CADTH Health Technology Review
Patient Navigation Programs for People With Dementia
Rapid Review
Authors: Srabani Banerjee, Charlene Argáez
Abbreviations
DCM
dementia care management
FCC
facilitated case conferencing
QALY
quality-adjusted life-years
QoL
quality of life
RCT
randomized controlled trial
WTP
willingness to pay
Key Messages
Patient navigation programs are generally community-based service delivery interventions (such as collaborative care, coordinated care, and case management) intended to enhance timely access to the diagnosis and treatment of individuals with chronic conditions, including dementia.
Overall, for coordinated care compared with usual care, clinical findings were either mixed or there were no between-group differences in terms of hospitalization, institutionalization, or nursing home admissions; quality of life; or symptoms. For coordinated care compared with usual care, there was no statistically significant between-group difference in mortality. However, there was evidence of improvement in terms of behaviour with coordinated care compared with usual care.
According to 1 economic evaluation, for patients with dementia, with the majority having no or mild cognitive impairment, collaborative dementia care management provided increased benefit (quality-adjusted life-years gained) at decreased cost.
Three guidelines were identified that provided recommendations for care coordination. One guideline recommends coordinated care for people living with dementia that is organized by a single-named health or social care professional. The second guideline recommends the use of digital technology to enhance care coordination in persons with mental illness. The third guideline recommends coordinated care for people with delirium, dementia, and depression.
Findings need to be interpreted in the light of limitations (such as lack of information in the study populations’ type of dementia, mixed findings in outcomes, and lack of information beyond 24 months of follow-up).
Context and Policy Issues
Dementia is a condition that includes a range of cognitive and behavioural symptoms that can include memory loss; problems with reasoning and communication, and change in personality; and a reduction in a person’s ability to carry out daily activities, such as shopping, washing, dressing, and cooking.1,2 The most common types of dementia are Alzheimer disease, vascular dementia, mixed dementia, dementia with Lewy bodies, and frontotemporal dementia.1,2 Dementia is a progressive condition (i.e., the symptoms gradually worsen). This progression varies from person to person and each person’s experiences may be different — people may often have some of the same general symptoms, but the degree to which these affect each person may vary.1 Dementia impacts physical, psychological, social, and economic aspects not only for the individual with dementia but also caregivers, families, and society at large.3
A WHO publication reported that, worldwide, there are approximately 50 million people living with dementia and approximately 10 million new cases are diagnosed every year.3 According to a 2012 to 2013 estimate in Canada, 402,000 seniors, or 7.1% of all people 65 years and older, were living with dementia and of these two-thirds were females.4 In Canada, approximately 76,000 new cases of dementia are diagnosed every year, which is about 14.3 new cases per 1,000 people 65 years and older.4 It is projected that by 2031, the total annual health care costs for Canadians with dementia will likely double compared to that from 2 decades earlier — from $8.3 billion to $16.6 billion.5
Individuals with dementia experience changes in behaviour, mood, memory, and physical disability, and need help in managing these challenging changes.6 Generally, people with dementia are cared for by a family member. It is often a challenge for the caregiver and the burden of caregiving can affect the caregiver’s mental and physical health. The needs of the individual with dementia and their caregivers are complex and appropriate approaches for care organization and delivery are important. Patient navigation approaches are generally community-based service delivery interventions intended to enhance timely access to the diagnosis and treatment of individuals with chronic conditions.7 Patient navigators help patients and their caregivers through the health care system and help them in understanding diagnoses, treatment options, and available resources.8 Patient navigators are the main point of contact for the patients and their caregivers.9 There is a growing interest in patient navigation approaches for organizing care for people with dementia and some suggestion that family physician and case manager collaboration may address the needs of the people with dementia and their caregivers.6
The purpose of this report is to review the clinical effectiveness and cost-effectiveness of patient navigation programs for people with dementia and, additionally, to review the evidence-based guidelines regarding patient navigation programs for people with any medical condition. This report is an upgrade from a recent CADTH Summary of Abstracts report published in November 2020.10 This report will summarize and critically appraise the relevant evidence identified from the previous report.10
Research Questions
What is the clinical utility of patient navigation programs for people with dementia?
What is the cost-effectiveness of patient navigation programs for people with dementia?
What are the evidence-based guidelines regarding the use of patient navigation services for people with any medical condition?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE and CINAHL, the Cochrane Library, the University of York Centre for Reviews and Dissemination (CRD) databases, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were patient navigation and dementia. For questions 1 and 2, no filters were applied to limit the retrieval by study type. For question 3, a filter was applied to limit the retrieval to guidelines only. The search was also limited to English-language documents published between January 1, 2010 and November 16, 2020.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
Population | Q1 and Q2: People with dementia (all types) Q3: People with any medical condition |
Intervention | Patient navigation programs or services (i.e., care coordination programs) |
Comparator | Q1 and Q2: Usual care; no coordination of care with patient navigation programs Q3: Not applicable |
Outcomes | Q1: Clinical utility (e.g., quality of life, time to diagnosis and treatment, disease severity, cognitive impairment [e.g., MMSE scores], mortality) Q2: Cost-effectiveness (e.g., cost per quality-adjusted life-year gained) Q3: Recommendations regarding best practices (e.g., appropriate patient populations, implementation considerations, appropriate clinical settings) |
Study Designs | HTAs, SRs, RCTs, non-randomized studies, economic evaluations, and evidence-based guidelines |
HTA = health technology assessment; MMSE = Mini-Mental State Examination; RCT = randomized controlled trial; SR = systematic review.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, or were published before 2010. Systematic reviews in which all relevant studies were captured in other more recent or more comprehensive systematic reviews were excluded. Primary studies retrieved by the search were excluded if they were captured in 1 or more included systematic reviews. Guidelines with unclear methodology were also excluded.
Critical Appraisal of Individual Studies
The included publications were critically appraised by 1 reviewer using the following tools as a guide: A MeaSurement Tool to Assess systematic Reviews 2 (AMSTAR 2)11 for systematic reviews, the Downs and Black checklist12 for randomized and non-randomized studies, the Drummond checklist13 for economic evaluations, and the Appraisal of Guidelines for Research & Evaluation (AGREE) II instrument14 for guidelines. Summary scores were not calculated for the included studies; rather, the strengths and limitations of each included publication were described narratively.
Summary of Evidence
Quantity of Research Available
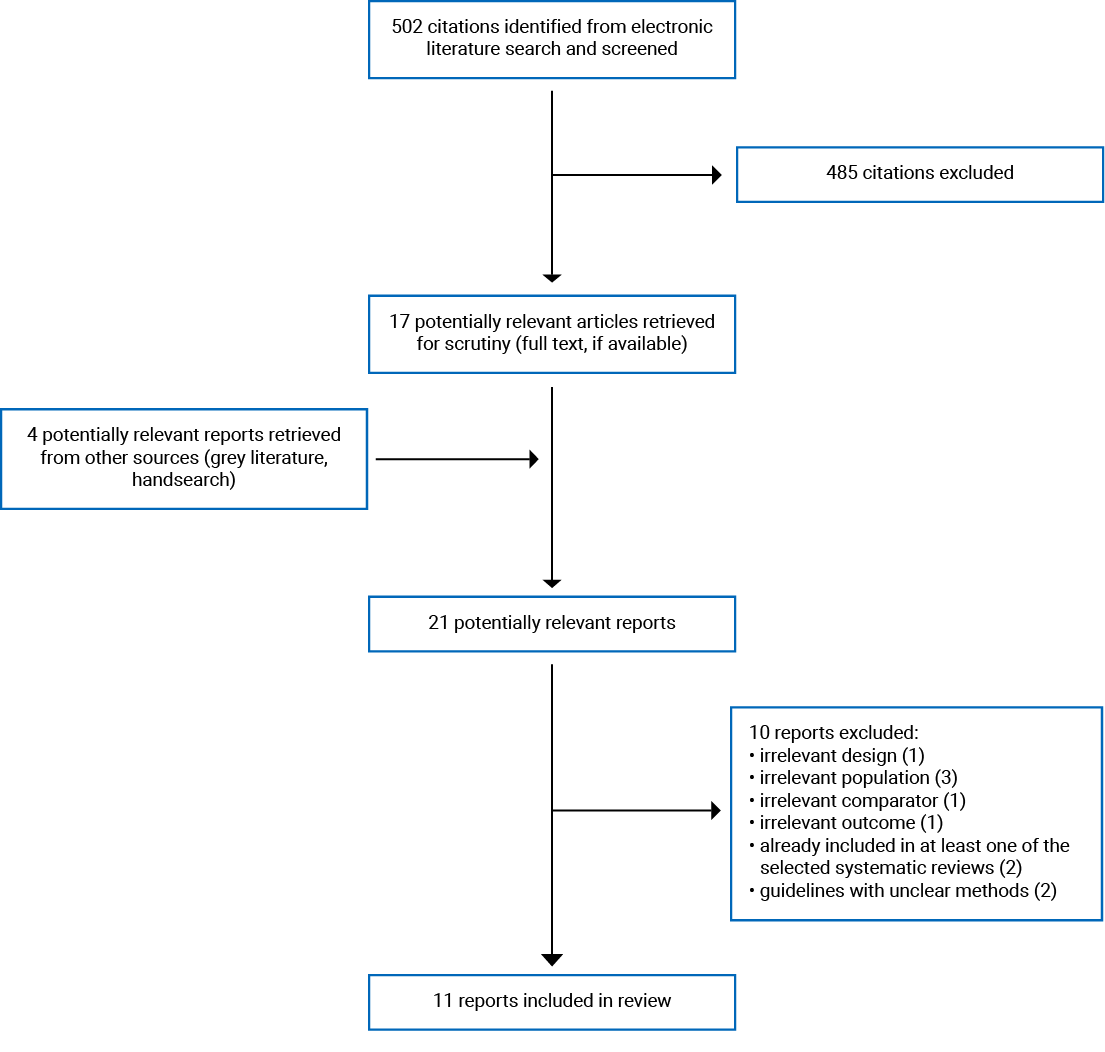
A total of 502 citations were identified in the literature search. Following the screening of titles and abstracts, 485 citations were excluded and 17 potentially relevant reports from the electronic search were retrieved for full-text review. Four potentially relevant publications were retrieved from the grey literature search for full-text review. Of these 21 potentially relevant articles, 10 publications were excluded for various reasons, and 11 publications met the inclusion criteria and were included in this report. These comprised 3 systematic reviews,15-17 2 randomized controlled trials (RCTs),18,19 1 non-randomized study,20 1 economic evaluation (presented in 2 publications [primary analyses21 and subgroup analyses22]), and 3 evidence-based guidelines.1,2,23 Appendix 1 presents the PRISMA24 flow chart of the study selection.
Summary of Study Characteristics
Three systematic reviews,15-17 2 RCTs,18,19 1 non-randomized study,20 1 economic evaluation (presented in 2 publications [primary analyses21 and subgroup analyses22]), and 3 evidence-based guidelines guidelines1,2,23 were included. Two systematic reviews15,16 had a broad focus and included studies on a variety of interventions for people with dementia; hence, only the subset of studies that are relevant for this current report are described in this report. For the third systematic review,17 all the included studies were relevant and included in this current report. The relevant primary studies in the included systematic reviews are listed in Appendix 5. There was some overlap in the studies included in the systematic reviews; it should therefore be noted that there is overlap of studies and that findings from the systematic reviews are not exclusive. Additional details regarding the characteristics of included publications are provided in Appendix 2 (Table 2, systematic reviews; Table 3, primary clinical studies; Table 4, economic evaluation; Table 5 and Table 6, guidelines).
Study Design
Of the 3 identified systematic reviews,15-17 2 systematic reviews16,17 included meta-analyses and 1 systematic review15 reported results narratively. Two systematic reviews15,16 were published in 2020 and 1 systematic review17 was published in 2017. The number of relevant primary studies reporting on patient navigation (i.e., collaborative care and/or case management) that were included in these systematic reviews ranged between 6 and 14; these studies were RCTs.
Two RCTs18,19 and 1 non-randomized study20 were included. These studies were published in 2017.
The included economic evaluation21 was a cost-utility analysis. A public payer perspective and a time horizon of 24 months were used. It was assumed that the change in health-related quality of life was linear, which seemed appropriate considering the nature of dementia. Sensitivity analysis using a societal perspective was conducted. The authors reported that there were no conflicts of interest. Clinical and utility data were obtained from a cluster RCT that was conducted by the authors. This RCT involved predominantly patients with dementia who had mild cognitive impairment. Cost data were obtained from market prices, the Pharmaceutical Index of the Scientific Institute of allgemeine Ortskrankenkasse, and the literature
The included guidelines were from the NICE–National Institute for Health and Care Excellence (NICE)1 in the UK, the RNAO–Registered Nurses’ Association of Ontario,2 and the EPA–European Psychiatric Association.23 For the 3 included guidelines1,2,23 the guideline development group conducted - literature searches to identify evidence and had a method to grade the levels of the evidence. For 1 guideline,1 the guideline development group comprised a multidisciplinary team with experts in various relevant health-related areas, and also lay persons. For the second guideline,2 the guideline development group comprised individuals holding clinical, administrative, and academic positions in various health care organizations. The third guideline23 did not report on the guideline development group. In 2 guidelines,1,23 recommendations were formulated based on consensus; in the third guideline, the method of formulating the recommendations was unclear.
Country of Origin
Of the 3 systematic reviews,15-17 1 systematic review15 was from the US, 1 systematic review16 was from Australia, and 1 systematic review17 was from the UK. The RCTs included in these systematic reviews were conducted in various countries (Canada, the US, the UK, Germany, France, the Netherlands, Finland, Norway, Australia, and China).
Of the 3 included primary studies, the countries of the first authors were the US19,20 and Australia.18 The studies were conducted in their respective countries.
The economic evaluation21 was from Germany.
The 3 guidelines1,2,23 were from various countries: Canada,2 the UK,1 and European countries.23
Patient Population
All 3 systematic reviews15-17 reported on individuals with dementia; the number of individuals ranged between 1958 and 10,392. In 1 systematic review,15 mean ages in the included studies ranged between 68 years and 83 years, and in the other 2 systematic reviews,16,17 age was not reported. None of the systematic reviews15-17 reported the type of dementia.
One RCT18 involved 286 individuals with advanced dementia living in a nursing home; the mean age was 85 years and 60% were females. The second RCT19 involved 75 veterans with dementia; the mean age was 79 years and were mainly males (the exact proportion of female to male participants was not reported). The non-randomized study20 involved 440 individuals with dementia; the age and proportion of female participants were not presented.
The patient population considered in the economic evaluation21,22 was persons with dementia, the majority with no or mild cognitive impairment. The subgroups considered were different age groups, female and male, different comorbidity levels, different levels of impairment, and patients living alone and patients not living alone.
In 1 guideline,1 the target population was people with dementia and their caregivers and the intended users were people with dementia and those involved in the care of people with dementia. In the second guideline,2 the target population was people with delirium, dementia, or depression, and the intended users were primarily nurses providing direct clinical care to older adults. In the third guideline,23 the target population was people with mental health issues and the intended users were those involved in the care of people with mental health issues.
Interventions and Comparators
In the 3 included systematic reviews,15-17 the intervention was some form of patient navigation program (case management and/or coordinated care). Case management involved care coordinated by a single individual, and collaborative or coordinated care involved a team coordinating care. In all 3 systematic reviews,15-17 the comparator was usual care; details were not presented. Additionally, 1 systematic review17 also included alternative dementia care interventions or waiting-list controls as comparators.
The 2 RCTs18,19 and 1 non-randomized study20 compared coordinated care, or case management with usual care. One RCT18 compared case management (facilitated case conferencing [FCC]) with usual care (no particular guidance or training around the approach to care planning). For this FCC care plan, a nurse who was trained as a palliative care planning coordinator organized and implemented the care plan. The second RCT19 compared a care management plan with usual care (usual care details were not presented). The coordinated care plan was the responsibility of a care manager, who was responsible for coordinating primary and specialty care, as well as the telephone education program. The non-randomized study19,20 compared a coordinated care plan with usual care (clinical assessment and referral). The coordinated care plan comprised the usual care and in addition included service connection, psychoeducation, and support.
In the economic evaluation21,22 the intervention investigated was collaborative dementia care management and the comparator was usual care (not otherwise described).
One guideline1 considered interventions such as care planning and coordination of care, inpatient care, and pharmacological and non-pharmacological treatment. The second guideline2 considered approaches needed for nurses to assess and manage older adults. The third guideline23 considered care coordination.
Outcomes
Outcomes reported in the 3 selected systematic reviews15-17 included hospitalization,16,17 institutionalization,15,17 nursing home admission,15,16 depression,15,17 neuropsychiatric symptoms,15 quality of life (QoL),15,17 function,15,17 cognitive ability,17 and mortality.17 Follow-up times ranged between 6 months and 36 months.
The types of outcomes reported in the 3 included primary studies (2 RCTs18,19 and 1 non-randomized study20) varied. One RCT18 reported on QoL, dementia symptoms, quality of care, hospital stay, and death. The second RCT19 reported on symptoms. The non-randomized study20 reported on symptoms and behaviour. Follow-up times in the primary studies ranged between 6 months to 18 months.
The economic evaluation21,22 reported on the incremental cost-effectiveness ratio expressed as cost per quality-adjusted life-year (QALY) gained.
One guideline23 considered health-related and social outcomes (e.g., symptom severity and QoL). The second guideline1 considered outcomes such as patient and caregiver preferences; QoL; and behavioural, psychological, and depressive symptoms; and caregiver burden and depression. The third guideline2 considered outcomes such as health-related and social outcomes, and caregiver burden.
Summary of Critical Appraisal
An overview of the critical appraisal of the included publications follows. Additional details regarding the strengths and limitations of included publications are provided in Appendix 3 (Table 7 [systematic reviews], Table 8 [primary clinical studies], Table 9 [economic evaluation], and Table 10 [guidelines]).
Systematic Reviews
In all 3 systematic reviews,15-17 the objective was stated, multiple databases were searched, article selection was described and was conducted independently by 2 reviewers, data extraction was conducted, lists of included articles were presented, and study characteristics were described. In 1 systematic review,15 it was unclear if data extraction was done in duplicate and in the other 2 systematic reviews,16,17 data extraction was done by a single reviewer, hence the potential for errors cannot be ruled out. In 1 systematic review,17 the quality of the included primary studies was rated by the authors as moderate or high. In the second systematic review,15 the strength of evidence was graded by the authors as low or insufficient. In the third systematic review,16 the authors reported for the included primary studies scores between 6 to 11, with 11 being the highest score and higher scores indicating less bias. In 1 systematic review,15 results were presented narratively; meta-analysis was not conducted because of differences in outcome measures and intervention complexity. In the remaining 2 systematic reviews,16,17 meta-analyses were conducted and seemed appropriate. In all 3 systematic reviews,15-17 it was reported that the authors had no conflicts of interest.
Randomized Controlled Trials
In the 2 RCTs,18,19 the objectives and inclusion criteria were stated; the exclusion criteria were not explicitly stated; and the population characteristics, interventions, and outcomes were described. Both studies were RCTs and the randomization procedure seemed appropriate. Neither of the studies were blinded; hence, the potential for detection and performance bias cannot be ruled out. In 1 RCT,18 sample size calculations for an end-of-life dementia outcome were conducted and the appropriate number of patients recruited; however, due to greater than expected deaths, the study was underpowered. It is therefore unclear if there would have been a statistically significant difference in outcomes between the 2 interventions compared (statistical significance was not assessed). In the second RCT,19 it was unclear if sample size calculations had been undertaken. It is therefore unclear if the study had sufficient power to detect a statistically significant difference in between-group differences for dementia-related symptoms. In 1 RCT,18 missing data for family-rated end-of-life dementia outcomes varied between 7.1% and 16.2%; hence, this could impact the findings. However, as the proportions of missing data were not reported for the 2 groups separately, the direction of impact is unclear. In the second RCT,19 34% and 16% in the intervention and control (usual care) arms, respectively, did not complete the study. This could therefore impact the findings but the direction of impact is unclear. In 1 RCT,18 the authors reported that there were no conflicts of interest. In the second RCT,19 conflicts of interest were not reported, therefore the impact, if any, is unclear.
Non-Randomized Study
In the non-randomized study,20 the objective and inclusion criteria were stated; the exclusion criteria were not explicitly stated; and the population characteristics, interventions, and outcomes were described. Participants were not randomized; hence, there is the potential for selection bias. As well, participants and investigators were not blinded; there is therefore the potential for detection and performance bias. There was a substantial proportion (> 19%) who did not complete the study and this could impact findings; the direction of impact is unclear. The authors reported that attrition was not different between the 2 arms. The conflicts of interest of the authors were not presented; its impact if any, is therefore unclear.
Economic Evaluation
In the economic evaluation,21 the objective, strategies compared, perspective taken, time horizon, and sources for clinical, utility, and cost data were reported. Incremental analyses results were reported; and cost-effectiveness acceptability curves were plotted and probabilities of the intervention being cost-effective at a specific willingness-to-pay threshold were presented. Conclusions were consistent with the results reported. The authors reported that there were no conflicts of interest. Some limitations need to be considered when interpretating the results. The clinical data were obtained from an RCT involving mostly patients with no indication of cognitive impairment or mild cognitive impairment; hence, this limits the generalizability of the findings to other levels of dementia. The time horizon was 24 months, so outcomes beyond that time frame were unknown. Health care resource utilization was retrospectively collected through interviews and therefore subject to recall bias.
Guidelines
In all 3 guidelines,1,2,23 the scope and purpose were described, the target users were mentioned, systematic literature searches were conducted to identify evidence, and the recommendations were clearly presented. The 3 guidelines were externally reviewed. The guideline development groups comprised individuals from various relevant areas and the views and preferences of the target users were sought in 2 guidelines.1,2 But this was not so in the third guideline.23 In all 3 guidelines,1,2,23 supporting evidence on which recommendations were based were presented. In 2 guidelines,1,23 recommendations were formulated based on consensus and in 1 guideline2 the method for formulating the recommendations was not presented. Two guidelines1,2 had a process for updating the guidelines and 1 guideline2,23 did not. In 1 guideline,1 the applicability of the guidelines was not specified in the document; however, according to the guideline development manual26 cited by the authors, consideration of applicability is required. In the other 2 guidelines, it was unclear if applicability was considered. In 1 guideline,1 a declaration of conflicts of interest was not presented in the guideline document; however, according to the guideline development manual26 cited by the authors, a process is in place to record and address conflicts. In the other 2 guidelines,2,23 the authors declared that they had no conflicts of interest.
Summary of Findings
The main findings from the included publications are subsequently summarized. Appendix 4 presents the main study findings and authors’ conclusions.
Clinical Utility of Patient Navigation Programs
Three systematic reviews,15-17 2 RCTs,18,19 and 1 non-randomized study20 reported on outcomes of patient navigation programs (care coordination and/or case management) compared with usual care. Case management is sometimes also referred to as coordinated care, so the terminology “coordinated care” will be used to present findings.
Hospitalization, Nursing Home Admission, or Institutionalization
There was no significant difference in hospitalization with coordinated care compared to usual care (2 systematic reviews16,17). Length of hospital stay was less with coordinated care compared with usual care, but statistical significance was not reported (1 RCT18). For coordinated care compared with usual care regarding emergency room visits, 1 systematic review15 reported that coordinated care had benefit. For coordinated care compared with usual care regarding nursing home admissions, 1 systematic review15 reported mixed findings (benefit in some studies and no difference in other studies) and 1 systematic review16 reported no statistically significant between-group differences for the multifactorial treatment and assessment, and statistically significant reduction for community care coordination. For coordinated care compared with usual care, in terms of institutionalization, 1 systematic review15 reported mixed findings and 1 systematic review17 reported no statistically significant between-group differences.
Quality of Life
For coordinated care compared with usual care, for QoL, 1 systematic review15 reported mixed findings and 1 systematic review17 and 1 RCT18 reported no statistically significant between-group difference.
Depression
For coordinated care compared with usual care, for depression, 2 systematic reviews15,17 reported no statistically significant between-group differences.
Symptoms
For coordinated care compared with usual care, regarding neuropsychiatric symptoms (assessed using the Neuropsychiatric Inventory), 1 systematic review15 reported mixed findings. One RCT19 and 1 non-randomized study20 reported no significant difference in dementia-related or neuropsychiatric symptoms, respectively, with coordinated care compared with usual care. One RCT18 reported improvement in symptom management with coordinated care compared with usual care, but statistical significance was not reported.
Behaviour
For coordinated care compared with usual care, regarding behaviour, 1 systematic review17 showed statistically significant improvement in behaviour and 1 non-randomized study20 showed a significant reduction in dementia-related behaviour.
Function and Cognition
For coordinated care compared with usual care, 1 systematic review17 showed that in function and cognition there were no statistically significant between-group differences; and 1 systematic review15 showed that in function there was no between-group difference, but the statistical significance was not reported.
Mortality
One systematic review17 reported no difference in mortality with coordinated care compared with usual care. One RCT18 reported that there was no difference in the time period to death with coordinated care compared with usual care.
Cost-Effectiveness of Patient Navigation Programs
One economic evaluation21,22 comparing dementia care management (DCM) with usual care for patients with dementia was included. For patients with dementia, with DCM there was an increased QALY ( + 0.05) and a decreased cost (–€569) compared with usual care; i.e., DCM dominated usual care. For several subgroup analyses (age, female sex, living situation, deficits in daily living activities, different levels of cognitive deficits, different comorbidities) DCM was either dominant or cost-effective relative to usual care. The probability of DCM being cost-effective was 88% at a willingness-to-pay (WTP) threshold of €40,000 per QALY gained. The probabilities of DCM being cost-effective at a WTP of €40,000 per QALY gained, for the various subgroups, were 96% and 26% for patients living alone and patients not living alone, respectively; 87% and 48% for those aged ≥ 80 years and aged < 80 years, respectively; 96% and 16% in females and males, respectively; 97% and 16% for high and low deficit in daily activities, respectively; and 96% and 26% for high and low levels of comorbidities, respectively.
Guidelines
Three relevant guidelines1,2,23 were included. One guideline23 recommends the use of digital technology such as electronic health records to improve care coordination in persons with mental illness (recommendation grade C/D based on level 1 evidence; i.e., weak [details in Table 6]). It also recommends providing components of case management to persons with mental illness after discharge from inpatient treatment (recommendation grade C based on level 3 evidence; i.e., weak [details in Table 5]). A second guideline1 recommends, in the care of persons with dementia, providing a single-named health or social care professional who should be responsible for care coordination to ensure that information can be transferred between different settings, to maximize continuity and consistency, and to ensure relevant information is shared and recorded in the person’s care plan (recommendation: strong, based on moderate-level evidence). The third guideline2 recommends ensuring that “relevant information and care planning for older adults with delirium, dementia, and depression is communicated and coordinated over the course of treatment and during care transitions” (p. 91)2 (strength of recommendation not presented).
Limitations
Findings from the systematic reviews and primary studies were mixed for most outcomes and so definitive conclusions cannot be made. There was either considerable variability in what comprised coordinated care among the studies, or details were not available. The level of dementia was not always reported. Long-term effects are not known, as the follow-up times were generally less than 24 months. The majority of the studies were conducted in the US or Europe; therefore, generalizability of the findings to the Canadian context is unclear. However, there may be some similarities, as the majority of studies were conducted in developed countries.
The economic evaluation was based on a clinical data from a single study that involved people with dementia, the majority with no or mild cognitive impairment. The generalizability of the findings to a broader patient population is therefore limited. Furthermore, this was conducted in Germany and so the applicability to the Canadian context is unclear.
Conclusions and Implications for Decision- or Policy-Making
Three systematic reviews,15-17 2 RCTs,18,19 1 non-randomized study,20 1 economic evaluation (presented in 2 publications [primary analyses21 and subgroup analyses22]), and 3 evidence-based guidelines guidelines1,2,23 were included.
Three systematic reviews,15-17 2 RCTs,18,19 and 1 non-randomized study20 reported on the clinical utility of patient navigation programs (coordinated care) for the care of people with dementia. Overall, for coordinated care compared with usual care, findings were either mixed (i.e., in the systematic reviews, some studies showed a benefit and some showed no benefit) or there were no between-group differences in hospitalization, institutionalization, or nursing home admissions (3 systematic reviews15-17); QoL (2 systematic reviews15,16 and 1 RCT18); and symptoms (1 systematic review,15 1 RCT,19 and 1 non-randomized study20). For coordinated care compared with usual care, there were no statistically significant between-group differences in depression (2 systematic reviews15,17) and mortality (1 systematic review17 and 1 RCT18). There was improvement in behaviour with coordinated care compared with usual care (1 systematic review17 and 1 non-randomized study20). Details of patient characteristics were not always reported; it is possible that variation in patient characteristics could have contributed to mixed findings.
According to 1 economic evaluation for patients with dementia, compared with usual care, collaborative DCM was dominant; i.e., provided increased benefit (QALYs) and decreased cost. The probability of DCM being cost-effective was 88% at a WTP of €40,000 per QALY gained. This analysis was based on clinical data from a single RCT in which the majority of patients had no or mild cognitive impairment. It is possible, therefore, that the findings may not apply for a broader patient population.
Three guidelines1,2,23 were identified that reported on care coordination for people with dementia. One guideline1 recommends for the care of persons with dementia and the provision of a single named health or social care professional who should be responsible for the various aspects of care coordination (moderate evidence, strong recommendation). The second guideline23 recommends the use of digital technology to enhance care coordination in persons with mental illness (evidence level: 1; recommendation grade: C/D). The third guideline2 recommends coordinated care for people with delirium, dementia, and depression.
Findings need to be interpreted in the light of limitations reported.
As individuals with dementia have a variety of symptoms, providing care and support is very complex. Several factors such as implementation issues, health care resources required, and accessibility and training requirements need to be considered. Care programs may need to be adjusted to individual patient needs. Further research on the types of patient navigation programs that may be best suited to specific types and levels of dementia would provide additional insights into care management.
References
1.National Institute for Health Care and Excellence. Dementia: assessment, management and support for people living with dementia and their carers. (NICE guideline 97) 2018; https://www.nice.org.uk/guidance/ng97/evidence/full-guideline-pdf-4852695709 Accessed 2021 Jan 22.
2.Delerium, dementia, and depression in older adults: assessment and care. Toronto (ON): Registered Nurses' Association of Ontario; 2016: https://rnao.ca/sites/rnao-ca/files/bpg/RNAO_Delirium_Dementia_Depression_Older_Adults_Assessment_and_Care.pdf. Accessed 2021 Jan 22.
3.World Health Organization. Dementia. 2020; https://www.who.int/news-room/fact-sheets/detail/dementia. Accessed 2021 Jan 25.
4.Canadian Institute for Health Information. How dementia impacts Canadians. 2018; https://www.cihi.ca/en/dementia-in-canada/how-dementia-impacts-canadians. Accessed 2021 Jan 25.
5.Dementia in Canada, including Alzheimer's disease. Ottawa (ON): Public Health Agency of Canada; 2017: https://www.canada.ca/en/public-health/services/publications/diseases-conditions/dementia-highlights-canadian-chronic-disease-surveillance.html. Accessed 2021 Jan 25.
6.Khanassov V, Vedel I. Family physician-case manager collaboration and needs of patients with dementia and their caregivers: a systematic mixed studies review. Ann Fam Med. 2016;14(2):166-177. PubMed
7.Freeman HP, Rodriguez RL. History and principles of patient navigation. Cancer. 2011;117(15 Suppl):3539-3542. PubMed
8.YorkU. Patient navigator certificate. https://hlln.info.yorku.ca/fundamentals-of-patient-navigation/. Accessed 2021 Feb 3.
9.University of California San Francisco. Building a care ecosystem. 2021; https://memory.ucsf.edu/research-trials/professional/care-ecosystem. Accessed 2021 Feb 3.
10.Patient navigation programs for people with dementia: clinical utility, cost-effectiveness, and guidelines. (CADTH Rapid response report: summary of abstracts). Ottawa (ON): CADTH; 2020: https://www.cadth.ca/sites/default/files/pdf/htis/2020/RB1530%20Patient%20Navigation%20Final.pdf Accessed 2020 Jan 6.
11.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
12.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377-384. PubMed
13.Higgins JPT, Green S, editors. Figure 15.5.a: Drummond checklist (Drummond 1996). Cochrane handbook for systematic reviews of interventions. London (GB): The Cochrane Collaboration; 2011: http://handbook-5-1.cochrane.org/chapter_15/figure_15_5_a_drummond_checklist_drummond_1996.htm. Accessed 2021 Jan 22.
14.Agree Next Steps Consortium. The AGREE II Instrument. Hamilton (ON): AGREE Enterprise; 2017: https://www.agreetrust.org/wp-content/uploads/2017/12/AGREE-II-Users-Manual-and-23-item-Instrument-2009-Update-2017.pdf. Accessed 2021 Jan 22.
15.Minnesota Evidence-based Practice Center, Butler M, Gaugler JE, et al. Care interventions for people living with dementia and their caregivers (Comparative effectiveness review, no. 231). Rockville (MD): Agency for Healthcare Research and Quality; 2020: https://effectivehealthcare.ahrq.gov/sites/default/files/pdf/cer-231-dementia-interventions-final.pdf. Accessed 2021 Jan 5.
16.Lee DA, Tirlea L, Haines TP. Non-pharmacological interventions to prevent hospital or nursing home admissions among community-dwelling older people with dementia: a systematic review and meta-analysis. Health Soc Care Community. 2020;28(5):1408-1429. PubMed
17.Backhouse A, Ukoumunne OC, Richards DA, McCabe R, Watkins R, Dickens C. The effectiveness of community-based coordinating interventions in dementia care: a meta-analysis and subgroup analysis of intervention components. BMC Health Serv Res. 2017;17(1):717. PubMed
18.Agar M, Luckett T, Luscombe G, et al. Effects of facilitated family case conferencing for advanced dementia: a cluster randomised clinical trial. PLoS ONE. 2017;12(8):e0181020. PubMed
19.Mavandadi S, Wright EM, Graydon MM, Oslin DW, Wray LO. A randomized pilot trial of a telephone-based collaborative care management program for caregivers of individuals with dementia. Psychol Serv. 2017;14(1):102-111. PubMed
20.Mavandadi S, Wray LO, DiFilippo S, Streim J, Oslin D. Evaluation of a telephone-delivered, community-based collaborative care management program for caregivers of older adults with dementia. Am J Geriatr Psychiatry. 2017;25(9):1019-1028. PubMed
21.Michalowsky B, Xie F, Eichler T, et al. Cost-effectiveness of a collaborative dementia care management-results of a cluster-randomized controlled trial. Alzheimer's dement. 2019;15(10):1296-1308. PubMed
22.Radke A, Michalowsky B, Thyrian JR, Eichler T, Xie F, Hoffmann W. Who benefits most from collaborative dementia care from a patient and payer perspective? A subgroup cost-effectiveness analysis. J Alzheimers Dis. 2020;74(2):449-462. PubMed
23.Gaebel W, Kerst A, Janssen B, et al. EPA guidance on the quality of mental health services: a systematic meta-review and update of recommendations focusing on care coordination. Eur Psychiatry. 2020;63(1):e75. PubMed
24.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
25.Ramsey S, Willke R, Briggs A, et al. Good research practices for cost-effectiveness analysis alongside clinical trials: the ISPOR RCT-CEA Task Force report. Value Health. 2005;8(5):521-533. PubMed
26.National Institute for Health Care and Excellence. Developing NICE guidelines: the manual. 2014; https://www.nice.org.uk/process/pmg20/resources/developing-nice-guidelines-the-manual-pdf-72286708700869 Accessed 2021 Jan 22.
27.Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-926. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Table 2: Characteristics of Included Systematic Reviews
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
Butler et al.(2020)15 US Funding: sponsored by the Agency for Healthcare Research and Quality | Systematic review Number of primary studies included: 627, of which 10 RCTs (the studies were conducted in the US [4], Germany [1], Finland [2], Norway [1], and China [2]) were relevant for this current report. These studies were published between 2001 and 2019. Setting: community, except nursing home for 1 RCT Inclusion criteria: adults with possible or diagnosed AD/ADRD RCTs, and prospective studies with concurrent comparator arms. Only English publications due to resource limitations Exclusion criteria: before and after studies with no comparator arm. Fewer than 10 patients in each treatment group Aim: to assess care interventions for people with dementia and their caregivers that link to benefits | Individuals living with dementia The authors categorized different care options. The 2 options (case management and collaborative care) that are relevant for the current review are presented here. Case management (3 RCTs) N = 294 Age (range for mean) (years): 68 to 79 % Female (range): 44% to 58% Dementia type: unspecified Collaborative care (7 RCTs) N = 2,641 Age (range for mean) (years): 73 to 83 % Female (range): 43% to 77% Dementia type: unspecified | Interventions: Case management: health and social services, to support individuals with dementia and their caregivers, were coordinated by case manager. The case and case manager were not reported to be part of a team-based care approach. Collaborative care: A multidisciplinary team integrated medical and psychological approaches for health care of the individuals with dementia. The team members were at the same location or at different locations with a common hub. Support was provided to the individual with dementia and to the caregiver. Comparator Usual care: description not presented | Institutionalization, QoL, depression, neuropsychiatric symptoms, function, quality indicators, ER visits, nursing home placement Follow-up: for case management, 12 to 24 months For collaborative care, 6 to 18 months |
Lee et al. (2020)16 Australia Funding: not reported | Systematic review and meta-analysis Number of primary studies included: 20 studies of which 6 studies (RCTs) were relevant for this current report. These RCTs were conducted in the US (2), UK (1), Australia (1), Germany (1), and France (1). These were published between 1999 to 2014. Setting: community Inclusion criteria: RCT and non-randomized controlled studies with a control or comparison group Population: persons with dementia Outcomes reported: hospital and nursing home admissions QoL of persons with dementia and their caregivers Exclusion criteria: mean age less than 65 years, persons in nursing home or hospital, non-English publications Aim: to assess non-pharmacological interventions that can minimize hospital or nursing home admissions for people with dementia | Individuals with dementia N = 1,958 Age (in years): not reported (65 or older according to the inclusion criteria) % Female: not reported. Dementia type: not reported | Interventions: coordinated care Multifactorial assessment and treatment clinics/services Comparator: Usual care | Hospital admission, nursing home admission Follow-up: 9 to 12 months |
Backhouse et al. (2017)17 UK Funding: funded by the National Institute for Health Research (NIHR) Collaboration for Leadership in Applied Health Research and Care South West Peninsula | Systematic review and meta-analysis Number of primary studies included: 14 RCTs. These RCTs were conducted in the US (6), China (3), Finland (2), Canada (1), the Netherlands (1), and India (1). The RCTs were published between 2000 and 2014 Setting: community Inclusion: RCTs Population: individuals with diagnosis of dementia, no restrictions on age or gender. No restrictions on language of publication Exclusion: non-randomized experimental studies, studies that focused only on informal caregivers Aim: to assess effectiveness of community-based coordinating interventions for people with dementia | Individuals with dementia N = 10,392 Age (years): not reported (no restrictions according to the inclusion criteria) % Female: not reported. Dementia type: not reported | Interventions: interventions delivered by a single identified professional who was responsible for the provision and management of care (i.e., planning, facilitating and/or coordinating care) Comparators: usual care, standard community treatment, alternative dementia care interventions or waiting-list controls | Hospitalization, institutionalization, mortality, QoL, cognition, function, behaviour, and depression Follow-up (months): 12 to 36 |
AD = Alzheimer disease; ADRD = Alzheimer disease–related dementia; ER = emergency room; QoL = quality of life; RCT = randomized controlled trial.
Table 3: Characteristics of Included Primary Clinical Studies
Study citation, country, funding source | Study design | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
Randomized controlled trials | ||||
Agar et al. (2017)18 Australia Funding: not reported | RCT cluster design, single-blinding. The nursing homes were randomized. The staff, residents, and families at each nursing home were blinded. Participating investigators, project managers, and nursing home managers were not blinded because of the system-level nature of the intervention. Setting: nursing home (20 nursing homes in 2 major Australian cities) Inclusion criteria: nursing home designated as facility providing intensive level of care and having ≥ 100 beds and with ≥ 50% residents with dementia Exclusion criteria: not specified Aim: to assess the efficacy of FCC in improving EOL care for individuals with advanced dementia in nursing homes | Individuals with advanced dementia living in nursing homes N = 286 (156 in FCC [in 10 nursing homes]; 130 in UC [in 10 nursing homes) — of these, 131 (67 in FCC, 64 in UC) were included in the EOLD analysis. Those alive throughout the study (89 in FCC, and 66 in UC) were excluded from the analysis. Age (years) (mean ± SD): 85.3 ± 8.0 % Female: 60% Dementia level: advanced | Intervention: FCC. A registered nurse was trained as a palliative care planning coordinator who worked 2 days per week or equivalent. Responsibilities included identifying residents with advanced dementia who are likely to benefit; organizing and documenting case conferences involving family, multidisciplinary nursing home staff, and external health professionals; developing and overseeing implementation of care plans; and training nursing and direct care staff. Comparator: UC. No staff education, training or support was provided. There were no restrictions in terms of approach to care planning and decision-making. | Family-rated EOLD scores; nurse-rated EOLD scores, pharmacological management, non-pharmacological management, hospital admissions and emergency department admissions Study period: 18 months |
Mavandadi et al. 2017 (US)19 Funding: not reported | RCT (pilot trial) Setting: primary care (p. 1) Inclusion criteria: age ≥ 18 years, patient living in the community, and patient had a dementia diagnosis Exclusion criteria: not specified Aim: to assess the extent to which care management is associated with changes in caregiver and patient outcomes compared with usual care Note: only details of the population (patients with dementia) relevant for the current report are described here. | Older veterans with dementia N = 75 (38 in CM, 37 in UC) Age (years) (mean ± SD):78.95 ± 8.96 % Female: not reported (it was reported that patients were mainly male veterans) Dementia level (severity of symptoms based on NPI-Q score) (mean ± SD): 7.62 ± 5.32 (higher score indicates more severity) | Intervention: CM; individualized dementia CM, which is delivered by either nurse or social work–trained clinicians. The care manager coordinated connection to VA and community programs. The care manager also helped to coordinate scheduling in primary and specialty care. It also included a telephone education program. Comparator: UC; patients continued to receive standard care through the VA. Caregivers were provided information about VA and community resources for patients with dementia and their caregivers. | Symptom change (using NPI-Q, RMBPC) Follow-up: 6 months |
Non-randomized studies | ||||
Mavandadi et al. (2017)20 US Funding: funded by the state of Pennsylvania’s PACE/PACENET Program, Pennsylvania Department of Aging, Harrisburg, PA. The work was further supported by US Department of Veterans Affairs VISN4 MIRECC and VA Center for Integrated Healthcare. The funding source and the U.S. Department of Veterans Affairs had no role in the study design, methods, subject recruitment, data collection, analysis, or preparation of this article | Non-randomized study, prospective Setting: general community Inclusion criteria: care recipients aged ≥ 65 years, living in a non-institutionalized setting, screened positive for dementia; had at least 1 prescription of an antidepressant, anxiolytic, or antipsychotic in the past 6 months; caregiver age ≥ 18 years Exclusion criteria: not specified Aim: to assess efficacy of telephone-delivered community-based collaborative care management program for caregivers and care recipients (older adults with dementia) Note: only details of the population (patients with dementia) relevant for the current report are described here. | Individuals with dementia N = 440 (150 in ECS, 290 in CAR) Age (years): not reported (age ≥ 65 years was mentioned in the inclusion criteria) % Female: not reported Dementia level: not reported | Intervention: ECS is a comprehensive dementia management program. It comprised all of the services provided to the CAR group, in addition to service connection, psychoeducation, and support delivered by BHPs. All interviews and care management activities were via phone. Comparator: CAR. This comprised a baseline clinical interview followed by a brief summary regarding case recipient’s symptoms and functional status, and reported service and resource needs, which were then sent to the prescribing clinicians for treatment planning, clinical assessment, and referral | Behaviour and symptom change (using NPIQ, RMBPC) Follow-up: 6 months |
BHPs = behavioural health providers; CAR = clinical assessment and referral; CM = care management; ECS = enhanced caregiver services; EOL = end-of-life; EOLD = end-of-life dementia; FCC = facilitated case conferencing; MIRECC = Mental Illness Research Education and Clinical Center; NPIQ = Neuropsychiatric Inventory Questionnaire; PACE/PACENET = Pharmaceutical Assistance Contract for the Elderly/ Pharmaceutical Assistance Contract for the Elderly Needs Enhancement Tier; RCT = randomized controlled trial; RMBPC = Revised Memory and Behavior Problems Checklist; UC = usual care; VA = Veterans Affair; VA CIH = VA Center for Integrated Healthcare.
Table 4: Characteristics of Included Economic Evaluation
Study citation country, funding source | Type of analysis, time horizon, perspective | Population characteristics | Intervention and comparator(s) | Approach | Source of clinical, cost, and utility data used in analysis | Main assumptions |
Michalowsky et al. (2019)21 Germany Funding: German research foundation | Cost-utility analysis Time horizon: 24 months Perspective: public payer perspective (excluding informal care and caregiver QALYs) Discounting: 5% per year | Individuals with dementia, majority with no or mild cognitive impairment, and their caregivers | Intervention: community-based collaborative DCM to support PWD and their caregivers; targeted at the individual participant level delivered in the person’s home by a nurse with dementia-specific training. The nurse developed the care plan based on discussion with treating GP and implemented it with cooperation from the GP and various health care and social services Comparator: usual care | The economic evaluation was conducted alongside the clinical trial (cluster RCT) As there were differences in sample characteristics and dependency of observation to cluster (GPs), incremental cost and QALYs were estimated using linear regression models. To handle uncertainty in ICER, a nonparametric bootstrapping was used creating 1,000 resamples that were stratified for the cluster and group distribution. The probability of DCM being cost-effective was calculated using these resamples and different WTP thresholds. The authors reported that methods used for this analysis were consistent with those of the published guidelines for undertaking economic evaluations25 Sensitivity analyses were conducted | Clinical and utility data were from a cluster RCT (Delphi trial conducted in Germany involving patients who were mildly cognitively impaired) Preference weights used to calculate health utilities were obtained from a sample of the general population in the UK Cost data were obtained from market prices, the pharmaceuti-cal index of the scientific research institute of the AOK, and the literature | A linear change in HRQoL was assumed, considering the nature of dementia; i.e., increasing cognitive and functional deficits that affect HRQoL |
AOK = Allgemeine Ortskrankenkasse; DCM = dementia care management; GP = general practitioner; HRQoL = health-related quality of life; ICER = incremental cost-effectiveness ratio; PWD = person with dementia; QALY = quality-adjusted life-year; RCT = randomized controlled trial; WTP = willingness to pay.
Table 5: Characteristics of Included Guidelines
Intended users, target population | Intervention and practice considered | Major outcomes considered | Evidence collection, selection, and synthesis | Evidence quality assessment | Recommendations development and evaluation | Guideline validation |
EPA (2020),23 Europe | ||||||
Intended users: those involved in the care of people with mental health issues and organizers of mental health services Target population: people with mental health issues | Care coordination | Symptom change, quality of life, and service-related outcomes | Systematic literature search was conducted to identify relevant evidence. The authors reported that they conducted a systematic meta-review, which included a systematic overview of systematic reviews, meta-analyses, and evidence-based guidelines | Evidence was graded on a 4-point scale Recommendations were graded based on criteria reported in a European Psychiatric Association publication, with some modifications | GDG composition was not presented Recommendations were formulated by consensus There was a method for grading the recommendations | Externally reviewed |
NICE (2018),1 UK | ||||||
Intended users: People with dementia and those involved in the care of people with dementia Target population: people with dementia and their caregivers | Dementia diagnosis, care planning and coordination of care, inpatient care, pharmacological and non-pharmacological treatment, staff training, palliative care, and support for informal caregivers | Behavioural symptoms, cognitive impairment, health service usage, hospitalization, caregiver depression, caregiver satisfaction, and resource use and cost | Systematic literature search was conducted to identify relevant evidence (both quantitative and qualitative) Meta-analysis was conducted where possible or evidence was summarized narratively. This guideline was developed using a rigorous process (NICE manual)26 | Evidence quality was assessed using GRADE | GDG comprised a multidisciplinary team (such as psychiatrist, psychologist, nurse, occupational therapist, consultant, and health economist), social worker, and lay persons Recommendations were developed by consensus There was a method for grading the recommendations | Externally reviewed |
RNAO (2016),2 Canada | ||||||
Intended users: primarily for nurses providing direct clinical care to older adults. May also be used by other members of the interprofessional team who collaborate with nurse to provide comprehensive care, educators, administrators, and policy-makers Target population: people with delirium, dementia, or depression | Approaches needed for nurses to assess and manage older adults Nurse practice | Hospitalization, behavioural and psychological symptoms, depression management, communication, and care planning | Systematic literature search was conducted to identify relevant evidence (from relevant peer-reviewed literature and guidelines; additional details regarding study designs that were eligible were not specified) | Evidence quality was graded using a method based on that of SIGN and a published framework for evaluating evidence | The guideline program team and the expert panel included individuals holding clinical, administrative, and academic positions in various health care organizations and practice areas Method for formulating the recommendations was unclear It was unclear if there was a method for grading recommendations | Externally reviewed |
EPA = European Psychiatric Association; GDG = guideline development group; GRADE = Grading of Recommendations Assessment, Development and Evaluations; NICE = National Institute for Health and Care Excellence; RNAO = Registered Nurses’ Association of Ontario; SIGN = Scottish Intercollegiate Guidelines Network.
Table 6: Ratings Used in the Guidelines
Ratings for the evidence and associated recommendation |
EPA (2020),23 Europe |
Grade of evidence:
Grade of recommendations:
|
NICE (2018),1 UK |
NICE,1 used the GRADE methodology for assessing evidence quality. GRADE methodology:27
|
RNAO (2016),2 Canada |
RNAO, 20162
|
AMSTAR = A MeaSurement Tool to Assess systematic Reviews; EPA = European Psychiatric Association; GRADE = Grading of Recommendations Assessment, Development and Evaluations; NICE = National Institute for Health and Care Excellence; RCT = randomized controlled trial; RNAO = Registered Nurses’ Association of Ontario.
Appendix 3: Critical Appraisal of Included Publications
Table 7: Strengths and Limitations of Systematic Reviews Using AMSTAR 211
Strengths | Limitations |
Butler et al. (2020),15 US | |
|
|
Lee et al. (2020),16 Australia | |
|
|
Backhouse et al. (2017),17 UK | |
|
|
Table 8: Strengths and Limitations of Clinical Studies Using the Downs and Black Checklist12
Strengths | Limitations |
Randomized controlled trial | |
Agar et al. (2017)18 Australia | |
|
|
Mavandadi et al. (2017), US19 | |
|
|
Non-randomized study | |
Mavandadi et al. (2017),20 US | |
|
|
CAR = clinical assessment and referral; CM = care management; ECS = enhanced caregiver services; EOLD = end-of-life dementia; FCC = facilitated case conferencing; ITT intent-to-treat; UC = usual care.
Table 9: Strengths and Limitations of Economic Evaluation Using the Drummond Checklist13
Strengths | Limitations |
Michalowsky et al. (2019),21 Germany | |
|
|
Table 10: Strengths and Limitations of Guidelines Using AGREE II14
Item | EPA (2020),23 Europe | NICE (2018),1 UK | RNAO (2016),2 Canada |
Domain 1: Scope and Purpose | |||
1. The overall objective(s) of the guideline is (are) specifically described. | Yes | Yes | Yes |
2. The health question(s) covered by the guideline is (are) specifically described. | Yes | Yes | Yes |
3. The population (patients, public, and so forth.) to whom the guideline is meant to apply is specifically described. | Yes | Yes | Yes |
Domain 2: Stakeholder Involvement | |||
4. The guideline development group includes individuals from all relevant professional groups. | Unclear | Yes | Yes |
5. The views and preferences of the target population (patients, public, and so forth) have been sought. | Unclear | Yes | Yes (stakeholder input was sought) |
6. The target users of the guideline are clearly defined. | Yes | Yes | Yes |
Domain 3: Rigour of Development | |||
7. Systematic methods were used to search for evidence. | Yes | Yes | Yes |
8. The criteria for selecting the evidence are clearly described. | Yes | Yes | Unclear |
9. The strengths and limitations of the body of evidence are clearly described. | Unclear | Yes | Yes |
10. The methods for formulating the recommendations are clearly described. | Yes | Yes | Unclear |
11. The health benefits, side effects, and risks have been considered in formulating the recommendations. | Unclear | Yes | Unclear |
12. There is an explicit link between the recommendations and the supporting evidence. | Yes | Yes | Yes |
13. The guideline has been externally reviewed by experts before its publication. | Yes | Yes | Yes |
14. A procedure for updating the guideline is provided. | no | Yes | Yes |
Domain 4: Clarity of Presentation | |||
15. The recommendations are specific and unambiguous. | Yes | Yes | Yes |
16. The different options for management of the condition or health issue are clearly presented. | Yes | Yes | Yes |
17. Key recommendations are easily identifiable. | Yes | Yes | Yes |
Domain 5: Applicability | |||
18. The guideline describes facilitators and barriers to its application. | No | Uncleara | No |
19. The guideline provides advice and/or tools on how the recommendations can be put into practice. | No | Uncleara | To some extent (mentions implementation strategies) |
20. The potential resource implications of applying the recommendations have been considered. | No | Uncleara | No |
21. The guideline presents monitoring and/or auditing criteria. | No | Uncleara | No |
Domain 6: Editorial Independence | |||
22. The views of the funding body have not influenced the content of the guideline. | For the research, no specific grant from any funding agency, commercial, or not-for-profit organization were received. The authors reported that they had no conflicts of interest. | Uncleara | The authors reported that no limiting conflicts were identified. |
23. Competing interests of guideline development group members have been recorded and addressed. | The process of recording and addressing conflicts of interest were not presented. | Uncleara | It was reported that conflicts of interest were recorded and there was a process in place to address conflicts. |
AGREE II = Appraisal of Guidelines for Research and Evaluation II; EPA = European Psychiatric Association; NICE = National Institute for Health and Care Excellence; RNAO = Registered Nurses’ Association of Ontario.
aUnclear = It was not explicitly mentioned in the guideline report, but it was mentioned that the guidelines were developed according to the NICE guideline development manual,26 which requires that applicability is considered and editorial independence is ascertained.
Appendix 4: Main Study Findings and Authors’ Conclusions
Summary of Findings of Included Systematic Reviews
Butler et al. (2020),15 US
Main study findings
Outcomes with (1) case management and (2) collaborative care for individuals with dementia
Outcomes with case management compared to usual care
QoL
(1 randomized controlled trial [RCT], 102 individuals with dementia)
No between-group difference (1 RCT, P = not significant)
Strength of evidence: insufficient
Neuropsychiatry Inventory
(2 RCTs, 194 individuals with dementia)
Benefit (1 RCT, P < 0.01)
No between-group difference (1 RCT, P = not significant)
Strength of evidence: insufficient
Depression
(1 RCT, 102 individuals with dementia)
No benefit (1 RCT. P = not significant)
Strength of evidence: insufficient
Institutionalization rate
(2 RCTs, 192 individuals with dementia)
Benefit (1 RCT, P < 0.01)
No between-group difference (1 RCT, P = not significant)
Strength of evidence: insufficient
Collaborative care compared to usual care
QoL
(4 RCTs; 1,746 individuals with dementia)
Benefit (2 RCTs)
No between-group difference (2 RCTs)
Strength of evidence: low
Neuropsychiatry symptoms
(1 RCTs, 152 individuals with dementia)
Benefit (1 RCT)
Strength of evidence: insufficient
Depression
(1 RCT, 152 individuals with dementia)
No between-group difference (1 RCT)
Strength of evidence: insufficient
Function
(2 RCT, 560 individuals with dementia)
No between-group difference (2 RCTs)
Strength of evidence: insufficient
Quality indicators
(2 RCT, 559 individuals with dementia)
Benefit (2 RCTs)
Strength of evidence: low
Nursing home placement
(3 RCTs, 794 individuals with dementia)
Benefit found at 1.6 years but not at 2 years (1 RCT)
No between-group difference (2 RCTs)
Strength of evidence: insufficient
Emergency room visit
(1 RCT, 780 individuals with dementia)
Benefit (1 RCT)
Strength of evidence: low
Authors’ conclusions
Case management compared to usual care
“Evidence was insufficient to draw conclusions about the effect of case management for persons living with dementia and their caregivers.” (p. 85)15
Collaborative care
“Collaborative care models (i.e., Care Ecosystems or discrete adaptations of the ACCESS [Alzheimers Disease Coordinated Care for San Diego Seniors] models) may improve persons living with dementia quality of life. (low-strength evidence) This improvement may be very small to small, or it may be larger but concentrated in some not yet identified subgroup of people.
Collaborative care models (i.e., discrete adaptations of the ACCESS model) may improve system-level markers, including guideline-based quality indicators and reduction in emergency department visits. (low-strength evidence)
Evidence was insufficient to draw conclusions about all other outcomes for both persons living with dementia and caregiver/partner.” (p. 93)15
Lee et al. (2020),16 Australia
Main study findings
Outcomes with (1) community care coordination and (2) multifactorial assessment and treatment clinics/services for individuals with dementia:
Outcomes with community care coordination compared with usual care
Rate of nursing home admission (2 RCTs): effect size (ES), 0.66; 95% confidence interval (CI), 0.45 to 0.97 (statistically significant reduction with community care coordination); heterogeneity, I2 = 0%
Outcomes with multifactorial assessment and treatment clinics/services compared with usual care
Risk of hospital admission (2 RCTs): ES, 1.29; 95% CI, 0.67 to 2.51 (no statistically significant between-group difference); heterogeneity, I2 = 0%
Risk of nursing home admission (2 RCTs): ES, 2.83; 95% CI, 0.85 to 9.46 (no statistically significant between-group difference); heterogeneity, I2 = 0%
Rate (%) of nursing home admission (2 RCTs): ES, 1.95; 95% CI, 0.71 to 1.41 (no statistically significant between-group difference); heterogeneity, I2 = 0%
Authors’ conclusion
“The meta-analyses conducted in this review were mainly the combined results of two or three studies of an intervention. Therefore, caution needs to be taken in using these results. Current evidence suggests that policy-makers and service providers of home and community care programmes may consider using community care coordination to target a reduction in nursing home admission rate in people with dementia.” (p. 1428)16
Backhouse et al. (2017),17 UK
Main study findings
Outcomes with community-based care coordinating interventions for individuals with dementia:
Community-based care coordinating interventions compared with control (usual care, standard care, alternative dementia care interventions or waiting-list controls)
Hospitalization (6 RCTs): odds ratio (OR), 0.89; 95% CI, 0.64 to 1.24 (no statistically significant between-group difference); heterogeneity, I2 = 0%
Institutionalization (9 RCTs): OR, 0.60; 95% CI, 0.32 to 1.11 (no statistically significant between-group difference); heterogeneity, I2 = 48%
Mortality (9 RCTs): OR, 0.97; 95% CI, 0.81 to 1.16 (no statistically significant between-group difference); heterogeneity, I2 = 0%
QoL (3 RCTs): OR, 0.09; 95% CI, −0.09 to 0.27 (no statistically significant between-group difference); heterogeneity, I2 = 0%
Cognition (4 RCTs): OR, −0.09; 95% CI, −0.29 to 0.11 (no statistically significant between-group difference); heterogeneity, I2 = 0%
Function (3 RCTs): OR, −0.08; 95% CI, −0.30 to 0.14 (no statistically significant between-group difference); heterogeneity, I2 = 0%
Depression (3 RCTs): OR, 0.60; 95% CI, −1.08 to 2.27 (no statistically significant between-group difference); heterogeneity, I2 = 66%
Behaviour (4 RCTs): OR, −9.52; 95% CI, −18.05 to −1.00 (statistically significant improvement with coordinated care compared to control); heterogeneity, I2 = 88%
Authors’ conclusions
“The results of our review have shown that coordinating interventions have some potential for positive impact on selected outcome measures, but the evidence is inconsistent.
“The differences across models of coordinating interventions in dementia care are substantial, and this has made it difficult to identify what should be considered core components. However, with the rising prevalence of dementia, it is likely that complex interventions will be necessary to provide high quality and effective care for patients, and facilitate collaboration of health, social and third sector services. Furthermore, although there are challenges to the implementation of coordinating interventions, addressing those and incorporating more stakeholder preferences may produce more consistent results and increase the likelihood of success.” (p. 9 of 10)17
Summary of Findings of Included Randomized Controlled Trials
Agar et al. (2017),18 Australia
Main study findings
Outcomes with facilitate case conferencing (FCC) compared with usual care (UC), for individuals with advanced dementia:
Family-rated end-of-life dementia (EOLD) care scores with respect to the individuals with advanced dementia who died.
CAD-EOLD care (mean ± SD): 34.7 ± 5.9 for FCC and 35.5 ± 5.9 for UC; higher scores indicate more comfort
Symptom management-EOLD care (mean ± SD): 29.0 ± 9.5 for FCC, 31.7 ± 7.4 for UC; higher scores indicate lower symptom frequency
Satisfaction with care-EOLD care (mean ± SD): 31.0 ± 5.3 for FCC, 30.3 ± 4.2 for UC; higher scores indicate greater satisfaction
Nurse-rated EOLD care scores (mean ± SD) for the individuals with advanced dementia who died
CAD-EOLD care (mean ± SD): 32.1 ± 6.1 for FCC and 33.3 ± 5.7 for UC; higher scores indicate more comfort
Symptom management-EOLD care (mean ± SD): 22.4 ± 9.6 for FCC and 23.2 ± 8.3 for UC; higher scores indicate lower symptom frequency
Quality of life (QoL)-based generalized linear mixed models, with FCC and UC compared
For family-related satisfaction with care-EOLD (P = 0.91)
For staff-related CAD-EOLD (P = 0.13)
Care in the last month of life
Medication changes: 94% for FCC, 75% for UC, (P < 0.01)
Non-pharmacologic management: 85% for FCC and 68% for UC, (P < 0.05)
At least 1 hospital admission (median): 19% for FCC and 18% for UC, (P = not reported)
Hospital length of stay: 2 for FCC and 5 for UC (P = not reported); unit of length of stay was not specified
Emergency department presentation without hospital admission: 9% for FCC and 10% for UC (P = not reported)
Input from health professionals: 59% for FCC and 60% for UC (P = not reported)
Death
Median time to death: 7 months, with no difference between the FCC and UC arms, P = 0.27
Authors’ conclusion
“This study is one of the few RCTs of palliative care interventions in nursing homes worldwide, and appears to be the first to test efficacy of facilitated family case conferencing for people with advanced dementia. The study's primary endpoint of quality of EOL care was under-powered and did not show evidence of effect. In spite of these limitations, a systematic approach to facilitating a palliative approach and skills enhancement drove improvements in care. Given the growing burden of dementia globally, these data will be formative in interventions aimed to improving palliative care in nursing homes in the future.” (p. 12-13)18
Mavandadi et al. (2017), US19
Main study findings
Outcomes with care management compared with usual care for patients with dementia:
There was very little reported on patient outcomes. The main focus of this study was caregiver outcomes. “Caregiver” is not a relevant population for this current report; therefore findings pertaining to mainly the patients are presented here.
It was mentioned that, for care management compared to usual care, no significant between-group differences were found in caregiver burden or patients’ dementia-related symptoms. Additional details of patient outcomes were not presented.
Authors’ conclusion
“Findings suggest that CGs of veterans with dementia may benefit from a telephone-delivered, care management program in improving CG-related outcomes. Further research of care management program for caregivers of veterans with dementia in addressing barriers to care and reducing caregiver burden is warranted. These findings highlight the potential for such programs as adjuncts to dementia care offered in primary care practices. (p. 1)19
Summary of Findings of Included Non-Randomized Study
Mavandadi et al. (2017),20 US
Main study findings
Findings (from a non-randomized study) for care management program (enhanced caregiver service) compared with usual care (CAR) for patients with dementia:
There was very little reported on patient outcomes. The main focus of this study was caregiver outcomes. “Caregiver” is not a relevant population for this current report; therefore findings pertaining mainly to the patients are presented here.
Longitudinal mixed-effects analyses suggested no significant time x intervention group interaction effect (P = 0.94) in neuropsychiatric symptoms (based on Neuropsychiatric Inventory Questionnaire) in the full sample over time (i.e., baseline, 3-month, and 6-month follow-up).
However, for a subset of caregivers (N = 116 in the enhanced caregiver service arm), revised memory and behaviour problems checklist data were available, which showed there was a statistically significant reduction in frequency of dementia-related behaviours (P = 0.03)
Authors’ conclusion
“A community-based, telephone-delivered care management program for caregivers of individuals with dementia is associated with favorable caregiver and care-recipient-related outcomes.” (p. 1019)20
Summary of Findings of Included Economic Evaluation
Michalowsky et al. (2019),21 and Radke et al. (2020),22 Germany
Main study findings
Comparison of dementia care management (DCM) (intervention) with usual care (control) for care of individuals with dementia:
Primary analyses21
For persons living with dementia who are not living alone:
Incremental cost (€) (mean [standard error; SE]): 1,799 (3,020)
Incremental quality-adjusted life-year (QALY) (mean [SE]): 0.067 (0.06)
Incremental cost per QALY gained: €26,851 per QALY
For PWD who are living alone:
Incremental cost (€) (mean [SE]): −3,642 (3,938)
Incremental QALY (mean [SE]): 0.034 (0.06)
Incremental cost per QALY gained: DCM dominates
For all PWD (complete case analysis):
Incremental cost (€) (mean [SE]): −569 (24,91)
Incremental QALY (mean [SE]): 0.049 (0.04)
Incremental cost per QALY gained: DCM dominates
Considering a societal perspective (i.e., including informal care and QALYs of the caregivers), the incremental cost (€) was −351 and the incremental QALY was 0.06; therefore still in favour of DCM and demonstrating that DCM dominates usual care
Probabilities of DMC being cost-effective at a WTP €40,000 per QALY gained (based on the cost-effectiveness acceptability curves):
For all persons living with dementia (the complete case), the probability was 88%
For the persons living with dementia who were living alone, the probability was 96%
For the persons living with dementia who were not living alone, the probability was 26%
Subgroup analyses22
Incremental cost-effectiveness ratio (ICER – cost per QALY gained) values for DCM compared with usual care, according to different patient characteristics (such as age, sex, living situation, cognitive ability, function, and comorbidities)
Age:
Age < 80 years, ICER = €35,145 per QALY gained
Age ≥ 80 years, ICER = −€95,282 per QALY gained; DCM dominates
Sex:
Female, ICER = −€199,639 per QALY gained; DCM dominates
Male, ICER = €71,173 per QALY gained
Living situation:
Living alone, ICER = −€107,118 per QALY gained; DCM dominates
Not living alone, ICER = €26,851 per QALY gained
Deficits in daily living activities (B − ADL):
None, ICER = €20,414 per QALY gained
Mild, ICER = −€50,528 per QALY gained; DCM dominates
High, ICER = €21,836 per QALY gained; DCM dominates
Cognitive deficits (Mini-Mental State Examination):
No indication, ICER = −€37,313 per QALY gained (incremental cost with DCM compared to usual care was €5,485 and the corresponding incremental benefit with DCM was −0.147; i.e., usual care has less cost and more benefit); usual care dominates
Mild, ICER = −€5,945 per QALY gained; DCM dominates
Moderate to severe, ICER = −€54,647/ per QALY gained; DCM dominates
Comorbidity (Charleston Comorbidity Index):
Low, ICER = −€39,520 per QALY gained (i.e., with usual care, less cost and more benefit compared with DCM); usual care dominates
High, ICER = −€104,451 per QALY gained; DCM dominates
Very high, ICER = €26,694 per QALY gained
Probabilities of DMC being cost-effective at a WTP €40,000 per QALY gained for the various subgroups (based on the cost-effectiveness acceptability curves):
For subgroup: age < 80 years, probability was 48%
For subgroup: age ≥ 80 years, probability was 87%
For subgroup: females, probability was 96%
For subgroup: males, probability was 16%%
For subgroup: living alone, probability was 96%
For subgroup: not living alone, probability was 26%
For subgroup: high deficit in daily living activities (Bayer Activities of Daily Living Scale), probability was 97%
For subgroup: low deficit in daily living activities (Bayer Activities of Daily Living Scale), probability was 16%
For subgroup: with moderate cognitive deficits (Mini-Mental State Examination), probability was 100%
For subgroup: with no cognitive deficits (Mini-Mental State Examination), probability was 3%
For subgroup: high comorbidity, probability was 96%
For subgroup: low comorbidity, probability was 26%
Authors’ conclusions
Primary analyses21
“DCM is likely to be a cost-effective strategy in treating dementia and thus beneficial for public health-care payers and patients, especially for those living alone.” (p. 1296)21
Subgroup analyses22
“Patient characteristics significantly affect the cost-effectiveness. Females, patients living alone, patients with a high comorbidity, and those being moderately cognitively and functionally impaired benefit most from DCM. For those subgroups, healthcare payers could gain the highest cost savings and the highest effects on QALYs when DCM will be implemented.” (p. 449)22
Summary of Recommendations in Included Guidelines
European Psychiatric Association (2020),23 Europe
Recommendations, supporting evidence, quality of evidence, and strength of recommendations
Recommendation:
“Use digital technology such as electronic health records to enhance care coordination.” (p. 9)23
Evidence
Care coordination with the use of electronic health records allowed easier patient access to health care and enhanced communication between the caregiver and the patient (1 systematic review).
Grade of recommendation: C/D
Level of evidence: 1
Recommendation:
“Provide elements of case management to persons with mental illness after discharge from inpatient treatment.” (p. 8)23
Evidence
Limited evidence suggested that some components of case management (e.g., transition managers and timely communication between inpatient staff and outpatient care) may have positive effects on health-related and social outcomes, such as symptom severity and quality of life, for example (1 systematic review).
Grade of recommendation: C
Level of evidence: 3
National Institute for Health and Care Excellence (2018),1 UK
Recommendations, supporting evidence, quality of evidence, and strength of recommendations
Recommendations:
Care coordination
“Provide people living with dementia with a single named health or social care professional who is responsible for coordinating their care.” (p. 157)1
“Named professionals should:
arrange an initial assessment of the person's needs, which should be face to face, if possible
provide information about available services and how to access them
involve the person's family members or carers (as appropriate) in support and decision-making
give special consideration to the views of people who do not have capacity to make decisions about their care, in line with the principles of the Mental Capacity Act 2005
ensure that people are aware of their rights to and the availability of local advocacy services, and if appropriate to the immediate situation an independent mental capacity advocate
develop a care and support plan” (p.157)1
“When developing care and support plans and advance care and support plans, request consent to transfer these to different care settings as needed.” (p.157)1
“Service providers should ensure that information (such as care and support plans and advance care and support plans) can be easily transferred between different care settings (for example home, inpatient, community and residential care).” (p.157)1
“Staff delivering care and support should maximise continuity and consistency of care. Ensure that relevant information is shared and recorded in the person’s care and support plan.” (p.157)1
“Service providers should design services to be accessible to as many people living with dementia as possible, including:
people who do not have a carer or whose carer cannot support them on their own
people who do not have access to affordable transport, or find transport difficult to use
people who have responsibilities (such as work, children or being a carer themselves)
people with learning disabilities, sensory impairment (such as sight or hearing loss) or physical disabilities
people who may be less likely to access health and social care services, such as people from black, Asian and minority ethnic groups.“(p.158)1
Supporting evidence was obtained from systematic reviews conducted.
“Moderate-quality evidence from up to 8 RCTs containing 2,474 people living with dementia found improvements in quality of life and rates of entry into long stay care for the person living with dementia and carer burden for people offered case management versus usual care, but could not differentiate cognition, depressive symptoms or behavioural and psychological symptoms of dementia for the person living with dementia, or carer depressive symptoms or quality of life.” (p. 151)1
Qualitative evidence was also considered. Case managers were thought to be good at identifying needs and providing support (moderate confidence). Caregivers of people with dementia expected case managers to provide information regarding dementia and available services (moderate confidence). Face-to-face and telephone contact were both considered acceptable; however, face-to-face contact was the preferred choice, as it provided more opportunity for relationship-building (moderate confidence).
Quality of evidence: the term “should” is used in the recommendation statement when the recommendation is strong, based on the certainty of the evidence.
Level of evidence: moderate
Registered Nurses’ Association of Ontario (2016),2 Canada
Recommendations, supporting evidence, quality of evidence, and strength of recommendations
Recommendation:
“Establish processes within organizations to ensure that relevant information and care planning for older adults with delirium, dementia, and depression is communicated and coordinated over the course of treatment and during care transitions.” (p. 91)2
Evidence:
For the care of people with dementia, case management can be useful for addressing both health and social changes, offering comprehensive support and enhancing delivery of multicomponent interventions (1 citation).
Potential benefits of case management include reduced institutionalization and decreased caregiver burden (1 citation).
According to 1 report, evidence on the outcomes of case conferencing was not proven, whereas according to another report there was suggestion of benefits such as improved communication and care planning, and the prevention of unnecessary hospitalization and improved care transition.
It has been suggested that case management may be more successful if the caseload is reasonable, if roles are clearly articulated within the interprofessional team, and if it is reserved for people with prominent symptoms of dementia (1 citation).
It has been suggested that case management may be more successful if there is an integration between health and social professionals (1 citation).
Level of evidence: 1a and V
The authors reported that multiple levels indicate that there were varied study designs that support the different components of the recommendation.
Strength of recommendation: not reported
Appendix 5: Overlap Between Included Systematic Reviews
Table 11: Overlap in Relevant Primary Studies Between Included Systematic Reviews
Primary study citation | Butler et al. (2020),15 US | Lee et al. (2020),16 Australia | Backhouse et al. (2017),17 UK |
Bass et al. Alzheimer research and Therapy. 2014, 69 (9) | No | No | Yes |
Bass et al. The Gerontologist. 2003, 43 (1): 73-85 | No | No | Yes |
Callahan et al. JAMA. 2006, 295 (18): 2148-57 | Yes | Yes | Yes |
Challis et al. Journal of Geriatric Psychiatr., 2002, 17(4), 315-325 | No | Yes | No |
Chien et al. J Adv Nurs. 2011, 67(4): 774-87 | Yes | No | Yes |
Chien et al. Psychiatr Serv. 2008, 59(4): 433-6 | No | No | Yes |
Chodosh et al. Journal of Aging and Health. 2015, 27(5):864-93 | Yes | No | No |
Chu et al. Am J Alzheimers Dis. Other Dement. 2000, 15(5): 284-90 | No | No | Yes |
Dias et al. PLoSOne. 2008, 4(6): e2333 | No | No | Yes |
Eloniemi-Sulkava et al. J Am Geriatr Soc. 2009, 57:2200-8 | Yes | No | Yes |
Eloniemi-Sulkava et al. J Am Geriatr Soc. 2001, 49: 1282-7 | Yes | No | Yes |
Jansen et al. Int J Nurs Stud. 2011, 48: 933-43 | No | No | Yes |
Kohler et al. Current Alzheimer Research. 2004, 11(6), 538-548 | No | Yes | No |
Lam et al. Int J Geriatr Psychiatry. 2010, 25: 395-402 | Yes | No | Yes |
Lichtwarck et al. American Journal of Geriatric Psychiatry. 2018, 26(1):25-38 | No | No | No |
Logiudice et al. International Journal of Geriatric Psychiatry, 14(8), 626-632 | No | Yes | No |
Newcomer et al. Health Care Financ Rev. 1999, 20(4): 45-65 | No | No | Yes |
Nourhashemi et al. BMJ. 2010,340. doi:10.1136/bmj.c2466 | No | Yes | No |
Possin et al. JAMA Internal Medicine. 2019, 179(12):1658-1667 | Yes | No | No |
Samus et al. Am J Geriatr Psychitr.2014, 22(4): 398-414 | No | Yes | Yes |
Thyrian et al. JAMA Psychiatry. 2017, 74(10):996-1004 | Yes | No | No |
Vichrey et al. Ann Intern Med. 2006, 145(10): 713-26 | Yes | No | Yes |
No = the study was not included in the systematic review; Yes = the study was included in the systematic review.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca