CADTH Health Technology Review
Peripherally Inserted Central Catheters in Pediatric Patients Performed by Radiologists Using Fluoroscopy
Rapid Review
Authors: Amanda Shane, Zahra Premji
Abbreviations
AIEOP
Italian Association of Pediatric Hematology and Oncology
CVAD
central venous access device
CVC
central venous catheter
IR
interventional radiology
miniMAGIC
Michigan Appropriateness Guide for Intravenous Catheters in Pediatrics
PICC
peripherally inserted central catheter
PICU
pediatric intensive care unit
Key Messages
Evidence from 2 clinical studies showed that there was no difference in the rates of infection and complications between peripherally inserted central catheter (PICC) insertion at the bedside and insertion in Interventional Radiology (IR) suites. However, each of these studies focused on small subgroups of the larger pediatric population and had other methodological limitations.
Evidence from 1 clinical study in a single quaternary, non-cardiac, pediatric intensive care unit suggested that the median time from PICC line order to successful insertion was longer for lines placed in the IR compared to at the bedside.
Two guidelines with numerous quality limitations were identified that recommend ultrasound guidance for insertion of central venous access devices (CVAD), including PICCs: 1 was aimed at all pediatric patients and 1 was aimed at onco-hematological pediatric patients.
Context and Policy Issues
Peripherally inserted central catheters (PICC) are a type of central venous catheter that is inserted into peripheral veins in order administer medication over the intermediate to long-term (e.g., prolonged antibiotic administration).1 Several methods exist for the insertion of PICC lines including blind insertion (using anatomic landmarks), fluoroscopy, and most commonly, ultrasound guidance.2 PICC lines may be inserted by various medical personnel (e.g., nurses, non-radiologist physicians, interventional radiologists) and in various locations (e.g., at the bedside or in interventional radiology [IR] suites). The choice of insertion technique and location may depend on several factors including clinical indication (e.g., patients with critical airways could be considered for bedside placement; patients with a history of deep vein thrombosis could be considered for placement in IR),3 facility availability, personnel availability, and institutional guidelines.3,4
There is varying practice with respect to PICC line insertion in the Canadian pediatric population. A review and summary of the relevant literature may help inform process decisions within Canadian health care institutions.
The aim of this report is to summarize the evidence regarding the comparative clinical effectiveness of PICC insertions performed by radiologists in IR suites, compared to nurses at the bedside, and to summarize the evidence-based guidelines regarding the optimal approach for PICC insertions in pediatric patients.
Research Questions
What is the comparative clinical effectiveness of peripherally inserted central catheter (PICC) insertions performed by radiologists in interventional radiology suites versus PICC insertions performed by nurses at the bedside/unit in pediatric patients?
What are the evidence-based guidelines regarding the optimal approach for PICC insertions in pediatric patients?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, the Cochrane Database of Systematic Reviews, the international HTA database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. For question 1, the main search concepts were peripherally inserted central catheters, pediatric populations, and relevant radiologic techniques. No filters were applied to this search to limit retrieval by study type. Comments, newspaper articles, editorials, and letters were excluded. For question 2, the main search concept was peripherally inserted central catheters. Search filters were applied to this search to limit retrieval to guidelines. Where possible, retrieval was limited to the human population. The searches were also limited to English-language documents published between January 1, 2015 and December 12, 2020.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
Population | Pediatric patients undergoing PICC insertions |
Intervention | PICC insertions performed by radiologists in IR suites using fluoroscopy for insertion guidance Include: Studies using any method of catheter tip location confirmation (e.g., fluoroscopy, X-ray, ultrasound) performed by the radiologist |
Comparator | Q1: PICC insertions performed by nurses at the bedside using chest X-ray for catheter tip location confirmation Include: Studies involving nurses using no imaging (i.e., blind catheter placement) or another imaging modality (e.g., ultrasound) for insertion guidance Exclude: Studies where nurses use electrocardiograph for insertion guidancea Q2: Not applicable |
Outcomes | Q1: Clinical effectiveness (e.g., procedure length and success rates, multiple attempts or failure to access vessel, patient satisfaction, length of sedation, risk of over-sedation and aspiration, length of hospital stay, ED visits due to cardiovascular or other adverse events, insertion site infection, catheter fracture, air embolism, catheter misplacement, repeat procedure, referral to IR after nurse-performed insertion) Q2: Recommendations regarding the optimal approach for PICC insertions in pediatric patients (e.g., performed by radiologist vs. nurse, imaging modality for catheter tip location confirmation) |
Study Designs | HTAs, systematic reviews, RCTs, non-randomized studies, guidelines |
ED = emergency department; HTA = health technology assessment; IR = interventional radiology; PICC = peripherally inserted central catheter; RCT = randomized controlled trial.
aNot approved by Health Canada for this population and procedure.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, or were published before 2015. Systematic reviews in which all relevant studies were captured in other more recent or more comprehensive systematic reviews were excluded. Primary studies retrieved by the search were excluded if they were captured in 1 or more included systematic reviews. Guidelines with unclear methodology were also excluded.
Critical Appraisal of Individual Studies
The included publications were critically appraised by 1 reviewer using the following tools as a guide: the Downs and Black checklist5 for randomized and non-randomized studies, and the Appraisal of Guidelines for Research & Evaluation (AGREE) II instrument6 for guidelines. Summary scores were not calculated for the included studies; rather, the strengths and limitations of each included publication were described narratively.
Summary of Evidence
Quantity of Research Available
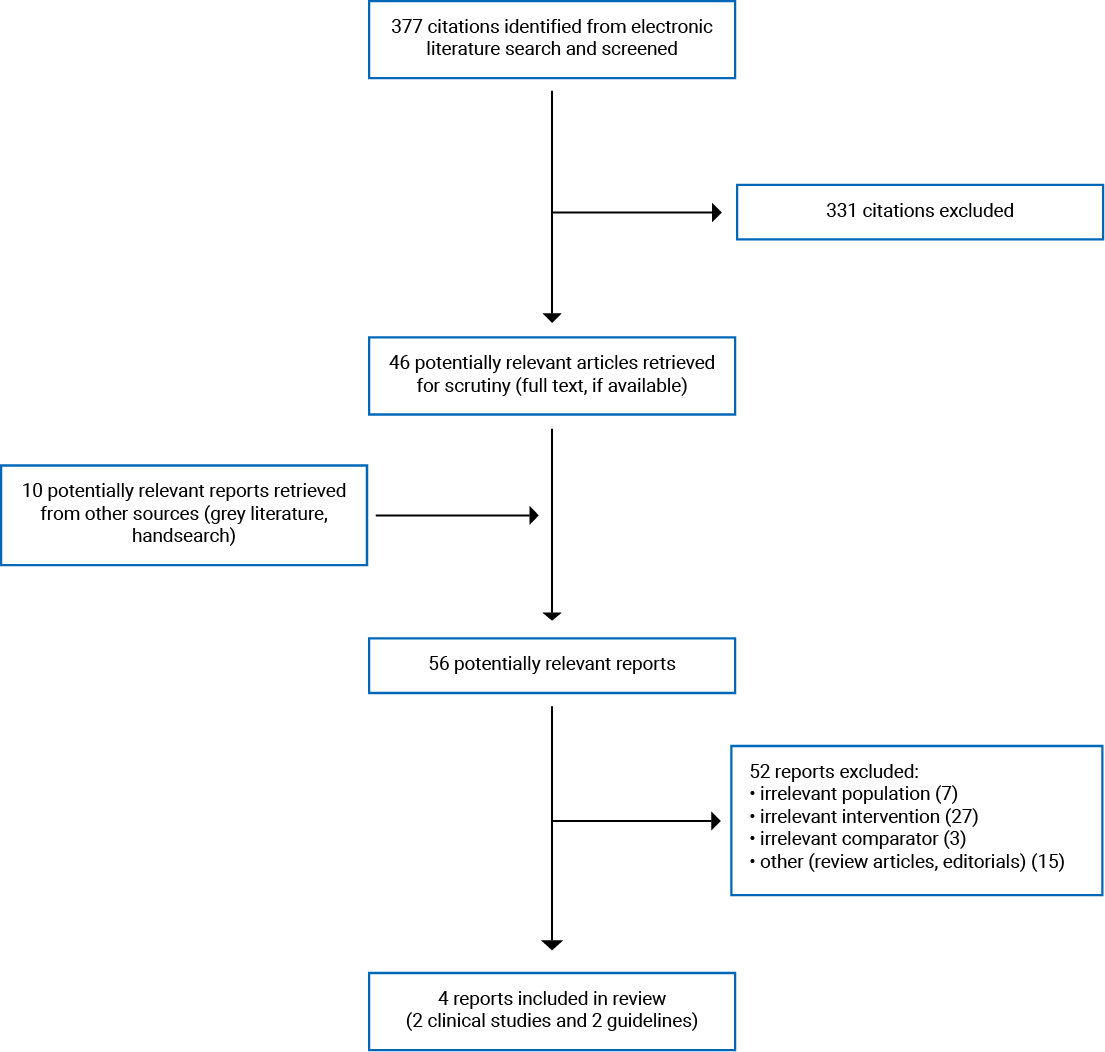
A total of 377 citations were identified in the literature search. Following the screening of titles and abstracts, 331 citations were excluded and 46 potentially relevant reports from the electronic search were retrieved for full-text review. Ten potentially relevant publications were retrieved from the grey literature search for full-text review. Of these potentially relevant articles, 52 publications were excluded for various reasons and 4 publications met the inclusion criteria and were included in this report. These comprised 2 non-randomized studies and 2 evidence-based guidelines. Appendix 1 presents the PRISMA7 flow chart of the study selection.
Additional references of potential interest are provided in Appendix 5.
Summary of Study Characteristics
Two primary clinical studies3,4 were identified that met the inclusion criteria for research question 1 and 2 guidelines8,9 were identified that met the inclusion criteria for research question 2. Additional details regarding the characteristics of included publications are provided in Appendix 2.
Study Design
Both primary clinical studies had retrospective cohort designs. The study authored by Conlon et al.3 included data from 472 PICC line placements from June 1, 2015 to May 31, 2017. The study authored by Chau et al.4 included data from patients who received common femoral tunnelled central venous catheters (CVC) from January 2014 to December 2015.
Both guidelines were published in 2020. The first was produced by the Italian Association of Pediatric Hematology and Oncology (AIEOP).8 The second is referred to as the Michigan Appropriateness Guide for Intravenous Catheters in Pediatrics (miniMAGIC) and was produced by a group of researchers and clinicians in the US and Australia.9 Literature for the AIEOP guideline was identified through 3 literature searches, each covering 1 research question; miniMAGIC was informed by a systematic review.10
To rate the quality of evidence and strength of recommendations, the AIEOP used an evidence grading system developed by the ESCMID — the European Society of Clinical Microbiology and Infectious Diseases. Each recommendation statement was assigned 1 of 3 quality levels based on the quality of the supporting evidence (where level I was “evidence from at least 1 properly designed [randomized controlled trial]” (p3)8 and level III was expert opinion, case studies, or reports) and a strength of recommendation (1 of 4 from A to D, where A indicates a strong recommendation for use and D represents a strong recommendation against use). The miniMAGIC guideline used the RAND/UCLA Appropriateness Method,11 which resulted in clinical indications classified as “appropriate,” “uncertain,” or “inappropriate.”
The miniMAGIC recommendations were generated through a modified Delphi process. The method of recommendation development was not reported for the AIEOP document.
Country of Origin
The country of origin for the first authors of both primary clinical studies3,4 was the US and both studies were conducted in the US. The AIEOP guidelines8 are intended for Italy and the miniMAGIC guidelines9 are intended for Australia and the US.
Patient Population
The study authored by Conlon et al.3 included 472 PICC line placements among a retrospective cohort of patients admitted to a quaternary care, medical-surgical, pediatric intensive care unit (PICU) in the US. This was described as a large centre (n = 55 beds) with more than 3,800 admissions per year. The Chau et al.4 study included 244 pediatric patients (specified as those aged 0 to 18 years) who received a femoral tunnelled CVC between January 2014 and December 2015. The study population was obtained from a subset of 2,375 pediatric patients who received a PICC line during this time period. This study took place at a tertiary care pediatric hospital in the US.
The intended users of both included guidelines8,9 were clinicians. The target population of the miniMAGIC9 guideline was pediatric patients aged 0 to 18 years, who were hospitalized or in ambulatory care in Australia or the US. The AIEOP8 guideline was specific to pediatric onco-hematology patients.
Interventions and Comparators
Conlon et al.3 compared PICC line placement at the bedside in the PICU to placement in IR suites. This was a 2-phase study. Phase 1 involved the implementation of a quality improvement intervention that aimed to establish criteria to identify patients for bedside PICC insertion, and then implementation of a bedside PICC service in the PICU. Phase 2 aimed to increase bedside PICC service providers and evaluate the initiative’s outcomes. The bedside PICC service consisted of clinicians trained in PICC placement (2 physicians in phase 1, expanded to 4 vascular access service physicians and 1 nurse practitioner in phase 2). “Trained” was defined as the completion of more than 10 ultrasound-guided PICC placements supervised by IR physicians. PICC line insertion was guided through ultrasound. The method of tip location confirmation was not reported. Indications for bedside placement included unstable transportation and hemodynamic instability (the full list is provided in Table 2). The comparator in this study was PICC line placement in IR. Indications for insertion in IR by fluoroscopy included known history of deep vein thrombosis, history of difficult upper extremity PICC placement, and repositioning of a PICC line using a rewiring technique. No further details were provided about the comparator.
The intervention in the Chau et al.4 publication was ultrasound-guided placement of primary femoral vein-tunnelled CVC at the bedside. The tip position was confirmed with ultrasound. Indications for bedside placement included unstable transportation (e.g., airway compromise). This was compared to the placement of primary femoral vein-tunnelled CVC in the IR suite, confirmed with fluoroscopy. Indications for bedside insertion in Chau et al.4 included unstable transportation, extracorporeal membrane oxygenation, or oscillator ventilation.
Both guidelines considered a variety of interventions. AIEOP8 made recommendations on the management of CVAD, indications for insertion and selection of CVADs, positioning of CVADs, infusion line management, exit/insertion site management, external and implantable CVADs, choice of and use of securement device, and recommendations on flushing and locking CVADs. miniMAGIC9 made recommendations on venous access device (VAD) selection characteristics and insertion technique.
Outcomes
The relevant outcomes from Conlon et al.3 included process measures (time, in hours, to PICC placement) and safety outcomes (rate per 1,000 line days of central line-associated bloodstream infections and rate per 1,000 line days of central line-associated venous thromboembolism). Two relevant outcomes were reported from Chau et al.4: the number and rate per 1,000 line days of central line-associated bloodstream infection (“laboratory-confirmation of bloodstream infection, central venous catheter indwelling for 48h before the bloodstream infection and without any other source of infection”4 [p. 809]) and the number and rate per 1,000 line days of complications. A composite complications variable, as defined by the Society of Interventional Radiology Reporting Standards for Thoracic Central Vein Obstruction,12 was used (bleeding, malposition, symptomatic venous thrombosis, catheter occlusion, and infection). The composite complications variable was further subdivided into 2 groups: early complications (the number of complications occurring 30 days or less from time of PICC insertion) and late complications (the number of complications occurring more than 30 days after PICC insertion).
The outcomes considered in the AIEOP guideline8 were not explicitly described. The outcomes considered in the miniMAGIC document9 included device and insertion characteristics that impact the success of VAD insertion and VAD failure, as well as complications. They are listed in Table 3.
Summary of Critical Appraisal
Additional details regarding the strengths and limitations of included publications are provided in Appendix 3.
Primary Clinical Studies
The 2 primary clinical studies shared some strengths, as per the Downs and Black checklist.5 In terms of reporting, both of the included studies3,4 clearly described the objectives, the main outcomes, patient characteristics, and the main findings. Estimates of the random variability in the data were reported, adverse events were included as outcomes (both studies reported complications and/or safety outcomes), no patients were lost to follow-up, and actual P values were reported. Regarding external validity, both studies created retrospective cohorts that comprised the entire source population (i.e., pediatric patients who received PICCs) during the study period and the study subjects were representative of the source population from which they were recruited. With respect to bias, both studies adjusted for length of follow-up (line days), the statistical tests used were appropriate, compliance with the interventions was reliable, and the outcome measures were accurate. In terms of confounding, the study subjects in the intervention and comparator groups were recruited over the same time periods and there were no patients lost to follow-up.
Both studies3,4 shared common limitations. These were both non-randomized studies. Study subjects were not blinded to the interventions and it is unknown whether those measuring the main outcomes were blind to the intervention. The study subjects were assigned to receive the intervention based on clinical criteria; it is unclear whether or how these clinical criteria may have affected the results in each group. Neither study presented a sample size calculation or described the clinically important differences in the main outcome measures, so it is difficult to determine whether sufficient power existed to detect differences across groups.
Differences in methodological quality were, as follows: Chau et al.4 described the intervention of interest, provided a small list of confounders (i.e., line days and lumen number; this may not have been complete), took place in a setting representative of the location that most pediatric patients receiving PICC lines would attend (i.e., tertiary care hospital), and made some adjustment for confounding in the analysis, whereas Conlon et al.3 did not.
The lack of randomization to the intervention and comparator groups in both these studies means that the patients in these groups may differ by important clinical or other factors and that the distribution of these factors across the 2 groups may be systematically different. Both studies3,4 outlined clinical criteria for receipt of PICC insertion at bedside. There was no discussion about how or whether these clinical characteristics may be associated with the measured outcomes. If an association existed, it was not adjusted for in the analyses. Lack of discussion by both authors about the impact of the clinical criteria on the selection of patients for the intervention makes interpretation of the findings difficult. Further, because sample size calculations were not provided, our confidence in the lack of statistical significance is decreased. Because no power calculation was provided, it is not possible to determine whether the lack of significant difference observed across comparisons is valid.
Guidelines
The objectives and target population were specifically described in both guidelines.8,9 The health questions were clear in the miniMAGIC guideline but were missing in the AIEOP document. Individuals from all relevant professional groups, views from the target population, and the target users were clearly defined in the miniMAGIC9 guideline but not in the AIEOP8 guideline. Both guidelines used systematic methods to identify evidence, but neither presented inclusion and exclusion criteria for the selection of evidence, described the strengths and limitations of the body of evidence, provided explicit links between the recommendations and the supporting evidence, had the guideline reviewed by external experts, or provided a procedure for updating the guideline. Whereas miniMAGIC9 described the methods for formulating the recommendations and considered the harms and benefits in formulating the recommendations, AIEOP8 did not. The recommendations were clearly written and easily identifiable, and different options for management were considered in both guidelines. Neither guideline included facilitators and barriers to implementation, provided knowledge translation tools, addressed resource implications, or included monitoring criteria. It was unclear whether competing interests of the guideline development groups were addressed in both guidelines. The AIEOP8 includes a statement declaring that the funding bodies have not influenced the content of the guideline but miniMAGIC9 does not.
There were similar limitations across both included guidelines. Neither described how the strengths and limitations of the body of evidence were considered in formulation of the recommendations. However, miniMAGIC9 was developed using a published method — the RAND/UCLA Appropriateness Method11 — and was accompanied by separate publications summarizing the systematic review10 conducted to inform these guidelines, and a methods paper.13 Further, the AIEOP guidelines are specific to pediatric oncology-hematology patients, which represents a subset of those of interest in the present review.
Summary of Findings
Appendix 4 presents the main study findings and authors’ conclusions.
Clinical Effectiveness of PICC Insertion by Radiologists Using Fluoroscopy in Pediatric Patients
Conlon et al.3 compared PICC insertion at the bedside using ultrasound guidance by a bedside PICC service team to PICC line insertion in the IR suite using fluoroscopic guidance. The bedside PICC service was developed in 2 phases with different practitioners participating in each: phase 1 — 2 trained physicians (n = 282 PICC lines); phase 2 — 2 trained physicians plus 4 vascular access service providers and 1 nurse practitioner (n = 211 PICC lines). The method of tip confirmation at bedside was not reported.
Chau et al.4 compared placement of femoral-tunnelled CVC at the bedside by ultrasound guidance and tip confirmation by ultrasound to placement in the IR using fluoroscopic guidance. The health care professional inserting the catheter at bedside (e.g., nurse, physician) was not reported.
The 2 included clinical studies3,4 reported a total of 4 relevant outcomes.
Infection
Both studies3,4 reported no significant difference in the rate per 1,000 line days of central line-associated bloodstream infections between patients with PICC insertion at bedside compared to those in the IR suite. In addition, Conlon et al.3 reported no significant difference in the rate of non-mucosal barrier injury laboratory-confirmed bloodstream infection per 1,000 line days between patients with PICC insertion at bedside compared to those in the IR suite.
Venous Thromboembolism and Other Clinical Complications
Conlon et al.3 reported no significant difference in the rate of venous thromboembolism per 1,000 line days between patients with PICC insertion at bedside compared to those in the IR suite. In Chau et al.,4 1.9% of bedside insertions were associated with venous thrombosis compared to 0.7% of IR insertions, but statistical significance of this comparison was not reported. Chau et al.4 reported that there was no significant difference in overall complications, early complications, or late complications between patients with PICC insertion at bedside compared to those in the IR suite, even after adjustment for line days and lumen number.
Process Outcomes
As per Conlon et al.,3 the median time from PICC line order to successful insertion was longer for IR placement compared to bedside.
Guidelines on PICC Insertion in Pediatric Patients
The AIEOP8 concluded that there is insufficient evidence to recommend 1 particular CVAD (i.e., PICC, centrally inserted central catheter or femoral-inserted central catheter). This guideline also made a strong recommendation based on level I evidence that ultrasound-guided insertion represents the standard procedure for insertion of CVADs.
The miniMAGIC9 considers it appropriate to insert all devices (including PICCs) by using ultrasound guidance. It also concludes that the appropriateness of electrocardiography-guided insertion of PICCs is uncertain because of limited evidence of benefit in the pediatric population.
Neither guideline provided a preferential recommendation for the location of insertion (i.e., bedside versus IR) or health care professional performing insertion (i.e., nurse versus physician).
Limitations
The 2 clinical studies had highly specialized populations3 or interventions.4 The patients in Conlon et al.3 were those admitted to a non-cardiac PICU (i.e., a specialized quaternary care unit). Chau et al.4 focused on a small subset of patients with PICC lines — those who received femoral-tunnelled CVCs. This narrow scope limits the generalizability of the findings to the broader population.
The method of tip location confirmation was either not reported3 or confirmed via ultrasound.4 The health care professionals performing the insertions were either not reported4 or included a group of professionals, of which 1 was a nurse practitioner who was involved in a subset of insertions.3 Therefore, both studies provided indirect evidence to address the research question.
Both studies reported non-significant differences between intervention and comparator groups across all clinical outcomes. However, the validity of these findings is uncertain because of the small number of outcome observations, the lack of power calculations, and limited discussion on the potential impact of confounding due to the non-randomization of patients to the intervention group.
Two guidelines were identified. The quality of the AIEOP guideline8 was affected by the following limitations: lack of transparency in reporting of the literature search or method of formulating recommendations, limited scope to pediatric onco-hematology patients, and specific to the Italian context.8 Further, neither guideline addressed the recommended location of PICC insertion or the recommended provider for insertion.
Gaps in the literature exist. No publications were identified that studied PICC insertion by radiologists using fluoroscopy as the intervention; instead, both studies were designed with PICC insertions by radiologists using fluoroscopy as the comparator. No evidence was identified that studied all PICC insertions in the general hospitalized pediatric population. There were no studies or guidelines produced in Canada, making generalizability to the context of the Canadian health care system indirect.
Conclusions and Implications for Decision- or Policy-Making
Two retrospective cohort studies3,4 were identified that compared PICC insertion in pediatric patients performed in IR suites versus at the bedside. Two guidelines8,9 were identified that addressed the optimal approach for PICC insertions in pediatric patients.
The studies summarized in this review did not report a difference in the rates of infection, venous thromboembolism, or other clinical complications between methods and locations of PICC insertion and 1 study3 reported a longer time to PICC line insertion with IR placement than with placement at the bedside. However, the identified evidence was limited in quality and quantity. It was also limited in the directness to which it addresses the research questions. It is not specific to nurses performing PICC insertions at bedside with X-ray for tip confirmation. Instead, it is general to bedside PICC insertion with ultrasound or no mention of tip confirmation.
Further, existing guidelines recommend the use of ultrasound guidance for insertion of PICC lines in pediatric patients but did not address recommendations for location of insertion or health care professional performing the insertion.
Future research should focus on a broader pediatric population, instead of population and hospital unit subgroups, to generate more generalizable results. If ethically appropriate, researchers should consider conducting randomized studies to reduce bias when comparing PICC insertion in various settings and by various health care professionals. Additional considerations beyond the evidence of clinical effectiveness and safety that may inform the choice of setting for PICC insertion may include logistical barriers and cost.
References
1.Cho SB, Baek HJ, Park SE, et al. Clinical feasibility and effectiveness of bedside peripherally inserted central catheter using portable digital radiography for patients in an intensive care unit: a single-center experience. Medicine (Baltimore). 2019;98(26):e16197. Medline
2.Cotogni P, Pittiruti M. Focus on peripherally inserted central catheters in critically ill patients. World J Crit Care Med. 2014;3(4):80-94. Medline
3.Conlon TW, Himebauch AS, Cahill AM, et al. Development and implementation of a bedside peripherally inserted central catheter service in a PICU. Pediatr Crit Care Med. 2019;20(1):71-78. Medline
4.Chau A, Hernandez JA, Pimpalwar S, Ashton D, Kukreja K. Equivalent success and complication rates of tunneled common femoral venous catheter placed in the interventional suite vs. at patient bedside. Pediatr Radiol. 2018;48(6):889-894. Medline
5.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377-384. Medline
6.Agree Next Steps C. The AGREE II Instrument. [Hamilton, ON]: AGREE Enterprise; 2017: https://www.agreetrust.org/wp-content/uploads/2017/12/AGREE-II-Users-Manual-and-23-item-Instrument-2009-Update-2017.pdf. Accessed 2021 Jan 14.
7.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34.Medline
8.Cellini M, Bergadano A, Crocoli A, et al. Guidelines of the Italian Association of Pediatric Hematology and Oncology for the management of the central venous access devices in pediatric patients with onco-hematological disease. J Vasc Access. 2020:1129729820969309. Medline
9.Ullman AJ, Bernstein SJ, Brown E, et al. The Michigan Appropriateness Guide for Intravenous Catheters in pediatrics: miniMAGIC. Pediatrics. 2020;145(Suppl 3):S269-s284. Medline
10.Paterson RS, Chopra V, Brown E, et al. Selection and insertion of vascular access devices in pediatrics: a systematic review. Pediatrics. 2020;145(Suppl 3):S243-s268. Medline
11.Fitch K, Bernstein S, Aguilar M, et al. The RAND/UCLA appropriateness method user's manual. Santa Monica (CA): RAND Corporation; 2001: https://www.rand.org/pubs/monograph_reports/MR1269.html. Accessed 2021 Jan 14.
12.Silberzweig JE, Sacks D, Khorsandi AS, Bakal CW. Reporting standards for central venous access. J Vasc Interv Radiol. 2003;14(9 Pt 2):S443-452. Medline
13.Ullman A, Chopra V. Developing pediatric appropriateness criteria for intravenous catheters. Pediatrics. 2020;145(Suppl 3):S231-s233. Medline
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Table 2: Characteristics of Included Primary Clinical Studies
Study citation, country, funding source | Study design | Population characteristics | Intervention and comparator(s) | Clinical outcomes |
Conlon et al. (2019)3 US Funding Sources: Children’s Hospital of Philadelphia, University of Pennsylvania National Institutes for Health, Society of Critical Care Medicine | Study design:Retrospective cohort study Setting:Single quaternary non-cardiac PICU in the US Objectives:
| Patients admitted to the PICU with PICC lines Number of observations: N = 472 (342 IR vs. 130 bedside) Median age (IQR):NR for overall study population but reported separately for intervention and comparator:
| Intervention: PICC line placement at the bedside of PICU patients by trained providers (physicians and a nurse practitioner) using ultrasound guidance Indications for bedside placement:
Comparator: Other locations (including IR suite) Indications for insertion in IR suite by fluoroscopy: | Relevant outcomes: Process measures (time to PICC placement) Safety outcomes: CLABSI and central line-associated VTE |
Chau et al. (2018)4 US Funding Source: Texas Children’s Hospital | Study design: Retrospective cohort study Setting: Single tertiary hospital in the US Objective: To compare bedside and IR suite placement of femoral vein-tunnelled CVCs | Pediatric patients (0 to 18 years) that met the criteria for femoral vein-tunnelled CVC:
Number of patients: N = 244 (140 IR vs. 104 bedside) Age range:1 to 3,531 days | Intervention: Bedside-placed primary femoral vein-tunnelled CVC. Bedside placement occurred in the ICU or ER, guided by ultrasound. Indications for bedside placement included “unstable transportation such as airway or cardiopulmonary compromise, extracorporeal membrane oxygenation, or oscillator ventilation.” (p890)4 Comparator: Placement of primary femoral vein-tunnelled CVC in the IR suite, confirmed with fluoroscopy. | Relevant outcomes: Complications as defined by the Society of Interventional Radiology Reporting Standards12: bleeding, malposition, symptomatic venous thrombosis, and catheter occlusion. Complications assigned as early (≤ 30 days) and late (> 30 days) Infection: laboratory-confirmation of bloodstream infection |
CLABSI = central line-associated bloodstream infection; CVC = central venous catheter; ER = emergency room; ICU = intensive care unit; IQR = interquartile range; IR = interventional radiology; NR = not reported; PICC = peripherally inserted central catheter; PICU = pediatric intensive care unit; vs. = versus; VTE = venous thromboembolism.
Table 3: Characteristics of Included Guidelines
Intended users, target population | Intervention and practice considered | Major outcomes considered | Evidence collection, selection, and synthesis | Evidence quality assessment | Recommendations development and evaluation | Guideline validation |
Italian Association of Pediatric Hematology and Oncology (2020)8 | ||||||
Intended users: clinicians Target population: pediatric onco-hematology patients | Management of CVADs (including PICCs), indications for insertion and selection of CVADs, positioning of CVADs, infusion line management, exit/insertion site management, recommendations for external CVADs, recommendations for implantable CVADs, choice of securement device, recommendations on the use of securement devices, recommendations on flushing and locking CVADs | The authors did not explicitly describe the outcomes of interest. | 3 literature searches:
No information about the method of evidence synthesis was provided. | Evidence quality was assessed via method proposed by the European Society of Clinical Microbiology and Infectious Disease (unpublished). Evidence grouped into 3 quality levels: evidence from at least 1 well-designed RCT (I); evidence from at least 1 well-designed clinical trial without randomization and/or observational studies (II); expert opinion or case studies (III) | Recommendations were proposed by topic work groups; discussed and approved by full guideline panel. The specific method used to generate the recommendation statements (e.g., consensus, Delphi method) was not described. | The guideline document was reviewed by members of the AIEOP. There was no mention of external review by other experts. |
miniMAGIC (2020)9 | ||||||
Intended users: clinicians Target population: pediatric patients aged 0 to 18 years (hospitalized and in ambulatory care) in Australia and the US | VAD selection, characteristics, and insertion technique | “Device characteristics included VAD type, device catheter-to-vein ratio, and device lumens. Insertion characteristics included insertion site and location and the use of vessel visualization technology. Complications included but were not limited to central line-associated bloodstream infection (CLABSI), VAD-associated thrombosis, occlusion, catheter dislodgement, catheter tip migration, catheter breakage or rupture, local infection, and phlebitis.” (pS244)10 | Systematic review with duplicate evidence screening and selection; triplicate data extraction | Duplicate quality assessment of included studies using validated tools | Members of the guideline panel independently and anonymously rated clinical scenarios (n = 1,234) on a scale of 1 to 9 (1 being “harm outweighs benefit” and 9 being “benefit outweighs harm”). A second round of ratings occurred after in-person discussion (modified Delphi process). Scenarios were classified to 3 levels of appropriateness as per the RAND/UCLA method:
|
|
AIEOP = Italian Pediatric Hematology Oncology Association; CVAD = central venous access device; miniMAGIC = the Michigan Appropriateness Guide for Intravenous Catheters in Pediatrics; PICC = peripherally inserted central catheter; NR = not reported; RAND/UCLA = RAND Corporation and University of California; RCT = randomized controlled trial; VAD = venous access device.
Appendix 3: Critical Appraisal of Included Publications
Table 4: Strengths and Limitations of Clinical Studies Using the Downs and Black Checklist5
Strengths | Limitations |
Conlon et al. (2019)3 | |
|
|
Chau et al. (2018)4 | |
|
|
IR = interventional radiology; NR = not reported; PICC = peripherally inserted central catheter; PICU = pediatric intensive care unit; vs. = versus.
Table 5: Strengths and Limitations of Guidelines Using AGREE II6
Item | Italian Association of Pediatric Hematology and Oncology (2020)8 | miniMAGIC (2020)9 |
Domain 1: Scope and purpose | ||
1. The overall objective(s) of the guideline is (are) specifically described. | Yes | Yes |
2. The health question(s) covered by the guideline is (are) specifically described. | No | Yes |
3. The population (patients, public, and so forth) to whom the guideline is meant to apply is specifically described. | Yes | Yes |
Domain 2: Stakeholder involvement | ||
4. The guideline development group includes individuals from all relevant professional groups. | Unsure | Yes |
5. The views and preferences of the target population (patients, public, and so forth) have been sought. | No | Yes |
6. The target users of the guideline are clearly defined. | No | Yes |
Domain 3: Rigour of development | ||
7. Systematic methods were used to search for evidence. | Yes | Yes |
8. The criteria for selecting the evidence are clearly described. | No | No |
9. The strengths and limitations of the body of evidence are clearly described. | No | No |
10. The methods for formulating the recommendations are clearly described. | No | Yes |
11. The health benefits, side effects, and risks have been considered in formulating the recommendations. | Unsure | Yes |
12. There is an explicit link between the recommendations and the supporting evidence. | No | No |
13. The guideline has been externally reviewed by experts before its publication. | No | No |
14. A procedure for updating the guideline is provided. | No | No |
Domain 4: Clarity of presentation | ||
15. The recommendations are specific and unambiguous. | Yes | Yes |
16. The different options for management of the condition or health issue are clearly presented. | Yes | Yes |
17. Key recommendations are easily identifiable. | Yes | Yes |
Domain 5: Applicability | ||
18. The guideline describes facilitators and barriers to its application. | No | No |
19. The guideline provides advice and/or tools on how the recommendations can be put into practice. | No | No |
20. The potential resource implications of applying the recommendations have been considered. | No | No |
21. The guideline presents monitoring and/or auditing criteria. | No | No |
Domain 6: Editorial independence | ||
22. The views of the funding body have not influenced the content of the guideline. | Yes | Unsure |
23. Competing interests of guideline development group members have been recorded and addressed. | Unsure | Unsure |
AGREE II = Appraisal of Guidelines for Research and Evaluation II; miniMAGIC = Michigan Appropriateness Guide for Intravenous Catheters in Pediatrics.
Appendix 4: Main Study Findings and Authors’ Conclusions
Table 6: Summary of Findings of Included Primary Clinical Studies
Main study findings | Authors’ conclusion |
Conlon et al. (2019)3 | |
Safety outcomes: The study evaluated 130 BPS PICC lines (2,447 line days) and 342 IR PICC lines (8,270 line days) [P = NR] All CLABSI (includes non-MBI-LCBI) • BPS PICC: n = 3; ratea = 1.23 • IR PICC group: n = 18; ratea = 2.18 • P = 0.37 Non-MBI-LCBIs • BPS PICC: n = 2; ratea = 0.82 • IR PICC: n = 18; ratea = 2.18 • P = 0.17 VTE • BPS PICC: n = 4; ratea = 1.63 • IR PICC group: n = 13; ratea: 1.57 • P = 0.91 Process outcomes: “The time from order to successful PICC placement was significantly reduced in patients with BPS line placement [n = 121, median 6h (IQR 2h-23h)] compared with IR line placement [n = 326, median 34h (IQR 19h-61h); P < 0.001]” (p5)3 | “We demonstrated that the creation and spread of a team comprised of trained physicians, nurse practitioners, and vascular access nurses specialized in bedside PICC techniques can result in a sustained increase in PICC lines placed at the bedside in critically ill children.” (p5)3 “Process measures and balancing metrics highlight programmatic timeliness with shorter time to PICC insertion and safety with no significant difference in CLABSI and VTE rates compared to PICCs placed in IR.” (p5)3 |
Chau et al. (2018)4 | |
Distribution of catheter complications by insertion location:
Complication and infection outcomes (P values NR)
Univariate logistic regression for complications:
Multivariate logistic regression:
| “The most common complication with IR suite placement “We found no statistically significant differences in the total complication rate, early or late complications, or infection rate between the IR suites and bedside-placed lower extremity tunneled CVC, despite the bedside group having statistically significant longer mean line days.” (p892)4 “The total complications per 1,000 line days for IR suite and bedside in this study (4.3 and 3.6, respectively) is within range of published literature for mechanical complications in all central venous access in the pediatric population 3.8–14 per 1,000 line days…” (p893)4 |
BPS = bedside PICC service; CI = confidence interval; CLABSI = central line-associated bloodstream infection; CVC = central venous catheter; h = hours; IQR = interquartile range; IR = interventional radiology; non-MBI-LCBI = non-mucosal barrier injury laboratory-confirmed bloodstream infection; NR = not reported; OR = odds ratio; PICC = peripherally inserted central catheter; vs. = versus; VTE = venous thromboembolism.
aRate as a measure of infections per 1,000 line days.
Table 7: Summary of Recommendations in Included Guidelines
Recommendations | Quality of evidence and strength of recommendations |
Italian Association of Pediatric Hematology and Oncology (2020)8 | |
“There is not sufficient evidence to absolutely recommend one device over another… usually it is recommended to use the device with the lease number of lumens…for therapeutic needs.” (p4)8 The authors listed PICCs, CICCs, and FICCs as the “devices” covered by the guideline. “The ultrasound-guided technique represents the current standard for venipuncture and venous cannulation for insertion of CVAD.” (p6)8 | Insufficient evidence Level of recommendation: AIt (Strong recommendation, with evidence from at least 1 well-designed RCT. The authors report that “t” refers to “transferred evidence…from different patient cohorts.” (p3)8) |
miniMAGIC (2020)9 | |
“…panelists rated it appropriate to insert all devices [PIVCs, PICCs and non-tunnelled CVADs] by using ultrasound guidance.” (pS279)9 “…electrocardiography guided insertion of PICCs across populations was rated as uncertain because (unlike the adult population) the evidence in pediatrics for benefit of this technology is limited.” (pS280)9 “Evaluation of the venous anatomy using ultrasound before placement of all central devices, and placement of VADs in neonates and pediatric patients with long-term vascular access-dependent conditions, was rated as appropriate by the panel.” (pS280)9 | Appropriate (based on the RAND/UCLA levels of appropriateness) Uncertain (based on the RAND/UCLA levels of appropriateness) Appropriate (based on the RAND/UCLA levels of appropriateness) |
CICC = centrally inserted central catheter; CVAD = central venous access device; FICC = femoral-inserted central catheter; miniMAGIC = Michigan Appropriateness Guide for Intravenous Catheters in Pediatrics; NR = not reported; PICC = peripherally inserted central catheter; PIVC = peripheral intravenous catheter; RAND/UCLA = RAND Corporation and University of California; RCT = randomized controlled trial; VAD = venous access device.
Appendix 5: Additional References of Potential Interest
Guidelines With Unclear Methodology
1.Queensland Health. Peripherally inserted central venous catheters (PICC). Brisbane, Queensland, Australia: Queensland Health; 2015 Jan: https://www.health.qld.gov.au/__data/assets/pdf_file/0032/444497/icare-picc-guideline.pdf. Accessed 2021 Jan 14.
• “For potentially difficult/complicated insertions due to patient factors (e.g. impalpable vessels, morbid obesity, pre-existing injury), clinicians should consider using ultrasound guided access for PICC placement.” (p4)
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca