencing acute exacerbation. Both the PRISMA-3 trial and its extension study, the PRISMA-3 OLE trial, were conducted at 26 centres in Ukraine and the US. In the PRISMA-3 trial, patients were randomized 1:1:1 to receive risperidone ISM 75 mg, risperidone ISM 100 mg, or matched placebo by IM injection every 4 weeks for a total of 3 injections during the 12-week treatment phase. The primary end point was the PANSS total score mean change from baseline at day 85, and the key secondary end point was the CGI-S score mean change from baseline at day 85. Other secondary end points from the trial that are of interest to this discussion are ORR at day 85, defined as a PANSS total score greater than or equal to a 30% decrease from baseline or a CGI-I score of 2 (much improved) or 1 (very much improved); PANSS positive subscale score mean change from baseline and PANSS negative subscale score mean change from baseline, each at day 85; and HRQoL at the end of the trial, as measured by the SWN-20 total score change from baseline. Harms outcomes were also assessed. End points of discussion for the pivotal studies in this review are based on outcomes included in the Sponsor’s Summary of Clinical Evidence as well as outcomes identified as important to this review according to the clinical expert consulted for this review and input from patient and clinician groups. Using the same considerations, end points were selected that were considered to be most relevant to inform expert committee deliberations, and this list of end points was finalized in consultation with members of the expert committee.
Patients eligible for participation in the PRISMA-3 trial were aged 18 years to 65 years, with a current diagnosis of schizophrenia, who were actively experiencing an acute exacerbation or relapse with a PANSS total score between 80 and 120 and a score of greater than or equal to 4 points for 2 or more of the following positive symptom items: delusions, conceptual disorganization, hallucinatory behaviour, and suspiciousness and/or persecution. All patients had a CGI-S score of greater than or equal to 4 (moderately ill or worse). Patients previously had a clinically significant beneficial response after treatment with an antipsychotic other than clozapine, had no history of inadequate clinical response to treatment with risperidone or paliperidone, and no history of treatment resistance. The mean age of patients in the PRISMA-3 trial was 42.0 years. Approximately two-thirds of the patients were male (67.0%) and one-third were female (33.0%). Approximately half of the patients were white (48.5%) and half were Black or African American (49.9%). The majority of patients were in the US (61.1%) and the remainder were in Ukraine (38.9%). The mean BMI of patients was 28.33 kg/m2. The mean time since acute exacerbation or relapse was 0.4 weeks, and the mean time since schizophrenia diagnosis was 15.5 years.
The PRISMA-3 OLE trial (N = 215) was an open-label extension of the PRISMA-3 trial that evaluated the long-term efficacy and safety of risperidone ISM in adult patients with schizophrenia. All patients received either risperidone ISM 75 mg or risperidone ISM 100 mg by IM injection every 4 weeks over 12 months. PRISMA-3 OLE trial end points that are of interest to this discussion included those for the PRISMA-3 trial (but at the OLE end point) as well as relapse (defined as a PANSS total score increase of ≥ 30% from baseline; or rehospitalization for psychotic symptoms or use of adjunctive antipsychotic medication after stabilization) and remitters (defined as the simultaneous attainment of a score of ≤ 3 for 6 months or longer on 8 main items of the PANSS [delusions, conceptual disorganization, hallucinations, blunted affect, passive apathetic social withdrawal, lack of spontaneity and flow of conversations, mannerisms and posturing, unusual thought content]).
Patients eligible for participation in the PRISMA-3 OLE trial included rollover patients who had completed the PRISMA-3 trial; those who had been receiving placebo (“unstable patients”) were randomly assigned to receive either risperidone ISM 75 mg or 100 mg and those patients who had been receiving risperidone ISM (“stabilized patients”) continued to receive risperidone ISM at the same dose as during the DB phase. De novo patients were aged 18 years to 65 years, with a current diagnosis of schizophrenia that was clinically stable at screening (“stable patients”), and with a PANSS total score of less than 70 and a CGI-S score of less than or equal to 3 (mild). De novo patients previously had a clinically significant beneficial response after treatment with an antipsychotic other than clozapine, no history of inadequate clinical response to treatment with risperidone or paliperidone, and no history of treatment resistance. These patients were also taking a stable dose of 4 mg/day to 6 mg/day of oral risperidone; patients taking oral risperidone at 4 mg/day received risperidone ISM 75 mg, and those taking oral risperidone at doses greater than 4 mg/day and up to 6 mg/day received risperidone ISM 100 mg.
The mean age of patients in the PRISMA-3 OLE trial was 39.3 years. Male patients comprised 60.9% and female patients 39.1% of the participants; the majority (84.7%) were white and 14.9% were Black or African American. The majority of patients were in Ukraine (81.4%) and the remainder were in the US (18.6%); all de novo patients were in Ukraine.40 The mean BMI was 26.88 kg/m2, and the mean time since schizophrenia diagnosis was 10.9 years.
Interpretation of Results
Efficacy
The efficacy outcomes of most importance in the treatment of schizophrenia, as identified by the clinical expert and patient and clinician groups, are improvement in functioning, symptom reduction or control (including preventing relapse), improved medication adherence, and improved quality of life. As such, many of the end points assessed in the PRISMA-3 and PRISMA-3 OLE studies align with key outcomes of importance to patients and clinicians.
Regarding the primary end point in the PRISMA-3 trial, patients with acute exacerbation of schizophrenia treated with risperidone ISM had a greater reduction in PANSS total score from baseline to day 85 than patients treated with placebo. In the risperidone ISM 75 mg group, the within-group change from baseline was −24.6 points and the between-group difference compared to placebo was −13.0 points; results for the risperidone ISM 100 mg group were similar (−24.7 and −13.3, respectively). The within-group changes exceeded the PANSS within-group estimated MID of 15 points; however, given that the MID for PANSS is unclear and varies by baseline severity, and in the absence of an estimated between-groups MID, there is uncertainty as to whether the benefit of risperidone ISM over placebo in the PANSS total score is clinically meaningful. Further, the clinical expert commented that there is uncertainty in the clinical meaningfulness of these results because the trial participants comprise patients with schizophrenia that would be more likely to respond to treatment with risperidone ISM. At day 85, the PANSS positive and negative subscale scores were both improved for each of the risperidone ISM doses compared to placebo. The clinical meaningfulness of the between-group differences is uncertain as MID estimates were not available. At the PRISMA-3 OLE trial end point, the mean PANSS total score had decreased from baseline in the OLE population by 11.2 points and the positive and negative subscale scores had decreased from baseline by 3.4 and 2.2 points, respectively.
Results of the prespecified subgroup analyses of the primary end point suggested smaller benefit among patients receiving risperidone ISM 100 mg who were in the US and who had a lower baseline PANSS score. Although credible effect modification could not be inferred from these analyses, the results raise uncertainty as to whether the magnitude of benefit observed in the PRISMA-3 trial can be generalized to these groups. The clinical expert noted that baseline PANSS scores are a known effect modifier as patients with higher baseline scores experience greater reductions in score.
In the PRISMA-3 trial, patients treated with risperidone ISM had a greater reduction in mean CGI-S score from baseline to day 85 than patients treated with placebo (key secondary end point). In the risperidone ISM 75 mg group, the within-group change from baseline was −1.3 points and the between-group difference compared to placebo was −0.7 points; results for the risperidone ISM 100 mg group were similar (−1.3 and −0.7, respectively). The within-group changes exceeded the 1-point improvement on the CGI-S that is considered clinically meaningful (in clinical trials and as noted by the clinical expert); however, in the absence of an estimated between-group MID, there is uncertainty as to whether the benefit of risperidone ISM over placebo in the CGI-S score is clinically meaningful. Further, as with the primary end point, the clinical expert noted that there is uncertainty in the clinical meaningfulness of these results due to the selected patient population in the trial. At PRISMA-3 OLE trial end point, the mean CGI-S score decreased from baseline in the OLE population by 0.5 points.
In the PRISMA-3 trial, at day 85, the proportion of patients with overall response was greater for both risperidone ISM groups compared to placebo. The clinical meaningfulness of these results is uncertain as MID estimates were not available. In the overall population in the PRISMA-3 OLE trial, at end point, the ORR was 45.1%. Given the lack of control group, it is not possible to discern to what degree the overall responses were attributable to risperidone ISM (i.e., overall response could be due to a placebo effect, natural history, or risperidone ISM).
Regarding HRQoL, the mean SWN-20 total score change from baseline to the end of the PRISMA-3 trial was higher for patients who received risperidone ISM than for those in the placebo group. At the end of the PRISMA-3 OLE trial, the mean SWN-20 total score change from baseline varied across groups, with de novo patients having the least change and rollover placebo patients having the greatest change. There is potential for bias in the SWN-20 results due to missing outcome data. No MID has been established for the SWN-20 scale and the clinical expert could not comment on the clinical meaningfulness of these results.
Relapse and remittance were assessed in the PRISMA-3 OLE trial; at the end of the trial, the relapse rate in the overall population was 10.7% and the remittance rate in the overall population was 61.0%. The clinical expert commented that these results appear to be clinically meaningful.
Interpretation of the efficacy results should take into account certain considerations. The primary and key secondary end points in PRISMA-3 were included in the multiple testing procedure whereas other secondary end points (e.g., PANSS subscales, overall response) and SWN-20 were not. Although the sponsor-provided summary of results notes the statistical significance of other secondary end points, this cannot be concluded and the results are considered as supportive evidence. The results of the PRISMA-3 OLE trial suggest long-term improvement or stability in symptoms or disease severity for patients in this study; however, the lack of a control arm precludes drawing conclusions regarding the magnitude of effect attributable to risperidone ISM and evaluating the clinical meaningfulness of the results. Interpretation of the PRISMA-3 OLE trial results should take into account that stabilized and stable patients were included in the overall OLE trial population and that these patients may not be expected to have the same magnitude of change in symptom and disease severity scores as patients who are acutely ill. Due to the open-label design of the PRISMA-3 OLE trial, results for subjective end points may have been biased due to patients and investigators knowing the treatment being received. Estimated between-group MIDs were not available in the literature for any efficacy end point, so there is uncertainty as to the clinical meaningfulness of between-group effects.
The clinical expert noted that clinical interviews are often used to assess treatment response in clinical practice (rather than the tools used in clinical trials) and commented that meaningful response to treatment (e.g., improvement in symptoms, stabilization of symptoms, improved functioning) is patient specific. Similarly, the clinician group noted that stability of illness and prevention of relapses are measures of successful treatment and that the magnitude of the response to treatment varies between patients and at different stages and times.
An important limitation of the PRISMA-3 trial is that risperidone ISM was compared to placebo, which does not represent the standard of care for treatment of patients with schizophrenia who are experiencing an acute exacerbation. As identified by the clinical expert, appropriate comparators to risperidone ISM would be risperidone LAI every 2 weeks (Risperdal Consta), aripiprazole LAI every month (Abilify Maintena), and the paliperidone LAIs; however, no evidence was available for this review comparing risperidone ISM to these LAIs.
Various factors were identified that may impact the generalizability of the PRISMA-3 and PRISMA-3 OLE trial results. As previously noted, due to the eligibility criteria, the trial includes a selected population of patients with schizophrenia that would be more likely to respond to treatment with risperidone ISM than the general population of patients with schizophrenia, which could inflate the results in favour of risperidone ISM. The population in the placebo-controlled PRISMA-3 trial comprised patients experiencing an acute exacerbation of schizophrenia. This population is more restricted than the indicated population in the reimbursement request. All the patients in the PRISMA-3 trial had previously received an antipsychotic and a large proportion had previously received risperidone, which impacts the generalizability of the results to patients who have never received an antipsychotic or who have previously received an antipsychotic that was not risperidone. The population in the PRISMA-3 OLE trial represents a broader population of patients with schizophrenia than in the DB trial as included were “stabilized” patients (who continued risperidone ISM treatment) as well as “stable” patients (who transitioned from a stable oral dose of risperidone to risperidone ISM). In addition, rollover patients in the PRISMA-3 OLE trial represent a selected population of patients who tolerated or continued to receive the study drug long enough to complete the DB phase. The clinical expert commented that the PRISMA-3 pivotal trials included patients with schizophrenia who were generally healthier than patients in clinical practice in Canada, who often have comorbidities (e.g., substance use disorder), and that Asian patients were under-represented in the enrolled trial populations.
De novo patients in the PRISMA-3 OLE trial had schizophrenia that was stabilized on higher doses of oral risperidone than the recommended conversion doses in the Okedi product monograph, which states that patients taking stable doses of oral risperidone outside of the range of 3 mg/day to 4 mg/day may not be candidates for risperidone ISM. The clinical expert noted that a significant proportion of patients would be expected have schizophrenia stabilized when receiving oral doses of risperidone greater than 4 mg/day and that these patients would receive treatment with a different LAI antipsychotic.
Harms
Management of side effects has been identified as an outcome of importance in the treatment of schizophrenia by the clinical expert and patient and clinician groups.
The sponsor-provided summary of harms results presented combined data for patients who received risperidone ISM in the PRISMA-3 and PRISMA-3 OLE trials. In both phases, the majority of patients (68.4%) experienced at least 1 TEAE. The most common TEAEs were headache (15.5%), hyperprolactinemia (10.4%), increased blood prolactin (9.3%), nasopharyngitis (7.8%), increased weight (7.8%), insomnia (6.5%), and akathisia (6.0%). In both phases, 4.7% of patients experienced at least 1 SAE, 7.8% of patients discontinued risperidone ISM due to a TEAE, and 1 patient (0.3%) experienced a TEAE that resulted in death.
AE data for risperidone ISM treatment compared to placebo from the PRISMA-3 trial were also considered by CDA-AMC. A higher proportion of patients experienced at least 1 TEAE in the risperidone ISM 75 mg (55.6%) and risperidone ISM 100 mg (64.4%) groups compared to the placebo group (44.2%). The most common TEAEs were increased blood prolactin (9.0% in the risperidone ISM 75 mg group and 14.4% in the risperidone ISM 100 mg group versus 0% in the placebo group); headache (10.4% in the risperidone ISM 75 mg group and 8.2% in the risperidone ISM 100 mg group versus 3.4% in the placebo group); hyperprolactinemia (5.6% in the risperidone ISM 75 mg group and 8.9% in the risperidone ISM 100 mg group versus 0.7% in the placebo group); akathisia (4.2% in the risperidone ISM 75 mg group and 7.5% in the risperidone ISM 100 mg group versus 2.0% in the placebo group); and increased weight (6.9% in the risperidone ISM 75 mg group and 5.5% in the risperidone ISM 100 mg group versus 2.0% in the placebo group). The proportion of patients who experienced at least 1 SAE was 1.4% in the risperidone ISM 75 mg group, 3.4% in the risperidone ISM 100 mg group, and 3.4% in the placebo group, and the proportion of patients who discontinued risperidone ISM due to a TEAE were 4.2%, 6.2%, and 6.8% in the risperidone ISM 75 mg, risperidone ISM 100 mg, and placebo groups, respectively. No patient died during the PRISMA-3 trial.40
According to the clinical expert, the types of AEs reported in the PRISMA-3 pivotal trials are as expected with risperidone treatment and would be manageable in clinical practice. The clinical expert highlighted that there are limitations to the generalizability of the harms results as the patients in the PRISMA-3 pivotal trials may have been more likely to tolerate risperidone than the general population of patients with schizophrenia; as such, higher rates of AEs may be expected to occur in practice. In addition, the clinical expert commented that under-representation of Asian patients is a consideration given that AE data for this population is not available.
According to the clinical expert, hyperprolactinemia, akathisia, and extrapyramidal symptoms are the AEs of most clinical relevance for risperidone ISM, because these can cause impairment and distress for patients.
Other Considerations
The sponsor submitted results from a bioequivalence study (BORIS) that evaluated the comparative bioavailability of risperidone ISM 100 mg every 4 weeks and oral risperidone 4 mg once daily. The results stated that steady-state total (AUCtau) and peak (Cmax ss) plasma exposures to risperidone active moiety were only slightly higher following repeat 100 mg risperidone ISM compared to once-daily 4 mg oral risperidone. Bioequivalence and pharmacokinetic parameters are not for CDA-AMC to evaluate, but for Health Canada to assess during formal review. Okedi has received a Notice of Compliance from Health Canada.83 The Health Canada Regulatory Decision Summary for Okedi states the following:
“A bioavailability study was conducted to demonstrate similar steady-state plasma concentrations of risperidone active moiety (risperidone + metabolite 9-OH-risperidone) achieved with Okedi 100 mg compared to 4 mg oral risperidone. Dose similarity between Okedi 75 mg and 3 mg oral risperidone was supported by population pharmacokinetic (popPK) models and extrapolation.”84
Patient group input (from a submission for a previous review) stated that patients need access to a wide range of treatments that are convenient and affordable to suit unique needs and improve adherence. In consideration of the limitations of current treatments identified by the sponsor, the clinical expert consulted for this review acknowledged that currently available LAI antipsychotics require dose titration as well as oral supplementation, loading doses, or frequent administrations to achieve steady-state plasma levels, noting that reduced injection frequency is preferable for patients and from a health care resources perspective. According to the Okedi product monograph, a loading dose should not be administered and risperidone ISM should not be supplemented with oral risperidone.2 Improved adherence was identified as an outcome of importance to patients and clinicians; however, no evidence was available from the PRISMA-3 pivotal trials that assessed this outcome for risperidone ISM and no evidence was submitted that evaluated adherence to risperidone ISM compared to other LAI antipsychotics.
Cost
The sponsor submitted a cost comparison evaluating the annual drug and administration costs associated with risperidone ISM compared to other LAI antipsychotics approved for the treatment of adults with schizophrenia, as well as to oral risperidone. At the submitted prices of $468.32 and $624.40 for the 75 mg and 100 mg vials, the annual drug cost of risperidone ISM is $6,088 or $8,117 per patient per year, with an estimated annual administration cost of $88. Based on publicly available list prices, the annual drug costs for the other antipsychotic LAIs (i.e., risperidone, aripiprazole, and paliperidone) ranged from $3,815 to $10,834 per patient, with annual administration costs ranging from $81 to $175. The incremental cost of risperidone ISM every 4 weeks compared to the recommended doses of the LAI comparators ranged from a savings of $4,746 annually to an additional cost of $4,302 annually, per patient, depending on the doses being compared. The annual drug acquisition cost of risperidone ISM is $5,644 to $7,821 more expensive than that of risperidone tablets ($222 to $445 per patient).
The sponsor’s cost comparison was associated with limitations including uncertainty in the assumption of clinical similarity, uncertainty in the equivalency of doses across comparators, and the potential confidential prices of the comparators. All incremental costs or savings are based on publicly available list prices and may not reflect actual prices paid by public drug plans in Canada.
Conclusions
Evidence from the PRISMA-3 trial demonstrated that, in patients with schizophrenia experiencing acute exacerbation, treatment with risperidone ISM resulted in greater improvements in symptoms and disease severity compared to placebo, as measured by the change in PANSS and CGI-S total scores after 12 weeks of treatment. In both risperidone ISM groups (75 mg and 100 mg), improvements in PANSS and CGI-S total scores exceeded the within-group MIDs estimated in the literature. However, in the absence of estimated between-group MIDs, there is uncertainty regarding the clinical meaningfulness of the advantage of risperidone ISM over placebo. In the PRISMA-3 OLE trial, improvements in PANSS and CGI-S scores were observed after 12 months in a population of patients with both acute and stable disease; however, the ability to draw conclusions is limited by the open-label study design and lack of comparator arm. No conclusions can be made regarding the effects of risperidone ISM on HRQoL in patients with schizophrenia based on the evidence available from the trials due to missing data and study design limitations. According to clinical expert input, the types of AEs that occurred in the PRISMA-3 trial are as expected for risperidone and considered manageable; however, higher rates of AEs may be expected in clinical practice. Improved adherence was identified as an outcome of importance to patients and clinicians; however, no evidence assessing this outcome for risperidone ISM was available from the PRISMA-3 pivotal trials and no evidence evaluating adherence to risperidone ISM compared to other LAI antipsychotics was submitted. In the absence of any submitted direct or indirect evidence, the comparative efficacy and harms of risperidone ISM relative to relevant comparators, including oral risperidone and other LAI antipsychotics, remain unknown.
At the sponsor’s submitted price, the annual cost of risperidone ISM is $6,088 or $8,117 per patient for the 75 mg and 100 mg every 4 weeks doses, respectively, which is more expensive than that of risperidone tablets ($222 to $445 per patient), but within the range of other LAI atypical antipsychotics ($3,815 to $10,834 per patient). The sponsor’s cost comparison was associated with limitations, including uncertainty in the assumption of clinical similarity (and therefore the appropriateness of conducting a cost comparison), uncertainty in the equivalency of doses across comparators, and the potential confidential prices of the comparators. Overall, given the lack of any submitted direct or indirect comparative efficacy data for risperidone ISM versus appropriate comparators, there is insufficient evidence for risperidone ISM to have a price premium over other LAIs reimbursed for the treatment of schizophrenia.
References
1.Bausch Health, Canada Inc. Drug Reimbursement Review sponsor submission: Okedi (risperidone long-acting injection), 75 mg or 100 mg, powder for extended-release suspension, intramuscular injection [internal sponsor's package]. March 21, 2025.
2.Bausch Health, Canada Inc. Okedi (Risperidone for extended-release injectable suspension): Powder for extended-release suspension, 75 mg or 100 mg, intramuscular injection [product monograph]. March 21, 2024.
3.Owen MJ, Sawa A, Mortensen PB. Schizophrenia. Lancet. 2016;388(10039):86-97. doi:10.1016/s0140-6736(15)01121-6 PubMed
4.Patel KR, Cherian J, Gohil K, Atkinson D. Schizophrenia: overview and treatment options. P t. 2014;39(9):638-45. PubMed
5.Fischer BA, Buchanan RW. Marder S, Friedman M, eds. Schizophrenia in adults: Clinical features, assessment, and diagnosis. UpToDate; March 14, 2025. Accessed March 21, 2025. https://www.uptodate.com
6.Morrissette DA, Stahl SM. Affective symptoms in schizophrenia. Drug Discov Today Ther Strateg. 2011;8(1):3-9. doi:10.1016/j.ddstr.2011.10.005
7.Barabassy A, Szatmári B, Laszlovszky I, Németh G. Negative Symptoms of Schizophrenia: Constructs, Burden, and Management. In: Durbano F, ed. Psychotic Disorders - An Update. IntechOpen; 2018.
8.National Institute for Health and Care Excellence. Psychosis and schizophrenia in adults: prevention and management Clinical guideline. 2014. Accessed April 30, 2025. https://www.nice.org.uk/guidance/cg178/resources/psychosis-and-schizophrenia-in-adults-prevention-and-management-pdf-35109758952133
9.Fellner C. New Schizophrenia Treatments Address Unmet Clinical Needs. P t. 2017;42(2):130-134. PubMed
10.Dong M, Lu L, Zhang L, et al. Quality of Life in Schizophrenia: A Meta-Analysis of Comparative Studies. Psychiatr Q. 2019;90(3):519-532. doi:10.1007/s11126-019-09633-4 PubMed
11.Strassnig M, Signorile J, Gonzalez C, Harvey PD. Physical performance and disability in schizophrenia. Schizophr Res Cogn. 2014;1(2):112-121. doi:10.1016/j.scog.2014.06.002 PubMed
12.Hjorthøj C, Stürup AE, McGrath JJ, Nordentoft M. Years of potential life lost and life expectancy in schizophrenia: a systematic review and meta-analysis. Lancet Psychiatry. 2017;4(4):295-301. doi:10.1016/s2215-0366(17)30078-0 PubMed
13.Piotrowski P, Gondek TM, Królicka-Deręgowska A, Misiak B, Adamowski T, Kiejna A. Causes of mortality in schizophrenia: An updated review of European studies. Psychiatr Danub. 2017;29(2):108-120. doi:10.24869/psyd.2017.108 PubMed
14.Government of Canada. Schizophrenia in Canada. July 7, 2020. Accessed May 1, 2025. https://health-infobase.canada.ca/datalab/schizophrenia-blog.html?=undefined&wbdisable=true
15.Government of Canada. Canadian Chronic Disease Surveillance System (CCDSS) - Schizophrenia. Accessed May 1, 2025. https://health-infobase.canada.ca/ccdss/data-tool/
16.Chokhawala K, Stevens L. Antipsychotic Medications. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. February 26, 2023. Accessed May 2, 2025. https://www.ncbi.nlm.nih.gov/books/NBK519503/
17.Marder S. Stein MB, Friedman M, eds. Psychosis in adults: Initial management. UpToDate; November 13, 2024. Accessed March 21, 2025. https://www.uptodate.com
18.Kane JM, Rubio JM, Kishimoto T, Correll CU. Marder S, Friedman M, eds. Treatment-resistant schizophrenia. UpToDate; August 15, 2024. Accessed March 21, 2025. https://www.uptodate.com
19.Stroup TS, Marder S. Stein MB, Friedman M, eds. Schizophrenia in adults: Maintenance therapy and side effect management. UpToDate; June 27, 2024. Accessed March 21, 2025. https://www.uptodate.com
20.Remington G, Addington D, Honer W, Ismail Z, Raedler T, Teehan M. Guidelines for the Pharmacotherapy of Schizophrenia in Adults. Can J Psychiatry. 2017;62(9):604-616. doi:10.1177/0706743717720448 PubMed
21.Keepers GA, Fochtmann LJ, Anzia JM, et al. The American Psychiatric Association Practice Guideline for the Treatment of Patients With Schizophrenia. Am J Psychiatry. 2020;177(9):868-872. doi:10.1176/appi.ajp.2020.177901 PubMed
22.Norman R, Lecomte T, Addington D, Anderson E. Canadian Treatment Guidelines on Psychosocial Treatment of Schizophrenia in Adults. Can J Psychiatry. 2017;62(9):617-623. doi:10.1177/0706743717719894 PubMed
23.Health Quality Ontario. Schizophrenia Care in the Community for Adults. Accessed May 30, 2025. https://www.hqontario.ca/Evidence-to-Improve-Care/Quality-Standards/View-all-Quality-Standards/Schizophrenia-Care-in-the-Community/The-Quality-Standard-In-Brief
24.Government of Canada. Notices of Compliance Prescription Products for Human Use. January - December, 1993. Accessed April 16, 2025. https://www.canada.ca/content/dam/hc-sc/migration/hc-sc/dhp-mps/alt_formats/hpfb-dgpsa/txt/prodpharma/pre93et-eng.txt
25.Janssen Inc. Risperdal (risperidone tablets House Standard): 0.25, 0.5, 1, 2, 3 and 4 mg [product monograph]. April 14, 1993. Updated September 10, 2018. Accessed April 16, 2025. https://pdf.hres.ca/dpd_pm/00047330.PDF
26.Government of Canada. Notice of Compliance Information Risperdal Consta, Risperdal Consta 3N. November 25, 2022. Accessed April 16, 2025. https://health-products.canada.ca/noc-ac/nocInfo?no=4799
27.Canada's Drug Agency. SR0671-000 risperidone (Perseris). November 9, 2021. Accessed April 16, 2025. https://www.cda-amc.ca/risperidone
28.HLS Therapeutics Inc. Perseris (risperidone for extended-release injectable suspension): powder for suspension, 90 mg or 120 mg subcutaneous injection [product monograph]. November 19, 2020. Updated May 25, 2023. Accessed April 16, 2025. https://pdf.hres.ca/dpd_pm/00070975.PDF
29.Government of Canada. Drug Product Database Product information Perseris. August 14, 2024. Accessed April 16, 2025. https://health-products.canada.ca/dpd-bdpp/info?lang=eng&code=99721
30.Canada's Drug Agency. CDA-AMC Reimbursement Review Risperidone (Longavo) SR0811-000. 2025. Accessed August 13, 2025. https://www.cda-amc.ca/risperidone-0
31.Canada's Drug Agency. CADTH Reimbursement Review Risperidone for Extended Release Injectable Suspension (Perseris). November 2021. Accessed April 4, 2025. https://www.cda-amc.ca/sites/default/files/DRR/2021/SR0671-Perseris-Combine.pdf
32.Canadian Institute for Health Information. Ontario Mental Health Reporting System Metadata. Accessed May 30, 2025. https://www.cihi.ca/en/ontario-mental-health-reporting-system-metadata#:~:text=Data%20source&text=The%20tool%20was%20developed%20by,(SCIPP)%20case%2Dmix%20methodology
33.Marder SR, Cannon TD. Schizophrenia. N Engl J Med. 2019;381(18):1753-1761. doi:10.1056/NEJMra1808803 PubMed
34.Schmitt A, Falkai P, Papiol S. Neurodevelopmental disturbances in schizophrenia: evidence from genetic and environmental factors. J Neural Transm (Vienna). 2023;130(3):195-205. doi:10.1007/s00702-022-02567-5 PubMed
35.Addington D, Abidi S, Garcia-Ortega I, Honer WG, Ismail Z. Canadian Guidelines for the Assessment and Diagnosis of Patients with Schizophrenia Spectrum and Other Psychotic Disorders. Can J Psychiatry. 2017;62(9):594-603. doi:10.1177/0706743717719899 PubMed
36.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision (DSM-5-TR). Accessed April 30, 2025. https://www.psychiatryonline.org/doi/book/10.1176/appi.books.9780890425787
37.Gerlinger G, Hauser M, De Hert M, Lacluyse K, Wampers M, Correll CU. Personal stigma in schizophrenia spectrum disorders: a systematic review of prevalence rates, correlates, impact and interventions. World Psychiatry. 2013;12(2):155-64. doi:10.1002/wps.20040 PubMed
38.Hoftman GD. The Burden of Mental Illness Beyond Clinical Symptoms: Impact of Stigma on the Onset and Course of Schizophrenia Spectrum Disorders. American Journal of Psychiatry Residents' Journal. 2016;11(4):5-7. doi:10.1176/appi.ajp-rj.2016.110404
39.Health Quality Ontario. Schizophrenia Care in the Community for Adults 2023 Update. Accessed June 3, 2025. https://www.hqontario.ca/Portals/0/documents/evidence/quality-standards/qs-schizophrenia-care-in-the-community-quality-standard-en.pdf
40.Laboratorios Farmacéuticos ROVI, S.A. Clinical Study Report: ROV-RISP-2016-01. Multicenter, Randomized, Double-Blind, Placebo-controlled Study to Evaluate the Efficacy and Safety of Intramuscular Injections of Risperidone ISM® in Patients with Acute Exacerbation of Schizophrenia (PRISMA-3) [internal sponsor's report]. June 2, 2020.
41.UpToDate. Risperidone: Drug Information. UpToDate; 2025. Accessed March 21, 2025. https://www.uptodate.com
42.Government of Canada. Drug Product Database Product information Abilify Asimtufii 720 mg / 2.4 mL. August 14, 2024. Accessed June 26, 2025. https://health-products.canada.ca/dpd-bdpp/info?lang=eng&code=104340
43.Government of Canada. Drug Product Database Product information Abilify Asimtufii 960 mg / 3.2 mL. August 14, 2024. Accessed June 26, 2025. https://health-products.canada.ca/dpd-bdpp/info?lang=eng&code=104341
44.Otsuka Canada Pharmaceutical Inc. Abilify Maintena (Aripiprazole for prolonged release injectable suspension): 300 and 400 mg per vial, intramuscular injection. Abilify Asimtufii (Aripiprazole prolonged release injectable suspension): 720 mg/2.4 mL and 960 mg/3.2 mL, intramuscular injection [product monograph]. February 10, 2014. Updated January 17, 2025. Accessed June 2, 2025. https://pdf.hres.ca/dpd_pm/00078326.PDF
45.Janssen Inc. Invega Trinza (paliperidone palmitate prolonged-release injectable suspension): Suspension in pre-filled syringes, 175 mg/0.875 mL, 263 mg/1.315 mL, 350 mg/1.75 mL, and 525 mg/2.625 mL paliperidone (as paliperidone palmitate), Intramuscular (3 months) [product monograph]. June 22, 2016. Updated September 28, 2023. Accessed April 16, 2025. https://pdf.hres.ca/dpd_pm/00072662.PDF
46.Janssen Inc. Invega Sustenna (paliperidone palmitate prolonged-release injectable suspension): Suspension in pre-filled syringes, 50 mg/0.5 mL, 75 mg/0.75 mL, 100 mg/1 mL, and 150 mg/1.5 mL paliperidone (as paliperidone palmitate), Intramuscular [product monograph]. June 30, 2010. Updated September 27, 2023. Accessed April 16, 2025. https://pdf.hres.ca/dpd_pm/00072646.PDF
47.Janssen Inc. Risperdal Consta (risperidone powder for Injectable Prolonged-Release Suspension): 12.5 mg, 25 mg, 37.5 mg and 50 mg Intramuscular Injection [product monograph]. December 18, 2020. Accessed April 16, 2025. https://pdf.hres.ca/dpd_pm/00059290.PDF
48.Sivem Pharmaceuticals ULC. Risperidone: Risperidone Tablets 0.25 mg, 0.5 mg, 1 mg, 2 mg, 3 mg and 4 mg [product monograph]. December 20, 2022. Accessed April 16, 2025. https://pdf.hres.ca/dpd_pm/00068857.PDF
49.Bausch Health, Canada Inc. Sponsor's Summary of the Clinical Evidence [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Okedi, 75 mg or 100 mg, powder for extended-release suspension, intramuscular injection. March 21, 2025.
50.Correll CU, Litman RE, Filts Y, et al. Efficacy and safety of once-monthly Risperidone ISM(®) in schizophrenic patients with an acute exacerbation. npj Schizophr. 2020;6(1):37. doi:10.1038/s41537-020-00127-y
51.Filts Y, Litman RE, Martínez J, Anta L, Naber D, Correll CU. Long-term efficacy and safety of once-monthly Risperidone ISM® in the treatment of schizophrenia: Results from a 12-month open-label extension study. Schizophr Res. 2022;239:83-91. doi:10.1016/j.schres.2021.11.030 PubMed
52.Laboratorios Farmacéuticos, ROVI. Global Value Dossier of Okedi® for the treatment of schizophrenia in Europe [sponsor supplied reference]. 2022 Jan 26.
53.Bausch Health, Canada Inc. Bausch Health, Canada Inc. response to Canada's Drug Agency request for additional information regarding Okedi review on June 4, 2025 [internal additional sponsor's information]. June 6, 2025.
54.Busner J, Targum SD. The clinical global impressions scale: applying a research tool in clinical practice. Psychiatry (Edgmont). 2007;4(7):28-37. PubMed
55.Laboratorios Farmacéuticos ROVI, S.A. Statistical Analysis Plan: ROV-RISP-2016-01. Multicenter, Randomized, Double-Blind, Placebo-controlled Study to Evaluate the Efficacy and Safety of Intramuscular Injections of Risperidone ISM® in Patients with Acute Exacerbation of Schizophrenia (PRISMA-3) [internal sponsor's report]. February 15, 2019.
56.Laboratorios Farmacéuticos ROVI, S.A. Clinical Study Protocol: ROV-RISP-2016-01. Multicenter, Randomized, Double-Blind, Placebo-Controlled Study to Evaluate the Efficacy and Safety of Intramuscular Injections of Risperidone ISM® in Patients with Acute Exacerbation of Schizophrenia (PRISMA-3) [internal sponsor's report]. March 22, 2018.
57.Laboratorios Farmacéuticos ROVI, S.A. Clinical Study Protocol: ROV-RISP-2016-02. An Open-Label, One-Sequence Study to Evaluate the Steady-State Comparative Bioavailability of Intramuscular Risperidone ISM® and Oral Risperidone [internal sponsor's report]. March 23, 2018.
58.Laboratorios Farmacéuticos ROVI, S.A. Clinical Study Report: ROV-RISP-2016-02. An Open-Label, One-Sequence Study to Evaluate the Steady-State Comparative Bioavailability of Intramuscular Risperidone ISM® and Oral Risperidone [internal sponsor's report]. July 3, 2019.
59.Walling DP, Hassman HA, Anta L, et al. The Steady-State Comparative Bioavailability of Intramuscular Risperidone ISM and Oral Risperidone: An Open-Label, One-Sequence Study. Drug Des Devel Ther. 2021;15:4371-4382. doi:10.2147/DDDT.S332026 PubMed
60.Laboratorios Farmacéuticos, ROVI. CTD 2.7.2 Summary of Clinical Pharmacology Studies [sponsor supplied reference]. July 2021.
61.Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261-76. doi:10.1093/schbul/13.2.261 PubMed
62.Peralta V, Cuesta MJ. Psychometric properties of the positive and negative syndrome scale (PANSS) in schizophrenia. Psychiatry Res. 1994;53(1):31-40. doi:10.1016/0165-1781(94)90093-0 PubMed
63.Leucht S, Kane JM, Etschel E, Kissling W, Hamann J, Engel RR. Linking the PANSS, BPRS, and CGI: Clinical Implications. Neuropsychopharmacology. 2006;31(10):2318-2325. doi:10.1038/sj.npp.1301147 PubMed
64.Rabinowitz J, Mehnert A, Eerdekens M. To what extent do the PANSS and CGI-S overlap? J Clin Psychopharmacol. 2006;26(3):303-7. doi:10.1097/01.jcp.0000218407.10362.6e PubMed
65.European Medicines Agency. Guideline on clinical investigation of medicinal products, including depot preparations in the treatment of schizophrenia. September 20, 2012. Accessed May 31, 2025. https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-clinical-investigation-medicinal-products-including-depot-preparations-treatment-schizophrenia-revision-1_en.pdf
66.de Haan L, Weisfelt M, Dingemans P, Linszen D, Wouters L. Psychometric properties of the Subjective Well-Being Under Neuroleptics scale and the Subjective Deficit Syndrome Scale. Psychopharmacology (Berl). 2002;162(1):24-28. doi:10.1007/s00213-002-1054-x PubMed
67.Bausch Health, Canada Inc. Bausch Health, Canada Inc. response to Canada's Drug Agency request for additional information regarding Okedi review on May 22, 2025 [internal additional sponsor's information]. May 26, 2025.
68.Lieberman JA, Stroup TS, McEvoy JP, et al. Effectiveness of Antipsychotic Drugs in Patients with Chronic Schizophrenia. N Engl J Med. 2005;353(12):1209-1223. doi:10.1056/NEJMoa051688 PubMed
69.Ontario Ministry of Health. Ontario drug benefit formulary/comparative drug index. Accessed May 15, 2025. https://www.formulary.health.gov.on.ca/formulary/
70.Ontario Ministry of Health. Schedule of Benefits: Physician Services Under the Health Insurance Act [sponsor supplied reference]. 2024.
71.Rothe PH, Heres S, Leucht S. Dose equivalents for second generation long-acting injectable antipsychotics: The minimum effective dose method. Schizophr Res. 2018;193:23-28. doi:10.1016/j.schres.2017.07.033 PubMed
72.pan-Canadian Pharmaceutical Alliance. Abilify Maintena (aripiprazole) for schizophrenia. 2015. Accessed April 27, 2025. https://www.pcpacanada.ca/negotiation/20847
73.pan-Canadian Pharmaceutical Alliance. Invega Trinza (paliperidone palmitate) for schizophrenia. 2018. Accessed April 27, 2025. https://www.pcpacanada.ca/negotiation/20996
74.Bausch Health, Canada Inc. Pharmacoeconomic evaluation [internal sponsor's report]. Drug Reimbursement Review sponsor submission: PrOKEDI® (risperidone long-acting injection), 75 mg and 100 mg as a sterile two-syringe mixing system 2025.
75.Government of Saskatchewan. Saskatchewan Drug Plan: search formulary. 2025. Accessed May 21 2025. http://formulary.drugplan.ehealthsask.ca/SearchFormulary
76.Otsuka Canada Pharmaceutical Inc. PrABILIFY MAINTENA® (aripiprazole) for Prolonged Release Injectable Suspension 300 mg and 400 mg [product monograph] [sponsor supplied reference]. 2021. March 2, 2021.
77.Janssen Inc. PrRISPERDAL CONSTA® (risperidone powder) for Injectable Prolonged-Release Suspension 12.5 mg, 25 mg, 37.5 mg and 50 mg [Product Monograph] [sponsor supplied reference]. December 18, 2020.
78.Janssen Inc. PrINVEGA SUSTENNA® (paliperidone palmitate) Prolonged Release Injectable Suspension 50 mg/0.5 mL, 75 mg/0.75 mL, 100 mg/1 mL, and 150 mg/1.5 mL [Product Monograph] [sponsor supplied reference]. September 27, 2023.
79.Apotex Inc. Apo-Aripiprazole Depot (Aripiprazole for prolonged release injectable suspension): Powder for suspension, prolonged release, 300 mg / vial, 400 mg / vial, intramuscular injection [product monograph]. 2025. Accessed May 12, 2025. https://health-products.canada.ca/dpd-bdpp/info?lang=eng&code=104498
80.Pan-Canadian Pharmaceutical Alliance. Generic Drugs. 2023. Accessed May 12, 2025. https://www.pcpacanada.ca/generic-drug-framework
81.Canada's Drug Agency. CDA-AMC Reimbursement Review Risperidone (SR0811-000). 2025. Accessed June 2, 2025. https://www.cda-amc.ca/risperidone-0
82.Teva Canada Limited. Longavo (Risperidone extended-release injectable suspension): 50 mg / 0.14 mL, 75 mg / 0.21 mL, 100 mg / 0.28 mL. 125 mg / 0.35 mL in a single-dose pre-filled syringe for subcutaneous use [product monograph]. June 18, 2025. Accessed June 24, 2025. https://pdf.hres.ca/dpd_pm/00080831.PDF
83.Government of Canada. Notice of Compliance Information Okedi. Accessed June 3, 2025. https://health-products.canada.ca/noc-ac/nocInfo?no=32932
84.Government of Canada. Regulatory Decision Summary for Okedi. Accessed June 3, 2025. https://dhpp.hpfb-dgpsa.ca/review-documents/resource/RDS1722527517248
85.Ontario. Ontario Drug Benefit Formulary/Comparative Drug Index [sponsor supplied reference]. 2024.
86.Government of Saskatchewan. Saskatchewan Formulary [sponsor supplied reference]. 2024.
87.Ontario Ministry of Health. Schedule of benefits for physician services under the Health Insurance Act: (February 14, 2025 (effective March 3, 2025)). 2025. Accessed June 3, 2025. https://www.ontario.ca/files/2025-03/moh-schedule-benefit-2025-03-19.pdf
88.IQVIA. PharmaStat. 2025. Accessed June 3, 2025. https://www.iqvia.com/
Appendix 1: Additional Clinical Information
Please note that this appendix has not been copy-edited.
Table 21: PANSS Total Score Mean Change From Baseline to Day 85 — ANCOVA Sensitivity Analysis Using Observed Data (mPP Population)
ANCOVA Model | Placebo N = 109 | Okedi 75 mg N = 114 | Okedi 100 mg N = 112 |
|---|---|---|---|
LSM (SE; 95% CI) | −10.5 (1.56; −13.6 to −7.5) | −23.1 (1.52; −26.1 to −20.2) | −23.7 (1.53; −26.7 to −20.7) |
Okedi vs. Placebo | |||
LSMD (SE; 95% CI) | NA | −12.6 (2.17; −16.9 to −8.3) | −13.1 (2.18; −17.4 to −8.9) |
P value | NA | < 0.0001 | < 0.0001 |
LH MD (SE; 95% CI)a | NA | −12.6 (2.25; −17.0 to −8.2) | −12.6 (2.27; −17.1 to −8.2) |
CHW-adjusted P valuea | NA | < 0.0001 | < 0.0001 |
Hommel-adjusted P valueb | NA | < 0.0001 | < 0.0001 |
Model effects (P value) | |||
Treatment | < 0.0001 | NA | NA |
Baseline PANSS | < 0.001 | NA | NA |
Country | 0.242 | NA | NA |
ANCOVA = analysis of covariance; CHW = Cui, Hung, Wang; CI = confidence interval; LH = Lawrence and Hung; LSM = least squares means; LSMD = least squares means difference; MD = mean difference; mPP = modified per protocol, NA = not applicable; PANSS = Positive and Negative Syndrome Scale; SE = standard error; vs. = versus.
aCHW and LH methods combine results from stage 1 and stage 2.
bHommel adjustment of CHW P values.
Source: PRISMA-3 Clinical Study Report40
Table 22: PANSS Total Score Mean Change From Baseline to Day 85 — ANCOVA Model Sensitivity Analysis Using MI (Randomized, Modified Randomized, mPP, and PP Populations)
Outcome | Placebo | Okedi 75 mg | Okedi 100 mg | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
RP N = 147 | mRP N = 134 | mPP N = 109 | PP N = 119 | RP N = 145 | mRP N = 131 | mPP N = 114 | PP N = 126 | RP N = 146 | mRP N = 130 | mPP N = 112 | PP N = 121 | |
LSM (SE; 95% CI) | −12.3 (1.46; −15.2 to −9.4) | −11.4 (1.53; −14.4 to −8.3) | −11.5 (1.62; −14.7 to −8.4) | −12.5 (1.54; −15.5 to −9.5) | −22.7 (1.46; −25.5 to −19.8) | −23.2 (1.51; −26.1 to −20.2) | −23.6 (1.55; −26.6 to −20.5) | −23.0 (1.5; −25.9 to −20.0) | −23.2 (1.47; −26.1 to −20.3) | −23.0 (1.53; −26.0 to −20.0) | −24.0 (1.58; −27.1 to −20.9) | −24.1 (1.51; −27.1 to −21.2) |
Okedi vs. Placebo | ||||||||||||
LSMD (SE; 95% CI) | NA | NA | NA | NA | −10.4 (2.07; −14.4 to −6.3) | −11.8 (2.13; −16.0 to −7.6) | −12.0 (2.25; −16.4 to −7.6) | −10.5 (2.15; −14.7 to −6.3) | −10.9 (2.07; −15.0 to −6.9) | −11.7 (2.15; −15.9 to −7.5) | −12.4 (2.26; −16.9 to −8.0) | −11.6 (2.17; −15.9 to −7.4) |
P value | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
LH MD (SE; 95% CI)a | NA | NA | NA | NA | −10.2 (2.08; −14.3 to −6.2) | −11.6 (2.21; −15.9 to −7.3) | −11.9 (2.26; −16.3 to −7.4) | −10.3 (2.16; −14.5 to −6.1) | −10.6 (2.08; −14.7 to −6.6) | −11.4 (2.17; −15.6 to −7.1) | −11.9 (2.28; −16.3 to −7.4) | −11.1 (2.15; −15.3 to −6.9) |
CHW-adjusted P valuea | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
Hommel-adjusted P valueb | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 |
ANCOVA = analysis of covariance; CHW = Cui, Hung, Wang; CI = confidence interval; LH = Lawrence and Hung; LSM = least squares means; LSMD = least squares means difference; MD = mean difference; MI = multiple imputation; mPP = modified per protocol; mRP = modified randomized population; NA = not applicable; PANSS = Positive and Negative Syndrome Scale; PP = per protocol; RP = randomized population; SE = standard error; vs. = versus.
aCHW and LH methods combine results from stage 1 and stage 2.
bHommel adjustment of CHW P values. These are the primary P values of the study and are used to assess confirmatory superiority of each Okedi dose over placebo if P < 0.05 and if the corresponding P value from the ITT analysis is also P < 0.05.
Source: PRISMA-3 Clinical Study Report40
Table 23: PANSS Total Score Mean Change From Baseline to Day 85 — Subgroup Analysis by Country and Baseline PANSS Score (mITT Population)
MMRM | Placebo | Okedi 75 mg | Okedi 100 mg | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Country | Baseline PANSS | Country | Baseline PANSS | Country | Baseline PANSS | ||||||||
Ukraine N = 56 | US N = 76 | < 95 N = 60 | ≥ 95 N = 72 | Ukraine N = 57 | US N = 72 | < 95 N = 59 | ≥ 95 N = 70 | Ukraine N = 57 | US N = 72 | < 95 N = 58 | ≥ 95 N = 71 | ||
LSM (SE; 95% CI) | −11.5 (2.20; −15.8 to −7.2) | −10.8 (2.10; −14.9 to −6.7) | −7.6 (2.28; −12.1 to −3.2) | −14.1 (2.07; −18.2 to −10.0) | −25.0 (2.14; −29.2 to −20.8) | −24.3 (2.02; −28.3 to −20.3) | −20.7 (2.19; −25.0 to −16.4) | −27.9 (2.02; −31.9 to −23.9) | −29.0 (2.17; −33.3 to −24.8) | −20.8 (2.08; −24.9 to −16.7) | −18.7 (2.24; −23.1 to −14.3) | −29.8 (2.05; −33.8 to −25.7) | |
Okedi vs. Placebo | LSMD (SE; 95% CI) | NA | NA | NA | NA | −13.5 (3.06; −19.5 to −7.5) | −13.5 (2.91; −19.2 to −7.8) | −13.0 (3.15; −19.2 to −6.9) | −13.8 (2.90; −19.5 to −8.1) | −17.5 (3.08; −23.6 to −11.5) | −10.0 (2.95; −15.8 to −4.2) | −11.1 (3.18; −17.4 to −4.8) | −15.6 (2.91; −21.4 to −9.9) |
P value | NA | NA | NA | NA | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.0001 | < 0.001 | < 0.001 | < 0.0001 | |
CI = confidence interval; mITT = modified intention to treat; LSM = least squares means; LSMD = least squares means difference; MMRM = mixed model with repeated measures; PANSS = Positive and Negative Syndrome Scale; SE = standard error; vs. = versus.
Source: PRISMA-3 Clinical Study Report40
Figure 3: LS Mean Change From Baseline at Each Time Point in PANSS (mITT Population)
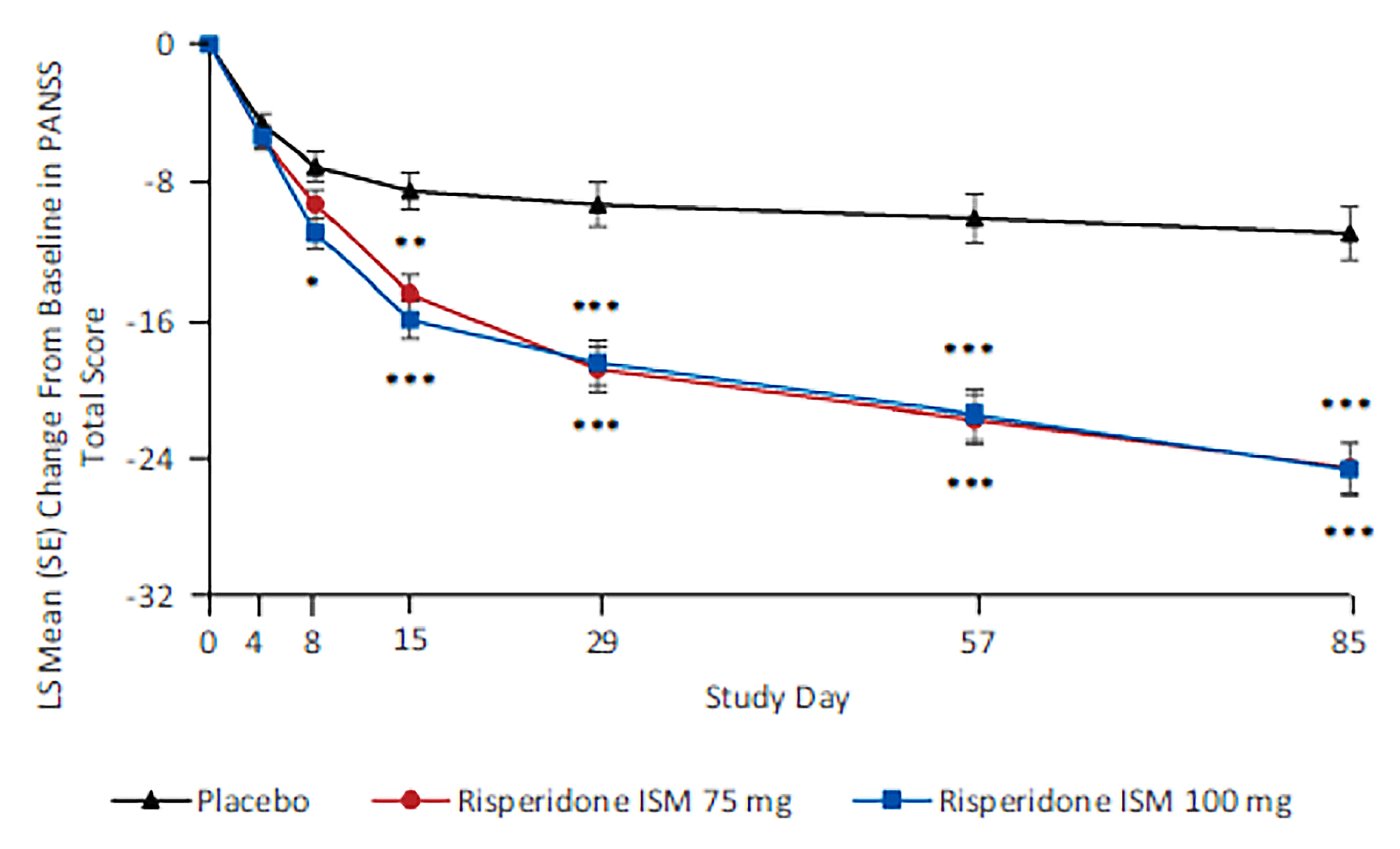
ISM = in situ microparticle; LS = least squares; mITT = modified intention to treat; PANSS = Positive and Negative Syndrome Scale; SE = standard error.
* P < 0.01
** P < 0.001
*** P < 0.0001
Source: PRISMA-3 Clinical Study Report,40 Correll et al. (2020).50 Correl CU, Litman RE, Filts Y, Llaudo J, Naber D, Torres F, Martinez J. Copyright © 2020, Correl, Litman, Filts, Llaudo, Naber, Torres, Martinez. Used under Creative Commons Licensing CC BY 4.0 https://creativecommons.org/licenses/by/4.0/ Please find material here: https://www.nature.com/articles/s41537-020-00127-y
Figure 4: LS Mean Change From Baseline at Each Time Point in CGI-S (mITT Population)
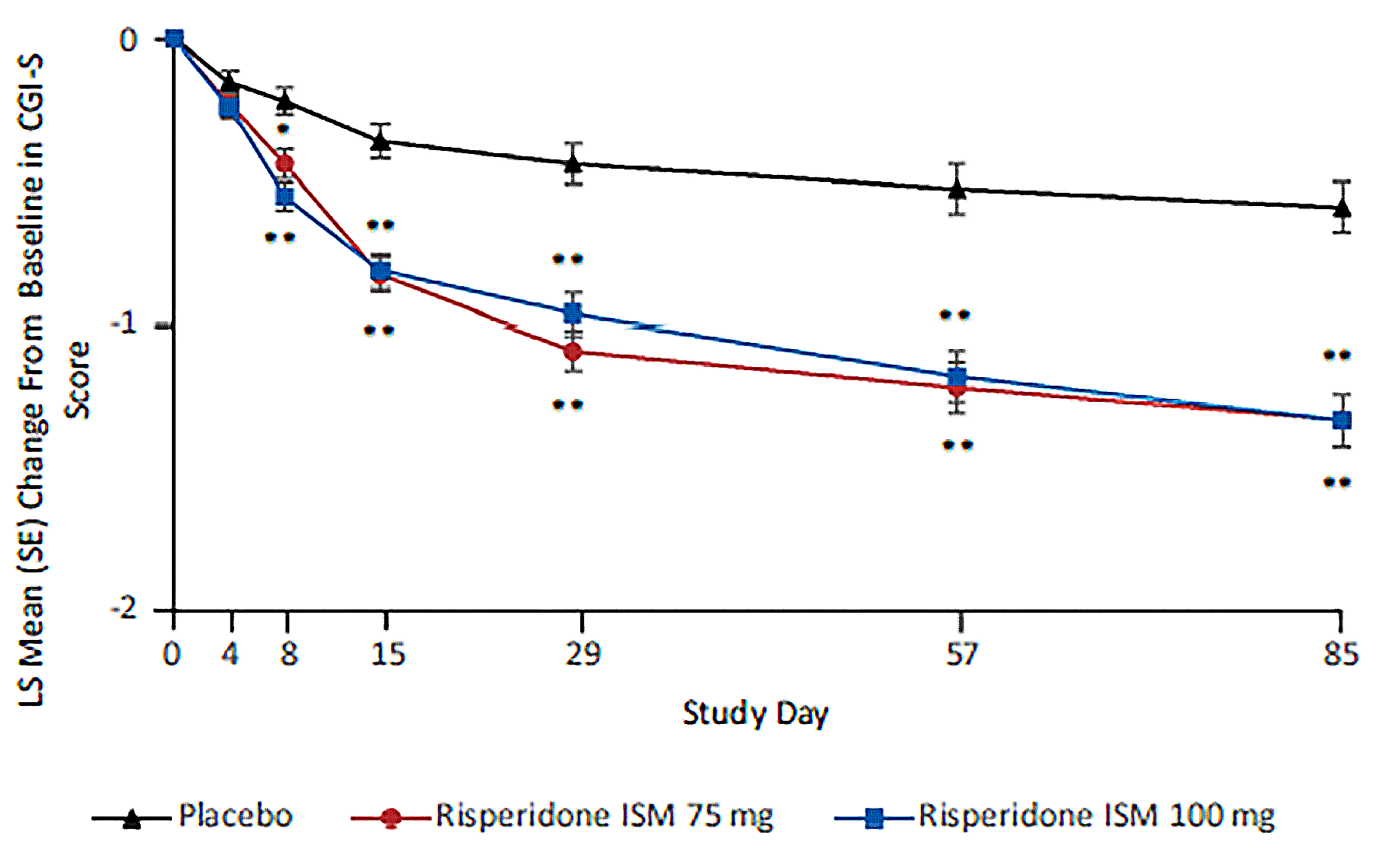
CGI-S = Clinical Global Impression-Severity; ISM = in situ microparticle; LS = least squares; mITT = modified intention to treat; SE = standard error.
* P < 0.01
** P < 0.0001
Source: PRISMA-3 Clinical Study Report,40Correll et al. (2020).50 Correl CU, Litman RE, Filts Y, Llaudo J, Naber D, Torres F, Martinez J. Copyright © 2020, Correl, Litman, Filts, Llaudo, Naber, Torres, Martinez. Used under Creative Commons Licensing CC BY 4.0 https://creativecommons.org/licenses/by/4.0/ Please find material here: https://www.nature.com/articles/s41537-020-00127-y
Table 24: Other Secondary End Points at Day 85 — PRISMA-3 (mITT Population)
Efficacy assessment | Placebo N = 132 | Okedi 75 mg N = 129 | Okedi 100 mg N = 129 |
|---|---|---|---|
CGI-I score | |||
LS mean (SE; 95% CI) | 3.3 (0.10; 3.1 to 3.5) | 2.5 (0.10; 2.3 to 2.7) | 2.5 (0.10; 2.3 to 2.7) |
LS mean difference (SE; 95%CI) | NA | −0.8 (0.14; −1.0 to −0.5) | −0.7 (0.14; −1.0 to −0.4) |
P value | NA | < 0.0001 | < 0.0001 |
Overall response | |||
Responders, n (%) | 24 (18.2) | 69 (53.5) | 60 (46.5) |
95% CI (%) | 12.0 to 25.8 | 44.5 to 62.3 | 37.7 to 55.5 |
Difference in proportions (%) | NA | 35.3 | 28.3 |
95% CI | NA | 23.7 to 45.2 | 16.7 to 38.3 |
P value | NA | < 0.0001 | < 0.0001 |
PANSS positive subscale | |||
LSM (SE; 95% CI) | −4.1 (0.50; −5.1 to −3.1) | −8.0 (0.49; −9.0 to −7.1) | −8.7 (0.50; −9.7 to −7.8) |
LSMD (SE; 95% CI) | NA | −3.9 (0.70; −5.3 to −2.5 | −4.6 (0.71; −6.0 to −3.2 |
P value | NA | < 0.0001 | < 0.0001 |
PANSS negative subscale | |||
LSM (SE; 95% CI) | −1.7 (0.40; −2.5 to −0.9) | −3.8 (0.38; −4.5 to −3.0) | −3.7 (0.39; −4.4 to −2.9) |
LSMD (SE; 95% CI) | NA | −2.1 (0.55; −3.1 to −1.0) | −2.0 (0.55; −3.1 to −0.9) |
P value | NA | < 0.001 | < 0.001 |
PANSS general psychopathology subscale | |||
LSM (SE; 95% CI) | −5.6 (0.82; −7.2 to −4.0) | −12.8 (0.79; −14.4 to −11.3) | −12.4 (0.81; −14.0 to −10.8) |
LSMD (SE; 95% CI) | NA | −7.3 (1.13; −9.5 to −5.0) | −6.8 (1.15; −9.1 to −4.6) |
P value | NA | < 0.0001 | < 0.0001 |
CGI-I = Clinical Global Impression-Improvement; CI = confidence interval; mITT = modified intention to treat; LS = least squares; LSM = least squares means; LSMD = least squares means difference; NA = not applicable; PANSS = Positive and Negative Syndrome Scale; SE = standard error.
Source: PRISMA-3 Clinical Study Report40
Figure 5: LS Mean Change From Baseline in PANSS Positive Subscale (mITT Population)
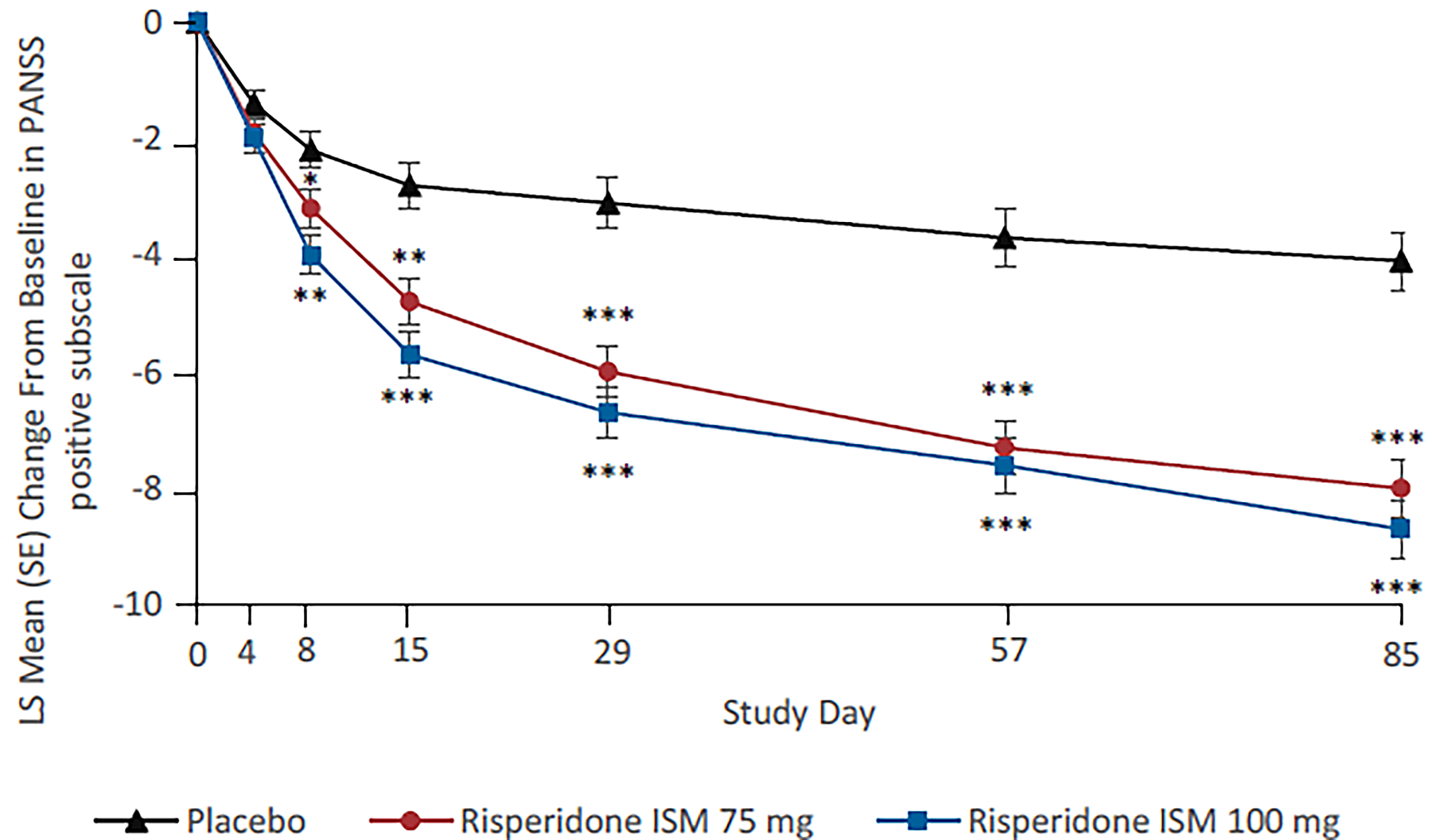
ISM = in situ microparticle; LS = least squares; mITT = modified intention to treat; PANSS = Positive and Negative Syndrome Scale; SE = standard error.
* P < 0.05
** P < 0.001
*** P < 0.0001
Source: PRISMA-3 Clinical Study Report,40 Correll et al. (2020).50 Correl CU, Litman RE, Filts Y, Llaudo J, Naber D, Torres F, Martinez J. Copyright © 2020, Correl, Litman, Filts, Llaudo, Naber, Torres, Martinez. Used under Creative Commons Licensing CC BY 4.0 https://creativecommons.org/licenses/by/4.0/ Please find material here: https://www.nature.com/articles/s41537-020-00127-y
Figure 6: LS Mean Change From Baseline in PANSS Negative Subscale (mITT Population)
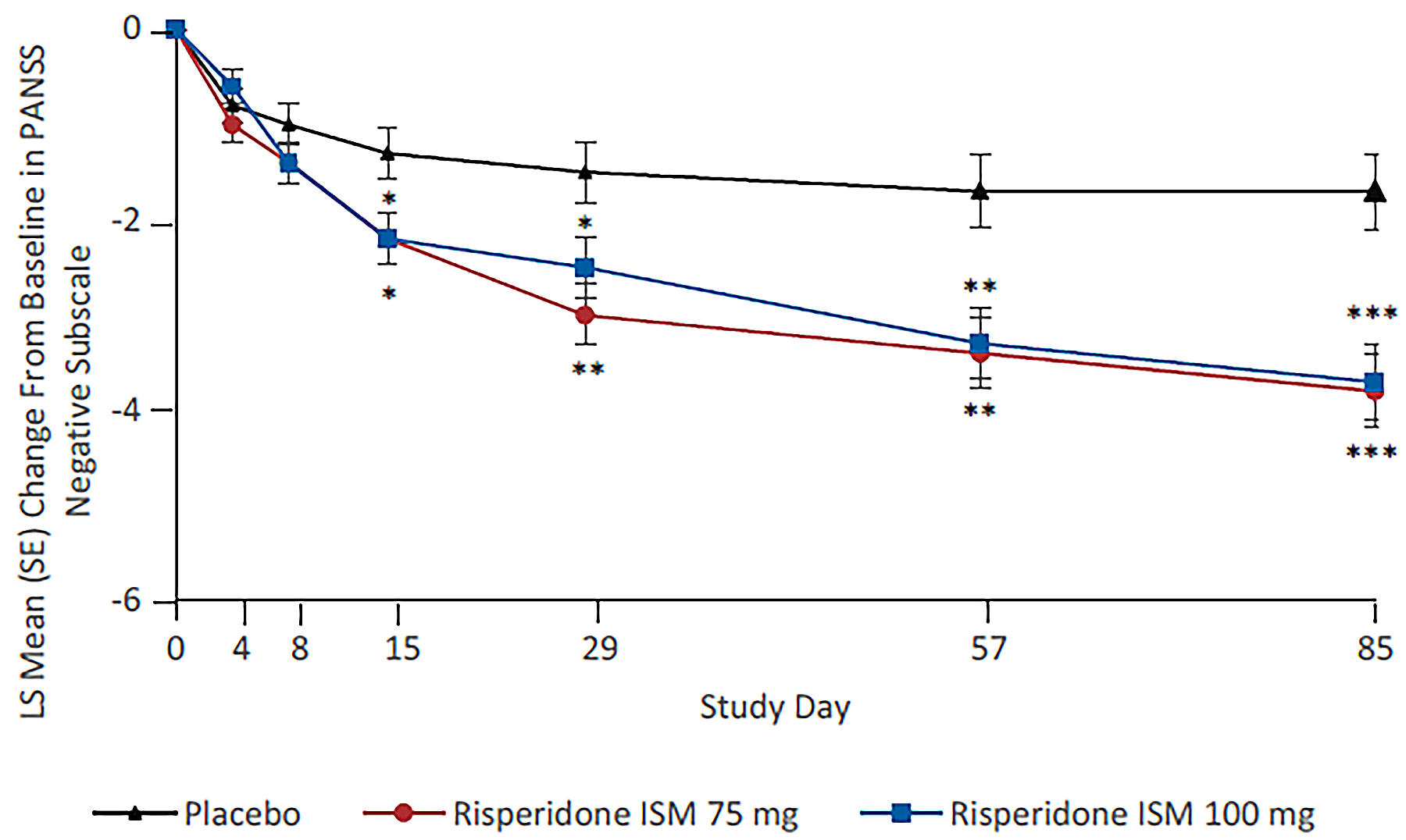
ISM = in situ microparticle; LS = least squares; mITT = modified intention to treat; PANSS = Positive and Negative Syndrome Scale; SE = standard error.
* P < 0.05
** P < 0.01
*** P < 0.001
Source: PRISMA-3 Clinical Study Report,40 Correll et al. (2020).50 Correl CU, Litman RE, Filts Y, Llaudo J, Naber D, Torres F, Martinez J. Copyright © 2020, Correl, Litman, Filts, Llaudo, Naber, Torres, Martinez. Used under Creative Commons Licensing CC BY 4.0 https://creativecommons.org/licenses/by/4.0/ Please find material here: https://www.nature.com/articles/s41537-020-00127-y
Figure 7: LS Mean Change From Baseline in PANSS General Psychopathology Subscale (mITT Population)
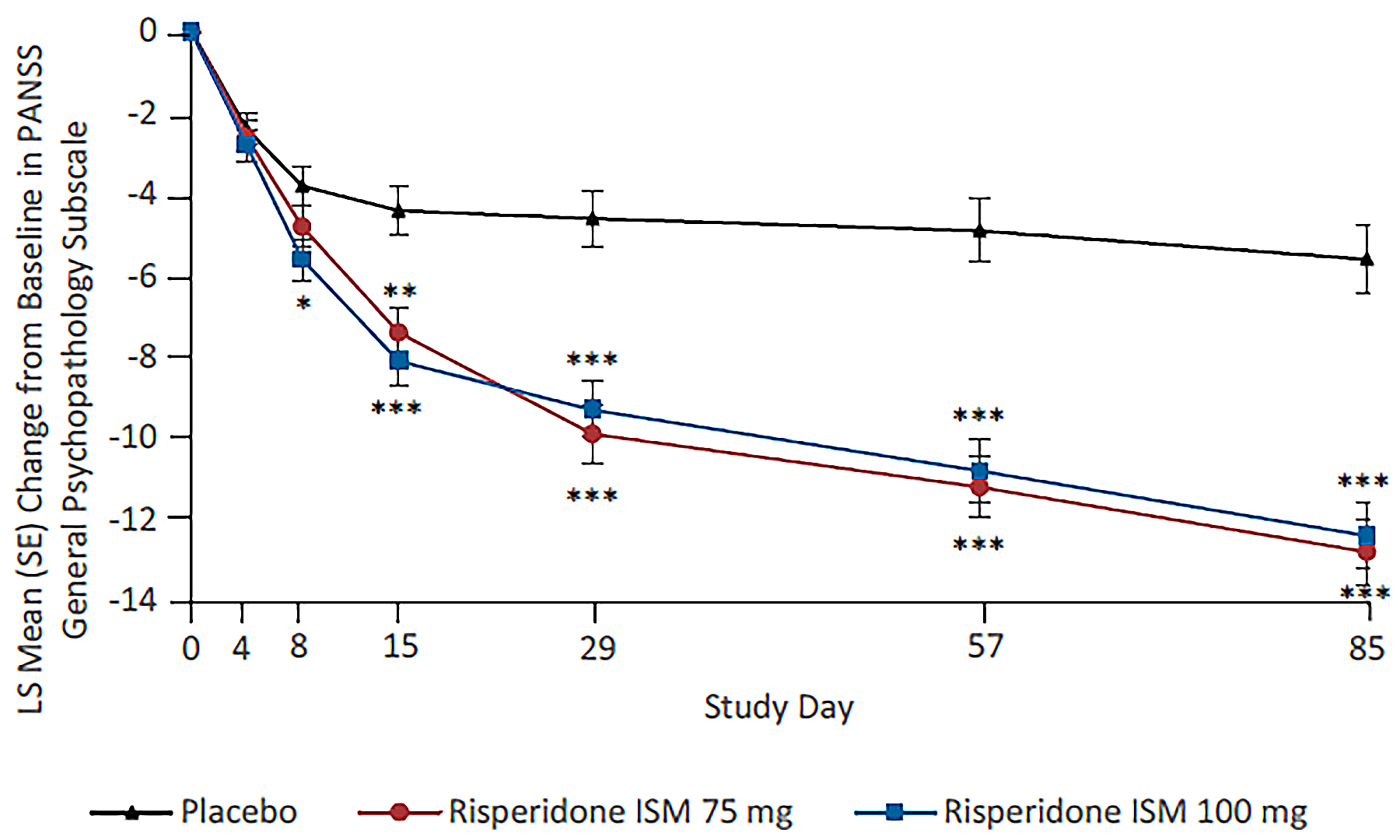
ISM = in situ microparticle; LS = least squares; mITT = modified intention to treat; PANSS = Positive and Negative Syndrome Scale; SE = standard error.
* P < 0.05
** P < 0.001
*** P < 0.0001
Source: PRISMA-3 Clinical Study Report,40 Correll et al. (2020).50 Correl CU, Litman RE, Filts Y, Llaudo J, Naber D, Torres F, Martinez J. Copyright © 2020, Correl, Litman, Filts, Llaudo, Naber, Torres, Martinez. Used under Creative Commons Licensing CC BY 4.0 https://creativecommons.org/licenses/by/4.0/ Please find material here: https://www.nature.com/articles/s41537-020-00127-y
Figure 8: ORR at Each Time Point (mITT Population)
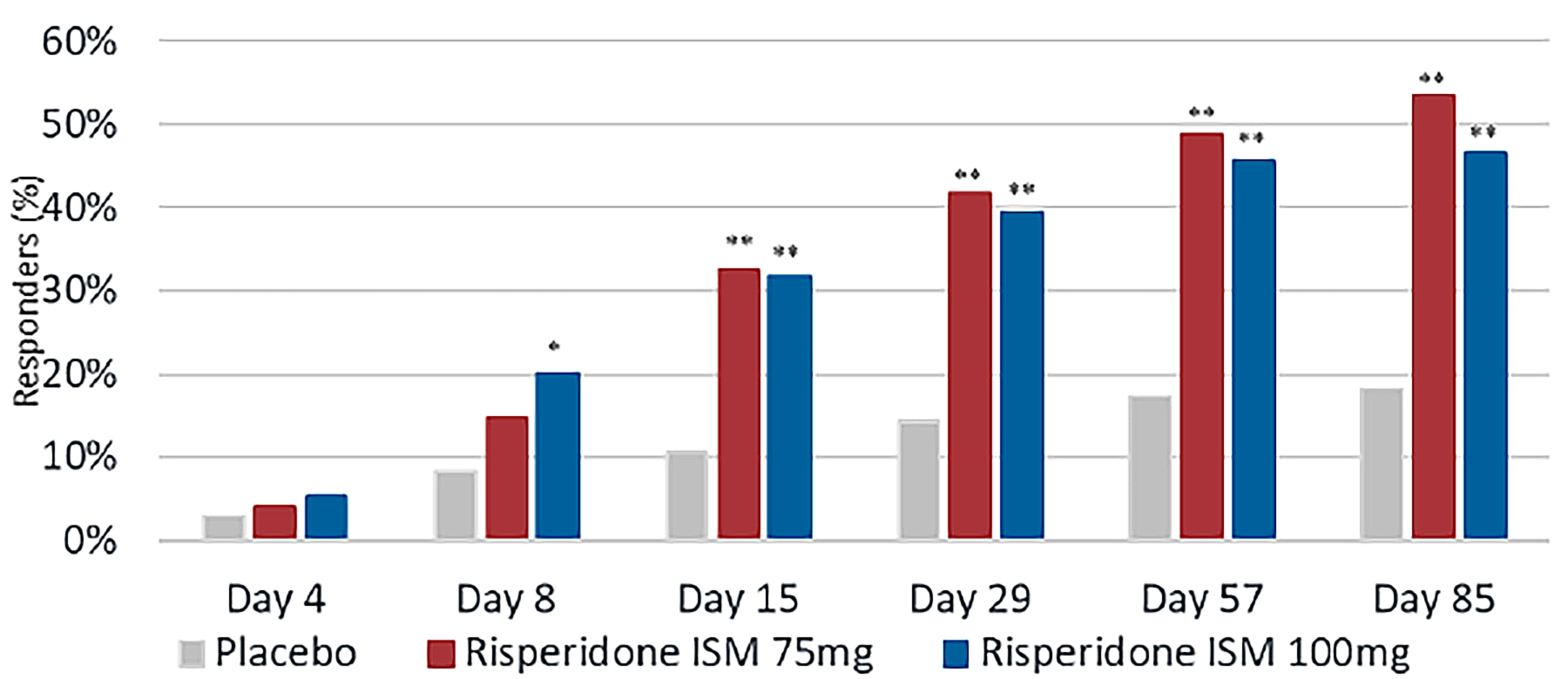
ISM = in situ microparticle; mITT = modified intention to treat; ORR = overall response rate.
* P < 0.01.
** P < 0.0001.
Source: PRISMA-3 Clinical Study Report,40 Correll et al. (2020).50 Correl CU, Litman RE, Filts Y, Llaudo J, Naber D, Torres F, Martinez J. Copyright © 2020, Correl, Litman, Filts, Llaudo, Naber, Torres, Martinez. Used under Creative Commons Licensing CC BY 4.0 https://creativecommons.org/licenses/by/4.0/ Please find material here: https://www.nature.com/articles/s41537-020-00127-y
Figure 9: Kaplan-Meier Plot of Time to PANSS Response at Each Time Point (mITT Population)
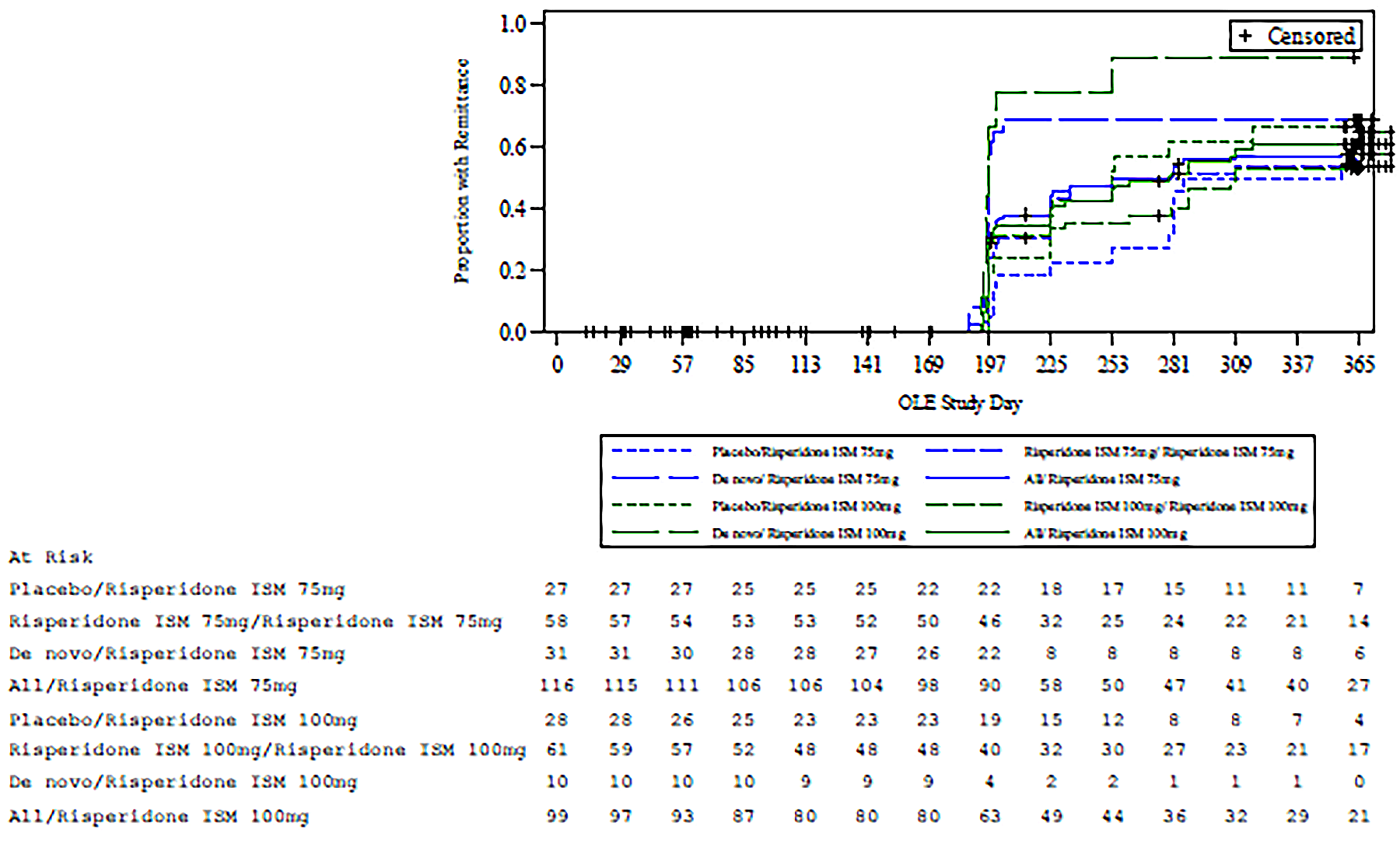
ISM = in situ microparticle; mITT = modified intention to treat; PANSS = Positive and Negative Syndrome Scale.
Source: PRISMA-3 Clinical Study Report40
Figure 10: Kaplan-Meier Plot of Time to Remittance in the OLE Phase (OLP)
ISM = in situ microparticle; OLE = open-label extension; OLP = open-label population.
Source: PRISMA-3 Clinical Study Report40
Figure 11: Kaplan-Meier Plot of Time to Discontinuation in the OLE Phase (OLP)
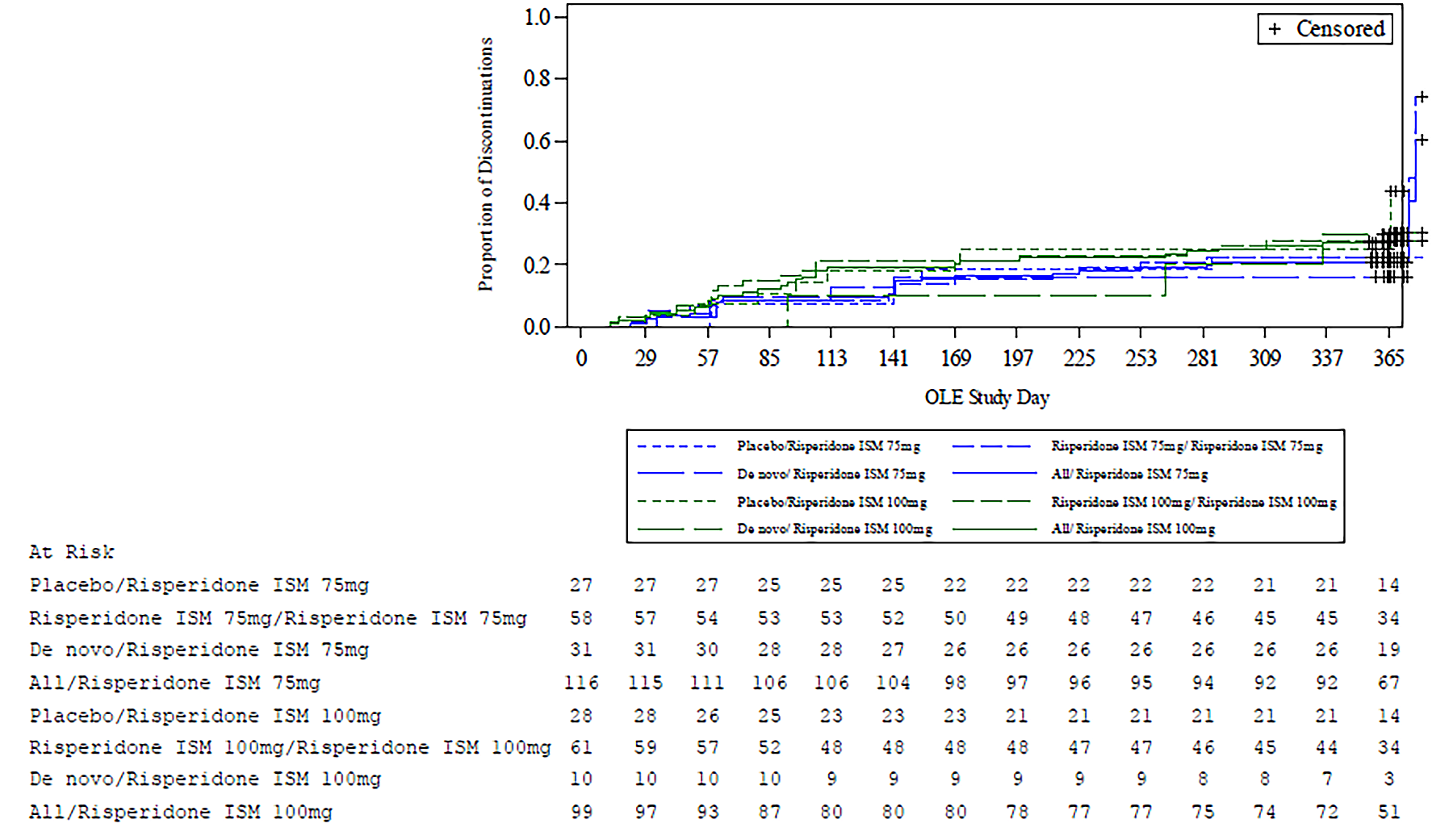
ISM = in situ microparticle; OLE = open-label extension; OLP = open-label population.
Source: PRISMA-3 Clinical Study Report.40
Appendix 2: Additional Economic Information
Please note that this appendix has not been copy-edited.
Additional Details on the Sponsor’s Submission
Table 25: Sponsor’s Submitted Drug Acquisition Cost Comparison
Generic name (brand name) | Strength or concentration | Dosage form | Price ($) | Recommended dosage regimen | Annual drug cost ($) | Difference in annual cost vs. OKEDI 75, $ (%) | Difference in annual cost vs. OKEDI 100, $ (%) |
|---|---|---|---|---|---|---|---|
Risperidone (Okedi) | — | 75 mg 100 mg Powder for extended-release suspension | 468.3200 624.4000 | 75 mg or 100 mg every 4 weeks | 6,088.16 8,117.20 | Reference | Reference |
Comparators | |||||||
Aripiprazole (Abilify Maintena) | 200 mg/mL | 300 mg 400 mg Powder for injection | 487.2002 487.2002 | 300 mg to 400 mg monthly | 5,846.40 5,846.40 | −241.76 (−4.0%) NC | NC −2,270.80 (−28.0%) |
Paliperidone (Invega Sustenna) | 100 mg/mL | 50 mg 75 mg 100 mg 150 mg Prefilled syringe | 327.4500 491.1800 544.4400 725.9100 | 50 mg to 150 mg monthly | 3,929.40 5,894.16 6,533.28 8,710.92 | NC -194.00 (−3.2%) NC NC | NC NC -1,583.92 (−19.5%) 593.72 (7.3) |
Paliperidone (Invega Trinza) | 200 mg/mL | 175 mg 263 mg 350 mg 525 mg Pre-filled syringe | 953.7300 1430.6100 1430.6100 1907.4900 | 175 mg to 525 mg every 3 months | 3,814.92 5,722.44 5,722.44 7,629.96 | NC -365.72 (−6.0%) NC NC | NC NC -2,394.76 (−29.5%) -487.24 (−6.0%) |
Risperidone (Risperdal Consta) | 6.25 mg/mL 12.5 mg/mL 18.75 mg/mL 25 mg/mL | 12.5 mg 25 mg 37.5 mg 50 mg Powder for injection | 100.3400a 208.3500 312.5100 416.6800 | 25 mg to 50 mg every 2 weeks | 2,608.84 5,417.10 8,125.26 10,833.68 | NC -671.06 (−11.0%) NC NC | NC NC 8.06 (0.1) 2,716.48 (33.5%)b |
Risperidone (generics) | 0.25 mg 0.5 mg 1 mg 2 mg 3 mg 4 mg | Tablet | 0.0878 0.1470 0.2031 0.4062 0.6083 0.8111 | 4 mg to 6 mg per dayc | 296.25 to 444.62 222.18 296.25 | NC NC NC NC −5,865.98 (−96.4%) NC | NC NC NC NC NC −7,820.95 (−96.4%) |
NC = not comparable; vs. = versus.
Note: All prices are from the Ontario Drug Benefit Formulary, accessed January 2025, unless otherwise specified.85
aCost obtained from the Saskatchewan Drug Plan Formulary as Risperdal Consta 12.5 mg/vial is not listed in Ontario.86
bCorrected from the sponsor’s submitted incremental cost of $4,987.28 (61.4%), which inadvertently compared the annual cost of risperidone ISM 75 mg every 4 weeks to the annual cost of risperidone 50 mg every 2 weeks in this column.
cThe recommended dose for Risperidone (generics) based on the product monograph is 4 mg to 6 mg per day. OKEDI injection 75 mg once every four weeks is similar to oral risperidone of 3 mg/day and OKEDI injection 100 mg once every four weeks is similar to oral risperidone of 4 mg/day. Patients who are stable on oral risperidone doses lower than 3 mg/day or higher than 4 mg/day may not be candidates for OKEDI. Therefore, the cost comparison vs OKEDI 75 mg and OKEDI 100 mg assumes risperidone doses of 3 mg/day and 4 mg/day, respectively.
Source: Sponsor’s submitted economic evaluation.74
Table 26: Sponsor’s Estimated Health Care Costs
Generic name (Brand name) | Number of injections per year | Aggregated health care cost per year ($)a | Difference in health care costs per year ($) |
|---|---|---|---|
Risperidone ISM (Okedi) | 13 | 88 | Reference |
Long-acting injectable comparators | |||
Risperidone (Risperdal Consta) | 26 | 175 | 88 |
Aripiprazole (Abilify Maintena) | 12 | 81 | –7 |
Paliperidone (Invega Sustenna) | 12 | 81 | –7 |
Paliperidone (Invega Trinza) | 4 | 27 | –61 |
Oral comparator | |||
Risperidone tablets (generics) | NA | 0 | –88 |
ISM = in situ microparticle; NA = not applicable.
aBased on health care components included in the table. Health care costs assumed an administration fee of 6.75 per injection.87
Additional Details on the CDA-AMC Reanalyses and Additional Analyses
CDA-AMC did not conduct any additional pharmacoeconomic analyses in the review of risperidone ISM.
Appendix 3: Submitted BIA and CDA-AMC Appraisal
Please note that this appendix has not been copy-edited.
Table 27: Summary of Key Take-Aways
Key take-aways of the BIA |
|---|
|
BIA = budget impact analysis; CDA-AMC = Canada's Drug Agency; ISM = in situ microparticle; LAI = long-acting injection.
Summary of Sponsor’s BIA
The sponsor submitted a BIA that estimated the expected incremental budgetary impact of reimbursing risperidone ISM for the treatment of adults with schizophrenia.
The BIA was conducted from the perspective of public drug plan payers over a 3-year time horizon (2026 to 2028), with 2025 as the base year. The sponsor’s estimate reflects the aggregated results from the jurisdictional provincial budgets (excluding Quebec) as well as the Non-Insured Health Benefits program. The sponsor used IQVIA Pharmastat data from 2022 to 2024 to estimate market size. The sponsor’s base case included drug acquisition costs, dispensing fees, and markups. The market uptake for risperidone ISM was based on internal forecasting conducted by the sponsor. Key inputs to the BIA are documented in Table 28.
The sponsor estimated the 3-year incremental budgetary savings associated with risperidone ISM for the treatment of schizophrenia in adults would be $875,856 (savings in year 1 = $104,333, year 2 = $324,167, year 3 = $447,356).
Table 28: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as year 1 / year 2 / year 3 if appropriate) |
|---|---|
Proportion of total claims attributable to patients with schizophreniaa | |
Risperidone LAI q.2.w. (Risperdal Consta) | 75% |
Paliperidone LAI q.m (Invega Sustenna) | 75% |
Paliperidone LAI q.3.m. (Invega Trinza) | 75% |
Aripiprazole LAI q.m. (Abilify Maintena) | 60% |
Risperidone tablets (generic) | 41% |
Market uptake proportionsb | |
Market uptake of risperidone ISM from other LAIs | 2.5% / 8.5% / 13.0% |
Market uptake of risperidone ISM from oral risperidone | 0% / 0% / 0% |
Market uptake, reference scenario, patient-years of treatmentc | |
Risperidone ISM q.4.w. (Okedi) | 0 / 0 / 0 |
Risperidone LAI q.2.w. (Risperdal Consta) | 1,183 / 1,096 / 1,011 |
Paliperidone LAI q.m. (Invega Sustenna) | 12,850 / 13,387 / 13,925 |
Paliperidone LAI q.3.m. (Invega Trinza) | 5,214 / 5,526 / 5,840 |
Aripiprazole LAI q.m. (Abilify Maintena) | 9,588 / 10,432 / 11,277 |
Risperidone tablets (generic) | 79,994 / 95,561 / 111,583 |
Market uptake, new drug scenario, patient-years of treatmentc | |
Risperidone ISM q.4.w. (Okedi) | 721 / 2,588 / 4,167 |
Risperidone LAI q.2.w. (Risperdal Consta) | 1,153 / 1,003 / 879 |
Paliperidone LAI q.m. (Invega Sustenna) | 12,529 / 12,249 / 12,115 |
Paliperidone LAI q.3.m. (Invega Trinza) | 5,083 / 5,057 / 5,081 |
Aripiprazole LAI q.m. (Abilify Maintena) | 9,348 / 9,546 / 9,811 |
Risperidone tablets (generic) | 79,994 / 95,561 / 111,583 |
Cost of treatment (weighted average cost per patient-year)d | |
Risperidone ISM q.4.w. (Okedi) | $6,564 |
Risperidone LAI q.2.w. (Risperdal Consta) | $8,521 |
Paliperidone LAI q.m. (Invega Sustenna) | $7,417 |
Paliperidone LAI q.3.m. (Invega Trinza) | $6,562 |
Aripiprazole LAI q.m. (Abilify Maintena) | $4,510 |
Risperidone tablets (generic) | $32 to $1,941, depending on brand and dose |
ISM = in situ microparticle; LAI = long-acting injectable; q.m. = every month; q.3.m. = every 3 months; q.2.w. = every 2 weeks; q.4.w. = every 4 weeks.
aCited as data on file from clinical expert.1
bCited as the sponsor’s internal product forecast.1
cTreatment patient-years were forecast from the number of public claims for each product in 2022 to 2024 as reported by IQVIA Pharmastat and linearly projected through to 2028, then divided by the standard number of doses patients receive each year (e.g., 26 for risperidone LAI [Risperdal Consta], 4 for paliperidone LAI [Invega Trinza]).1 For oral risperidone, the number of publicly funded units for each product were divided by 365.
dWeighted average cost per patient-year is based on the proportion of claims for each dose of a product based on IQVIA Pharmastat public claims data, multiplied by the annual cost of treatment with that dose. The sponsor assumed that 75% of patients receiving risperidone ISM (Okedi) would use the 100 mg every 4 week dose, with the remaining 25% using the 75 mg every 4 weeks dose.1 The weighted average doses of each comparator used to inform comparator costs were not constrained to those the sponsor considered clinically similar to doses of risperidone ISM (Okedi).
CDA-AMC Appraisal of the Sponsor’s BIA
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Use of a claims-based approach to estimate market size introduces uncertainty: The sponsor estimated market size based on a projection of 2022 to 2024 public claims data for the comparator LAI antipsychotics from IQVIA Pharmastat,88 and then estimating the proportion of claims that were attributable to schizophrenia with expert opinion (Table 28). The number of claims were then converted to the number of patient-years of treatment based on the assumed number of injections per year or tablets per day. This method introduces uncertainty as the proportion of claims for each comparator that are attributable to schizophrenia is uncertain and may differ between different products. The method also assumes that administration frequency is similar between indications. Finally, the estimation of patient-years of therapy is particularly uncertain for oral risperidone, where patients may take multiple tablets of the same or different strengths daily to achieve their target dose. The sponsor’s BIA forecasts that the number of person-years of treatment with oral risperidone that patients with schizophrenia in the pan-Canadian population will receive will increase from approximately 66,600 in the base year to 111,500 in year 3, a growth rate not explained by the sponsor.
CDA-AMC was unable to address this limitation in reanalysis.
Uncertainty in the uptake of risperidone ISM and comparators being displaced: The sponsor assumed that risperidone ISM will displace all LAI antipsychotics in proportion to their reference scenario market share. Clinical expert input obtained by CDA-AMC did not find this assumption to be plausible, instead indicating that the uptake of risperidone ISM would for the most part displace risperidone biweekly (i.e., Risperdal Consta), with patients either switching to less frequent risperidone dosing or be those who were going to switch to a risperidone product anyway due to unsatisfactory response or intolerance to another antipsychotic, who would otherwise begin using risperidone biweekly. Additionally, clinical expert opinion obtained by CDA-AMC found the sponsor’s projection that risperidone ISM would capture 2.5%, 8.5%, and 13% of the LAI antipsychotic market in years 1, 2, and 3 of its potential funding to be optimistic, noting that the sponsor’s projected market share for risperidone biweekly was only 4%, 3.6% and 3.2% of the LAI antipsychotic market in years 1, 2, and 3, respectively (e.g., risperidone biweekly has 1,183 of 28,834 antipsychotic LAI patient-years of treatment in year 1). It was deemed implausible that the funding of risperidone ISM would more than quadruple the risperidone proportion of the LAI antipsychotic market. Finally, clinical expert input obtained by CDA-AMC indicated that the availability risperidone ISM might increase the proportion of patients using LAI risperidone rather than oral risperidone by as much as 5%, due to the reduced injection frequency of risperidone ISM relative to risperidone biweekly.
In its base case, CDA-AMC halved the uptake of risperidone ISM from the LAI comparators. In a scenario analysis, CDA-AMC assumed that risperidone ISM would capture an additional 2%, 3%, and 5% of risperidone biweekly’s patient-years of treatment in the base year from oral risperidone in years 1, 2, and 3, respectively. Due to the structure of the submitted BIA, CDA-AMC was unable to alter the proportion of risperidone ISM uptake coming from each LAI comparator.
Dosing of displaced comparators is not consistent with the sponsor’s estimated dose equivalencies: In the submitted cost comparison, the sponsor assumed that risperidone ISM 75 mg every 4 weeks was comparable only to risperidone 25 mg biweekly, aripiprazole 300 mg monthly, paliperidone 75 mg monthly, and paliperidone 263 mg quarterly, while risperidone ISM 100 mg every 4 weeks was considered comparable to risperidone 37.5 or 50 mg biweekly, aripiprazole 400 mg monthly, paliperidone 100 or 150 mg monthly, and paliperidone 350 and 525 mg quarterly. However, in the BIA, the sponsor assumed that the weighted average cost per dose of all available strengths of each comparator would be displaced by the weighted average cost of risperidone ISM, where 25% of patients were assumed to receive the 75 mg dose with the other 75% receiving the 100 mg dose). In doing so, the sponsor underestimated the cost of risperidone biweekly and overestimated the relative cost of paliperidone monthly and quarterly relative to that of risperidone ISM.
Due to the structure of the BIA, CDA-AMC was unable to address this limitation in reanalysis. If all LAI antipsychotics are assumed to be displaced in proportion to their reference scenario market share and if doses of risperidone ISM are assumed to displace only those doses of comparators assumed equivalent by the sponsor, this limitation would be expected to lead to an underestimation of the budgetary impact of reimbursing risperidone ISM (i.e., the incremental cost of risperidone ISM is underestimated or the incremental savings are overestimated).
Dispensing fees and markups were inappropriately calculated: The sponsor’s base case included dispensing fees and markups. Typically, CDA-AMC removes these when conducting reanalyses but does not consider their inclusion to be a limitation of the sponsor’s submission. However, in the submitted BIA, the sponsor calculated the pan-Canadian (labelled “national”) dispensing fees and markups by taking a simple average of those of the other jurisdictions without weighting by the proportion of patient-years each jurisdiction represented. Additionally, dispensing fees and markups for each jurisdiction appear to be included in the total cost calculations for those jurisdictions, which are then summed to yield national costs for the reference and new drug scenarios. This implies that all such fees are already included within the national analysis. However, when CDA-AMC tested these parameters by changing the jurisdictional fee and markup inputs while holding the national average fees and markups steady, and vice versa, the analyses yielded unpredictable and discrepant results (e.g., setting the national fees and markups to 0 without altering the other jurisdictional values resulted in a 3-year pan-Canadian budget impact of –$4.6 million, while holding the national fees and markups at their submitted value and altering all other jurisdiction’s fees and markups to 0 resulted in a 3-year pan-Canadian budget impact of +$2.1 million). As such, the sponsor’s analysis appears to be miscalculating and/or double-counting at least some of such costs. Due to the way in which the BIA is programmed, CDA-AMC was unable to trace the cause of these discrepancies within the time frame of the review.
CDA-AMC excluded dispensing fees and markups from all reanalyses to remove unclear errors in their calculation.
Confidential pricing agreements: While the submitted price for risperidone ISM is within the range of the publicly available list prices of other LAI antipsychotics, these list prices are likely higher than the price paid by jurisdictional drug plans due to confidential pricing agreements for at least some of the comparators.72,73 Therefore, the submitted price of risperidone ISM likely requires a price reduction to avoid incurring additional costs relative to its comparators.
CDA-AMC was unable to address this limitation in reanalyses, as the negotiated prices of the comparators are unknown.
CDA-AMC Reanalyses of the BIA
CDA-AMC revised the sponsor’s submitted analysis by making changes in model parameter values and assumptions, in consultation with clinical experts, as outlined in Table 29.
Table 29: Revisions to the Submitted BIA
Stepped analysis | Sponsor’s value or assumption | CDA-AMC value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
1. Remove dispensing fees/markups | Included | Excluded |
Changes to derive the CDA-AMC base case | ||
1. Risperidone ISM uptake (Y1/Y2/Y3) | 2.5% / 8.5% / 13% | 1.25% / 4.25% / 6.50% |
CDA-AMC base case | Correction 1 + Reanalysis 1 | |
BIA = budget impact analysis; CDA-AMC = Canada's Drug Agency; ISM = in situ microparticle.
The results of the CDA-AMC step-wise reanalysis are presented in summary format in Table 30 and a more detailed breakdown is presented in Table 31. In the CDA-AMC base case, the 3-year budget savings associated with reimbursing risperidone ISM for the treatment of adult patients with schizophrenia was $792,827 (year 1 = $85,078, year 2 = $283,557, year 3 = $424,193).
Table 30: Summary of the CDA-AMC Reanalyses of the BIA
Stepped analysis | Three-year total |
|---|---|
Submitted base case | -$875,856 |
Sponsor’s corrected base case, markups and dispensing fees removed | -$1,585,654 |
CDA-AMC reanalysis 1: risperidone ISM uptake halved | -$792,827 |
CDA-AMC base case (reanalysis 1) | -$792,827 |
BIA = budget impact analysis; CDA-AMC = Canada's Drug Agency; ISM = in situ microparticle.
CDA-AMC used the CDA-AMC base case to conduct scenario analyses to explore uncertainty in the estimated budget impact of reimbursing risperidone ISM. The results are provided in Table 31.
Assuming risperidone ISM captures market share from oral risperidone equivalent to 2%, 3%, and 5% of the patient-years of treatment estimated for risperidone biweekly in the base year in years 1, 2, and 3, respectively.
Assuming generic aripiprazole LAI becomes available at 55% of the list price of the originator brand (refer to the Issues for Consideration section of this report).
Table 31: Disaggregated Summary of the BIA
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference total | $213,280,924 | $227,028,783 | $241,225,605 | $255,568,397 | $723,822,785 |
Risperidone ISM (Okedi) | $0 | $0 | $0 | $0 | $0 | |
All other comparators | $213,280,924 | $227,028,783 | $241,225,605 | $255,568,397 | $723,822,785 | |
New drug total | $213,280,924 | $226,924,450 | $240,901,438 | $255,121,042 | $722,946,930 | |
Risperidone ISM (Okedi) | $0 | $5,086,305 | $18,257,292 | $29,401,196 | $52,744,794 | |
All other comparators | $213,280,924 | $221,838,144 | $222,644,146 | $225,719,846 | $670,202,136 | |
Budget impact | $0 | -$104,333 | -$324,167 | -$447,356 | -$875,856 | |
CDA-AMC base case | Reference total | $190,386,948 | $201,654,636 | $213,094,043 | $224,598,525 | $639,347,204 |
Risperidone ISM (Okedi) | $0 | $0 | $0 | $0 | $0 | |
All other comparators | $190,386,948 | $201,654,636 | $213,094,043 | $224,598,525 | $639,347,204 | |
New drug total | $190,386,948 | $201,569,559 | $212,810,486 | $224,174,332 | $638,554,377 | |
Risperidone ISM (Okedi) | $0 | $2,365,752 | $8,491,866 | $13,675,138 | $24,532,756 | |
All other comparators | $190,386,948 | $199,203,807 | $204,318,621 | $210,499,194 | $614,021,621 | |
Budget impact | $0 | -$85,078 | -$283,557 | -$424,193 | -$792,827 | |
CDA-AMC scenario analyses | ||||||
Scenario 1: risperidone ISM displaces oral risperidone | Reference total | $190,386,948 | $201,654,636 | $213,094,043 | $224,598,525 | $639,347,204 |
New drug total | $190,386,948 | $201,767,793 | $213,165,737 | $224,865,720 | $639,799,250 | |
Budget Impact | $0 | $113,157 | $71,694 | $267,195 | $452,046 | |
Scenario 2: generic aripiprazole LAI | Reference total | $167,383,748 | $170,841,476 | $179,033,998 | $187,244,591 | $537,120,065 |
New drug total | $167,383,748 | $171,071,709 | $179,916,919 | $188,748,831 | $539,737,459 | |
Budget impact | $0 | $230,233 | $882,921 | $1,504,240 | $2,617,394 | |
BIA = budget impact analysis; CDA-AMC = Canada’s Drug Agency; ISM = in situ microparticle; LAI = long-acting injectable.
Note: The CDA-AMC reanalysis is based on the publicly available prices of the comparator treatments.
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.