CADTH Health Technology Review
Transcatheter Mitral Edge-to-Edge Repair in Patients with Primary or Secondary Mitral Regurgitation
Rapid Review
Authors: Kwakye Peprah, Holly Gunn, Melissa Walter
Abbreviations
AKI
acute kidney injury
CCI
Charlson Comorbidity Index
IQR
interquartile range
LOS
length of stay at the hospital
MI
myocardial infarction
MR
mitral regurgitation
NIS
National Inpatient Sample
NYHA
New York Heart Association
RCT
randomized controlled trial
SD
standard deviation
SMVR
surgical mitral valve repair or replacement
SR
systematic review
STS
Society of Thoracic Surgeons
TMVR
transcatheter mitral valve repair
Key Messages
Four systematic reviews (SRs) and 6 retrospective cohort studies provided evidence for the clinical effectiveness of transcatheter mitral valve repair (TMVR) versus open heart conventional surgical mitral valve repair or replacement (SMVR) in patients with primary or secondary mitral regurgitation (MR). No relevant evidence regarding the cost-effectiveness of TMVR versus SMVR in patients with primary or secondary MR was identified; therefore, no summary can be provided.
There was evidence indicating a statistically significant difference in favour of TMVR over SMVR regarding the odds of post-procedure bleeding, need for permanent pacemaker implantation, 30-day readmission, and a shorter duration of hospitalization.
There was evidence suggesting a statistically significant difference in favour of SMVR over TMVR regarding the odds of recurrent MR, the need for reoperation, and mortality rate (i.e., during hospitalization, at 1 year, and > 3 years). Also, compared with TMVR, the likelihood of residual MR grade > 2 or freedom from MR grade ≥ 2 or ≥ 3 at 4 years was statistically significantly lower or higher, respectively, with SMVR.
Evidence regarding the comparative clinical effectiveness of TMVR versus SMVR concerning stroke, acute kidney injury (AKI), cardiogenic shock, and death during hospitalization was conflicting and inconclusive.
There was no evidence of a significant difference between the 2 interventions regarding overall mortality or mortality at 5 years, overall survival, freedom from cardiac death at 4 years, cardiac arrest, acute myocardial infarction (MI), and respiratory or vascular complications.
A major limitation of the evidence was that it derives from studies of low or unknown quality and risk of bias, Furthermore, all the findings are confounded by differences in patient selection, which reflect the approved indications for the interventions but prevent a direct comparison between the TMVR and SMVR groups.
Context and Policy Issues
The mitral valve is a valve located between the heart’s 2 left chambers where its 2 leaflets or flaps open and close in a manner that ensures a unidirectional blood flow from the atrium to the ventricle. Mitral regurgitation (MR) is a condition of the malfunctioning mitral valve that allows a backward blood leak in the heart because the mitral valve does not close properly.1 The disease is categorized into primary (also called degenerative) or secondary (also called functional), where primary MR is caused by an abnormality of 1 or more components of the valve apparatus (e.g., leaflets, papillary muscles, annulus); whereas, secondary MR is due to another heart disease such as coronary heart disease, cardiac ischemia, and heart failure.2,3
MR is a progressive disease and considered the second most frequent valve disease.1,4 The incidence of MR increases with age, with a prevalence of over 9% in patients ≥ 75 years of age.4 Common symptoms of MR include fatigue, shortness of breath, coughing, an irregular heartbeat, and swollen feet or ankles.1 The disease is frequently associated with decreased quality of life, increased difficulty performing regular daily activities,1 ventricular dysfunction, and high mortality.5 Without appropriate treatment the annual mortality rate in symptomatic patients exceeds 5% and may be up to 20% in those with severe MR.6
The goal of treatment is to decrease MR severity and improve quality of life.1 The main treatment options include guideline-directed medical therapy, surgical mitral valve repair or replacement (SMVR), as well as heart transplant and left ventricular assist devices for advanced heart failure.5 Medical management of MR has been reported to have poor outcomes, with a high hospitalization rate due to heart failure, and mortality rates of 20% at 1-year and 50% at 5-years following treatment.7 In contrast, SMVR is considered the gold-standard treatment for patients with symptomatic, severe MR and is associated with high rates of MR relief (> 95%) and mortality rates of 1% to 3%.6,8 However, approximately 50% of patients with severe MR are considered to have a prohibitive risk for surgery and are not qualified as candidates for SMVR.4,6-8 High-risk factors leading to SMVR ineligiblity include frailty, old age (> 80 years), left ventricular dysfunction with severely reduced (< 30%) left ventricular ejection fraction, and other associated comorbidities. Therapeutic alternatives for patients with severe MR who are ineligible for SMVR are limited, and most of them traditionally receive medical management although it has been proven to be less effect.7,8
In 2014 Health Canada–approved transcatheter mitral valve repair (TMVR) using MitraClip for percutaneous reduction of severe symptomatic primary MR in patients with prohibitive risk for SMVR.9 The device had received the US FDA approval for a similar indication in 2013.4,6,8 In 2019, the FDA approved MitraClip for use in patients with moderate-to-severe or severe functional MR, who have heart failure symptoms despite optimal medical therapy.
A CADTH report on TMVR published in June 2020 reviewed the clinical effectiveness of the MitraClip device for the treatment of tricuspid regurgitation but did not include any evidence about its clinical or cost-effectiveness in MR.10 The objective of the current report is to identify and summarize evidence of clinical and cost-effectiveness of TMVR versus open heart conventional SMVR in patients with primary or secondary MR.
Research Questions
What is the clinical effectiveness of transcatheter edge-to-edge repair in patients with primary or secondary mitral regurgitation?
What is the cost-effectiveness of transcatheter edge-to-edge repair in patients with primary or secondary mitral regurgitation?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, the Cochrane Database of Systematic Reviews, the international HTA database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search strategy comprised controlled vocabularies, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were transcatheter mitral edge-to-edge repair and mitral valves. No filters were applied to limit the retrieval by study type. Comments, newspaper articles, editorials, letters, and case reports were excluded. Where possible, retrieval was limited to the human population. The search was also limited to documents in English published between January 1, 2016, and June 4, 2021.
Selection Criteria and Methods
In the first screening level, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. One reviewer screened citations and selected studies. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | Adults with primary or secondary mitral regurgitation (functional or degenerative; e.g., mitral insufficiency, mitral incompetence) |
Intervention | Transcatheter mitral edge-to-edge repair (e.g., with MitraClip G4 system or PASCAL Transcatheter Valve Repair System) alone (i.e., not combined with other repair strategies such as annuloplasty) |
Comparator | Open heart conventional surgical mitral valve repair or replacement |
Outcomes | Q1: Clinical effectiveness (e.g., complications [all-stroke, major vascular complications, life-threatening and/or major bleed], hospital and intensive care unit stay or readmission, new permanent pacemaker implantation, health-related quality of life measures, mitral regurgitation grade) Q2: Cost-effectiveness (e.g., incremental cost-effectiveness ratios) |
Study designs | Health Technology Assessments, Systematic Reviews, Randomized Controlled Trials, Non-Randomized Studies, Economic Evaluations |
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1 or if they were duplicate publications or were published before 2016. Primary studies retrieved by the search were excluded if captured in 1 or more of the included SRs. SRs in which all relevant studies were included in other more recent or more comprehensive systematic reviews were excluded.
Critical Appraisal of Individual Studies
The included publications were critically appraised by 1 reviewer using the following tools as a guide: A MeaSurement Tool to Assess Systematic Reviews 2 (AMSTAR 2)11 for SRs and the Downs and Black checklist12 for non-randomized studies. Summary scores were not calculated for the included studies; instead, a narrative review of the strengths and limitations of each included study was provided.
Summary of Evidence
Quantity of Research Available
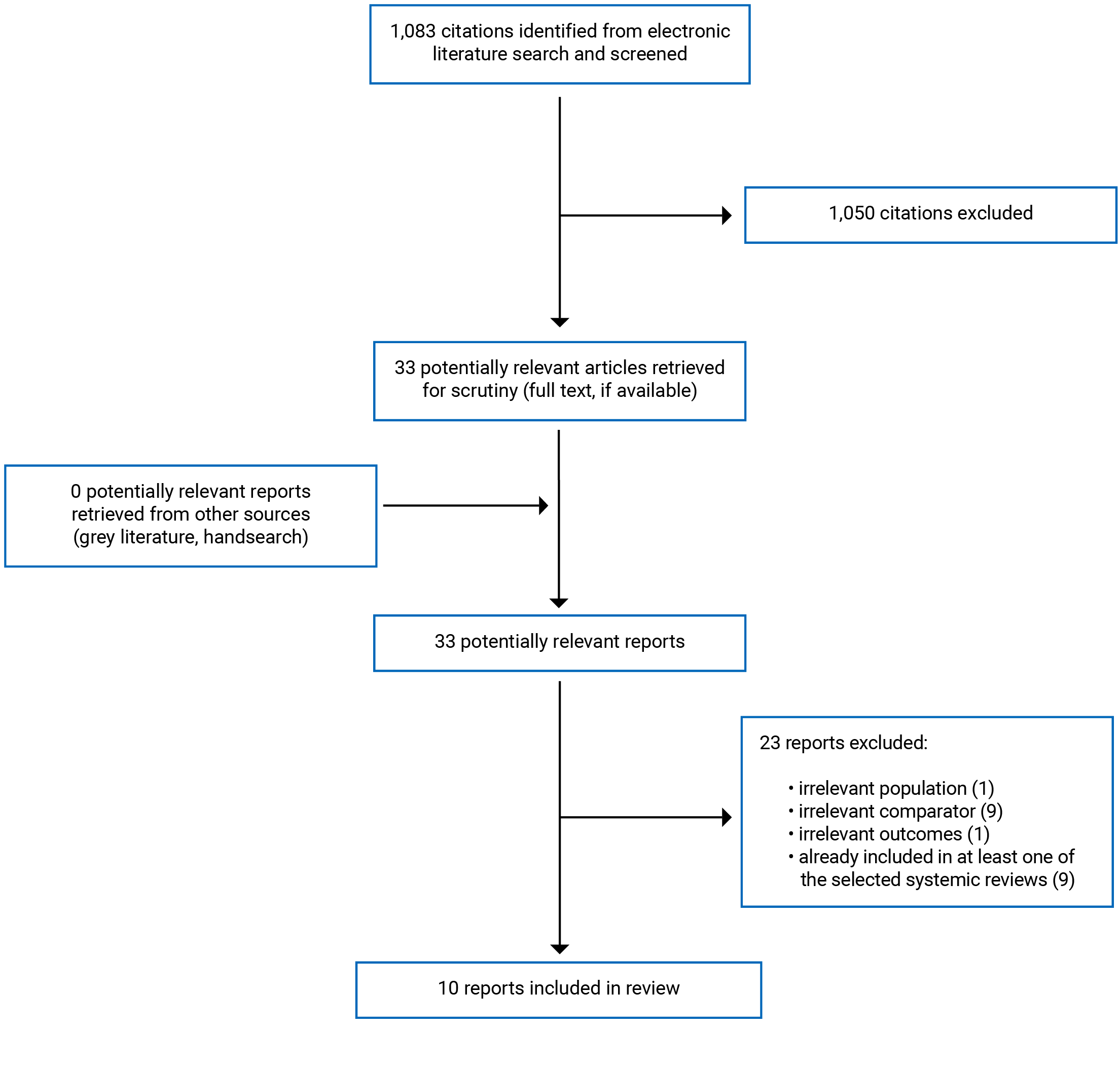
A total of 1,083 citations were identified in the literature search. After screening of titles and abstracts, 1,050 citations were excluded, and 33 potentially relevant reports from the electronic search were retrieved for full-text review. The grey literature search did not identify any additional potentially relevant studies. Of 33 articles reviewed in full-text, 23 were excluded for various reasons, while 10 that met the inclusion criteria were included in this report. These comprised 4 SRs3,4,8,13 with meta-analyses and 6 retrospective cohort studies6,14-18 relevant for clinical effectiveness of TMVR. No studies relevant for cost-effectiveness of TMVR were identified. Appendix 1 presents the PRISMA19 flow chart of the study selection.
Summary of Study Characteristics
Study Design
Systematic reviews
Three of the included SRs were published in 20213,4,8 while 1 was published in 2020.13 Each SR was based on relevant literature retrieved by systematic searches conducted in multiple databases (i.e., 2 or more) between December 2018 and June 2020. The number of primary studies included in the individual SRs ranged from 9 to 14. In 1 SR of 12 studies,13 which had a broader scope with an additional comparator of interest, 7 retrospective cohort studies were relevant to this report, while 5 were out of the scope. The 4 included SRs reported including 42 primary studies. When overlapping included studies were accounted for, the 4 SRs involved 22 unique studies published from 2011 to 2020. Thus, there was a considerable overlap of primary studies across 2 or more SRs. A table describing the overlap is available in Appendix 5. The individual studies included in the SRs were retrospective cohort studies except for 2 RCTs, 1 of which was common to 3 of the SRs.3,4,8 Additional details regarding the characteristics of the included SRs have been provided in Appendix 2.
Primary clinical studies
Four of the included retrospective cohort studies were published in 20206,14,17,18 while 1 was published in 201815 and another in 2016.16 Three of the studies14,17,18 used data from the National Inpatient Sample database of the US, and another study6 was based on data from the Nationwide Readmission Database of the US. Each of the remaining 2 studies15,16 was conducted using patient data retrieved from records of 2 separate hospitals (one per each study). One study17 analyzed data for propensity score-matched patients from the TMVR and SMVR groups. Additional details regarding the characteristics of the included retrospective cohort studies have been provided in Appendix 2.
Country of Origin
Each of the 4 included SRs had lead authors from the UK,3 the US,4 China,8 or Portugal.13 Four of the 6 retrospective cohort studies were conducted in the US6,14,17,18 while 1 was conducted in Germany15 and another 1 in Italy.16
Patient Population
Additional details regarding the populations of the studies included in this report are available in Appendix 2.
Systematic reviews
The number of patients involved in the individual SRs ranged from 930 to 4,219. One SR3 included patients who underwent an intervention for any defined MR, whereas 2 another SRs included patients with severe MR.8,13 One SR4 did not specify the severity of MR, and another SR broadly classified MR grade as ≥ 2. The mean age of patients in each SR was between 67.0.and 67.3 years for those treated with TMVR and 53.4 and 63.0 for those treated with SMVR. All 4 SRs indicated that patients treated with TMVR were older and had more comorbidities. Three SRs evaluated patients’ surgical risk and found that patients in the TMVR group had a higher surgical risk than those in the SMVR group as measured by logistic Euroscore3,4,8 or the Society of Thoracic Surgeons (STS)4 scores.
Primary clinical studies
The number of patients in the retrospective cohort studies varied from hundreds for the single-centre studies15,16 (148 to 378) to thousands for the national database-dependent studies6,14,17,18 (2,910 to 19,580). One study used data for patients with severe MR14 and another was conducted in patients with severe or moderately severe secondary MR.16 The other studies did not provide information regarding the MR severity of the patients. The authors indicated that overall, patients in the TMVR groups were older and had more comorbidities or surgical risk than those in the SMVR group, except in 1 study17 that used a propensity score matching approach to match patients from the 2 groups whose data were used in analyses. The mean age of patients in 4 of the studies6,15,16,18 was between 67.0 and 77.5 years for those treated with TMVR compared with 61.2 and 63.7 for those treated with SMVR. One study18 reported age using median instead of mean, with a median age of 79.0 years and 62.0 years for the TMVR and SMVR groups, respectively. The study17 that used a propensity score matching approach involved elderly patients at least 80 years old. The mean age in that study was 83.7 years in both the TMVR and SMVR groups.17 Two studies evaluated comorbidity using the Charlson Comorbidity Index (CCI),6,14 and another 2 studies assessed surgical risk using logistic Euroscore16 or STS.15 In contrast, the other 2 studies reported itemized varying conditions considered risks factors for poor surgical outcomes.17,18
Interventions and Comparators
The relevant study comparisons were between TMVR and SMVR. One SR3 and 1 retrospective cohort study16 stated that the MitraClip procedural techniques were standard, with the clip device positioned at the place where regurgitation is occurring. In both publications, it was indicated that more than 1 clip was implanted when necessary. According to the SR,3 6 of its primary studies that provided SMVR procedure details used annuloplasty while 1 performed surgical replacement of the valve. In the cohort study,16 the SMVR approach was a surgical edge-to-edge repair together with annuloplasty using undersized rigid or semirigid rings. Nine of the included studies did not provide procedural details about TMVR or SMVR. All 4 SRs3,4,8,13 and 5 of the retrospective cohort studies6,14-16,18 stated directly or implied that the TMVR procedure used the MitraClip devices. However, the type of TMVR device was unclear from the retrospective cohort study conducted in Germany.15
Outcomes
Reported outcomes of interest to this review included, but not limited to, post-operative mortality,3,4,8,13,17,18 survival,3,16 length of stay (LOS) in hospital,3,4,6,14-18 recurrent/residual MR,3,4,8,15 readmission rate,6 reoperation rate,3,4 need for a pacemaker,14,17 and incidence of complications (e.g., major bleeding,13,14,17 stroke,3,4,6,13-15 acute kidney injury [AKI],4,14,15,17 and acute myocardial infarction [MI]).6,14
Summary of Critical Appraisal
Overall, the quality of evidence from the included SRs3,4,8,13 and retrospective cohort studies6,14-18 was limited by factors discussed in the appraisal below. Details regarding the strengths and limitations of individual studies are available in Appendix 3.
Systematic Reviews
Two SRs4,13 reported establishing review protocols a priori and registering them on PROSPERO; whereas, for the other 2 SRs,3,8 there was no indication that a protocol was prepared before the review started. Thus, in the absence of a published protocol for the latter 2,3,8 it could not be independently ascertained if there had been any significant deviations from the protocol that could have introduced risks of bias. Each SR3,4,8,13 had a clear objective and well-defined populations, interventions, and comparators under study, as well as the outcomes of interest. All the SRs3,4,8,13 included relevant primary studies retrieved from comprehensive literature searches of multiple databases. In 1 SR,4 the database search was supplemented by a handsearch of all references of relevant reviews and eligible articles for potentially missed eligible studies.
All the SRs3,4,8,13 performed abstract and citation screening and final study selection in duplicate according to prespecified inclusion and exclusion criteria to minimized selection bias. In 2 SRs,4,13 2 reviewers independently rated the quality or risk of bias of included primary studies with appropriate tools (Newcastle Ottawa Scale and the Cochrane method), resolving disagreements by consensus. One SR4 also evaluated the certainty in evidence for each outcome using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. However, 2 other SRs3,8 neither assessed the quality or risk of bias of their primary studies nor the certainty in evidence for the outcomes they measured, making it difficult to judge the quality and reliability of their reported estimates. It is noteworthy that across the 4 included SRs,3,4,8,13 all the primary studies were non-randomized studies, except 2 RCTs of unknown quality and risk of bias, 1 of which was common to 3 SRs,3,4,8 while the other was included in another SR.13 Therefore, for each SR,3,4,8,13 the individual studies providing input for analysis had inherent high-risk of selection bias. Indeed, there were imbalances in some patient characteristics, such as age, comorbidities, and surgical risk status, that could potentially bias the reported findings against TMVR. The authors did not explain their selection of the study designs for inclusion in the SRs. However, it appeared that the lack of relevant studies limited the options for study selection.
In 2 SRs,3,8 extracted data were independently checked for accuracy by other reviewers; however, it was unclear whether data extraction in 2 other SRs4,13 was performed in duplicate or independently confirmed for accuracy. All the SRs3,4,8,13 conducted meta-analyses using random-effects models on pooled data from multiple studies. Thus, it was expected that their reported outcomes, representing a summary of findings combined across multiple studies, would offer increased precision of effect estimates than the individual primary studies would. However, in 2 SRs,8,13 the overall effect estimates for some comparisons were derived from pooled data involving an RCT and non-randomized studies, meaning that the estimates may have been biased. For the meta-analyses, each of the SRs3,4,8,13 assessed between-study heterogeneity with the appropriate statistical methods.3,4,8,13 Three SRs3,4,13 assessed publication bias; whereas, 1 SR8 did not. For the SRs that reported assessing publication bias, 2 reported no evidence of significant publication bias; ,13 whereas, 1 found significant publication bias regarding 5 years mortality but not any other outcome.4
Primary Clinical Studies
All the retrospective cohort studies6,14-18 stated objectives and described patients’ characteristics, the interventions and comparators of interest, and outcomes to be measured clearly. Four studies analyzed data from large nationwide databases (the US NIS14,17,18 and the Nationwide Readmissions Database6). Thus, the data were likely to represent the targeted population of patients treated for MR, and the settings and staff who performed the procedures and provided care were likely to be representative of care settings and treatment received by most patients who underwent TMVR or SMVR across the country. The analytical methods were appropriate, and overall, the findings were reported clearly along with estimates of the random variability and P values reported. However, none of the cohort studies adjusted for multiplicity, even though they all conducted multiple statistical tests. As a result, there may be potential inflation of the type I error rate in these studies. Two studies6,17 reported results from propensity-score-matched populations across the comparing groups to minimize bias due to inter-group differences.
A key source of uncertainty in the included primary studies was that they were all retrospective cohort studies lacking randomization that reduces the risk of selection bias. Indeed, there were imbalances in some patient characteristics, such as age and comorbidities, surgical risk status, and general medical history that could potentially bias the reported findings against TMVR. The limitation applied even for the 2 studies6,17 that used a propensity score matching approach since the method adjusts only for parameters considered in a chosen model and does not rule out significant residual or unknown confounders. For 4 of the primary studies6,14,17,18 data were limited to the period of patients’ hospitalization. Therefore, information after discharged from the hospital, including intermediate and long-term outcomes, was not available for assessment. Also, 4 studies6,14,17,18 analyzed data pooled and coded from multiple hospitals across the US, increasing the likelihood that the differences in expertise and facilities at the various settings could influence their results. In 3 studies,15,16,18 patients in the TMVR group were compared to a historical cohort of SMVR-treated patients. Thus, it was unclear whether the results had been impacted by changes in techniques and health care delivery over time, and if the findings could be different had the procedures been performed within the same period. Relatedly, with a few exceptions, most of the data used in all the included studies came from the early years (up to 2016) after MitraClip received approval for use in patients with MR in the US (2013) and Canada (2014). Therefore, it is unclear if increased practitioner experience and skills with the procedure over time may have resulted in significant improvements in current outcomes compared to the earlier period. Moreover, all the studies were based on data from outside Canada, including 2 single-site studies conducted in Germany and Italy. Thus, the generalizability of the findings in the Canadian context is unknown.
Summary of Findings
Clinical Effectiveness of Transcatheter Mitral Edge-to-Edge Repair
The findings from the included studies have been summarized below by outcome. The main study-level findings and authors’ conclusions for the individual studies are available in Appendix 4 4. The included SRs had overlapping primary studies; therefore, the pooled estimates from the separate reviews contain much of the same data. A citation matrix illustrating the degree of overlap is presented in Appendix 5.
Complications
Post-procedural complications reported by the included studies include stroke, AKI, major bleeding, pacemaker implantation, cardiac arrest, cardiogenic shock, myocardia infraction, and respiratory and vascular complications. Details for each outcome are summarized below.
Stroke
Three SRs3,4,13 and 3 retrospective cohort studies6,14,15 reported on the odds ratio for stroke (Table 2 and Table 3). While all the SRs3,4,13 and 1 cohort study15 found no statistically significant difference between TMVR and SMVR regarding this outcome, 2 cohort studies6,14 reported a statistically significantly lower odds of having a stroke with TMVR than with SMVR. Thus, the results were inconclusive due to the inconsistency across the reporting SRs and studies.
Table 2: Odds Ratio for Stroke Following TMVR Versus SMVR — Systematic Reviews Results
Stroke | Khader et al., 20213 | Oh, et al., 20214 | Barros da Silva et al., 202013 |
|---|---|---|---|
Studies pooled | 8 | 5 | 7 |
N | 3,382 | 975 | 930 |
OR (95% CI), MitraClip vs. SMVR | 1.50 (0.62 to 3.64) | 0.68 (0.34 to 1.37) | 0.49 (0.17 to 1.42) |
P value | 0.370 | 0.28 | 0.19 |
CI = confidence interval; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Table 3: Odds Ratio for Stroke Following TMVR Versus SMVR — Retrospective Cohort Studies’ Results
Stroke | Jogu et al., 202014 | Lima et al., 20206 | Körber et al., 201815 | |||
|---|---|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,402 | 7,510 | 196 | 182 |
n (%) | < 11 (NR) | 137 (2.0) | < 10 (< 0.7) | 120 (1.8) | 2 (1.0) | 4 (2.4) |
OR (95% CI), MitraClip vs, SMVR | 0.37 (0.15 to 0.92) | 0.21 (0.08 to 0.54) | NR | |||
P value | 0.02 | 0.0012 | 0.43 | |||
CI = confidence interval; NR = not reported; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Acute kidney injury
One SR4 and 4 retrospective cohort studies6,14,15,17 reported on the odds ratio for AKI. Whereas the SR4 and 2 of the cohort studies6,14 found no statistically significant difference between TMVR and SMVR regarding this outcome, 2 other cohort studies15,17 reported a statistically significantly lower odds of having an AKI with TMVR than with SMVR (P < 0.001) Table 4 and Table 5). Thus, the evidence was inconclusive due to the inconsistency across the reporting SRs and studies.
Table 4: Incidence of Acute Kidney Injury Following TMVR Versus SMVR — A Systematic Reviews Results
Acute kidney injury | Oh, et al., 20214 |
|---|---|
Studies pooled | 6 |
N | 1,156 |
OR (95% CI) MitraClip vs, SMVR | 0.82 (0.35 to 1.92) |
P value | 0.65 |
CI = confidence interval; OR = odds ratio SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair
Table 5: Incidence of Acute Kidney Injury Following TMVR Versus SMVR — Retrospective Cohort Studies’ Results
Acute kidney injury | Jogu et al., 202014 | Lima et al., 20206 | Malik et al., 202017 | Körber et al., 201815 | ||||
|---|---|---|---|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,402 | 7,510 | 1,455 | 1,455 | 196 | 182 |
n (%) | 87 (11.0) | 732 (9.0) | 198 (8.0) | 694 (10.7) | NR (10.7) | NR (25.1) | 35 (17.9) | 47 (25.8) |
OR (95 CI) MitraClip vs. SMVR | 1.26 (0.99 to 1.60) | 0.72 (0.48 to 1.07) | NR | 0.22 (0.11 to 0.44) | ||||
P value | 0.06 | 0.10 | < 0.001 | < 0.001 | ||||
CI = confidence interval; NR = not reported; OR = odds ratio SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Bleeding
One SR13 and 3 retrospective cohort studies6,14,17 reported on the odds ratio for bleeding. They were consistent in finding a statistically significantly lower (P < 0.001) bleeding with TMVR than with SMVR (Table 6 and Table 7). The results were consistent with the understanding that TMVR is a less invasive procedure compared with SMVR.
Table 6: Incidence of Major Bleeding Following TMVR Versus SMVR — A Systematic Review Results
Bleeding | Barros da Silva et al., 202013 |
|---|---|
Studies pooled | 5 |
N | 626 |
OR (95% CI), MitraClip vs, SMVR | 0.25 (0.11 to 0.56) |
P value | < 0.0007 |
CI = confidence interval; OR = odds ratio SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Table 7: Incidence of Major Bleeding Following TMVR Versus SMVR — Retrospective Cohort Studies’ Results
Bleeding | Jogu et al., 202014 | Lima et al., 20206 | Malik et al., 202017 | |||
|---|---|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,402 | 7,510 | 1,455 | 1,455 |
n (%) | 157 (21.0) | 3,170 (40.0) | 269 (24.2) | 3,298 (43.0) | NR (9.3) | NR (39.5) |
OR (95% CI), MitraClip vs, SMVR | 0.30 (0.32 to 0.47) | 0.42 (0.32 to 0.56) | NR | |||
P value | < 0.0001 | < 0.0001 | < 0.001 | |||
CI = confidence interval; NR = not reported; OR = odds ratio SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Permanent Pacemaker implantation
Two retrospective cohort studies14,17 reported on implantation of permanent pacemakers. They were consistent in finding that the odds for needing a permanent pacemakers implantation was statistically significantly lower with TMVR than with SMVR (Table 8).
Table 8: Incidence of PPI Following TMVR Versus SMVR — Retrospective Cohort Studies’ Results
Pacemaker implantation | Jogu et al., 202014 | Malik et al., 202017 | ||
|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,455 | 1,455 |
n (%) | < 11 (NR) | 302 (4.0) | (NR) 0.7 | (NR) 5.8 |
OR (95% CI) | 0.23 (0.11 to 0.50)] | NR | ||
P value | < 0.0001 | < 0.001 | ||
CI = confidence interval; NR = not reported; OR = odds ratio; PPI = proton pump inhibitor; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Cardiac arrest
One retrospective cohort study14 reporting on the odds ratio for cardiac arrest found no statistically significant difference in the odds of having a cardiac arrest in patients treated with TMVR compared with those who had SMVR (Table 9).
Table 9: Incidence of Cardiac Arrest Following TMVR Versus SMVR — Retrospective Cohort Study Results
Cardiac arrest | Jogu et al., 202014 | |
|---|---|---|
TMVR | SMVR | |
N | 766 | 7,950 |
n (%) | < 11 (NR) | 112 (1.4) |
OR (95% CI) | 0.55 (0.24 to 1.26) | |
P value | 0.19 | |
CI = confidence interval; NR = not reported; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Cardiogenic shock
Two retrospective cohort studies6,17 reporting on the odds ratio for cardiogenic shock found a statistically significantly lower odds of having a cardiogenic shock with TMVR than with SMVR. However, results from another retrospective cohort study14 did not show a statistically significant difference in this outcome between TMVR and SMVR Table 10).
Table 10: Incidence of Cardiogenic Shock Following TMVR Versus SMVR — Retrospective Cohort Studies’ Results
Cardiogenic shock | Jogu et al., 202014 | Lima et al., 20206 | Malik et al., 202017 | |||
|---|---|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,402 | 7,510 | 1,455 | 1,455 |
n (%) | 17 (2.0) | 249 (3.0) | 33 (1.6) | 231 (3.3) | NR (2.1) | NR (7.9) |
OR (95% CI) | 0.70 (0.43 to 1.15) | 0.47 (0.27 to 0.82) | NR | |||
P value | 0.19 | 0.01 | < 0.001 | |||
CI = confidence interval; NR = not reported; OR = odds ratio SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Acute myocardial infarction
Two retrospective cohort study6,14 reported on the odds ratio for MI. They found no statistically significant difference between TMVR and SMVR regarding this outcome (Table 11).
Table 11: Incidence of Acute MI Following TMVR Versus SMVR — Retrospective Cohort Study Results
Acute MI | Jogu et al., 202014 | Lima et al., 20206 | ||
|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,402 | 7,510 |
n (%) | < 11 (NR) | 32 (0.004) | 17 (1.5) | 76 (1.2) |
OR (95% CI) | 1.30 (0.46 to 0.3.68) | 1.27 (0.36 to 4.41) | ||
P value | 0.55 | 0.71 | ||
CI = confidence interval; MI = myocardial infarction; NR = not reported; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Respiratory and vascular complications
One retrospective cohort study17 reporting on the percentage of patients who had respiratory or vascular complications found no statistically significant difference between TMVR and SMVR regarding these outcomes (Table 12).
Table 12: Incidence of Respiratory and Vascular Complications Following TMVR Versus SMVR — Retrospective Cohort Studies’ Results
Respiratory and vascular complications | Malik et al., 202017 | |||
|---|---|---|---|---|
Respiratory | Vascular | |||
TMVR | SMVR | TMVR | SMVR | |
N | 1,455 | 1,455 | 1,455 | 1,455 |
% | 1.7 | 4.1 | < 0.3 | 2.1 |
P value | 0.0807 | 0.0551 | ||
SMVR = surgical mitral valve repair. TMVR = transcatheter mitral valve repair.
Length of hospital stay
Two SRs3,4 and 6 retrospective cohort studies6,14-18 reported on the duration of hospital stay after TMVR versus SMVR. The units of measurement were days with the results presented as standardized mean difference,3 mean difference,4 median with interquartile rage (IQR),14-16 and mean with standard deviation (SD).6,17 All the SRs3,4 and cohort studies6,14-18 were consistent in showing that the LOS was statistically significantly shorter with TMVR than SMVR (Table 13 and Table 14).
Table 13: Post-Procedure Length of Hospital Stay Following TMVR Versus SMVR – Systematic Reviews Results
Length of hospital stay | Khader et al., 20213 | Oh, et al., 20214 |
|---|---|---|
Studies pooled | 8 | 7 |
N | 3,382 | 1,177 |
SMD or MD (95% CI) | 0.882 (0.77 to 0.99)a | –3.86 days (–4.73 to –2.99)b |
P value | < 0.001 | < 0.01 |
CI = confidence interval, MD = mean difference; SMD = standard mean difference; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
aReporting SMD (95% CI).
bReporting MD (95% CI).
Table 14: Post-Procedure Length of Hospital Stay Following TMVR Versus SMVR —Retrospective Cohort Studies Results
Length of hospital stay | Jogu et al., 202014 | Lima et al., 20206 | Malik et al., 202017 | Ullah et al., 202018 | Körber et al., 201815 | De Bonis et al., 201616 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,402 | 7,510 | 1,455 | 1,455 | 1,120 | 18,460 | 196 | 182 | 85 | 58 |
Mean (SD) | NR | 5.4 (0.32) | 7.9 (0.13) | 4.7 (6.1) | 10.4 (7.6) | 11.6 (10.0) | 14.0 (12.7) | NR | NR | |||
Median (IQR) | 2 (1 to 5) | 6 (4 to 7) | NR | NR | NR | 6 (4 to 8) | 10 (8 to 12) | 4.8 (3.8 to 7.0) | 10.0 (8.0 to 13.0) | |||
CIE (95% CI) | −3.26 (0.99 to 1.60) | NR | NR | NR | NR | NR | ||||||
P value | < 0.0001 | < 0.0001 | < 0.01 | < 0.0001 | < 0.001 | < 0.0001 | ||||||
CI = confidence interval; CIE = change in estimate; IQR = interquartile range; NR = not reported; SD = standard deviation; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Mortality
Three SRs3,4,8 and 3 retrospective cohort studies14,17,18 reported on the odds ratio for mortality after TMVR versus SMVR. While all the SRs3,4,8 and 2 cohort studies14,18 found no statistically significant difference in mortality during hospitalization between TMVR and SMVR, 1 cohort study17 reported a statistically significantly lower odds of dying for this outcome with TMVR than with SMVR (Table 15 and Table 16). Thus, the results concerning mortality during post-procedure hospitalization were inconclusive due to the inconsistency across the reporting SRs and cohort studies. Two SRs4,8 reporting 1 year outcomes were consistent in showing that the odds of dying were statistically significantly higher with TMVR than with SMVR. One of the SR8 also found that that the odds of dying were statistically significantly higher with TMVR than with SMVR at > 3 years (Table 15). Another SR4 found no statistically significant difference in the odds for overall mortality or mortality at 5 years between TMVR and SMVR. It should be noted that the SR4 reported a significant publication bias regarding the 5-year mortality, but not for other outcomes.
Table 15: Odds Ratio for Mortality Following TMVR Versus SMVR — Systematic Reviews Results
Mortality | Khader et al., 20213 | Oh, et al., 20214 | Yuan et al., 20218 |
|---|---|---|---|
Operative Mortality (i.e., death in-hospital and up to 30 days post-surgery) | |||
Studies pooled | 8 | 8 | 11 |
N | 3364 | 1435 | 2144 |
OR (95% CI)- MitraClip vs, SMVR | 1.63 (0.63 to 4.23) | 1.04 (0.58 to 1.88) | 0.88 (0.53 to 1.47) |
P value | 0.139 | 0.88 | 0.64 |
Mortality at 1 year | |||
Studies pooled | NR | 6 | 11 |
N | 1057 | 2984 | |
OR (95% CI) MitraClip vs, SMVR | 1.48 (1.03 to 2.16) | 1.79 (1.4 to 2.28) | |
P value | 0.04 | < 0.00001 | |
Mortality at > 3 years | |||
Studies pooled | NR | 7 | |
N | 1749 | ||
OR (95% CI) MitraClip vs. SMVR | 2.26 (1.04 to 4.92) | ||
P value | < 0.00001 | ||
Mortality at 5 years | |||
Studies pooled | NR | 3 | NR |
N | 756 | ||
OR (95% CI)- MitraClip vs, SMVR | 1.44 (0.56 to 3.70) | ||
P value | 0.44 | ||
Overall mortality | |||
Studies pooled | NR | 5 | NR |
N | NR | ||
OR (95% CI) MitraClip vs. SMVR | 2.06 a (0.98 to 4.28) | ||
P value | 0.06 | ||
CI = confidence interval; NR = not reported; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Table 16: Odds Ratio for Mortality Following TMVR Versus SMVR — Retrospective Cohort Studies Results
Mortality | Jogu et al., 202014 | Malik et al., 202017 | Ullah et al., 202018 | |||
|---|---|---|---|---|---|---|
TMVR | SMVR | TMVR | SMVR | TMVR | SMVR | |
N | 766 | 7,950 | 1,455 | 1,455 | 1,120 | 18,460 |
n (%) | 13 (2.0) | 88 (1.0) | NR (0.7) | NR (3.1) | 150 (13.3) | 985 (5.3) |
OR (95 CI) | 1.50 (0.84 to 2.70) | NR | 2.0 (0.93 to 1.68) | |||
P value | 0.17 | 0.0262 | 0.15 | |||
CI = confidence interval; NR = not reported; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Survival
One SR3 and 1 retrospective cohort study16 reported survival outcomes for TMVR versus SMVR. Of note, the SR3 reported on the odds ratio for survival both at midterm (1 to 2 years) and long-term (4 to 5 years) outcomes; whereas, the cohort study16 reported on the overall survival and freedom from cardiac death at 4 years, analyzed by Kaplan–Meier method with differences among groups evaluated with the log-rank test. However, mean percentage (SD) results were reported for the 2 outcomes in the cohort study.16 Both the SR3 and the cohort study16 were consistent in showing no statistically significant difference between the 2 procedures regarding all the assessed outcomes (Table 17 and Table 18). In the retrospective cohort study,16 the duration of follow-up was significantly shorter for the TMVR group compared with the SMVR group (median 2.5 years versus 7.2 years; P = 0.0001) (Table 25). Thus, it is unknown if the difference between the 2 groups will be reproducible in follow-ups of equal duration.
Table 17: Odds Ratio for Survival Following TMVR Versus SMVR — Systematic Review Results
Survival | Khader et al., 20213 | |
|---|---|---|
Midterm (1 to 2 years) | Long-term (4 to 5 years) | |
Studies pooled | 4 | 4 |
N | NR | 1216 |
OR (95% CI) MitraClip vs, SMVR | 1.69 (0.59 to 4.84) | 0.70 (0.35 to 1.41) |
P value | 0.332 | 0.323 |
CI = confidence interval; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Table 18: Survival Following TMVR Versus SMVR — Retrospective Cohort Study Results
Survival | De Bonis et al., 201616 | |||
|---|---|---|---|---|
Overall survival | Freedom from cardiac death at 4 years | |||
TMVR | SMVR | TMVR | SMVR | |
N | 85 | 58 | 85 | 58 |
Mean % (SD) | 77 (5.6) | 74 (5.1) | 82 (5.2) | 84 (5.6) |
P value | 0.2 | 0.5 | ||
SD = standard deviation; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Recurrent MR
Three SRs3,4,8 reported on the on the odds ratio for recurrent MR following TMVR versus SMVR, with post-procedural findings at the time of discharge from hospital,4 and at 30 days,8 1 year,8 midterm (i.e., 1 to 2 years),3 greater than 3 years,8 and 5 years.4 All the SRs3,4,8 were consistent in showing that the odds of recurrent MR were statistically significantly higher following treatment with TMVR compared with SMVR. The OR (95% CI) at the various assessment points were between 2.46 (1.54 to 3.94) and 9.51 (5.38 to 16.83) (Table 19). Also, 1 retrospective cohort study15 reporting the mean (SD) for post-operative residual MR greater than grade 2 found that the outcome was statistically significantly higher with TMVR than with SMVR (Table 20). Similarly, another retrospective cohort study16 reported that the mean percentage (SD) of patients with freedom from MR grade greater than or equal to 2 or greater than or equal to 3 at 4 years was statistically significantly lower in the TMVR group than in the SMVR group (Table 21).
Table 19: Odds Ratio for Recurrent MR Following TMVR Versus SMVR — Systematic Reviews Results
Recurrent MR | Khader et al., 20213 | Oh, et al., 20214 | Yuan et al., 20218 | |||
|---|---|---|---|---|---|---|
MR ≥ grade 3 | ||||||
Point of assessment | Midterm (1 to 2 years) | At discharge | At 5 years | 30 days | 1 year | > 3 years |
Studies pooled | 5 | 6 | 3 | 10 | 6 | 4 |
N | 520 | 685 | 684 | 1,193 | 726 | 756 |
OR (95% CI) MitraClip vs. SMVR | 3.45 (2.17 to 5.26) | 2.81 (1.39 to 5.69) | 2.46 (1.54 to 3.94) | 9.51 (5.38 to 16.83) | 3.14 (1.20 to 8.25) | 8.47 (4.76 to 15.10) |
P value | < 0.001 | < 0.01 | < 0.01 | < 0.00001 | 0.02 | < 0.00001 |
CI = confidence interval; MR = mitral regurgitation; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Table 20: Residual MR Following TMVR Versus SMVR — Retrospective Cohort Study Results
Post-operative MR > grade 2 | Körber et al., 201815 | |
|---|---|---|
TMVR | SMVR | |
N | 196 | 182 |
Mean (SD) | 14 (7.3) | 4 (2.4) |
P value | 0.033 | |
SD = standard deviation; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Table 21: Freedom From MR Grade Greater Than or Equal to 2 or Greater Than or Equal to 3 at 4 years Following TMVR Versus SMVR — Retrospective Cohort Studies Results
Freedom from MR | De Bonis et al., 2016 | |||
|---|---|---|---|---|
Freedom from MR grade ≥3 at 4 years | Freedom from MR grade ≥2 at 4 years | |||
TMVR | SMVR | TMVR | SMVR | |
N | 85 | 58 | 85 | 58 |
Mean % (SD) | 75 (7.6) | 94 (3.3) | 37 (7.2) | 82 (5.2) |
P value | 0.04 | 0.0001 | ||
MR = mitral regurgitation; SD = standard deviation; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Reoperation
Two SRs3,4 reported on the odds ratio for reoperation after TMVR versus SMVR. All the studies were consistent in showing that the odds of having a reoperation was statistically significantly higher with TMVR compared with the SMVR (Table 22).
Table 22: Odds Ratio for Reoperation Following TMVR Versus SMVR — Systematic Reviews Results
Reoperation | Khader et al., 20213 | Oh, et al., 20214 |
|---|---|---|
Studies pooled | 4 | 4 |
N | 559 | 530 |
OR (95% CI) MitraClip vs. SMVR | 2.551 (1.22 to 5.32) | 5.28 (3.43 to 8.11) |
P value | 0.012 | < 0.01 |
CI = confidence interval; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Readmission rate
One retrospective cohort study6 reported on the odds ratio for 30-day readmission rates between patients who underwent TMVR versus those who had SMVR. The unadjusted results showed no statistically significant difference in the odds of readmission between the 2 procedures. However, after a propensity score matching adjustment, TMVR was associated with statistically significantly lower odds for 30-day readmission compared with SMVR (Table 23). There were no data provided to independently assess the extent to which the adjustment reduced the imbalance between the TMVR and SMVR groups.
Table 23: Odds Ratio for Readmissions Following TMVR Versus SMVR — Retrospective Cohort Study Results
30-day readmission rate | Lima et al., 20206 | |
|---|---|---|
TMVR | SMVR | |
N | 1,402 | 7,510 |
% | 10.7 | 11.7 |
Unadjusted OR (95 CI) | 1.11 (0.89 to 1.39) | |
P value | 0.35 | |
Propensity score adjusted OR (95% CI) | 0.70 (0.51 to 0.95) | |
P value | 0.02 | |
CI = confidence interval; OR = odds ratio; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Cost-Effectiveness of Transcatheter Mitral Edge-to-Edge Repair
No relevant evidence regarding the cost-effectiveness of TMVR versus SMVR for the treatment of primary or secondary MR was identified; therefore, no summary can be provided.
Limitations
A fundamental limitation involves limited information about the included studies and differences in study characteristics. For example, most of the included studies provided insufficient information about the type (i.e., degenerative or functional MR) and severity of MR before the interventions were given. However, 1 study16 restricted enrolment to patients who had severe or moderately severe functional MR, while another study17 recruited only elderly patients who were 80 years or older (Table 24 and Table 29 for detailed patient characteristics), which could inform patient selection criteria. Nevertheless, the results from those studies may not be generalizable to patients who are different from the study participants. Five of the 6 included studies did not report a specific follow-up period. Also, there were inadequate details about how the TMVR and SMVR procedures were applied in the various studies, with the available information suggesting a lack of standardization for the interventions. For instance, while some patients in the TMVR groups were implanted with a single clip, others were treated with multiple clips at the discretion of the attending surgeon. Also, the reported SMVR methods differed between the studies that provided information about the procedure. Taken together with other limitations discussed in the critical appraisal section, the generalizability of the findings is unclear, and it is difficult to draw conclusions from the included studies with any certainty. Some retrospective cohort studies identified patients with relevant data using the International Classification of Disease, ninth revision, Clinical Modifications (ICD-9-CM); whereas others used the 10th revision (ICD-10-CM). Thus, there was a potential for misalignment in the definition of patient’s conditions. However, the impact of this on the reported outcomes was unknown. None of the included studies reported on health-related quality of life outcomes, an essential critical end point, especially for patients with chronic diseases. No relevant evidence on cost-effectiveness was identified.
Conclusions and Implications for Decision- or Policy-Making
Four SRs3,4,8,13 and 6 retrospective cohort studies6,14-18 provided evidence for the clinical effectiveness of TMVR versus SMVR in patients with MR. Most of the clinical effectiveness outcomes of interest fell under the category of complications. They included the incidence of stroke,3,4,6,13-15 AKI,4,6,14,15,17 major bleeding,6,13,14,17 cardiac arrest,14 cardiogenic shock,6,14,17 acute MI,6,14 as well as respiratory17 and vascular17 complications. The other outcomes were implantation of permanent pacemakers,14,17 post-procedure LOS3,4,6,14-18 mortality,3,4,8,14,17,18 survival,3,16 recurrent/residual MR,3,4,8,15 freedom from grade greater than or equal to 2 or greater than or equal to 3 MR at 4 years,16 reoperation rate3,4 and readmission rate.6 No relevant evidence regarding the cost-effectiveness of TMVR versus SMVR for the treatment of primary or secondary MR was identified; therefore, no summary can be provided.
Evidence from 1 SR13 and 3 retrospective cohort studies6,14,17 indicated that TMVR was associated with less frequent post-procedural bleeding than SMVR. Also, there was consistent evidence from 2 SRs and 6 retrospective cohort studies showing a shorter hospitalization duration following TMVR than after SMVR. Furthermore, in an analysis that applied a propensity score matching adjustment, 1 retrospective cohort study6 found that TMVR was associated with a statistically significantly lower odds of 30-day readmission compared with SMVR. However, there were no data provided to independently assess the extent to which the adjustment reduced the imbalance between the TMVR and SMVR groups. These findings may be explained because SMVR is a more invasive procedure likely to result in more bleeding and a longer recovery time than TMVR. Evidence from 2 retrospective cohort studies suggested that TMVR was associated with a significantly less frequent need for permanent pacemaker implantation compared with SMVR. This finding is in agreement with reports in the literature showing that the rate of new-onset atrial fibrillation was 3.8% after TMVR20 and 18.0% after SMVR,21 suggesting that patients who underwent SMVR are more likely to require permanent pacemaker implantation.
However, evidence from 3 SRs3,4,8 suggested a higher incidence of recurrent MR following TMVR compared with SMVR at the time of discharge from hospital,4 and at 30 days,8 1 year,8 midterm (i.e., 1 to 2 years),3 greater than 3 years,8 and 5 years4 after the procedures. Similarly, 1 retrospective cohort study15 found that postoperative residual MR greater than grade 2 occurred significantly more frequently after TMVR than SMVR. Another retrospective cohort study16 reported that the percentage of patients with freedom from MR grade greater than or equal to 2 or greater than or equal to 3 at 4 years was statistically significantly lower in the TMVR group than in the SMVR group. Two SRs3,4 found evidence of a significantly higher odds of reoperation after TMVR than after SMVR. However, the death rate at 1 year was statistically significantly higher with TMVR than SMVR in in 2 SRs4,8 including 1 SR8 with a similarly higher mortality rate for TMVR than SMVR at greater than 3 years. One SR4 found no statistically significant difference in overall mortality or mortality at 5 years between the 2 procedures. Also, there was no evidence of a statistically significant difference between TMVR and SMVR regarding overall survival,3,16 freedom from cardiac death at 4 years,16 cardiac arrests,14 acute MI6,14 and respiratory17 and vascular17 complications.
The evidence from comparisons between TMVR and SMVR regarding the incidence of stroke, AKI, cardiogenic shock, and death during hospitalization were inconclusive due to conflicting findings from the reporting SRs and studies. Three SRs3,4,13 and 1 retrospective cohort study found no difference between TMVR and SMVR in the incidence of stroke. In contrast, 2 retrospective cohort studies reported a significantly lower incidence of stroke with TMVR. For AKI, evidence from 1 SR4 and 2 retrospective cohort studies6,14 showed no significant difference between the 2 procedures, whereas 2 retrospective cohort studies15,17 reported a significantly lower incidence of stroke with TMVR. Similarly, evidence from 3 SRs3,4,8 and 2 retrospective studies suggested no significant difference in mortality rates during hospitalization after TMVR compared with SMVR. In contrast, the results from 1 retrospective cohort study indicated a statistically significantly lower percentage of in-hospital deaths among patients who underwent TMVR compared with those who had SMVR.
A key source of uncertainty in the SRs3,4,8,13 is the use of mostly retrospective cohort studies and 2 RCTs of unknown quality and risk of bias as primary studies. This implies that all the individual studies providing input for analyses in the SRs3,4,8,13 had an inherently high or unknown risk of bias, making it difficult to draw conclusions. Other limitations in the SRs include failure to evaluate the quality and risk of bias of their primary studies,3,8 and potentially biased pooled estimates due to meta-analyses combining data from an RCT and retrospective cohort studies.8,13 For the included primary studies, the major source of uncertainty was that they were all retrospective cohort studies lacking randomization that reduces the risk of selection bias. Thus, imbalances in baseline patient characteristics, such as surgical risk status and general medical history, potentially biased their reported findings against TMVR. Four of the retrospective cohort studies6,14,17,18 used data limited to patients’ hospitalization period. Therefore, information after hospital discharge, including intermediate and long-term outcomes, was not available for assessment. Also, 4 studies6,14,17,18 analyzed data pooled and coded from multiple hospitals across the US. Although that provides a more representative sample of practice in real life, it also increases the likelihood that the differences in expertise and facilities at the various settings could impact their results. Moreover, patients in the TMVR group in 3 studies15,16,18 were compared to a cohort of SMVR-treated patients from a different historical period, creating uncertainty about whether their findings would be replicated if the cohorts underwent the procedures performed within the same period.
Furthermore, all the included studies3,4,6,8,13-18 used data from outside Canada, and the generalizability of the findings in the Canadian context is unknown.
Future studies using more rigorous designs such as RCTs or other types of studies employing appropriate statistical methods to minimize confounding may provide a more definitive answer concerning the clinical effectiveness of TMVR versus SMVR in the treatment of patients with primary or secondary MR. Economic evaluations are also needed to provide cost-effectiveness evidence.
References
1.University of Ottawa Heart Institute. MitraClip. 2021; https://www.ottawaheart.ca/test-procedure/mitraclip. Accessed 2021 Jul 5.
2.Gaasch WH. Management of chronic primary mitral regurgitation. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2021: www.uptodate.com. Accessed 2021 Jul 5.
3.Khader AA, Allaf M, Lu OW, et al. Does the clinical effectiveness of Mitraclip compare with surgical repair for mitral regurgitation? J Card Surg. 2021;36(3):1103-1119. PubMed
4.Oh NA, Kampaktsis PN, Gallo M, et al. An updated meta-analysis of MitraClip versus surgery for mitral regurgitation. Ann Cardiothorac Surg. 2021;10(1):1-14. PubMed
5.Ali M, Shreenivas SS, Pratt DN, Lynch DR, Kereiakes DJ. Percutaneous interventions for secondary mitral regurgitation. Circ Cardiovasc Interv. 2020;13(8):e008998. PubMed
6.Lima FV, Kolte D, Rofeberg V, et al. Thirty-day readmissions after transcatheter versus surgical mitral valve repair in high-risk patients with mitral regurgitation: Analysis of the 2014-2015 Nationwide readmissions databases. Catheter Cardiovasc Interv. 2020;96(3):664-674. PubMed
7.Yandrapalli S, Biswas M, Kaplan J. Mitral valve minimally invasive surgical treatment. StatPearls. 2021.
8.Yuan H, Wei T, Wu Z, et al. Comparison of transcatheter mitral-valve repair and surgical mitral-valve repair in elderly patients with mitral regurgitation. Heart Surg Forum. 2021;24(1):E108-E115. PubMed
9.Health Canada. Summary Basis of Decision (SBD) for MitraClip Clip Delivery System. 2020; https://hpr-rps.hres.ca/reg-content/summary-basis-decision-medical-device-detailThree.php?linkID=SBD00402. Accessed 2021 Jul 6.
10.Li KX, Wright M-D. Transcatheter mitral valve repair device for the treatment of tricuspid or tricuspid and mitral regurgitation: a review of clinical effectiveness and cost-effectiveness. (CADTH Rapid response report: summary with critical appraisal). Ottawa (ON): CADTH; 2020: https://cadth.ca/sites/default/files/pdf/htis/2020/RC1282%20MitraClip%20Tricuspid%20Regurgitation%20Final.pdf. Accessed 2021 Jul 5.
11.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
12.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377-384. PubMed
13.Barros da Silva P, Sousa JP, Oliveiros B, et al. Stroke after transcatheter edge-to-edge mitral valve repair: a systematic review and meta-analysis. EuroIntervention. 2020;15(16):1401-1408. PubMed
14.Jogu HR, Arora S, Strassle PD, et al. Impact of age and comorbidities on the effect of transcatheter versus surgical mitral valve repair on inpatient outcomes. Catheter Cardiovasc Interv. 2020;95(6):1195-1201. PubMed
15.Korber MI, Scherner M, Kuhr K, et al. Acute kidney injury following percutaneous edge-to-edge vs. minimally invasive surgical mitral valve repair: incidence, predictors and prognostic value. EuroIntervention. 2018;13(14):1645-1651. PubMed
16.De Bonis M, Lapenna E, Buzzatti N, et al. Optimal results immediately after MitraClip therapy or surgical edge-to-edge repair for functional mitral regurgitation: are they really stable at 4 years? Eur J Cardiothorac Surg. 2016;50(3):488-494. PubMed
17.Malik AH, Zaid S, Yandrapalli S, et al. Trends and outcomes with transcatheter versus surgical mitral valve repair in patients >=80 years of age. Am J Cardiol. 2020;125(7):1083-1087. PubMed
18.Ullah W, Sattar Y, Mukhtar M, et al. Outcomes of open mitral valve replacement versus transcatheter mitral valve repair; insight from the National Inpatient Sample Database. Int J Cardiol Heart Vasc. 2020;28:100540. PubMed
19.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
20.Arora S, Vemulapalli S, Stebbins A, et al. The prevalence and impact of atrial fibrillation on 1-year outcomes in patients undergoing transcatheter mitral valve repair: results from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. JACC Cardiovasc Interv. 2019;12(6):569-578. PubMed
21.Kang MK, Joung B, Shim CY, et al. Post-operative left atrial volume index is a predictor of the occurrence of permanent atrial fibrillation after mitral valve surgery in patients who undergo mitral valve surgery. Cardiovasc Ultrasound. 2018;16(1):5. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Note that this appendix has not been copy-edited.
Table 24: Characteristics of Included Systematic Reviews and Meta-Analyses
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Khader et al., 20213 UK Funding: None The authors declared that they had no conflict of interest | A systematic review and meta-analysis of 12 primary studies (one RCT and 11 retrospective cohort studies) published between 2012 and 2019 | A total of 4,219 patients undergoing intervention for any type of defined MR (TMVR: 1210; SMVR: 3009). Age, years – Mean (SD) TMVR: 67.0 (12.7) to 82.9 (3.5) SMVR: 61.7 (13.7) to 78.8 (13.13). Sex – Male, % TMVR: 41.7% to 83.6% SMVR: 41.0% to 80.4% Risk assessment Logistic Euroscore I – Mean (SD) TMVR: 12.3 (3.7) to 33.7 (18.7) SMVR: 3.9 (3.7) to 14.2 (8.9) | TMVR with MitraClip vs. SMVR | Outcomes:
Follow-up: up to 5 years |
Oh et al., 2021,4 US Funding: None The authors declared that they had no conflict of interest | A systematic review and meta-analysis of 9 primary studies (one RCT and 8 retrospective cohort studies) published between 2013 and 2019 | A total of 1,873 patients undergoing intervention for MR grade ≥ 2 (TMVR: 997; SMVR: 876). Age, years – Mean (SD) TMVR: 67.0 (12.7) to 81.2 (6.3) SMVR: 62.6 (13.7) to 80.1 (6.9) Sex – Male, % TMVR: 42.0% to 83% SMVR: 44% to 77% Risk assessment Logistic Euroscore I – Range TMVR: 12.3 to 33.7 SMVR: 3.9 to 37.1 STS Risk score – Range TMVR: 2.48 to 8.5 SMVR: 0.7 to 5.3 | TMVR with MitraClip vs. SMVR | Outcomes:
Follow-up: up to 5 years (mean: 4.8 years) |
Yuan et al., 20218 China Funding: None reported. | A systematic review and meta-analysis of 14 primary studies (one RCTs and 13 retrospective cohort studies) published between 2012 and 2020. | A total of 3,355 patients with severe MR undergoing intervention treat MR (TMVR: 1,672; SMVR: 1,683). Age, years – Mean (SD) TMVR: 67.0 (12.7) to 84.5 (3.2) SMVR: 63.0 (12.0) to 82.9 (3.5) Sex – NR Risk assessment Logistic Euroscore I – Mean (SD) TMVR: 8.03 (7.23) to 33.5 (20.0). SMVR: 3.9 (3.7) to 25.0 (22.0). | TMVR with MitraClip vs. SMVR | Outcomes:
Follow-up: up to 5 years (range: 6 months to 5 years) |
Barros da Silva et al., 202013 Portugal Funding: None The authors declared that they had no conflict of interest | A systematic review and meta-analysis of 12 primary studies (3 RCTs and 9 retrospective cohort studies) published between 2011 and 2019; MitraClip vs. SMVR was compared in 7 retrospective studies; therefore 7 of the 12 studies were relevant to this report. | A total of 930 patients (from the 7 relevant studies) who underwent intervention to resolve severe MR (TMVR: 477; SMVR: 453). Age, years – Mean (SD) TMVR: 67.3 (12.8) to 82.2 (1.65) SMVR: 53.4 (13.0) to 81.7 (1.35) Sex – NR Risk assessment – NR | TMVR with MitraClip vs. SMVR or OMT alone; TMVR with MitraClip vs. SMVR was relevant to this review | Outcomes:
Follow-up: up to 5 years (range: 4 to 60 months) |
AF = atrial fibrillation; HF = heart failure; LOS = length of stay at the hospital; MR = mitral regurgitation; NR = not reported; NA = not applicable; NMA = network meta-analysis; NYHA = New York Heart Association; OMT = optimal medical treatment; RCT = randomized controlled trial; SD = standard deviation; SMVR = surgical mitral valve repair, STS = Society of Thoracic Surgeons: TMVR = transcatheter mitral valve repair.
Table 25: Characteristics of Included Primary Clinical Studies
Study citation, country, funding source | Study design | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Jogu et al., 202014 US Funding: Unclear. At least 1 author reported receiving support from external sources. However, it was unknown if the support was for this study. | A retrospective cohort study based on the US NIS database | A total of 8,716 patients who underwent elective procedure to correct severe MR (TMVR 766 [9%] and SMVR 7,950 [91%]) Age, years – Median (IQR) TMVR: 79.0 (69 to 85) SMVR: 62.0 (52 to 71) Sex – Male, % TMVR: 55.0 SMVR: 60.0 Risk assessment CCI – Median (IQR) TMVR: 79 (69 to 85) SMVR: 1 (0 to 1) Inclusion Criteria: Patients hospitalized after elective TMVR or elective SMVR between January 1, 2012, and September 30, 2015 Exclusion criteria: Data of patients were excluded if they underwent
| TMVR with MitraClip vs. SMVR | Outcomes:
Follow-up: Not clearly reported. |
Lima et al., 20206 US Funding: None The authors declared that they had no conflict of interest | A retrospective cohort using the 2014-2015 NRD | A total of 8,912 patients who underwent TMVR (n = 1,402) or SMVR (n = 7,510) were included in the analysis. Age, years – mean (SD) TMVR: 76.7 (0.6) SMVR: 61.2 (0.2) Sex – Male, % TMVR: 52.3 SMVR: 62.0 Risk Assessment CCI – Mean (SD) TMVR: 2.84 (0.07) SMVR: 1.25 (02) P value < 0.0001 Inclusion Criteria: Patients with a primary diagnosis of MR who were
| TMVR (with unnamed device) vs. SMVR | Outcomes:
Follow-up: The participants were followed for 30 days. |
Exclusion criteria: Patients were excluded if
| ||||
Malik et al., 202017 US Funding: None | A retrospective cohort study with propensity score matching based on the US NIS database | A total of 2,910 propensity score-matched elderly patients (1,455 in each group) who underwent mitral valve repair Age, years – Mean (SD) years TMVR: 83.7 (2.84) SMVR: 83.7(2.5) Sex – Male, % TMVR: 46.7 SMVR: 49.1 Risk assessment Elixhauser comorbidity score – Mean (SD) TMVR: 5.5 (2.0) TMVR: 5.6 (2.0) Inclusion Criteria Patients ≥ 80 years of age who underwent TMVR or SMVR between 2012 and 2016 Exclusion criteria Data of patients were excluded if they underwent concomitant cardiac surgery or other valvular procedures including aortic, pulmonary, or tricuspid, or if they had MV replacement. | TMVR with MitraClip vs. SMVR | Outcomes:
Follow-up: Not clearly reported. |
Ullah et al., 202018 US Funding: None reported | A retrospective cohort study based on the US NIS database | A total of 19, 580 patients treated for MR with either TMVR (n = 1,120 [6%]) or OMVR (n = 18,460 [94%]) Age, years – Mean (SD) TMVR: 67.0(13) OMVR: 63.7(14) Sex – Male, % TMVR: 29.4 OMVR: 37.4 Risk assessment – NR Inclusion Criteria: Adult patients (> 18 years old) who underwent TMVR or SMVR between October 2016 and September 2017 Exclusion criteria: Patients were excluded if they under 18 years old. | TMVR with MitraClip vs. SMVR | Outcomes:
Follow-up: Not clearly reported. |
Körber et al., 201815 Germany Funding: None reported. The authors stated that have no conflicts of interest to declare. | A retrospective cohort study analyzing patients’ data from a single health care institution | A total of 378 patients receiving treatment for MR using TMVR (n = 196) and SMVR (n = 182). Age, years – Mean (SD) TMVR: 77.5 (7.7) SMVR: 62.6 (13.7) Sex – Male, n (%) TMVR: 110 (56.1) SMVR: 111 (61.0) Risk assessment score: STS score, Median (IQR) TMVR: 2.5 (1.6-4.1) SMVR: 0.7 (0.3-1.4) | TMVR (with unnamed device) vs. SMVR | Outcomes:
Follow-up, median (range): 428 days (2 to 1,092 days) |
De Bonis et al., 201616 Italy Funding: None The authors stated that they had no conflict of interests to declare. | A retrospective cohort study based on analysis of data from a single health care institution | A total of 143 treated for moderate-to-severe MR secondary to both ischemic or non-ischemic dilated cardiomyopathy using MitraClip therapy (n = 85) or SMVR (n = 58) Age, years – Mean (SD) TMVR: 69 (9.4) SMVR: 62 (10.1) Sex – Male, n (%) TMVR: 70 (82.0) SMVR: 40 (69.0) Risk assessment score: Logistic Euroscore II – mean (SD) TMVR: 19.0 (15.9) SMVR: 11.4 (3.2) NYHA class II – n (%): TMVR: 13 (15) SMVR: 9 (15) NYHA class III – n (%): TMVR: 57 (67) SMVR: 36 (62) | TMVR with MitraClip vs. surgical SMVR with edge-to-edge technique combined with annuloplasty | Outcomes:
Follow-up, median (IQR): 2.5 (1.5 to 3.6) years for MitraClip vs. 7.2 (3.4 to 9.9) years for SMVR; (P = 0.0001). |
NYHA class IV – n (%): TMVR: 15 (17) SMVR: 13 (22) Inclusion Criteria:
Exclusion criteria: Data of patients were excluded if they
|
CCI = Charlson comorbidity index; IQR = interquartile range; LOS = length of stay at hospital; LV = left ventricular; MI = myocardial infarction; MR = mitral regurgitation; MV = mitral valve; NRD = National Readmissions Database; NIS = National Inpatient Sample; NR = not reported; NYHA = New York Heart Association; OMVR = conventional open heart mitral valve replacement; SD = standard deviation; SMVR = surgical mitral valve repair; STS = Society of Thoracic Surgeons; TIA = transient ischemic attack; TMVR = transcatheter mitral valve repair.
Appendix 3: Critical Appraisal of Included Publications
Note that this appendix has not been copy-edited.
Table 26: Strengths and Limitations of Systematic Reviews and Meta-Analyses Using AMSTAR 211
Strengths | Limitations |
|---|---|
Khader et al., 20213 | |
The authors specified objectives of the SR and PICO clearly. A systematic literature search was conducted in multiple databases for relevant published papers. Titles and abstracts were screened based on specified inclusion and exclusion criteria by 3 authors. Eligible studies for inclusion were cross-checked by 2 others, who evaluated and resolved discrepancy. Accuracy of extracted data were cross-checked by 2 reviewers. The meta-analysis used a random-effect model, with heterogeneity investigated by Cochrane's test and the I2 statistic, and publication bias was assessed by funnel plots. There was no funding for the study, and the authors declared that they had no conflict of interest. | It was unclear if a protocol was prepared before the review started, and a protocol for SR was not published. Thus, it could not be ascertained if there had been any significant deviations from the protocol that could have introduced risk of bias. Most of the included primary studies were retrospective cohort studies (except 1 RCT) with inherent risk of selection bias. The authors did not explain their selection of the study designs for inclusion in the SR. However, it appeared that the paucity of relevant studies limited the options for study selection. A list of excluded studies was not provided. However, there was a PRISMA flow diagram indicating the number and reasons for exclusion. The risk of bias in the SR’s included primary studies was not evaluated in a systematic manner. The overall effect estimates for some comparisons were derived from pooled estimates involving data from an RCT and non-randomized studies, meaning that the results may have been biased. |
Oh, et al., 20214 | |
A SR protocol was established a priori and registered on PROSPERO. The authors specified objectives of the SR and PICO clearly. A systematic literature search was conducted in 2 databases for relevant published papers. It was supplemented by a handsearch of all references of relevant reviews and eligible articles for potentially missed eligible studies. Two reviewers independently screened titles and abstracts based on specified study eligibility criteria. Eligible studies for inclusion were cross-checked by 2 others, who evaluated and resolved discrepancy. Two reviewers independently rated the quality of included studies using the NOS and the Cochrane method, as applicable, reaching a final decision by consensus. The quality of the evidence for each outcome was summarized with the GRADE. The meta-analysis used a random-effect model to calculate pooled effect estimates. Heterogeneity was assessed by Cochrane's Q test and the I2 statistic, and publication bias was evaluated using regression-based Egger’s test. Predefined sensitivity analyses were performed for outcomes, removing 1 study at a time. The interpretation and discussion of the results considered that the significantly different comorbidity burden among the non-randomized patients across the study groups could potentially impact the conclusions. There was no funding for the study, and the authors declared that they had no conflict of interest. | All of the primary studies included in the SR were judged to be at unclear risk of bias, and most of them were retrospective cohort studies (except 1 RCT), with inherent risk of selection bias. Data extraction was not performed in duplicate, and it was unknown if the accuracy of extracted data were independently confirmed. There was inconsistent data reporting in different parts of the article. It is unclear if the conflicting data may be explained by the author-reported “ongoing evaluation” and incomplete updates in some parts of the paper. The authors did not explain their selection of the study designs for inclusion in the SR. However, it appeared that the paucity of relevant studies limited the options for study selection. A list of excluded studies was not provided, although a PRISMA flow diagram indicating the number and reasons for exclusion was available. |
Yuan et al., 20218 | |
The authors specified objectives of the SR and PICO clearly. A systematic literature search was conducted in multiple databases for relevant published papers. Two reviewers independently screened titles and abstracts to select studies for inclusion based on specified study eligibility criteria. Any disagreement was resolved through discussion involving a third author. Data extraction was performed in duplicate, with disagreement resolved through discussion involving a third reviewer. The meta-analysis used a random-effect model to calculate pooled effect estimates. Heterogeneity was assessed by Cochrane's Q test and the I2 statistic, There was no funding for the study. | It was unclear if a protocol was prepared before the review started, and a protocol for the SR was not published. Thus, it could not be ascertained if there had been any significant deviations from the protocol that could have introduced risk of bias. The quality or risk of bias of the primary studies was not evaluated. All but 1 of the 14 primary studies were retrospective cohort studies with inherent risk of selection bias. The included studies were not evaluated for publication bias. The authors did not explain their selection of the study designs for inclusion in the SR. However, it appeared that the paucity of relevant studies limited the options for study selection. A list of excluded studies or reasons for exclusion was not provided. The reporting was suboptimal, with some forest plots including data from an author not listed in the original included studies. |
Barros da Silva et al., 202013 | |
A SR protocol was established a priori and registered on PROSPERO. The authors specified objectives of the SR and PICO clearly. A systematic literature search was conducted in 2 databases for relevant published papers. Two reviewers independently screened titles and abstracts, as well as full- text of potentially eligible articles based on pre-specified inclusion and exclusion criteria. Disagreements were resolved through discussions involving a third author. Two reviewers independently rated the quality of included studies using the NOS for non-randomized studies and the Cochrane RoB tool for an RCT. A random-effects and cumulative meta-analysis was performed to calculate pooled effect estimates. Heterogeneity was assessed using the I2 statistic and assumed to be significant if it exceeded 50%. Publication bias was evaluated using Egger’s test and both Galbraith and normalized Galbraith plots. There was no funding for the study, and the authors declared that they had no conflict of interest. | All but 1 of the 7 relevant primary studies included in the SR and comparing MitraClip and SMRV were retrospective cohort studies without randomization to minimize the risk of selection bias. It was unclear if data extraction was performed in duplicate, and it was unknown if the accuracy of extracted data were independently confirmed. The authors did not explain their selection of the study designs for inclusion in the SR. However, it appeared that the paucity of relevant studies limited the options for study selection. The authors did not provide a list of excluded studies. However, they provided a PRISMA flow diagram indicating the number and reasons for exclusion. The overall effect estimates were derived from pooled estimates that included an RCT and non-randomized studies, meaning that the results may have been biased. |
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; GRADE = Grading of Recommendations Assessment, Development and Evaluation; NOS = Newcastle Ottawa Scale; PICO = population, intervention, control group and outcome; PRISMA = Preferred Reporting Items for Systematic Reviews and Meta-Analyses; PROSPERO = International prospective register of systematic reviews; RoB = risk of bias; RCT randomized controlled trial; SR = systematic review.
Table 27: Strengths and Limitations of Clinical Studies Using the Downs and Black checklist12
Strengths | Limitations |
|---|---|
Jogu et al., 202014 | |
The objective of the study was stated clearly. Patients’ characteristics were described. Data for the study were likely to be representative of the population of interest since they were obtained from the NIS database, which is the largest publicly available all-payer health care database in the US. The interventions of interest were well-described. The settings and staff who performed the procedures and provided care were likely to be representative of treatment received by most patients in the US given that data were collected across the country. The outcomes to be measured and the findings were reported clearly along with estimates of the random variability and P values. | It is a non-randomized study lacking randomization that reduces the risk of selection bias. Risk factors for surgery, such as age and comorbidities, were higher for patients in the TMVR group compared with those in the SMVR group. The study used inpatient data only. Therefore, it had no information about outcomes and complications after the patients were discharged. Thus, intermediate and long-term results were lacking. Information concerning the history of mitral valve repair (i.e., first time or reoperation) was not provided. The procedures were performed in a variety of settings by different surgeons and staff. Therefore, it was unknown whether the reported results were influenced by the differences in expertise and facilities. The study was based on data from the US, and it is unclear if its findings would be generalizable in the Canadian context. Source of funding was unclear. At least 1 author reported receiving support from external sources. However, it was unknown if the support was for this study. |
Lima et al., 20206 | |
The authors specified objectives of the study and outcomes to be measured. The analysis was based on data the US Nationwide Readmissions Database, making it likely to be representative of the population of interest, at least in that country. Patients’ characteristics and interventions of interest were well-described. Results were analyzed with appropriate statistical methods and findings were well- reported. There was no funding, and the authors declare no conflicts of interest. | It is a non-randomized study lacking randomization that reduces the risk of selection bias. Risk factors for surgery, such as age and comorbidities, were significantly higher for patients in the TMVR group compared with those in the SMVR group. For patients with multiple readmissions withing 30 days, only the first event was included in analysis. Thus, the results do not represent a complete readmission history within 30 days, and readmission data beyond 30 days were censored. The study used inpatient data only. Therefore, it had no information about outcomes and complications after the patients were discharged. Thus, intermediate and long-term results were lacking. The procedures were performed in a variety of settings by different surgeons and staff. Therefore, it was unknown whether the reported results were influenced by the differences in expertise and facilities. The study was based on data from the US, and it is unclear if its findings would be generalizable in the Canadian context. |
Malik et al., 202017 | |
The objective of the study was stated clearly. Patients’ characteristics were described. Data for the study were likely to be representative of the population of interest since they were obtained from the NIS database, which is the largest publicly available all-payer health care database in the US. Analyses were based on data from a propensity-score-matched population across the comparing groups to minimize bias due to differences. The interventions of interest were well-described The settings and staff who performed the procedures and provided care were likely to be representative of treatment received by most patients in the US given that data were collected across the country. The outcomes to be measured and the findings were reported clearly along with estimates of the random variability and P values. The authors stated that they had no conflict of interest to declare. | It is a non-randomized study lacking randomization that reduces the risk of selection bias. Thus, regardless of propensity score matching, the study is inherently likely to have more systemic biases than a RCT due to its retrospective cohort design. The inclusion criteria limited eligibility to participate in the study to patients who were at least 80 years old. Thus, the generalizability of the results in younger patients was unknown. The study used inpatient data only. Therefore, it had no information about outcomes and complications after the patients were discharged. Thus, intermediate and long-term results were lacking. Analyzed patient data were defined using 2 different versions of ICD clinical modification codes (i.e., the ICD-10-CM and ICD-9-CM). It was unknown whether the code definitions from the 2 sources aligned perfectly or if there were differences with potential to impact the findings. The procedures were performed in a variety of settings by different surgeons and staff. Therefore, it was unknown whether the reported results were influenced by the differences in expertise and facilities. The study was based on data from the US, and it is unclear if its findings would be generalizable in the Canadian context. |
Ullah et al., 202018 | |
The objective of the study was stated clearly. Patients’ characteristics were described. Data for the study were likely to be representative of the population of interest since they were obtained from the NIS database, which is the largest publicly available all-payer health care database in the US. The interventions of interest were well-described and outcomes to be measured were reported. The settings and staff who performed the procedures and provided care were likely to be representative of treatment received by most patients in the US given that data were collected across the country. The authors stated that they had no disclosures to make. | It is a non-randomized study lacking randomization that reduces the risk of selection bias. Risk assessment scores were not provided; therefore, it was unknown how the 2 groups compared in terms of risk that could potentially impact the results. Reporting was suboptimal. For example, it was unclear whether the length of hospital stay data referred only to the post-procedure period or also included the time on admissions before the procedure, and there was inconsistency between reported patients’ age in text and the corresponding data table. The study used inpatient data only. Therefore, it had no information about outcomes and complications after the patients were discharged. Thus, intermediate and long-term results were lacking. Information concerning the history of mitral valve repair (i.e., first time or reoperation) was not provided. The procedures were performed in a variety of settings by different surgeons and staff. Therefore, it was unknown whether the reported results were influenced by the differences in expertise and facilities. The study was based on data from the US, and it is unclear if its findings would be generalizable in the Canadian context. |
Körber et al., 201815 | |
The objective of the study was stated clearly. Patients’ characteristics were described. The interventions of interest were well-described. The outcomes to be measured and the findings were reported clearly along with estimates of the random variability and P values. The authors stated that they had no conflict of interest to declare. | It is a non-randomized study lacking randomization that reduces the risk of selection bias. Age, STS scores and comorbidities, were higher for patients in the TMVR group than for those in the SMVR group. A sample size calculation was not performed, and it was unclear if the study was adequately powered for all outcomes. Due to its relatively recent introduction, patients in the TMVR group received treatment during a different period than those in the and SMVR group, although with some overlap. Thus, it was unknown whether there were differences in health care delivery that could have potentially impacted the reported findings. The study was conducted in a single institution in Germany; therefore, the generalizability of the findings in the Canadian context is unknown. |
De Bonis et al., 201616 | |
The objective of the study was stated clearly. Patients’ characteristics were described. The interventions of interest were well-described. The outcomes to be measured and the findings were reported clearly along with estimates of the random variability and P values. The authors stated that they had no conflict of interest to declare. | It is a non-randomized study lacking randomization that reduces the risk of selection bias. Patients in the SMVR group were treated before the TMVR technique (i.e., MitraClip) became available. Thus, given the changes in techniques and health care delivery over time, it was unclear whether the results could have been different if the procedures were performed within the same period. Participation in the study was restricted to patients who had severe or moderately severe secondary (functional) MR. Thus, the generalizability of the results in patients with primary (degenerative) MR was unknown. Some patients received multiple MitraClips implantation when it was deemed necessary to achieve the best possible outcome. Also, some patients (predominantly in the SMVR group) were given post-operative prophylactic support with intra-aortic balloon pump. Thus, the lack of standardization in the delivery of the index interventions and immediate post-operative care made an even comparison difficult. Age and the risk of operative mortality, as measured by log Euroscore, were higher for patients in the TMVR group compared with those in the SMVR group. A sample size calculation was not performed, and it was unclear if the study was adequately powered for all outcomes. The study was conducted in a single institution in Italy; therefore, the generalizability of the findings in the Canadian context is unknown. |
CM = clinical modification; ICD = International Classification of Diseases; NA = not applicable; NIS = national inpatient sample; SMVR = surgical mitral valve repair; TMVR = transcatheter mitral valve repair.
Appendix 4: Main Study Findings and Authors’ Conclusion
Note that this appendix has not been copy-edited.
Summary of Findings from Included Studies by outcome
Table 28: Summary of Stroke Outcome
Study Citation and Study Design | Detailed Findings |
|---|---|
Stroke – TMVR versus SMVR | |
Khader et al., (2021)3 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 8 studies (n = 3,382) found that the odds for stroke were not statistically significantly different between the MitraClip and SMVR groups (OR = 1.50; 95% CI, 0.62 to 3.64; P = 0.370). |
Oh et al., (2021)4 SR (1 RCTs and 8 retrospective cohort studies) | Analysis of data from 5 studies (n = 975) found that the odds for stroke were not statistically significantly different between the MitraClip and SMVR groups (OR = 0.68; 95% CI, 0.34 to 1.37; P = 0.28). |
Barros da Silva et al., (2020)13 SR (3 RCTs and 9 retrospective cohort studies) | Analysis of data from 7 studies (n = 930) found that the odds for stroke were not statistically significantly different between the MitraClip and SMVR groups (OR = 0.49; 95% CI, 0.17 to 1.42; P = 0.19). |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: < 11 (NR) vs. SMVR: 137 (2.0). The difference was statistically significant in favour of MitraClip (OR = 0.37 [95 CI, 0.15 to 0.92]; P = 0.02). |
Lima et al., (2020)6 A retrospective cohort study | n (%) – TMVR: < 10 (< 0.7) vs. SMVR: 120 (1.8). The difference was statistically significant in favour of MitraClip (OR = 0.21 [95 CI, 0.08 to 0.54]; P = 0.0012). |
Körber et al., (2018)15 A retrospective cohort study | n (%) – TMVR: 2 (1.0) vs. SMVR: 4 (2.4). The difference was not statistically significant (P = 0.43). |
CI = confidence interval; OR = odds ratio; RCT = randomized controlled trial; SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 29: Summary of Acute Kidney Injury Outcome
Study Citation and Study Design | Detailed Findings |
|---|---|
AKI – TMVR versus SMVR | |
Oh et al., (2021)4 SR (1 RCTs and 8 retrospective cohort studies) | Analysis of data from 6 studies (n = 1,156) found that the odds for AKI events were not statistically significantly between the MitraClip and SMVR groups (OR = 0.82: 95% CI, 0.35 to 1.92; P = 0.65). |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: 87 (11.0) vs. SMVR: 732 (9.0). The difference was not statistically significant (OR = 1.26 [95 CI, 0.99 to 1.60]; P = 0.06). |
Lima et al., (2020)6 A retrospective cohort study | n (%) – TMVR: 198 (8.0) vs. SMVR: 694 (10.7). The difference was not statistically significant (OR = 0.72 [95 CI, 0.48 to 1.07]; P = 0.10). |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | n (%) – TMVR: NR (10.7) vs. SMVR: NR (25.1). The difference was statistically significant in favour of MitraClip (P < 0.001). |
Körber et al., (2018)15 A retrospective cohort study | n (%) – TMVR: 35 (17.9) vs. SMVR: 47 (25.8). A multivariable regression analysis showed that the risk of AKI was significantly lower with TMVR than with SMVR (OR 0.22, 95% CI, 0.11 to 0.44; P < 0.001, sensitivity analysis). |
AKI = acute kidney injury; CI = confidence interval; NR = not reported; OR = odds ratio; RCT = randomized controlled trial; SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 30: Summary of Bleeding Outcomes
Study Citation and Study Design | Detailed Findings |
|---|---|
Bleeding – TMVR versus SMVR | |
Barros da Silva et al., (2020)13 SR (3 RCTs and 9 retrospective cohort studies) | Analysis of data from 5 studies (n = 626) found that the odds for new-onset major bleeding after procedure were statistically significantly higher with SMVR than with TMVR using MitraClip (OR = 0.25; 95% CI, 0.11 to 0.56; P < 0.0007). |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: 157 (21.0) vs. SMVR: 3170 (40.0). The difference was statistically significant in favour of MitraClip (OR = 0.30 [95 CI, 0.32 to 0.47]; P < 0.0001). |
Lima et al., (2020)6 A retrospective cohort study | n (%) – TMVR: 269 (24.2) vs. SMVR: 3,298 (43.0). The difference was statistically significant in favour of MitraClip (OR = 0.42 [95 CI, 0.32 to 0.56]; P < 0.0001). |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | n (%) – TMVR: NR (9.3) vs. SMVR: NR (39.5). The difference was statistically significant in favour of MitraClip (P < 0.001). |
CI = confidence interval; HR = hazard ratio; NR = not reported; OR = odds ratio; SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 31: Summary of Heart-Related Outcomes
Study Citation and Study Design | Detailed Findings |
|---|---|
Permanent Pacemaker implantation – TMVR versus SMVR | |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: < 11 (NR) vs. SMVR: 302 (4.0). The difference was statistically significant in favour of MitraClip (OR = 0.23 [95 CI, 0.11 to 0.50]; P < 0.0001). |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | n (%) – TMVR: NR (0.7) vs. SMVR: NR (5.8). The difference was statistically significant in favour of MitraClip (P < 0.001). |
Cardiac arrest – TMVR versus SMVR | |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: < 11 (NR) vs. SMVR: 112 (1.4). The difference was not statistically significant (OR = 0.55 [95 CI, 0.24 to 1.26]; P = 0.19). |
Cardiogenic shock – TMVR versus SMVR | |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: 17 (2.0) vs. SMVR: 249 (3.0). The difference was not statistically significant (OR = 0.70 [95 CI, 0.43 to 0.1.15]; P = 0.19). |
Lima et al., (2020)6 A retrospective cohort study | n (%) – TMVR: 33 (1.6) vs. SMVR: 231 (3.3). The difference was statistically significant in favour of TMVR (OR = 0.47 [95 CI, 0.27 to 0.82]; P = 0.01). |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | n (%) – TMVR: NR (2.1) vs. SMVR: NR (7.9). The difference was statistically significant in favour of MitraClip (P < 0.001). |
Acute myocardial infarction – TMVR versus SMVR | |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: < 11 (NR) vs. SMVR: 32 (0.004). The difference was not statistically significant (OR = 1.30 [95 CI, 0.46 to 0.3.68]; P = 0.55). |
Lima et al., (2020)6 A retrospective cohort study | n (%) – TMVR: 17 (1.5) vs. SMVR: 76 (1.2). The difference was not statistically significant (OR = 1.27 [95 CI, 0.36 to 4.41]; P = 0.71). |
Atrial fibrillation – TMVR versus SMVR | |
Barros da Silva et al., (2020)13 SR (3 RCTs and 9 retrospective cohort studies) | Analysis of data from 4 studies (n = 498) found that the odds for new onset AF after procedure were statistically significantly higher with SMVR than with TMVR using MitraClip (OR = 0.20; 95% CI, 0.06 to 0.70; P = 0.01). |
CI = confidence interval; NR = not reported; OR = odds ratio; RCT = randomized controlled trial; SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 32: Summary of Respiratory and Vascular Complications Outcomes
Study Citation and Study Design | Detailed Findings |
|---|---|
Respiratory complications – TMVR versus SMVR | |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | % – TMVR: 1.7 SMVR: 4.1. The difference was not statistically significant (P = 0.0807). |
Vascular complications – TMVR versus SMVR | |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | % – TMVR: < 0.3 vs. SMVR: 2.1. The difference was not statistically significant P = 0.0551). |
SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 33: Summary of Length of Hospital Stay Outcomes
Study Citation and Study Design | Detailed Findings |
|---|---|
Length of hospital stay – TMVR versus SMVR | |
Khader et al., (2021)3 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 8 studies (n = 3,382) found that after the procedures, patients in the MitraClip group stayed in hospital for a statistically significantly shorter period than those in the SMVR group (SMD = 0.885; 95% CI, 0.77 to 0.99; P < 0.001). |
Oh et al., (2021)4 SR (1 RCTs and 8 retrospective cohort studies) | Analysis of data from 7 studies (n = 1,177) found that after the procedures, patients in the MitraClip group stayed in hospital for a statistically significantly shorter period than those in the SMVR group (MD = −3.86; 95% CI, −4.73 to 0.-2.99; P < 0.01). |
Jogu et al., (2020)14 A retrospective cohort study | Days, median (IQR)– TMVR: 2 (1 to 5) vs. SMVR: 6 (4 to 7). The difference was statistically significant in favour of MitraClip (CIE = −3.26 [95 CI, −3.72 to −2.80]; P < 0.0001). |
Lima et al., (2020)6 A retrospective cohort study | Days, mean (SD)– TMVR: 5.4 (0.32) vs. SMVR: 7.9 (0.13). The difference was statistically significant in favour of TMVR (P < 0.0001). |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | Days, mean (SD)– TMVR: 4.7 (6.1) vs. SMVR:10.4 (7.6). The difference was statistically significant in favour of MitraClip (P < 0.01). |
Ullah et al., (2020)18 A retrospective cohort study with propensity score matching | Days, mean (SD)– TMVR: 11.56 (10.0) vs. SMVR:14.01 (12.7). The difference was statistically significant in favour of MitraClip (P < 0.0001). |
Körber et al., (2018)15 A retrospective cohort study | Days, median (IQR)– TMVR: 6 (4 to 8) vs. SMVR: 10 (8 to 12). The difference was statistically significant in favour of TMVR (P < 0.001). |
De Bonis et al., (2016)16 A retrospective cohort study | Days, median (IQR)– TMVR: 4.8 (3.8 to 7.0) vs. SMVR: 10.0 (8.0 to 13.0). The difference was statistically significant in favour of MitraClip (P < 0.0001). |
CI = confidence interval; CIE = change in estimate; IQR = interquartile range; MD = mean difference; RCT = randomized controlled trial; SD = standard deviation; SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 34: Summary of Mortality and Survival Outcomes
Study Citation and Study Design | Detailed Findings |
|---|---|
Mortality – TMVR versus SMVR | |
Khader et al., (2021)3 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 8 studies (n = 3,364) found no statistically significant difference in the odds for an early mortality between MitraClip and SMVR (OR = 1.63; 95% CI, 0.63 to 4.23; P = 0.139). |
Oh et al., (2021)4 SR (1 RCTs and 8 retrospective cohort studies) | Analysis of data from 5 studies (n = NR) found no statistically significant difference in the overall mortality associated with MitraClip compared with SMVR after a mean follow-up period of 4.8 years (HR = 2.06; 95% CI, 0.98 to 4.28; P = 0.06). Analysis of data from 8 studies (n = 1,435) found no statistically significant difference in the odds for 30-day mortality associated with MitraClip compared with SMVR (OR = 1.04; 95% CI, 0.58 to 1.88; P = 0.88). Analysis of data from 3 studies (n = 756) found no statistically significant difference in the odds for 5-year mortality associated with MitraClip compared with SMVR (OR = 1.44; 95% CI, 0.56 to 3.70; P = 0.44). |
Yuan et al., (2021)8 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 11 studies (n = 2,144) found no statistically significant difference in the odds for 30-day mortality associated with MitraClip compared with SMVR (OR = 0.88; 95% CI, 0.53 to 1.47; P = 0.64). Analysis of data from 11 studies (n = 2,984) found that the odds for mortality at 1-year associated with MitraClip were statistically significantly higher compared with SMVR (OR = 1.79; 95% CI, 1.4 to 2.28; P < 0.00001). Analysis of data from 7 studies (n = 1,749) found that the odds for mortality associated with MitraClip were statistically significantly higher compared with SMVR > 3 years after the procedures (OR = 2.26; 95% CI, 1.04 to 4.92; P < 0.00001). |
Jogu et al., (2020)14 A retrospective cohort study | n (%) – TMVR: 13 (2.0) vs. SMVR: 88 (1.0). The difference was not statistically significant (OR = 1.50 [95 CI, 0.84 to 2.70]; P = 0.17). |
Malik et al., (2020)17 A retrospective cohort study with propensity score matching | n (%) – TMVR: NR (0.7) vs. SMVR: NR (3.1). The difference was statistically significant in favour of MitraClip (P = 0.0262 |
Ullah et al., (2020)18 A retrospective cohort study with propensity score matching | n (%) – TMVR: 150 (13.4) vs. OMVR: 985 (5.3). The adjusted odds of in-hospital mortality accounting for comorbidities showed no significant difference between the 2 groups (OR = 1.2, 95% CI, 0.93 to 1.68; P = 0.15). |
Survival – TMVR versus SMVR | |
Khader et al., (2021)3 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 4 studies (n = NR) found that the survival rate was not statistically significantly different between MitraClip and SMVR at both the midterm (HR = 1.69; 95% CI, 0.59 to 4.84; P = 0.332; n = NR) and long-term (HR = 0.70; 95% CI, 0.35 to 1.14; P = 0.323; n = 1,216) assessments. Midterm and long-term were defined as 1 to 2 years and 4 to 5 years, respectively. |
De Bonis et al., (2016)16 A retrospective cohort study | Overall survival, Mean % (SD) – TMVR: 77 (5.6) vs. SMVR: 74 (5.1). The difference was not statistically significant (P = 0.2). Freedom from cardiac death at 4 years, Mean % (SD) – TMVR: 82 (5.2) vs. SMVR: 84 (4.6), The difference was not statistically significant (P = 0.5). |
CI = confidence interval; HR = hazard ratio; MR = mitral regurgitation; NR = not reported; OR = odds ratio; RCT = randomized controlled trial; SD = standard deviation; SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 35: Summary of Recurrent or Residual MR Outcomes
Study Citation and Study Design | Detailed Findings |
|---|---|
Recurrent MR – TMVR versus SMVR | |
Khader et al., (2021)3 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 5 studies (n = 520) found that the odds of a recurrent moderate-to-severe MR (i.e., grade 3 or 4) 1 to 2 years after the procedure were significantly higher in the MitraClip group than in the SMVR group (OR = 0.29; 95% CI, 0.19 to 0.46; P < 0.001). |
Oh et al., (2021)4 SR (1 RCTs and 8 retrospective cohort studies) | Analysis of data from 6 studies (n = 685) found that the odds of a post-procedural recurrent severe MR (i.e., grade 3 or 4) were significantly higher in the MitraClip group then the SMVR group at the time of discharge (OR = 2.81; 95% CI, 1.39 to 5.69; P < 0.01). Analysis of data from 3 studies (n = 684) found that the odds of a post-procedural recurrent severe MR were significantly higher in the MitraClip group than in the SMVR group at 5 years after the procedures (OR = 2.46; 95% CI, 1.54 to 3.94; P < 0.01). |
Yuan et al., (2021)8 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 10 studies (n = 1,193) found that the odds for a recurrent moderate-to-severe MR (i.e., ≥ grade 3) 30 days after the procedure were significantly higher in the MitraClip group then the SMVR group (OR = 9.51; 95% CI, 5.38 to 16.83; P < 0.00001). Analysis of data from 6 studies (n = 726) found that the odds for a recurrent moderate-to-severe MR 1 year after the procedure were statistically significantly higher with MitraClip compared with SMVR (OR = 3.14; 95% CI, 1.20 to 8.25; P 0.02). Analysis of data from 4 studies (n = 756) found that the odds for a recurrent moderate-to-severe MR were statistically significantly higher with MitraClip compared with SMVR > 3 years after the procedures (OR = 8.47; 95% CI, 4.76 to 15.10; P < 0.00001). |
Körber et al., (2018)15 A retrospective cohort study | Post-operative MR > grade 2, mean (SD) – TMVR: 14 (7.3) vs. SMVR: 4 (2.4). The difference was statistically significant in favour of SMVR (P = 0.033). |
De Bonis et al., (2016)16 A retrospective cohort study | Freedom from MR ≥ grade 3 at 4 years, Mean % (SD) – TMVR: 75 (7.6) vs. SMVR: 94 (3.3). The difference was statistically significant in favour of SMVR (P = 0.004). Freedom from MR ≥ grade 2 at 4 years, Mean % (SD) – TMVR: 37 (7.2) vs. SMVR: 82 (5.2). The difference was statistically significant in favour of SMVR (P = 0.0001). |
CI = confidence interval; HR = hazard ratio; MR = mitral regurgitation; OR = odds ratio; RCT = randomized controlled trial; SD = standard deviation; SMVR = surgical mitral valve repair, TMVR = transcatheter mitral valve repair.
Table 36: Summary of Re-operation and Readmission Outcomes
Study Citation and Study Design | Detailed Findings |
|---|---|
Reoperation – TMVR versus SMVR | |
Khader et al., (2021)3 SR (1 RCT and 11 retrospective cohort studies) | Analysis of data from 4 studies (n = 559) found that the odds of reoperation were statistically significantly higher in the MitraClip group than in the SMVR group (OR = 0.392; 95% CI, 0.188 to 0.817; P = 0.012). |
Oh et al., (2021)4 SR (1 RCTs and 8 retrospective cohort studies) | Analysis of data from 4 studies (n = 530) found that the odds for reoperation were statistically significantly higher in the MitraClip group than in the SMVR group (OR = 5.28; 95% CI, 3.43 to 8.11; P < 0.01). |
Re-admission rate – TMVR versus SMVR | |
Lima et al., (2020)6 A retrospective cohort study | . 30-day readmission rate, % – TMVR: 10.7 vs. SMVR: 11.7. Unadjusted OR (1.11, 95% CI 0.89 to 1.39, P = 0.35). However, after propensity score adjustment, TMVR was associated with a lower risk of 30-day readmission compared with SMVR (adjusted OR 0.70 [95% CI, 0.51 to 0.95], P = 0.2). |
CI = confidence interval; MR = mitral regurgitation; OR = odds ratio; RCT = randomized controlled trial; SMVR = surgical mitral valve repair, SR = systematic review; TMVR = transcatheter mitral valve repair.
Appendix 5: Overlap Between Included Systematic Reviews
Note that this appendix has not been copy-edited.
Table 37: Overlap in Relevant Primary Studies Between Included Systematic Reviews
Primary study citation | Khader et al., 20213 | Oh et al., 20214 | Yuan et al., 20218 | Barros da Silva et al., 202013 |
|---|---|---|---|---|
Gyoten et al., ESC Heart Fail 2020;7:1781-1790 | No | No | Yes | No |
Niikura et al., J Am Heart Assoc 2020;9:e14874. | No | No | Yes | No |
Anwer et al., Ann Thorac Surg 2019;107:725-731 | Yes | Yes | Yes | Yes |
Buzzatti et al., J Thorac Cardiovasc Surg 2019;158:86-94 | Yes | Yes | Yes | No |
Doshi et al., Am J Cardiol. 2018;121:762‐767. | Yes | No | No | No |
Kamperidis et al., Eur Heart J Cardiovasc Imaging. 2018;19(2):208‐215. | Yes | No | No | No |
Körber et al., Am J Cardiol 2018;121:94-9. | No | Yes | No | No |
Kortlandt et al., EuroIntervention. 2019; 14(17):1733‐1739. | Yes | No | No | No |
Kortlandt et al., Circ Cardiovasc Interv 2018;11:e5985 | No | No | Yes | No |
Kreidel et al., Semin Thorac Cardiovasc Surg 2018;30:152-7. | No | Yes | No | No |
Ostovar et al., Thorac Cardiovasc Surg 2018;66:537-544 | No | No | Yes | No |
Alozie et al., BMC Cardiovasc Disor 2017;17:85 | No | No | Yes | Yes |
Toyama et al., Echocardiography. 2017;34(3):334‐339 | Yes | No | No | No |
De Bonis et al., Eur J Cardiothorac Surg. 2016;49(1):255‐262 | Yes | Yes | Yes | No |
Ondrus et al., Interact Cardiov Th 2016;23:784-789. | No | No | Yes | Yes |
Buzzatti et al., Am J Cardiol 2015;115:487-492 | No | No | Yes | No |
Feldman et al., J Am Coll Cardiol 2015;66:2844-2854 | Yes | Yes | Yes | No |
Swaans et al., JACC Cardiovasc Interv 2014;7:875-881 | Yes | Yes | Yes | No |
Paranskaya et al., Can J Cardiol 2013;29:452-459 | Yes | Yes | Yes | Yes |
Conradi et al., Eur J Cardio‐Thoracic Surg. 2013;44(3):490‐496. | Yes | Yes | No | Yes |
Taramasso et al., Eur J Cardio-Thorac 2012;42:920-926 | Yes | No | Yes | Yes |
Feldman et al., N Engl J Med. 2011; 364:1395-406. | No | No | No | Yes |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca